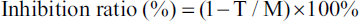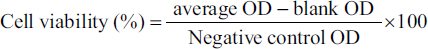Abstract
Background
Zingiber zerumbet (L.) Smith (lempoyang) has a traditional application in the treatment of indigestion, worm infestation, loss of appetite, and postpartum conditions.
Objectives
Extracts of Z. zerumbet (methanol, hexane, and ethyl acetate) were utilized to investigate the mutagenic, anti-mutagenic, and cytotoxic properties.
Materials and Methods
Initially, a mutagenicity test (Ames test) was conducted, followed by an anti-mutagenicity test, to assess the potential of Z. zerumbet extracts in inhibiting mutagenicity induced by sodium azide and 9-aminoacridine. Furthermore, the cytotoxic ability of the extracts was evaluated using the methyl thiazolyl tetrazolium (MTT) assay.
Results
The mutagenicity assessment revealed that the methanol extract of Z. zerumbet exhibited a twofold increase in the number of revertants in Salmonella typhimurium strain TA 1537. Similar results were observed for the hexane extract, except at a concentration of 6.25 mg/mL, where no significant increase in revertants was observed. On the other hand, the ethyl acetate extract demonstrated a twofold increase in revertants in S. typhimurium strain TA 1535. Notably, the ethyl acetate extract displayed remarkable anti-mutagenic activity against 9-aminoacridine, while the hexane extract exhibited strong anti-mutagenic activity against sodium azide. Regarding cytotoxicity assessment using the MTT assay, the methanol extract of Z. zerumbet exhibited the highest cytotoxicity with an IC50 value estimated at 388.50 ± 135.75 µg/mL. The hexane and ethyl acetate extracts showed IC50 values of 514.17 ± 135.75 and 589.67 ± 241.67 µg/mL, respectively.
Conclusion
The extracts displayed both mutagenic and cytotoxic activities. However, they also exhibited promising anti-mutagenic potential, which could be harnessed for cancer prevention purposes.
Introduction
Mutagens exist not only in plastics and natural environmental chemicals but also arise from metabolic processes within the human body, resulting in the production of significant amounts of mutagens such as nitrosamines, bisulfite, and hydroxylamine. The presence of endogenous mutagens associated with cancer is nearly unavoidable (Singer & Grunberger, 2012). Mutagens can induce changes through mutagenesis cycles caused by synthetic agents or the intrinsic properties of nucleic acids. At birth, infants already possess 2% of these changes. For instance, a single amino acid substitution in hemoglobin can lead to sickle cell anemia, while the absence of an enzyme or protein receptor can result in inborn errors of metabolism (Singer & Grunberger, 2012).
An ideal strategy to mitigate the harmful effects of mutagens is to harness the anti-mutagenic properties present in plants and reduce consumption from other sources that may possess defensive effects against mutagens. Natural anti-mutagens found in edible and medicinal plants hold great importance, as they could play a preventive role against cancer in humans (Bhattachar, 2011). Utilizing common anti-mutagens as a chemopreventive approach aims to hinder, suppress, reverse, or inhibit the progression of malignancy (Shureiqi et al., 2000). Traditional and complementary medicine (T&CM) has been making significant contributions to human healthcare for decades. The application of T&CM continues to expand worldwide, including in Malaysia, due to its rich tropical biodiversity. A study by Siti et al. (2009) revealed extensive usage of T&CM by the Malaysian population, particularly herbal-based therapies for health maintenance and treatment.
Extracts and juices derived from medicinal plants and fruits, such as Mitragyna speciosa (ketum) (Ghazali et al., 2011), Carica papaya (betik) (Rathnasamy et al., 2013), Myristica fragrans (buah pala) (Akinboro et al., 2012), and Psidium guajava (jambu batu) (Alsarhan et al., 2014), have demonstrated notable potential as anti-mutagens. Z. zerumbet (L.) Smith, a member of the Zingiberaceae family, is an important medicinal ginger widely used in various Asian countries. Species within this family exhibit potent pharmacological properties, including antioxidant, antibacterial, antipyretic, anti-inflammatory, immunomodulatory, and anticancer effects (Singh et al., 2019). Z. zerumbet possesses phytochemical constituents such as zerumbone (ZER), flavonoids, and other phenolic compounds, which are particularly associated with its mutagenic, anti-mutagenic, and cytotoxic potential. Various classes of bioactive compounds found in natural products exhibit robust chemopreventive effects, such as anti-inflammatory and antioxidant properties (George et al., 2021). Hence, the present study aims to investigate the mutagenic, anti-mutagenic, and cytotoxic abilities of Z. zerumbet extracts.
