Abstract
Background
Chikusetsusaponin IVa (CHS) is a triterpenoid saponin that has been demonstrated to possess multiple properties.
Objectives
This study focused on inhibiting H9N2 avian influenza virus (AIV) by CHS and investigated the underlying mechanisms, specifically targeting the nuclear factor erythroid2-related factor 2 (Nrf2) signaling pathway.
Materials and Methods
The antiviral activity of CHS was evaluated by determining its half inhibitory concentration and therapeutic index (TI) in vitro. Markers of oxidative stress, encompassing superoxide dismutase, glutathione peroxidase, and malondialdehyde levels, were assessed. Analyses of pathways from the Kyoto Encyclopedia of Genes and Genomes and Gene Ontology were conducted to elucidate the underlying mechanisms, followed by network investigation and simulations of molecular docking. Moreover, the effects of CHS on Nrf2 and mitogen-activated protein kinase (MAPK) signaling pathways were assessed in human lung carcinoma A549 cells and a mouse model of H9N2 AIV infection.
Results
CHS showed inhibitory effects on H9N2 AIV in vitro, with TI values of 4.00 (co-treatment) and 6.29 (posttreatment). CHS reduced H9N2 AIV-induced oxidative stress and was predicted to target Nrf2, MAPK/phosphoinositide 3-kinase (PI3K)-protein kinase B (Akt) signaling pathways, and oxidation–reduction processes. Molecular docking analysis revealed a high affinity between CHS and Nrf2. In A549 cells, CHS enhanced Nrf2 activation, inhibited MAPK signaling, and reduced H9N2 AIV-induced oxidative stress. The therapeutic mechanism of CHS was found to be mediated by Nrf2. In a mouse model, CHS mitigated pulmonary impairment, improving the organism’s antioxidant activity. CHS also regulated the Nrf2 and MAPK signaling pathways in lung tissue.
Conclusion
CHS inhibits H9N2 AIV and alleviates virus-induced oxidative stress by targeting the Nrf2 signaling pathway. These observations lay the groundwork for devising prospective therapeutic interventions targeting H9N2 AIV infection.
Introduction
Influenza A virus (IAV) is the main pathogen of pandemic influenza. The World Health Organization (WHO) estimates that IAV is responsible for 3–5 million severe illnesses and leads to approximately 300,000–650,000 deaths (Javanian et al., 2021). Based on the difference in surface proteins, hemagglutinin (HA) and neuraminidase (NA), IAVs are divided into a multitude of different subtypes, with 18 HA and 11 NA subtypes available (Wu et al., 2021). Among them, the H9N2 AIV is the most widely distributed in poultry, which can directly cause pathogenicity, co-infection, and immunosuppressive damage in birds, causing huge economic losses to the poultry industry (Ali et al., 2019; Sun & Liu, 2015; Zhang et al., 2008). In addition, H9N2 AIV can be transmitted from avians to mammals, including humans, which also provides internal genes to deadly avian influenza strains such as H7N9 and H5N6, causing acute lung injury (ALI) and acute respiratory distress syndrome (ARDS), which are serious health epidemics (Bi et al., 2020). Over 90% of human H9N2 infections worldwide stem from China, primarily due to vaccine and antiviral drug inefficacy alongside adverse reactions (Morens et al., 2023; Qi et al., 2021; Ye et al., 2020). Accordingly, it is crucial to continually develop new therapies for combating H9N2 viruses.
Alternanthera philoxeroides (Mart.) Griseb. belongs to the amaranth family and reportedly possesses diverse biological activities, such as antimicrobial, antioxidant, antitumor, anticancer, antiviral, and immunomodulatory properties. Chemically, multiple bioactive compounds (e.g., alkaloids, anthraquinones, flavonoids, and megastigmanes) have been extracted from Alternanthera philoxeroides (Mart.) Griseb. (Nahar et al., 2022). Among these components, chikusetsusaponin IVa (CHS), a triterpene saponin, is known for its beneficial properties, including oxidation, inflammation, tumor resistance, hypoglycemia, and lipid-lowering effects (Xu et al., 2021). Additionally, CHS exhibits strong antiviral capabilities against several viruses, including HSV-1 and -2 and cytomegalovirus (CMV). (Rattanathongkom et al., 2009). Despite its recognized effects, CHS’s specific roles and mechanisms in combating H9N2 AIV infection at the cellular level remain unclear.
The nuclear factor erythroid2-related factor 2 (Nrf2) signaling pathway is a vital target for oxidative stress-related diseases and regulates the intracellular oxidative stress response (Lu et al., 2019; Mills et al., 2018). Upregulation of heme oxygenase-1 (HO-1) through Nrf2 reduces reactive oxygen species (ROS) production (Ahmed et al., 2017; Zhao et al., 2013). Moreover, CHS can act on Nrf2 to improve oxidative stress. Therefore, we will investigate the antiviral effects of CHS from antioxidant aspects. This study aimed to evaluate both the antiviral and antioxidant properties of CHS in treating H9N2 AIV infections through both in vitro and in vivo approaches. We found that CHS inhibits H9N2 AIV replication and reduces virus-induced oxidative stress. CHS was predicted to target Nrf2 signaling and experimentally shown to activate Nrf2, inhibit mitogen-activated protein kinase (MAPK) signaling, and attenuate cellular and mouse oxygenated stress damage from H9N2 AIV infections. Our studies demonstrated that CHS could be a viable option for curing H9N2 AIV infections.
