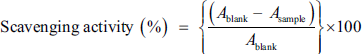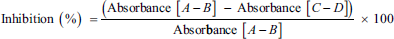Abstract
Background
Cynoglottis chetikiana subsp. chetikiana, an endemic Boraginaceae plant growing in Turkey, has not been previously studied in terms of pharmacognosy. However, related species, such as Anchusa, Symphytum, and Rindera, have been known for their pharmacological effects attributed to compounds such as flavonoids, terpenoids, and other phenol derivatives.
Objectives
This study aimed to reveal the phytochemical composition and potential biological activity of C. chetikiana.
Materials and Methods
Phytochemical profiling analysis was performed by using the liquid chromatography–high-resolution mass spectrometry (LC–HRMS) system. The cytotoxic effects of the extracts were evaluated on the MCF-7 mammalian breast cancer cell line by the MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) method. The in vitro α-glucosidase and tyrosinase enzyme inhibitory activities were assessed using literature methods. Additionally, antioxidant properties were investigated, leading to the bioactivity-guided isolation of the most active substance.
Results
LC–HRMS analysis revealed 26 major phenolic compounds and 1 pyrrolizidine alkaloid in C. chetikiana. Bioactivity-guided studies led to the isolation of a flavonoid, rutin. The n-butanol extract of the plant exhibited high antioxidant capacity and was rich in phenolic compounds. The methanol extract of C. chetikiana demonstrated low-moderate α-glucosidase inhibitory activity (IC50 = 608.62 ± 7.01 µg/mL). Moreover, the methanol extract showed significant anti-tyrosinase enzyme activity (195.84 ± 5.81 mg Kojic acid equivalent/g extract), and the petroleum ether extract displayed the highest cytotoxicity on MCF-7 cells (IC50 = 76.91 ± 3.12 µg/mL).
Conclusion
This is the first phytomedicinal study report for C. chetikiana, which gives promising insight for further pharmaceutical research.
Introduction
Boraginaceae, which is one of the largest angiosperm families, contains approximately 146 genera and almost 2,000 species distributed worldwide. In 1983, Vural and Tan defined a separate breed as Cynoglottis, which was previously known as a subgenus of the Anchusa (Boraginaceae) genus. It is a small genus with only two species, Cynoglottis barrelieri and Cynoglottis chetikiana, registered in the Flora of Turkey. C. chetikiana carries two endemic subspecies: C. chetikiana subsp. paphlagonica (Hausskn. Bornm.) Vural & Kit Tan and C. chetikiana subsp. chetikiana, locally known as “dağdarısı” (Selvi et al., 2004; Vural & Kit, 1988).
Most of the compounds that have been isolated and structurally identified from Boraginaceous species are related to phenolic compounds (such as phenolic acids, flavonoids, and naphthoquinones), sterols, saponins, pyrrolizidine alkaloids (PA), and fatty acids. Research on Boraginaceae plants has shown that they are rich sources of metabolites with a wide range of bioactivities, including anti-inflammatory, antimicrobial, antiseptic, diuretic, wound healing, and anticancer effects, which support their ethnopharmacological uses. However, some are also rich in PA, toxins produced by plants as a defense mechanism against insects and herbivores. Despite containing PAs known for their toxic effects, Boraginaceae plants have gained attention as promising drug leads owing to their vast biological activities and various secondary metabolites, necessitating careful evaluation of their safety (Chebaro et al., 2023; Ganos et al., 2020; Ramezani et al., 2020; Shilov et al., 2022; Trifan et al., 2021; Yuzbasioglu et al., 2020).
There are currently no available records on the phytochemical composition and biological activities of both sub-species of C. chetikiana. However, a single report indicated that the other species, Cynoglottis barrelieri, displayed robust antioxidant activity, along with the identification of phenolic compounds and PA (Varvouni et al., 2020). Furthermore, the closest species, Anchusa spp., has been widely used in traditional medicine for its antimicrobial, antioxidant, and wound-healing effects (Dresler et al., 2017; Kuruüzüm-Uz et al., 2013).
The medicinal potential of the endemic C. chetikiana subsp. chetikiana (CC) was investigated to assess its various biological activities. The α-glucosidase enzyme-inhibitory activity of the plant extract and sub-extracts was evaluated in diabetes drug discovery studies. Additionally, the tyrosinase enzyme-inhibitory activity was screened to address the conditions related to excessive melanogenesis. Furthermore, an MTT test was conducted on the MCF-7 human breast cancer cell line to explore the cytotoxic potential of CC against breast cancer. Phytochemical fingerprint analyses were performed using liquid chromatography–high-resolution mass spectrometry (LC–HRMS). Bioactivity-guided isolation studies based on radical scavenging activity and the antioxidant capacity of CC were also investigated. 2,2-Diphenyl-1-picrylhydrazyl (DPPH), SO–, and nitric oxide (NO)– radical scavenging activities, copper (II) reducing antioxidant capacity (CUPRAC), ferric reducing antioxidant power (FRAP), 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS)•+ antioxidant capacities, and total phenol quantities of CC were determined.
Materials and Methods
General Experimental Procedures
Thin-layer chromatography (TLC) was performed using a pre-coated silica gel aluminum plate with the fluorescent indicator F254 (Merck, Germany). Column chromatography was performed using silica gel 60, 70-230 mesh (Merck 7734, Germany), Sephadex LH20 (Fluka), and polyamide (Sigma–Aldrich, USA). 1D (1H and 13C) and 2D (COSY, HMBC, and HMQC) NMR spectra were recorded on a Bruker Avance DRX-500 using methanol-d4, calibration to 3.31 ppm (solvent signal). All solvents and commercially available reagents used in activity assays were purchased from Sigma–Aldrich, USA.
Plant Material
C. chetikiana subsp. chetikiana was collected on the road from Beyşehir, Konya, to Akseki, Antalya, 38 km before Akseki, Turkey, in May 2010 (37°11′53.6″ N 31°54′49.9″ E). The herbarium sample is stored in the Gazi University Science Faculty Herbarium (B. Bilgili 3804). Dried above-ground plant parts were used in this study.
Extraction and Isolation
Fresh plant material (~385 g) was air-dried in the shade. Approximately 345 g of the dried and powdered aerial parts were used to prepare the Cynoglottis chetikiana methanol extract (CC-MeOH) extract (80 g, 2.5 L MeOH × 5) at 40°C using a rotary evaporator. After evaporation, the CC-MeOH was dissolved in 250 mL of distilled water. It was fractionated with petroleum ether (PE) to remove lipids and pigments (300 mL × 5), and the remaining aqueous phase was extracted with n-butanol (300 mL × 5) at room temperature to yield 12 g of CC-nBuOH extract. Initially, the lyophilized nBuOH-soluble fraction (8 g) of the MeOH extract was chromatographed on a polyamide column eluted with H2O:MeOH (0 ↓ 100%, MeOH) and five fractions were obtained (Fr. A–E). Fr. E (540 mg) was further applied on a silica gel column (SK) using a CH2Cl2:MeOH:H2O (80:20:2 ↓ 70:30:3, each fraction was 50 mL) gradient elution. The obtained fraction (28-33, 80 mg) was further processed using a Sephadex-LH 20 column (SPH) with MeOH (100%) by isocratic elution. Fraction (21-31, 50 mg) of SPH was run on SK with the gradient elution of CH2Cl2:MeOH:H2O from (80:20:2) to (75:25:2.5) and SK (22-31) was obtained as a pure compound (CC-1, 23 mg).
LC–HRMS Analysis
The metabolomic profiles of the methanol extract were obtained using liquid chromatography quadrupole time-of-flight mass spectrometry (LC-qTOF-MS). Metabolites were separated using a C18 column (Poroshell HPH-C18 column, 100 mm, 2.1 m, 2.7 µm) and examined using an Agilent 6530 LC-qTOF-MS system. Gradient elution, solvent A (acetonitrile—0.1% formic acid) and solvent B (water—0.1% formic acid) were used in the mobile phase as follows: 0–1 min, 90% B; 1–7 min, 90–10% B; 7–10 min, 10% B; 10–11 min, 90% B; and 11–15 min, 90% B. The volume of flow was set to 0.3 mL/min. The injection volume was 10 µL. The ESI source was operated in positive and negative modes with the capillary voltage and temperature set at 3,500 V and 325 °C, respectively. The auto MS–MS data of the metabolites were collected over a 200-count threshold between 100 and 1,700 m/z. Plant metabolites were fragmented using a collision energy of 20 eV in the MS/MS process. Raw MS data were processed for peak detection, deconvolution, alignment, and identification using MS-Dial (Tsugawa et al., 2015). The minimum peak height for peak detection was set to an amplitude of 2000. The MS1 and MS2 limits were set as 0.01 and 0.05 Da. MS/MS data were used to identify the metabolites precisely. The annotation of the metabolites was achieved by comparing their mass spectra, fragmentation patterns, and retention times to those of standards and libraries. Only structures with a score of 70 or higher were considered to be correctly identified.
Biological Activity Assay
Antioxidant Capacity Assays
Total phenol determination
Samples were prepared at a 1,000 µg/mL concentration, and standard gallic acid was prepared as dilutions at 50–400 µg/mL concentrations. 10 µL solutions were transferred to microplate wells, followed by the addition of 150 µL of Folin–Ciocalteu reagent (diluted at a ratio of 1:4 with distilled water) and 50 µL of saturated Na2CO3 solution. Absorbance was measured at 725 nm after a 2 h incubation at room temperature (Zhang et al., 2008).
SO Radical Scavenging Activity
Samples and standard ascorbic acid were prepared in DMSO at concentrations ranging from 5 to 200 µg/mL. 10 µL of nitroblue tetrazolium (NBT) solution was added to each well of a 96-well plate, followed by the addition of 30 µL of the sample and standard solutions. Then, 100 µL of alkaline DMSO (0.9 mL DMSO + 0.1 mL of 5 mM NaOH) was added to each well, and absorbances were measured at 560 nm (Govindarajan et al., 2004).