Materials and Methods
Materials
The human lung carcinoma A549 cells were achieved from Procell in China and maintained in Ham’s F-12K medium enriched with 10% fetal bovine serum (FBS) from Gibco, USA and 1% penicillin/streptomycin (P/S) from Hyclone, USA. This cultivation took place at 37°C, 5% CO2. The H9N2 AIV was procured from the National Center for Veterinary Culture Collection in China and stored at –80 °C. The virus was propagated in embryonated chicken eggs (10 days) without specific pathogens (Beijing Meriavitong Laboratory Animal Technology Co., Ltd., China). The Reed–Muench method (50% tissue culture infective dose (TCID50) = 10–3.64/100 µL) was used to determine TCID50 (Ramakrishnan, 2016). The cell counting kit-8 (CCK-8) was obtained from Meilun Technology (Dalian, China). CHS (ChemFaces, Wuhan, China) with a purity of at least 98% (Cat. CFN92516) and oseltamivir phosphate (Lot 6002207009) was sourced from Yangtze River Pharmaceutical Group. Antibodies: Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) (bs-0755R) was purchased from Bioss Technology (Beijing, China). Extracellular regulated protein kinases (ERK) (A4782), phospho-extracellular regulated protein kinases (p-ERK) (AP0974), c-Jun N-terminal kinase (JNK) (AP4867), phospho-c-Jun N-terminal kinase (p-JNK) (AP1163), p38 mitogen activated protein kinases (p38) (A4771), and phospho-p38 mitogen activated protein kinases (p-p38) (AP0057) were purchased from ABclonal Technology. Nrf2 (16396-1-AP) was purchased from Proteintech Technology (Wuhan, China). The malondialdehyde (MDA) (A003-1), superoxide dismutase (SOD) assay kit (A001-3), and glutathione peroxidase (GSH-Px) assay kit (A005) were purchased from Nanjing Jiancheng Bioengineering Institute in Nanjing, China. ROS assay kit (S0033S) was purchased from Beyotime (Shanghai, China). Major instruments used were the Western blot (1658001, Bio-Rad), fluorescence microscopy (TH4-200, Olympus), and confocal microscopy (FV3000, Olympus).
Antiviral Effects of CHS
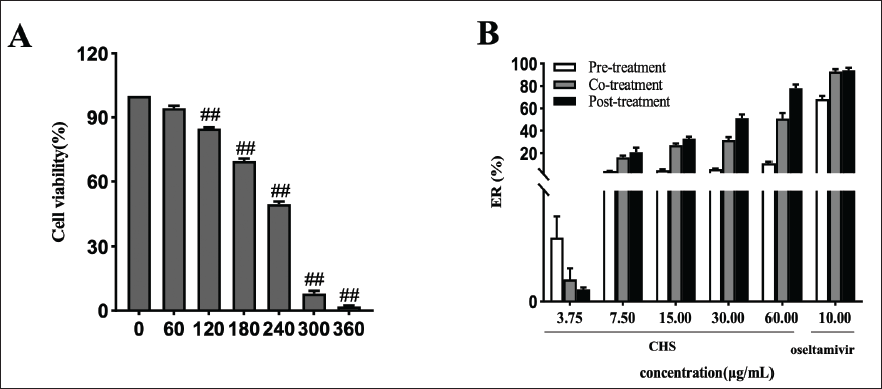
The A549 cells cultured on 96-well plates (2 × 104 cells/well) were subjected to treatment with CHS (0, 60, 120, 180, 240, 300, 360 µg/mL) for 48 hours, then with 10% CCK-8 (100 µL/well) for additional 2 hours. Cells’ absorbance was measured at 490 nm. The median toxic concentration (TC50) of CHS was reckoned by the probit regression method using SPSS 26.0 software. A549 cells were treated with CHS before, after, or simultaneously with H9N2 AIV infection. Cells were then cultured for 48 hours in a fresh medium. CHS (3.75, 7.5, 15, 30, 60 µg/mL) and oseltamivir (10 µg/mL) (100 µL/well) were applied in each group. Cell viability was assessed by the same method. The half inhibitory concentration (IC50) was determined by the probit regression method in SPSS26.0 software. The treatment index (TI) was used as an indicator of the viral suppressive efficacy of drugs (TI = TC50/IC50).
Antioxidant Assay
Concentration of 2 × 104 cells per well for A549 cells was incubated for 12 hours. Following this, the cells were in contact with 100TCID50 of H9N2 AIV and then cultured in either fresh medium alone or medium containing varying levels of CHS (15, 30, 60 µg/mL) for 48 hours. Assay kits were utilized to evaluate the activities of SOD, GSH-Px, MDA, and ROS, respectively.
Target Prediction and Enrichment Analysis
The potential targets of CHS resistant to H9N2 through oxidative stress were identified by STITCH (Search Tool for Interactions of Chemicals) (
Network Construction
The aggregated data, including gene names and scores, were uploaded into Cytoscape to generate a Compounds-Targets-Pathways network graph. According to the gene names, the pathway information, including the MAPK signaling pathway and Nrf2-mediated oxidative stress response, matches the KEGG database. Nodes were color-coded based on pathway information: MAPK pathway in red, Nrf2 pathway in green, and others in yellow.
Molecular Docking Analysis
Molecular docking studies were conducted to assess the potential binding affinity between CHS and Nrf2. The 3D crystal structures of Nrf2 were sourced from the Protein Data Bank (PDB) website (
Immunofluorescence (IF) Analysis
A549 cells underwent the same treatment as previously described and were prepared for IF analysis. After allowing the slides to dry partially, a tissue pen was used to mark a circular area on the cover slide containing evenly distributed cells. A 100 µL cell lysis solution was applied and left to react for 10 minutes. The slides were then rinsed with phosphate-buffered saline (PBS), and the labeled areas were added 3% hydrogen peroxide solution. The cells were covered with a primary antibody solution (1:200) diluted in 5% bovine serum albumin (BSA) and incubated at 4°C all night. Overnight, a secondary antibody (1:200) specific to the primary antibody’s species was applied and incubated for 50 minutes. Subsequently, 100 µL 4′,6-diamidino-2-phenylindole (DAPI) stain was added, and the slides were sealed with an antifade mounting medium before imaging with a fluorescence microscope.