DPPH Radical Scavenging Activity
Samples and standard ascorbic acid were diluted to concentrations of 5–100 µg/mL. These solutions were added to the wells of a 96-well plate, followed by the addition of 50 µL of 1 mM DPPH solution prepared in methanol. After incubation for 30 minutes with protection from light, absorbance was measured at 517 nm (Govindarajan et al., 2004).
NO Radical Scavenging Activity
Quercetin was used as a positive control. Sample and control solutions were prepared in distilled water and diluted into 25–200 µg/mL concentrations. 60 µL of these solutions were added to each well of a 96-well plate. 60 µL of 10 mM sodium nitroprusside (dissolved in PBS) was added, and after 150 minutes of incubation, 120 µL of Griess reagent (sulfanilamide 1%, naphtylethylenediamine dihydrochloride 0.1%, and phosphoric acid 2.5% distilled water) was added. The absorbance was measured at 577 nm (Govindarajan et al., 2004).
All free radical scavenging effects were calculated using the following equation:
CUPRAC
Samples were prepared at 50–400 µg/mL concentrations. 50 µL CuCl2, 50 µL neocuproine (2,9-dimethyl-1,10-phenanthroline), and 50 µL ammonium acetate (pH: 7) mixture were added onto wells that contain 25 µL sample and standard solutions. Then, 25 µL distilled water was added. After 30 minutes of incubation in the dark, absorbance was measured at 450 nm. The result was calculated with an increase in optical density and expressed as gallic acid equivalent/g extract (Apak et al., 2006).
FRAP
Samples were prepared at 1,000 µg/mL. Dilutions of standard Trolox were prepared. 15 µL of the sample/Trolox solutions were transferred to the wells in 96-well plates. 285 µL FRAP reagent (2.5 mL TPTZ dissolved in 10 mM 40 mM HCl), 25 mL acetate buffer, and 20 mL of 2.5 mL FeCl3·H2O were added to them. After incubating at 37°C for 30 minutes, the absorbance was measured at 593 nm. Based on the standard curve drawn for Trolox, the antioxidant effect value of the samples was calculated as Trolox equivalent per gram of extract (Özgen et al., 2006).
ABTS•+ Antioxidant Capacity Assay
10 µL samples and Trolox solutions were transferred into 96-well plates. They were treated with 240 µL ABTS •+ solution (7 mM ammonium ABTS salt and 2.45 mM potassium persulfate dissolved in water; after overnight incubation, this solution was then diluted with ethanol or pH 7.4 buffer until its OD was nearly 0.7, measured in a spectrophotometer at 734 nm), and absorbance was measured after 6 min incubation at room temperature. The total radical scavenging capacity was calculated in relation to Trolox’s absorbance reduction. The results were expressed in terms of Trolox equivalent antioxidant capacity per gram sample (TEAC/mg) (Büyüktuncel, 2013).
Enzyme Inhibitory Activities
Tyrosinase Enzyme Inhibition Assay
Tyrosinase inhibition was determined by the method described by Masuda et al. (2005). Kojic acid was used as a positive control. Stock solutions of all samples were prepared and then diluted with phosphate buffer (pH 6.8) to different concentrations. 40 µL of 100 U/mL tyrosinase enzyme and 120 µL of 100 mM phosphate buffer (A), and 160 µL of 100 mM phosphate buffer (B) were used as blank solutions. 40 µL of sample solution, 40 µL of 100 U/mL tyrosinase enzyme, and 80 µL of 100 mM phosphate buffer were added to the 96-well plate and incubated at room temperature for 10 minutes (C). 40 µL of sample solution and 40 µL of 100 U/mL tyrosinase enzyme were used (D). The reaction was initiated by the addition of 40 µL of 3 mM
α-Glucosidase Inhibition Assay
α-Glucosidase inhibitory activity was determined using a modified version of the procedure previously reported by Bachhawat et al. (2011). Each well in 96-well plates contained 10 µL enzyme solution (1 U/mL), 50 µL potassium phosphate buffer (0.1 M, pH 6.9), and 20 µL of samples in DMSO. These were pre-incubated at 37°C for 5 minutes. Then, 20 µL of 3 mM PNGP prepared in the phosphate buffer was added to initiate the reaction. The mixture was further incubated at 37°C for 30 minutes. The reaction was stopped by adding 50 µL of 0.1 M sodium carbonate solution. The absorbance of the mixture was measured at 405 nm by a spectrophotometer. The inhibition rate was calculated by comparing the increase in absorbance to that of the control. Acarbose was used as a positive control.
Cytotoxic Activity on MCF-7 Cells
Cytotoxic effects of C. chetikiana extracts were investigated against the MCF-7 human breast cancer cell line by using the MTT [3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide, Sigma–Aldrich] method. The cultured cells were trypsinized and seeded into 96 multi-well plates at a concentration of 1 × 105 cells/mL in DMEM (Biochrom, Berlin, Germany) and incubated for 24 h in a humidified 5% CO2 incubator at 37°C. Dilutions of test samples and standard doxorubicin, dissolved in a medium containing DMSO (not to exceed 0.1%), were added to cells and incubated for 48 h. After incubation, MTT solution in PBS (10 mL of 5 mg/mL) was added to each well and incubated for another 4 h until purple formazan crystals were visible under a microscope. Then, 100 µL DMSO was added to each well to dissolve the crystals. Absorbance was measured at 577/655 nm using a microplate reader, and the percentage of cell viability and IC50 (50 percent inhibition concentration) values were calculated (Mosmann, 1983).
Statistical Analysis
The results of the antioxidant capacity and radical scavenging activities, in vitro enzyme inhibitory activities, and cytotoxicity were expressed as the mean ± standard deviation (SD), (n = 3). Statistical differences between two groups were determined using Student’s t-test, whereas for three or more groups, comparisons were conducted using one-way analysis of variance (ANOVA) followed by appropriate multiple comparison tests (Dunnett’s or Tukey’s), all performed using GraphPad Prism v.9.0, with significance evaluated at p < 0.05.
Results
Structure Elucidation of CC-1
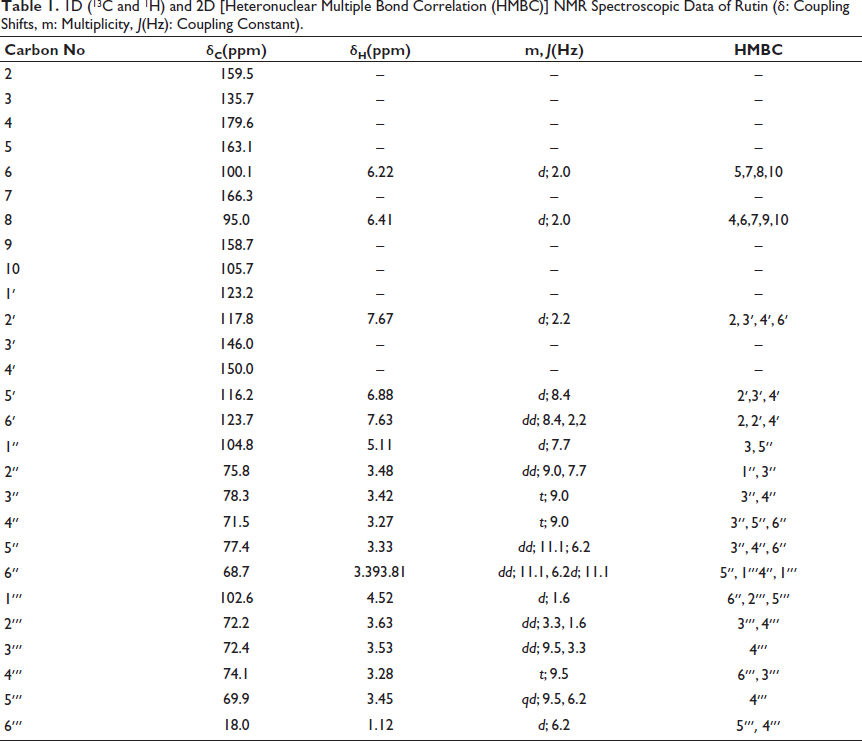
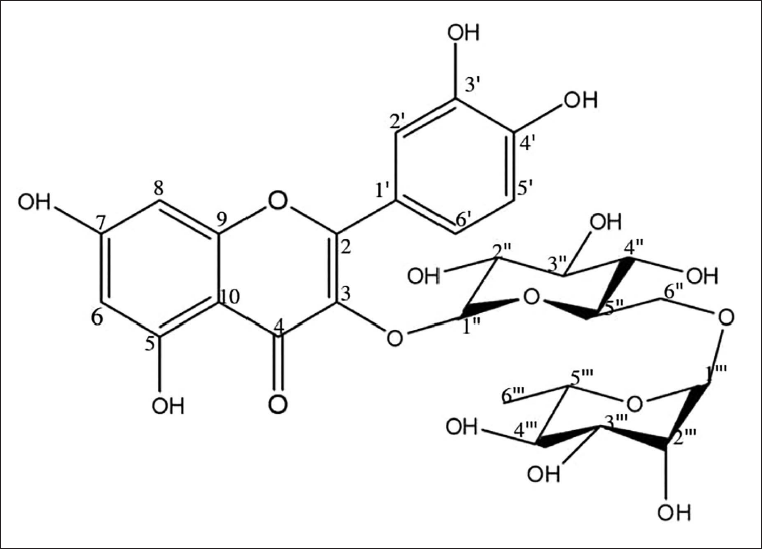
The yellow-colored amorphous CC-1 exhibited yellow fluorescence in daylight, a purple spot at UV254 nm, and yellow–brown fluorescence at UV366 nm on a thin layer chromatogram. The spot turned dark yellow in daylight when the vanillin/H2SO4 reagent was sprayed and heated at 105°C, suggesting that this compound may be a flavonoid with a sugar moiety. The closed formula was determined as C27H30O16 by using the m/z 611 [M+H]+ ion peak in the mass and NMR spectra (Table 1). Based on the 1D- and 2D-1H and 13C NMR spectral data compared with the literature, CC-1 was identified as rutin (Figure 1) (Matsuura et al., 2002).