Western Blot Analysis
Treated A549 cells were processed as before in Western blot analysis. RIPA lysis buffer, which contained protease inhibitors and phosphorylase inhibitors, was used to lyse the cells. Proteins were separated using SDS-polyacrylamide gel (SDS-PAGE) and transferred to polyvinylidene difluoride (PVDF) membranes. The membranes were blocked for 1 hour, and incubated overnight at 4°C with primary antibodies (1:1,000 dilution) and GAPDH (1:10,000 dilution). After three rinses with 1× TBST, they were incubated with a secondary antibody (1:3,000 dilution) for 1 hour. The protein bands were stained by enhanced chemiluminescence (ECL) chemiluminescent kits and subsequently quantified by ImageJ with GAPDH as the internal control.
siRNA Transfection
Nrf2 siRNA1 (sense: 5′-UGACAGAAGUUGACAAUUATT-3′, an-tisense: 5′-UAAUUGUCAACUUCUGUCATT-3′), Nrf2 siRNA2 (sense: 5′-GAGAAAGAAUUGCCUGUAATT-3′, an-tisense: 5′-UUACAGGCAAUUCUUUCUCTT-3’), Nrf2 siRNA3 (sense: 5′-GUAAGAAGCCAGAUGUUAATT-3′, an-tisense: 5′-GUAAGAAGCCAGAUGUUAATT-3′), and negative control siRNA (sense: 5′-UUCUCCGAACGU GUCACGUTT-3′, an-tisense: 5′-ACGUGACACGUUCGGAGAATT-3′) were synthesized and purchased from Siwega Bio technology Wuhan Co.
Cells were divided into six groups: control siRNA, control siRNA+H9N2, control siRNA+H9N2+CHS 60 µg/mL, Nrf2 siRNA, Nrf2 siRNA+H9N2, and Nrf2 siRNA+H9N2+CHS 60 µg/mL.
A549 cells (3 × 105 cells/well) were seeded in a six-well plate and cultured overnight with a serum-free medium. Subsequently, cells were transiently transfected with 100 nM Nrf2 siRNA or control siRNA for 48 h using the RiboFECTTM CP, according to the manufacturer’s protocol. After 48 hours, the cells were infected with 100TCID50 and treated with CHS at 37°C, 5% CO2.
Animal Experimental Design and Histopathological Examination
The assignment of mice to groups was completely randomized. The control group was not infected with H9N2 AIV, while the model and drug groups were intranasally inoculated with 50 µL of H9N2 AIV allantoic fluid (median lethal dose (3LD50)) for infection. Two hours after the virus infection, each administration group received CHS (15, 30, 60 mg/kg/d) or oseltamivir by gavage administration once daily for 5 consecutive days. Mice were monitored daily for 14 days to assess weight changes and observe any symptoms in the protection assay. Furthermore, to study the antioxidant effect of CHS in vivo, mice were sacrificed on the fifth day of postinfection. Lung tissues were collected and processed for histopathological examination using hematoxylin and eosin (H&E) staining.
Detection of SOD, GSH-PX, MDA, and ROS Levels in Lung Tissue
Lung tissues from each group of mice, homogenate supernatant were acquired. The MDA, SOD, and GSH-Px content were determined using thiobarbituric acid (TBA), WST-1, and colorimetric methods, respectively. The maximum absorbance of the compound was measured at 532, 550, and 412 nm, respectively, with a microplate reader. To detect lung tissue ROS levels, frozen lung tissue sections were thawed and dried at room temperature. A fluorescent quencher was added to the tissue perimeter, followed by ROS staining and DAPI staining. The slides were then covered and examined under a fluorescence microscope to capture images.
Western Blot and IF Staining for Protein Expression Analysis in Lung Tissue
Lung tissues collected from each group of mice, were homogenized and lysed in RIPA lysis buffer, and then analyzed to detect the protein concentration using a BCA kit. Western blot was used to detect the protein expression in lung tissue. IF staining was used for Nrf2 protein expression in lung tissues. For IF analysis, paraffin-embedded lung tissue samples from various groups of mice were sectioned and then deparaffinized. Antigen retrieval was performed using an EDTA buffer in a microwave. After three PBS washes, they were blocked with 5% BSA for 20 minutes. Post BSA removal, they were incubated with the primary antibody Nrf2 (dilution 1:400) at 4°C overnight in darkness in all sections. Following another set of PBS washes, they were incubated with a fluorescent secondary antibody (dilution 1:500) at 37°C for 50 minutes in darkness. After additional washes and drying, the sections were stained with DAPI (dilution 1:100) at 37°C for 10 minutes. The slices were sealed with antifade mounting medium and observed and photographed under a fluorescence microscope. Image merging was conducted using ImageJ software.
Statistical Analysis
Repeat all operations three times, with results expressed as the mean ± standard deviation (SD) (n = 3). Statistical analyses were performed using SPSS 26.0, employing a one-way analysis of variance (ANOVA) to determine the significance of differences among the groups, with p < 0.05 indicating statistical significance.
Results
Inhibitory Effects of CHS on H9N2 AIV In Vitro
The TC50 value was 215.53 ± 2.26 µg/mL for CHS. The IC50 of CHS in inhibiting H9N2 AIV was 53.84 ± 2.87 µg/mL and 34.29 ± 2.03 µg/mL for co-treatment and posttreatment, respectively. The TI of co-treatment and posttreatment was 4.00 and 6.29, respectively. The inhibition rate of pretreatment did not reach 50%, and no significant discrepancy was identified for the inhibition rates between co-treatment and posttreatment (Figure 1). Since the posttreatment showed a lower IC50 and a higher TI value, we speculated that the antiviral effect of posttreatment for 2 hours might be better than those of pretreatment and co-treatment.
CHS Reduced H9N2 AIV-induced Oxidative Stress
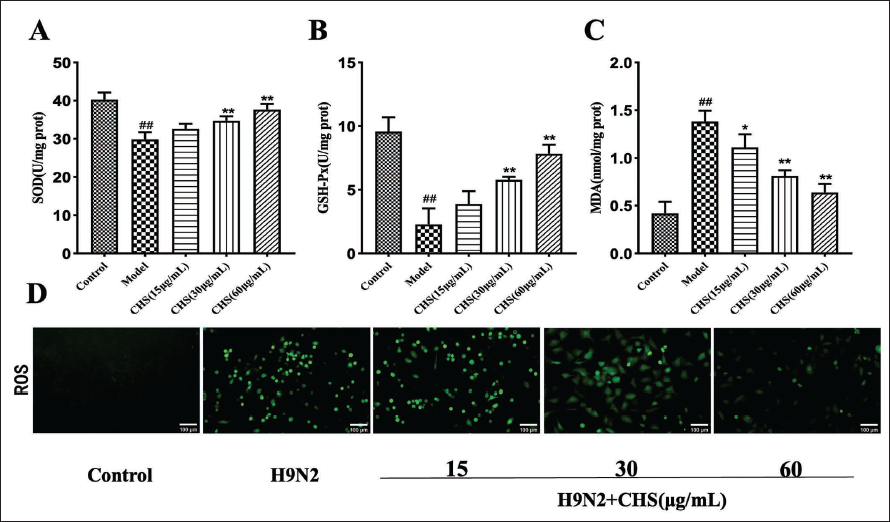
Herein, in vitro, compared to the control group, SOD and GSH-Px activities decreased notably (p < 0.01), and MDA levels increased appreciably (p < 0.01); these effects were significantly improved after CHS intervention (Figure 2A–C). Furthermore, the green fluorescence level dramatically increased when H9N2 AIV induced A549 cells, However, treatment with CHS decreased the green fluorescence level with increasing CHS concentrations (Figure 2D).