1D (13C and 1H) and 2D [Heteronuclear Multiple Bond Correlation (HMBC)] NMR Spectroscopic Data of Rutin (δ: Coupling Shifts, m: Multiplicity, J(Hz): Coupling Constant).
The Molecular Structure of Rutin.
LC–HRMS Analysis
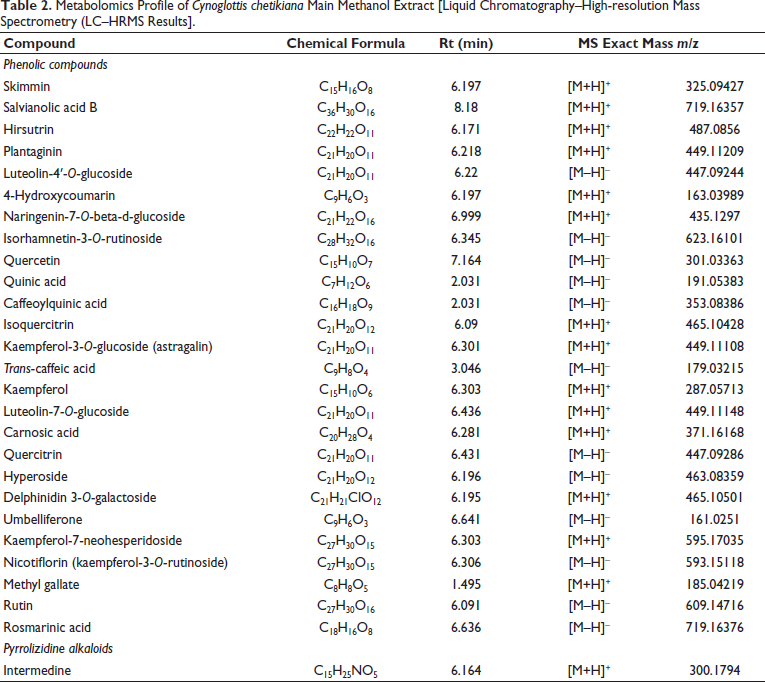
In this study, 27 compounds were identified by LC-qTOF-MS analysis (Table 2). Fifteen of them are flavonoids (hirsutrin, plantaginin, luteolin-4′-O-glucoside, naringenin-7-O-glucoside, isorhamnetin-3-O-rutinoside, quercetin, isoquercitrin, kaempferol-3-O-glucoside, kaempferol-7-neohesperidoside, nicotiflorin, rutin, quercitrin, hyperoside, kaempferol, luteolin-7-O-glucoside) and seven of them are phenolic acids (salvianolic acid B, quinic acid, caffeoylquinic acid, trans-caffeic acid, methyl gallate, rosmarinic acid, and carnosic acid), which are the majority. Notably, one anthocyanoside (delphinidin 3-galactoside), three coumarins (skimmin, 4-hydroxycoumarin, umbelliferone), and one PA (intermedine) were detected in the aerial parts of the CC.
Metabolomics Profile of Cynoglottis chetikiana Main Methanol Extract [Liquid Chromatography–High-resolution Mass Spectrometry (LC–HRMS Results].
Biological Assay Results
Antioxidant Capacity Assays
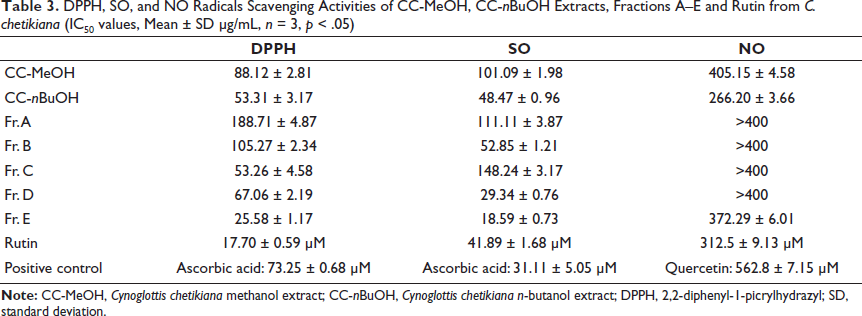
As a preliminary study, the antioxidant capacities of CC-MeOH and CC-nBuOH were determined, and the IC50 values were given as mean ± SD (Tables 3 and 4). The CC-nBuOH extract showed higher antioxidant activity and had more phenolic substances compared with the MeOH extract (p < 0.05). Since the CC-nBuOH extract has moderate antioxidant capacity, phytochemical studies have focused on the CC-nBuOH extract. Then, the antioxidant capacity of Fr. A–E was investigated using different tests. In most antioxidant capacity assays, Fr. E was the most active fraction (Tables 3 and 4, p < 0.0001). Several successive chromatographic studies were evaluated on Fr. E, resulting in the isolation of the active compound, CC-1. The antioxidant test results for extracts, fractions, and the pure compound (CC-1) are given in tables and figures.