Mechanism Prediction
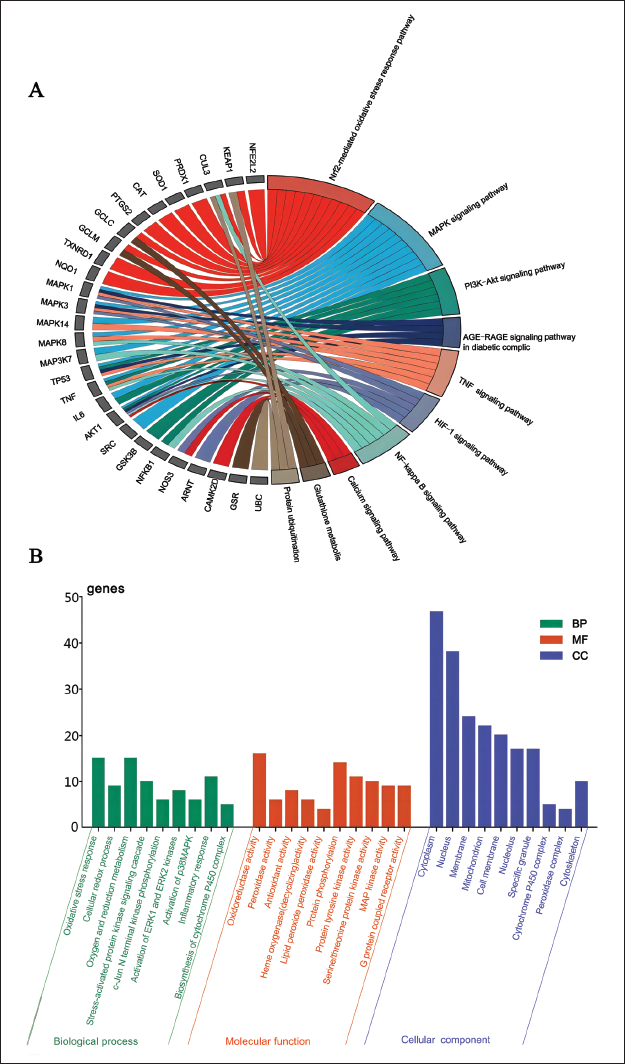
Integrated analysis of the p values, rich factor, number of genes included, and the biological roles played by each pathway was conducted to analyze the enrichment results. KEGG pathway analysis revealed that the 50 targets associated with CHS were predominantly enriched in pathways such as the Nrf2-mediated oxidative stress response, MAPK signaling, and phosphoinositide 3-kinase (PI3K)-protein kinase B (Akt) signaling (Figure 3A). Analysis of GO BP indicated that these targets are mainly involved in oxidation-reduction processes, response to oxidative stress, and cellular signal transduction. GO MF analysis suggested that the mechanisms of CHS antioxidant effects may be closely related to influencing oxidoreductase activity, protein phosphorylation, and kinase activity. GO CC analysis demonstrated that these targets are extensively located across various major cellular structures and organs, but are more concentrated in the cytoplasm, nucleus, cell membrane, and mitochondria (Figure 3B).
Analysis of Chikusetsusaponin IVa (CHS)-related Targets through Kyoto Encyclopedia of Genes and Genomes (KEGG) Pathway and Gene Ontology (GO) Analysis. (A) The Significant Enrichment of CHS-related Targets in Nuclear Factor Erythroid2-related Factor 2 (Nrf2)-mediated Oxidative Stress Response Pathway, Mitogen Activated Protein Kinase (MAPK) Signaling Pathway and Other Pathways by KEGG Pathway Analysis; (B) GO Analysis of CHS-related Targets Showing Enrichment in Biological Process (BP) Such As Oxidation–Reduction Process, Oxidative Stress Response, and Cellular Signal Transduction; Molecular Function (MF) Including Oxidoreductase Activity, Protein Phosphorylation, and Kinase Activity; and Cellular Component (CC) Such As the Cytoplasm, Nucleus, Cell Membrane and Mitochondria.
In summary, CHS may play an antioxidant role primarily by activating the Nrf2 pathway and MAPK/PI3K-Akt signaling pathways and influencing oxidation-reduction processes, protein phosphorylation, and kinase activities in the nucleus, mitochondria, cell membrane, and others. However, experimental studies are still needed to verify these analytical results.
Network Analysis
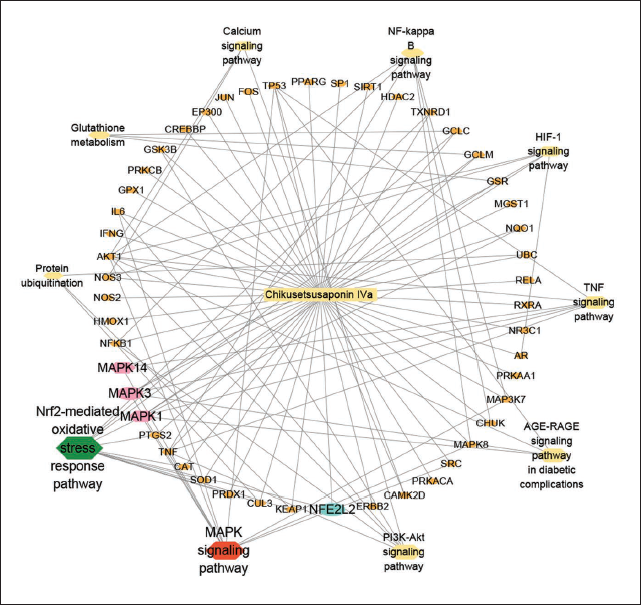
The network graph clearly shows the potential relationship between CHS, MAPK pathway, Nrf2 pathway, and oxidative stress-related genes. These pathways and genes may be important targets and mechanisms for CHS to anti-H9N2 AIV. As shown in the network graph, CHS may mainly play an anti-H9N2 AIV role by targeting Nrf2 to regulate the MAPK pathway and oxidative stress (Figure 4). It helps us systematically understand its mode of action and mechanism and provides a basis for further experimental design.
The “Compounds-Targets-Pathways” Network Illustrates the Potential Mechanisms of Chikusetsusaponin IVa (CHS) in Treating H9N2 Avian Influenza Virus (AIV) through the Regulation of Nuclear Factor Erythroid2-related Factor 2 (Nrf2) and Mitogen Activated Protein Kinase (MAPK) Pathways.
Molecular Docking Analysis
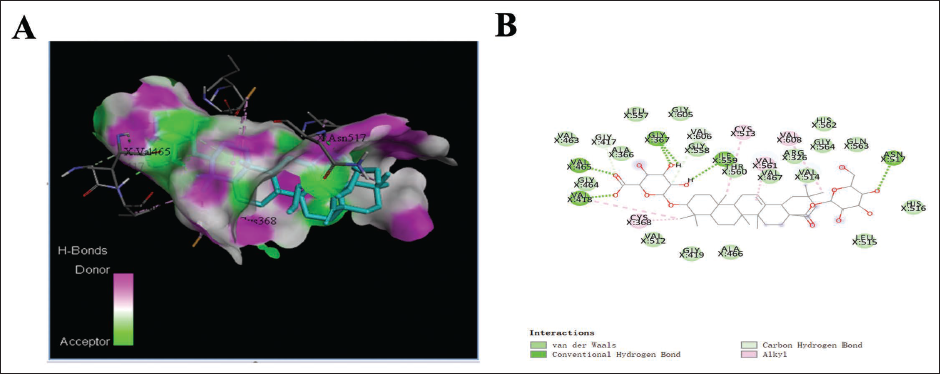
The binding energy <–5 kcal/mol indicates a high affinity between CHS and the target (Shi et al., 2021). Molecular docking simulations showed that there were multiple binding pockets for CHS in Nrf2. The binding energy of CHS with Nrf2 was –10.9 kcal/mol, indicating that CHS had great affinities with this core protein (Figure 5). This result suggested that CHS could interact with Nrf2 in both structure and electrochemistry, which provided evidence that CHS may treat H9N2 by targeting Nrf2.
The Molecular Docking Analysis of Chikusetsusaponin IVa (CHS) and Nuclear Factor Erythroid2-related Factor 2 (Nrf2) Using Discovery Studio Client. (A) The 3D Model of CHS and the Protein Nrf2; (B) The 2D Model of CHS and the Protein Nrf2.
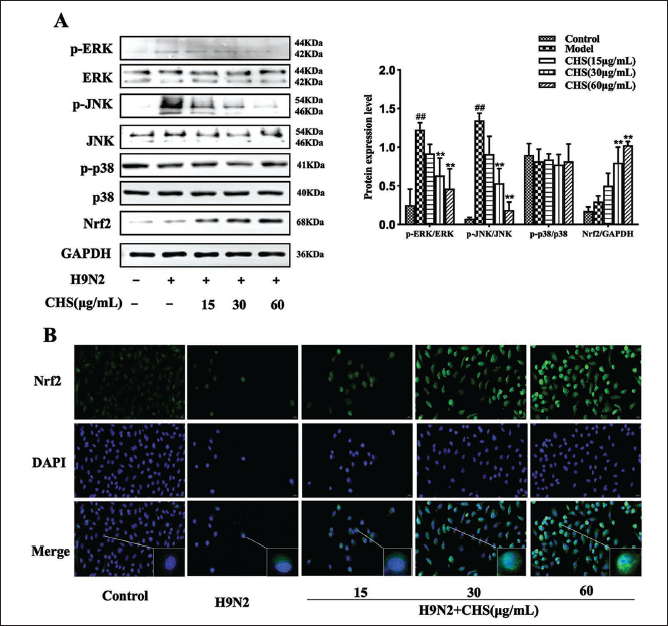
CHS Enhances Nrf2 to Inhibit MAPK and Oxidative Stress for anti-H9N2 AIV Activity
Following the obtained, the antioxidant effects of CHS post-H9N2 AIV infection were further clarified. As indicated by our results, CHS treatment significantly decreased the p-JNK and p-ERK levels and effectively increased the Nrf2 level (p < 0.01), while ERK, JNK, p-p38, and p38 expression were unchanged (Figure 6A). Furthermore, IF staining indicated an increase in nuclear Nrf2 accumulation due to CHS treatment (Figure 6B).
Nrf2 Mediates the Protective Effects of CHS Against H9N2 AIV
Western blot validation showed that siRNA successfully reduced Nrf2 expression in A549 cells, confirming the efficacy of this approach in silencing Nrf2 (Figure 7A and B).