DPPH, SO, and NO Radicals Scavenging Activities of CC-MeOH, CC-nBuOH Extracts, Fractions A–E and Rutin from C. chetikiana (IC50 values, Mean ± SD µg/mL, n = 3, p < .05)
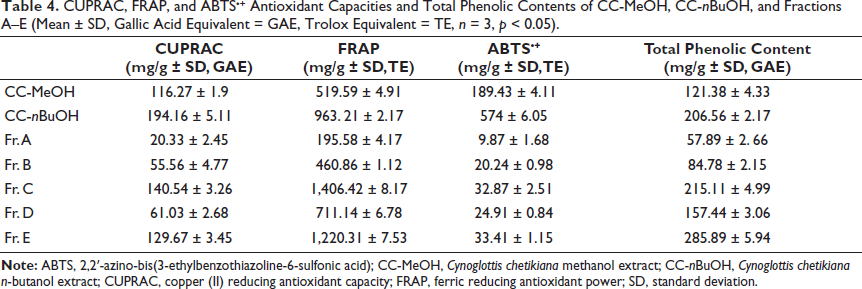
CUPRAC, FRAP, and ABTS•+ Antioxidant Capacities and Total Phenolic Contents of CC-MeOH, CC-nBuOH, and Fractions A–E (Mean ± SD, Gallic Acid Equivalent = GAE, Trolox Equivalent = TE, n = 3, p < 0.05).
Total Phenol Determination
Total phenol amounts were detected as 121.38 ± 4.33 and 206.56 ± 2.17 mg/g gallic acid equivalents (GAE) for CC-MeOH and CC-nBuOH extract, respectively. The results of the Fr. A–E yielded from CC-nBuOH were found to be 57.89 ± 2. 66 mg/g, 84.78 ± 2.15 mg/g, 215.11 ± 4.99 mg/g, 157.44 ± 3.06 mg/g, and 285.89 ± 5.94 mg/g GAE, respectively (Table 4). According to the results, Fr. E had the richest phenolic content (p < 0.0001).
DPPH Radical Scavenging Activity
IC50 values were expressed as mean ± SD and given in Table 3. According to the results, fraction E was the most active fraction among others (IC50 = 25.58 µg/mL ± 1.17) and the pure compound, rutin (CC-1), has more DPPH radical scavenging activity compared with the ascorbic acid (IC50 values are 17.70 ± 0.59 µM and 73.25 ± 0.68 µM, respectively, p < 0.0001).
SO Radical Scavenging Activity
IC50 values were calculated and are shown in Table 3 (all values are expressed as mean ± SD). According to the results, fraction E was found to be the most active fraction among others (IC50 = 18.59 ± 0.73 µg/mL), and the activity of the positive control (ascorbic acid) and rutin (CC-1) was comparable (IC50 values are 31.11 ± 5.05 µM and 41.89 ± 1.68 µM, respectively, p < 0.0001).
NO Radical Scavenging Activity
IC50 values were expressed as mean ± SD and are given in Table 3. The positive control (quercetin), fraction E, and CC-1 were detected as 170.11 ± 2.16 µg/mL (562.8 ± 7.15 µM), 372.29 ± 6.01 µg/mL, and 190.51 ± 5.57 (312.5 ± 9.13 µM) µg/mL, respectively (p < 0.0001). IC50 values of Fr. A–D could not be calculated (>400 µg/mL).
CUPRAC
The results are presented as mean ± SD, mg/g gallic acid equivalent (Table 4). Among these fractions, Fr. C and Fr. E showed the highest CUPRAC, with values of 140.54 ± 3.26 and 129.67 ± 3.45 mg/g GAE, respectively (p < 0.0001).
FRAP
The results are presented as the mean ± SD, mg/g Trolox equivalent (Table 4). Fr. C and Fr. E were found to have the most iron (III) reducing antioxidant capacity compared with the other fractions (1,406.42 ± 8.17 and 1,220.31 ± 7.53 mg/g TE, respectively, p < 0.0001)
ABTS•+ Antioxidant Capacity Assay
The results are given as given as mean ± SD, mg/g Trolox equivalent (Table 4). It was observed that Fr. C and Fr. E displayed the highest ABTS+ antioxidant capacity among the other fractions (32.87 ± 2.51 and 33.41 ± 1.15 mg/g TE, respectively, p < 0.0001).
Enzyme Inhibitory Activities
Tyrosinase Enzyme Inhibition Assay
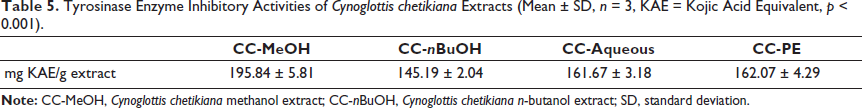
The calculated results indicated that the main CC-MeOH and other sub-extracts (CC-nBuOH, CC-aqueous, CC-PE) had similar and significant tyrosinase enzyme inhibitory activities. The results are given as kojic acid equivalent/g extract, with values of 195.84 ± 5.81, 145.19 ± 2.04, 161.67 ± 3.18, and 162.07 ± 4.29 respectively (p < 0.001, Table 5).