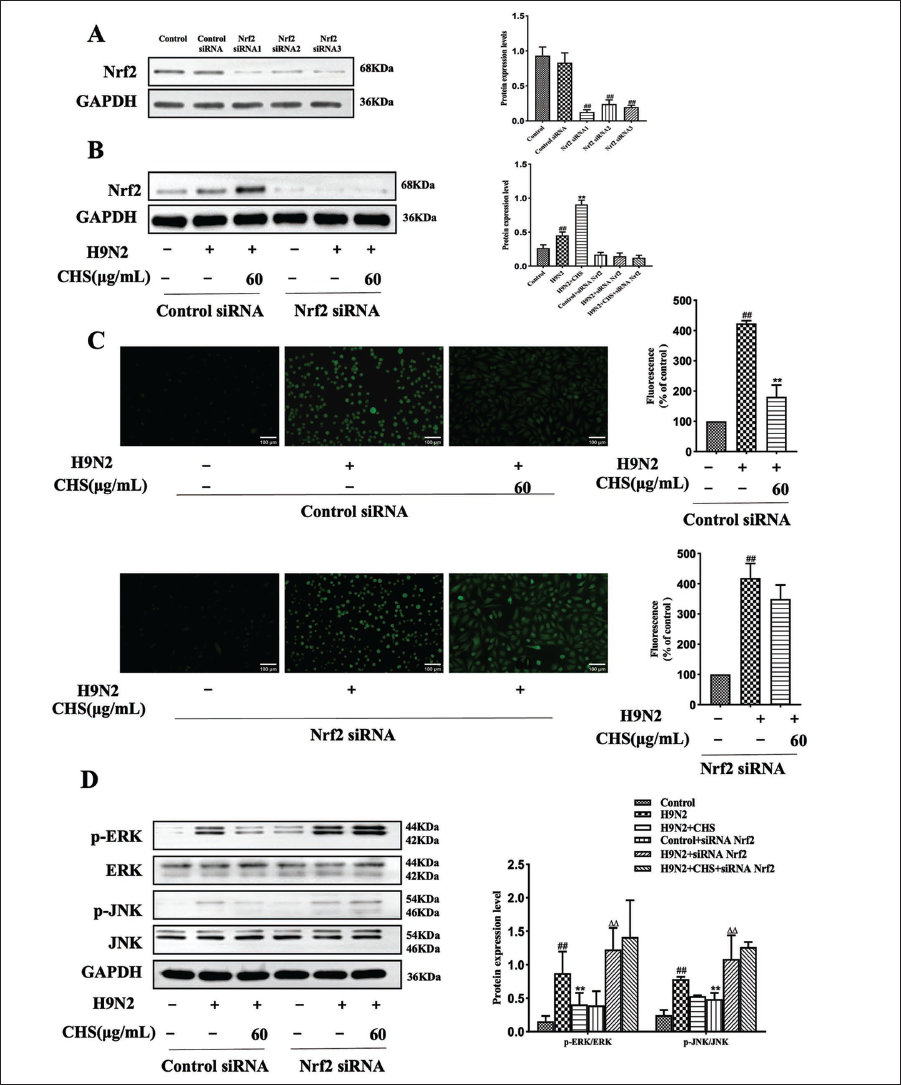
The expression of ROS levels in cells after Nrf2 silencing by siRNA is shown in Figure 7. In control siRNA cells, H9N2 AIV-induced ROS generation was significantly increased, but CHS impeded H9N2 AIV-induced ROS generation in A549 cells (p < 0.01). In A549 cells with Nrf2 downregulation, H9N2 AIV-induced ROS generation was significantly increased (p < 0.01), while CHS failed to impede ROS production induced by H9N2 AIV (Figure 7C).
To examine the role of Nrf2 in CHS’s inhibitory effect on H9N2 AIV-induced oxidative stress in A549 cells, we assessed MAPK pathway protein expression in control siRNA and Nrf2 siRNA cells after H9N2 AIV infection. The result shows that H9N2 AIV infection significantly increased p-ERK and p-JNK protein expression in both cell types. CHS (60 µg/mL) notably inhibited the upregulation of these proteins in control siRNA cells (p < 0.01) but not in Nrf2 siRNA cells (Figure 7D). These findings suggest Nrf2 is crucial for CHS’s antioxidant effect against H9N2 AIV in A549 cells, as CHS may suppress H9N2 AIV-activated MAPK signaling pathways via Nrf2 activation.
Protective Effect of CHS Against H9N2 AIV Infection in Mice
To evaluate the antiviral effects of CHS in mice infected with H9N2 AIV, changes in body weight and histological analyses of lung tissues were monitored. The control group showed no abnormalities, while the model group exhibited severe symptoms after H9N2 AIV infection. CHS and oseltamivir treatment improved the symptoms to varying degrees, and weight recovery began on the 8th day after treatment (Figure 8A).
The Protective Function of Chikusetsusaponin IVa (CHS) on H9N2 Avian Influenza Virus (AIV)-Infected Mice. (A) The Body Weight of Mice was Monitored Daily for 14 Days after Viral Infection; (B) Histological Observations of Lung Tissues.
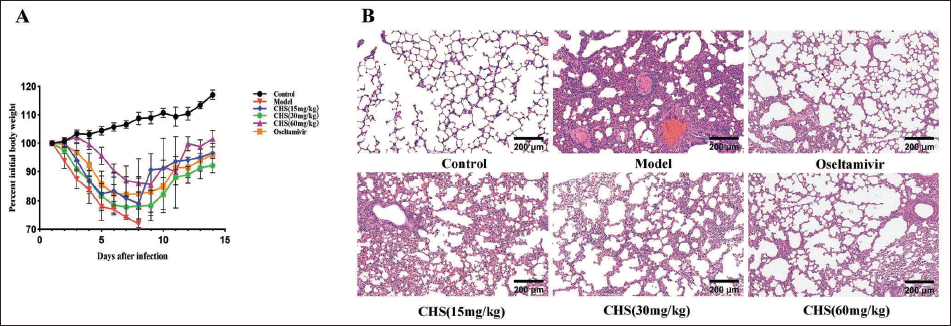
Histopathological examination revealed that lung tissue sections from the control group showed no pathological changes. Lung tissue sections from the model group displayed severe pulmonary lesions. CHS treatment (15, 30, 60 mg/kg) and oseltamivir treatment alleviated varying degrees of pulmonary lesions induced by virus infection. CHS (60 mg/kg) and oseltamivir demonstrated the most pronounced protective impact (Figure 8B). These findings indicate that CHS effectively guards against lung damage induced by H9N2 AIV.
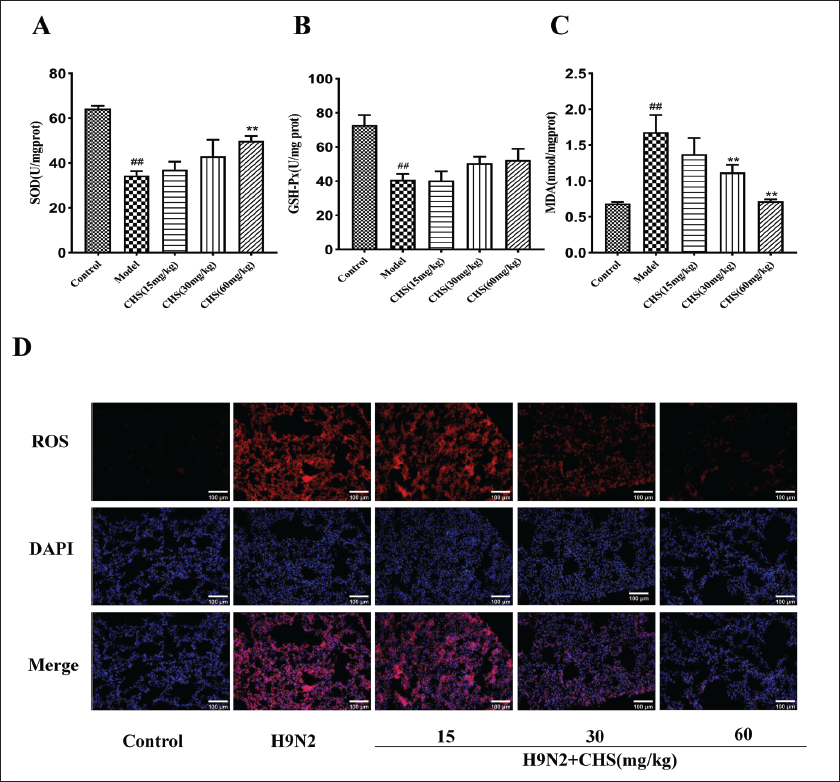
Effect of CHS on SOD, GSH-PX Activity, and MDA and ROS Levels in Lung Tissue
The impact of CHS (15, 30, 60 mg/kg) on SOD and GSH-PX activity, as well as MDA and ROS levels in lung tissues of H9N2 AIV-infected mice, was observed. Within the group of model mice, lung tissue SOD and GSH-Px activities notably decreased (p < 0.01), concomitant with a significant increase in MDA levels (p < 0.01). After CHS treatment, the SOD activity of mice in the CHS (60 mg/kg) group significantly increased (p < 0.01), and GSH-Px also increased but not significantly. MDA levels in the CHS (30 mg/kg) and CHS (60 mg/kg) groups exhibited a significant reduction (p < 0.01) (Figure 9A–C). Additionally, compared to the control group, red fluorescence was observed in the lung tissue of model mice. However, CHS treatment reduced the level of ROS fluorescence expression in the lung tissue of mice, indicating that CHS can alleviate H9N2 AIV-induced oxidative stress (Figure 9D).
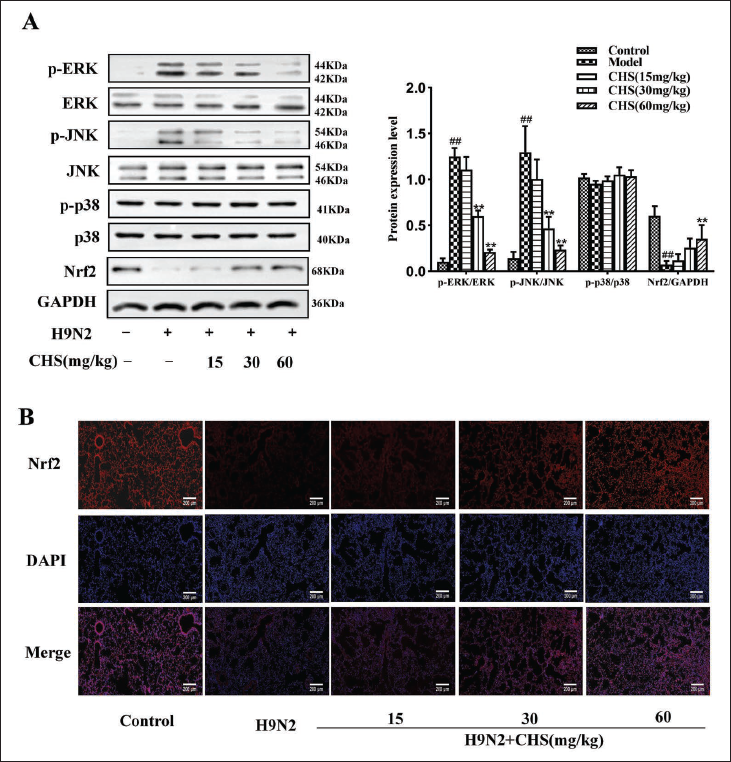
Impact of CHS on Nrf2 Signaling Pathway in H9N2 AIV-induced Lung Tissue
The impact of CHS on MAPK and Nrf2 signaling pathways in H9N2 AIV-induced lung tissue was investigated. Key protein expression was assessed via Western blot and IF. In contrast to the control group, a significant upregulation of p-JNK and p-ERK proteins was observed in the model group. CHS (60 mg/kg) effectively attenuated p-JNK and p-ERK protein expression (p < 0.01). Nrf2 protein expression was notably reduced in the model group but significantly enhanced in the CHS-treated group (60 mg/kg) (p < 0.01) (Figure 10A). Furthermore, IF staining indicated that Nrf2 expression was scarcely detectable in the model group. By contrast, Nrf2 expression was significantly elevated in the CHS-treated group (60 mg/kg) compared to the model group (Figure 10B). These findings imply that CHS may protect against the H9N2 AIV-induced lung injury caused by regulating the MAPK and Nrf2 signaling pathways.
Discussion
Natural Chinese medicine products have a promising future in the fight against influenza. CHS is the main active component of triterpene saponins, which was reported to possess antiviral activities against different viruses. For example, Nahar et al. (2022) reported that CHS can inhibit HSV-1 and -2 infections in vitro. This research discovered that CHS was antiviral effective against H9N2 AIV, indicating that CHS could serve as a novel anti-H9N2 AIV agent. The cycle of virus proliferation consists of six interrelated phases, including adsorption, penetration, shelling, biosynthesis, assembly, and release (Ampomah & Lim, 2020). The antiviral ability of CHS was investigated in three modes of action: pretreatment, co-treatment, and posttreatment. CHS showed the best antiviral effect in posttreatment by infecting A549 cells with H9N2 AIV. These findings indicate that CHS may possess a potent antiviral effect against H9N2 AIV during viral replication.
Interactions between the host and IAV can lead to varying degrees of oxidative stress due to disrupting the equilibrium between ROS and antioxidant factors (Vlahos et al., 2012). After CHS treatment, in both A549 cells and mouse lung tissue, there was an increase in the activities of SOD and GSH-Px, alongside a reduction in MDA and ROS levels. It is preliminarily concluded that CHS can improve the oxidative stress response caused by H9N2 AIV.