Tyrosinase Enzyme Inhibitory Activities of Cynoglottis chetikiana Extracts (Mean ± SD, n = 3, KAE = Kojic Acid Equivalent, p < 0.001).
α-Glucosidase Inhibition Assay
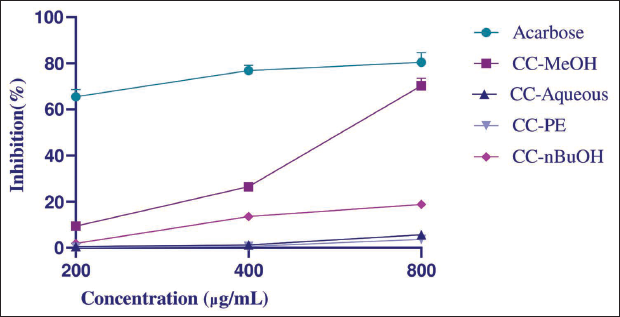
As for the α-glucosidase inhibitory activity, only the MeOH extract showed mild activity compared with the standard acarbose (IC50 values: 608.62 ± 7.01, 111.86 ± 2.16 µg/mL, respectively, p < 0.0001) (Figure 2).
α-Glucosidase Inhibitory Activities of Cynoglottis chetikiana Main Methanol (CC-MeOH), Petroleum Ether (CC-PE), n-Butanol (CC-nBuOH), and Remaining Aqueous (CC-Aqueous) Sub-extracts on 200, 400, and 800 µg/mL (Acarbose as Positive Control, Mean ± Standard Deviation (SD), n = 3, p < .001).
Cytotoxic Activity on MCF-7 Cells
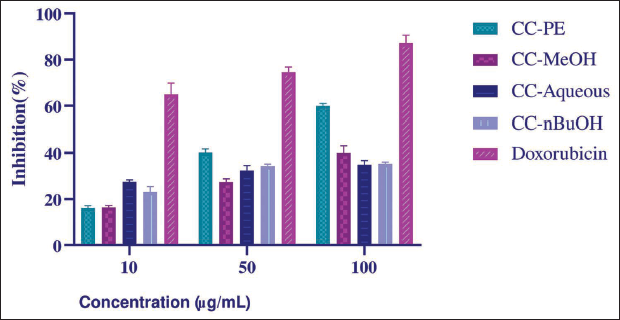
According to the activity results, CC-PE showed the highest activity (IC50 value = 76.91 ± 3.12 µg/mL) compared to other extracts (p < 0.0001, Figure 3). The IC50 value of the positive control, doxorubicin, was calculated to be 0.0512 ± 0.001 µg/mL.
Cytotoxic Effects of Cynoglottis chetikiana Extracts on MCF-7 Cell Line [Mean ± Standard Deviation (SD), n = 3].
Discussion
All phytochemical study and bioactivity test results are the first report for the C. chetikiana subsp. chetikiana. The antioxidant activity-guided isolation studies were applied to the CC. According to the antioxidant capacity test results, the CC-nBuOH extract showed higher antioxidant activity and a greater content of phenolic substances than the other CC extracts. Previous studies have consistently shown a positive correlation between the phenolic content and antioxidant activity (Stanković, 2020, pp. 275–307). The antioxidant capacities of the fractions obtained from CC-nBuOH (Fr. A–E) revealed that fraction E was the most active, resulting in the isolation of active compound, CC-1, from this fraction. Spectral results indicated that CC-1 is a flavonoid, rutin, which is mostly found in Boraginaceae (Masuda et al., 2005; Stanoeva et al., 2023). Rutin was isolated and identified in C. barrelieri, the Cynoglottis species, thus supporting our data. Moreover, our study assessed the radical scavenging activities of rutin. The results demonstrated the potent antioxidant properties of rutin compared to the positive controls, which is supported by the literature documenting the antioxidant potential of this flavonoid (Yang et al., 2008).
Metabolomics studies using LC-qTOF-MS revealed 27 compounds (Table 2). Salvianolic acid B, trans-caffeic acid, rosmarinic acid, isoquercitrin, nicotiflorin, rutin, astragalin, intermedine and hyperoside have been identified in C. barrelieri (Varvouni et al., 2020). Many of these metabolites have been previously reported in the related genus Anchusa (Al-Snafi, 2014). Notably, our study marks the first report of these compounds in C. chetikiana subsp. chetikiana. Beyond these, the remaining 18 compounds detected in this study are reported here for the first time not only in C. chetikiana but also in any Cynoglottis species.
Tyrosinase enzyme inhibition is significant in drug development research for conditions associated with pigmentation disorders (such as melasma, lentigo, and post-inflammatory hyperpigmentation). The test results for all extracts of CC indicate promising tyrosinase inhibitory effects (145.19 ± 2.04 – 162.07 ± 4.29 mg/g KAE). The closest species, Anchusa, demonstrated only 43.89–48.16 mg/g KAE (Sarikurkcu et al., 2015; Yang et al., 2008). Previous studies have indicated that some of the secondary metabolites of C. chetikiana, such as quercetin, kaempferol, and umbelliferone, inhibit tyrosinase by directly binding to the enzyme’s active site, thereby impeding substrate access (Kim & Uyama, 2005). Additionally, quercetin chelates copper ions that are essential for the catalytic activity of tyrosinase, further inhibiting the enzyme (Zolghadri et al., 2019). These compounds may have contributed to the observed activity in C. chetikiana.