Following this, network analysis was employed to further explore the protective mechanisms of CHS against H9N2 AIV. According to KEGG pathway analysis, the anti-H9N2 AIV effects of CHS are associated with targeting Nrf2 to regulate the MAPK pathway and oxidative stress. The Nrf2 pathway, an essential antioxidant signaling cascade, mitigates ROS-induced stress (Ahmed et al., 2017). It was shown that IAV infection elevates ROS levels and promotes Nrf2 nuclear translocation in isolated human alveolar type II cells and alveolar macrophages, while Nrf2 overexpression reduces viral replication and oxidative stress (Kosmider et al., 2012).
In accordance with these conclusions, molecular docking results showed that CHS had a good affinity with Nrf2. CHS enhances Nrf2 expression and fosters its accumulation in the nucleus both in vitro and in vivo. Therefore, our inference suggests CHS attenuated oxidative stress induced by H9N2 AIV via activating the Nrf2 signaling pathway. Moreover, CHS inhibited the phosphorylation level of ERK and JNK. According to previous reports, inhibitors of the MAPK pathway reduce AIV replication (MacFarlane & Todd, 2014). The RNP complex of influenza A virus is exported from the nucleus by phosphorylated ERK during infection. Upregulation of p-ERK inhibits viral replication by retaining RNP in the nucleus (Botwina et al., 2020). JNK kinase regulates antiviral chemokines and cytokines expression by mediating the viral sensor signaling cascade (Meineke et al., 2019).
The Nrf2 plays an essential regulatory role in oxidative stress, helping to maintain cellular balance under oxidative conditions (Huang et al., 2020). In the current study, silencing Nrf2 siRNA cells, CHS did not block the production of ROS. This suggests that Nrf2 is important in controlling ROS in H9N2 AIV-induced A549 cells. The expression of MAPK pathways was further investigated. It was found that the Nrf2 knockdown of siRNA blocked CHS from activating MAPK signals induced by H9N2 AIV.
In summary, CHS can inhibit the replication of H9N2 AIV and ameliorate H9N2 AIV-induced oxidative stress by activating Nrf2-related signaling pathways, thereby protecting the organism.
Conclusion
CHS inhibits H9N2 AIV and alleviates virus-induced oxidative stress by targeting the Nrf2 signaling pathway. These groundbreaking discoveries offer a solid foundation for exploiting promising therapeutic approaches to combat H9N2 AIV infection.
Summary
CHS restrained H9N2 AIV and reduced H9N2 AIV-induced oxidative stress. Bioinformatics and molecular docking analysis predicted the targeting of Nrf2 signaling pathways. Experiments confirmed CHS can activate the Nrf2 signal and stimulate the production of antioxidants in infected cells. In H9N2 AIV-infected mice, CHS decreased lung damage, upregulated pulmonary expression of Nrf2, and alleviated oxidative stress. In summary, Nrf2 is necessary for the oxidative stress induced by H9N2 AIV. CHS improves the oxidative stress response induced by H9N2 AIV by enhancing the activity of Nrf2, thereby assisting in protecting the body against the damaging effects of oxidative stress and supporting the health and function of the body. CHS could be a promising antiviral candidate to control H9N2 AIV infection.
Footnotes
Abbreviations
CHS: Chikusetsusaponin IVa; AIV: Avian influenza virus; IC50: Half inhibitory concentration; TC50: Median toxic concentration; TI: Therapeutic index; SOD: Superoxide dismutase; GSH-Px: Glutathione peroxidase; MDA: Malondialdehyde; KEGG: Kyoto Encyclopedia of Genes and Genomes; GO: Gene Ontology; CMV: Cytomegalovirus; Nrf2: Nuclear factor erythroid2-related factor 2; MAPK: Mitogen activated protein kinase; PI3K: Phosphoinositide 3-kinase; Akt: Protein kinase B; ROS: Reactive oxygen species; HA: Hemagglutinin; NA: Neuraminidase; ALI: Acute lung injury; ARDS: Acute respiratory embarrassment forced syndrome; HSV: Herpes simplex virus; HO-1: Heme Oxygenase-1; A549: Human lung carcinoma A549 cells ; FBS: Fetal bovine serum; PDB: Protein Data Bank; ERK: Extracellular regulated protein kinases; p-ERK: Phospho-extracellular regulated protein kinases; JNK: c-Jun N-terminal kinase; p-JNK: Phospho-c-Jun N-terminal kinase; p38: p38 mitogen activated protein kinases; p-p38: Phospho-p38 mitogen activated protein kinases; CCK-8: Cell Counting Kit-8; BP: Biological process; MF: Molecular function; CC: Cellular component; IF: Immunofluorescence; PBS: Phosphate-buffered saline; BSA: Bovine serum albumin; DAPI: 4′,6-Diamidino-2-phenylindole; SDS-PAGE: SDS-polyacrylamide gel; ECL: Enhanced chemiluminescence; TCID50: 50% tissue culture infective dose; LD50: Median lethal dose; EDTA: Ethylenediaminetetraacetic acid; GAPDH: Glyceraldehyde-3-phosphate dehydrogenase; WB: Western Blot.
Acknowledgments
The authors are indebted to Key Laboratory of Traditional Chinese Medicine Resources, Chemistry of Hubei Province and Hubei Shizhen Laboratory for providing convenience in the experimental process.
Authors’ Contributions
DQ and WST designed the experiments and wrote the original manuscript. ZX and YPT performed the experiment and polished the original manuscript. LYW coordinated and managed the conduct of experiments. ZY gathered background information for the experiment. ZXQ collected and visualized the experimental data. All the authors read and approved the final version of the manuscript.
Availability of Data and Materials
All data generated or analyzed during this study are included in this published article and its supplementary information files.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Ethical Committee of Experimental Animals.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Youth Project of Hubei University of Chinese Medicine (No.2021ZZX020) and the National Natural Science Foundation of China (Grant No.30470194).
Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Statement of Human and Animal Rights
All of the experimental procedures involving animals were conducted in accordance with the Institutional Animal Care and Use Committee guidelines.