α-Glucosidase inhibitory activity is a preliminary test for antidiabetic drug discovery research. Only the MeOH extract showed moderate activity compared to the standard acarbose, with IC50 values of 608.62 ± 7.01 and 111.86 ± 2.16 µg/mL, respectively. Quercetin and isoquercitrin are competitive inhibitors that fit into the α-glucosidase enzyme’s active site because of their structural similarity to the natural substrate (Dej-Adisai et al., 2021; Jhong et al., 2015). Studies have shown that these compounds, along with rutin, have potent inhibitory effects on α-glucosidase (Li et al., 2009). The activity observed in the methanol extract could be due to the presence of these compounds, suggesting that their inhibitory mechanisms may contribute to the observed effects.
Breast cancer is the most prevalent form of cancer among women. Evaluating the cytotoxic potential of plant extracts against different types of breast cancer cells can yield valuable insights (Chen et al., 2020). Our research focused on the cytotoxic activities of the CC extracts on the MCF-7 (estrogen receptor positive) breast cancer cell line. Only CC-PE showed notable activity (IC50 value = 76.91 ± 3.12 µg/mL). According to the National Cancer Institute (NCI), for anticancer drug research, plant extracts should exhibit cytotoxic activities below 30 µg/mL to be considered for further anticancer research (Boik, 2001). Although our results are below this threshold, CC-PE could be considered suitable for further isolation and mode-of-action studies. Among the compounds detected in this plant and that may contribute to its cytotoxic effects, quercetin and kaempferol have been shown to induce apoptosis in MCF-7 cells by activating caspases and upregulating pro-apoptotic proteins (Kang et al., 2009; Molani Gol & Kheirouri, 2022). Additionally, salvianolic acid B has been shown to induce cell cycle arrest in various cancer cells (Katary et al., 2019; Yang et al., 2011). The potential antitumor effects of PA have also been reported. Kurimoto et al. (2022) demonstrated the conversion of these compounds into active forms using a gold-based artificial metalloenzyme, bypassing liver metabolism and showing significant cytotoxicity in cancer cell lines without harming normal cells. This suggests the potential of using targeted therapies to alleviate genotoxicity and utilize PA in cancer treatment.
Conclusion
This is the first study to reveal the pharmacological and phytochemical potential of C. chetikiana. Our research identified significant antioxidant, α-glucosidase, and tyrosinase enzyme inhibitory activities, as well as cytotoxic activities of this plant. Notably, bioactivity-guided isolation led to the identification of rutin as the most potent antioxidant compound. LC-qTOF-MS analysis revealed the presence of 27 compounds, including phenolic acid derivatives, flavonoids, coumarins, and PA, which further enriched the chemical profile of the plant. These findings collectively underscore the pharmacological potential of C. chetikiana, positioning it as a promising candidate for further in vivo bioavailability and mode of action studies in drug development.
Abbreviations
ABTS: 2,2′-Azino-bis(3-ethylbenzothiazoline-6-sulfonic acid); CC-1: Compound 1 isolated from Cynoglottis chetikiana; CC-MeOH: Cynoglottis chetikiana methanol extract; CC-nBuOH: Cynoglottis chetikiana n-butanol extract; CUPRAC: Copper (II) reducing antioxidant capacity; d: Doublet; dd: Doublet of doublets; DMSO: Dimethyl sulfoxide; DPPH: 2,2-Diphenyl-1-picrylhydrazyl; FRAP: Ferric reducing antioxidant power; GAE: Gallic acid equivalent; HMBC: Heteronuclear multiple bond correlation; J: Coupling constant; LC–HRMS: Liquid chromatography–high-resolution mass spectrometry; LC-qTOF-MS: Liquid chromatography quadrupole time-of-flight mass spectrometry; m: Multiplet; MS/MS: Tandem mass spectrometry; NBT: Nitroblue tetrazolium; NMR: Nuclear magnetic resonance; NO: Nitric oxide; qd: Quartet of doublets; Rt: Retention time; SK: Silica gel column; SPH: Sephadex LH-20 column; t: Triplet; TE: Trolox equivalent; [M+H]+: Protonated molecular ion; [M–H]–: Deprotonated molecular ion.
Footnotes
Acknowledgments
The authors would like to thank TUBITAK for their support through the 2209/A- University Students Research Projects Support Program.
Authors’ Contribution
Design of the study: MYB, AKU, HD; experimental data collection: MYB, TR, SG; design and supervision of the phytochemical experiments: MYB, EN, TR, AKU, SG; supervision of the biological assays: MYB, SG; interpretation of the spectrometric data: MYB, AKU, AS; drafting the manuscript: all authors.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable (no humans/patients were involved in the research).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Türkiye Bilimsel ve Teknik Araştırma Kurumu (TÜBİTAK) 2209/A - University Students Research Projects Support Program.