Materials and Methods
Plant Extracts
Fresh rhizomes of Z. zerumbet (L.) Smith, identified with Voucher No. UKMB-29952, were sourced from an herb farm located in Temerloh, Pahang. These rhizomes were air-dried for a period of 3 days at room temperature. Subsequently, the dried rhizomes were powdered and subjected to maceration using methanol, hexane, and ethyl acetate, followed by sieving. The resulting filtrate was concentrated using a rotary evaporator under reduced pressure. The crude extracts obtained were then dissolved in 1 mL of 99% dimethyl sulfoxide (DMSO), resulting in five different concentrations of 100, 50, 25, 12.5, and 6.25 mg/mL (Florinsiah et al., 2014; Ghazali et al., 2011).
Bacterial Strains
Mutant strains of Salmonella typhimurium, namely TA 1535 and TA 1537 (Hayes et al., 2022; Muhammad et al., 2021), were employed in the mutagenicity test (Ames test) to assess mutagenicity and anti-mutagenicity. The genotypes of these test strains were screened for histidine properties, rfa mutation, uvrB mutation, and the presence of plasmid pKM101. S. typhimurium TA 1535 carries the hisC3706 base pair substitution mutation, while S. typhimurium TA 1537 is a frameshift strain with the hisG46 mutation.
Cell Line
Chinese hamster lung fibroblast cells (V79-4) (ATCC) (Guichard et al., 2016; Ooi et al., 2021) were employed for the cytotoxicity test using the methyl thiazolyl tetrazolium (MTT) assay. Cultivation of this cell line was carried out using Dulbecco’s Modified Eagle’s Medium (DMEM) supplemented with 10% fetal bovine serum (FBS).
Mutagenicity Assay
The mutagenic potential of Z. zerumbet extracts was assessed using the method described by Maron and Ames (1983) on S. typhimurium strains TA1535 and TA1537. A total of 500 µL of sodium phosphate buffer (PBS) without metabolic initiation, along with 100 µL of overnight cultured S. typhimurium TA1535 or TA1537 and 100 µL of the extract, were prepared and preincubated at 37°C for 20 min. Subsequently, 2 mL of melted top agar enriched with a histidine/biotin arrangement (0.5 mM) at 45°C was added to the mixture. The resulting mixture was gently mixed and poured onto glucose agar plates, which were then inverted and incubated at 37°C for 48 h. Each assay to determine the mutagenicity of the extracts was conducted three times. After 48 h of incubation, colonies were counted.
Anti-mutagenicity Assay
The anti-mutagenic properties of Z. zerumbet extracts were investigated using a modified version of the Ames test (Cui-cai et al., 1992), in which mutagens (sodium azide [NaN3] and 9-aminoacridine [9-AA]) were added prior to preincubation. In this assay, 500 µL of metabolic initiation-free PBS and 100 µL of the extracts at varying concentrations were combined with 100µL of mutagen. Subsequently, 100 µL of preincubated S. typhimurium was added to the mixture, followed by the gentle addition of 2 mL of melted top agar containing a histidine/biotin arrangement (0.5 mM) at 45°C. The resulting mixture was carefully mixed and poured onto glucose agar plates, which were then inverted and incubated at 37°C for 48 h. To evaluate the mutagenic potential of the extracts, each assay was conducted in triplicate. After 48 h, colonies were counted, and the inhibition ratio was calculated using the following equation:
Let T represent the number of revertants per plate when both the mutagen and plant extracts are present, and M represent the number of revertants per plate in the positive control. A value below 25% indicated no anti-mutagenic effect, a moderate effect fell within the range of 25%–40%, and a strong anti-mutagenicity value exceeded 40% (Negi et al., 2003; Ong et al., 1986). Each sample was assessed using three plates per run, and the data presented are the average values derived from three separate tests conducted with Z. zerumbet extracts obtained from different batches.
Cytotoxicity Assay
The Mosmann (1983) cell viability assay was conducted to assess the cytotoxic effects of methanol, hexane, and ethyl acetate extracts. V79-4 cells were cultured at a density of 1 × 103 cells/mL in 96-well plates using DMEM culture media supplemented with 1% penicillin–streptomycin and 10% fetal bovine serum (FBS). These cultured cells were then exposed to five different concentrations of methanol, hexane, and ethyl acetate extracts. After 24 h of treatment, the cells were incubated with 20 µL of 3-(4,5-dimethylthiazol-2-yl)-2,5-dipheniltetrazolium bromide for 4 h at 37°C and 5% CO2. The mitochondrial protein glucose-6-phosphate dehydrogenase (G6PD) converted MTT into formazan, resulting in the formation of purple crystals. To dissolve the cell membranes and formazan crystals, 200 µL of DMSO was added to each well. The absorbance (OD) of the purple formazan compound was measured at 570 nm to determine cell viability.
A dose–response curve, depicting the percentage of cell survival versus concentration, was constructed using linear regression interpolation analysis to determine the IC50, which represents the minimum concentration of methanol, hexane, and ethyl acetate extracts that leads to 50% survival of V79-4 cells.
Results
Mutagenicity Assay
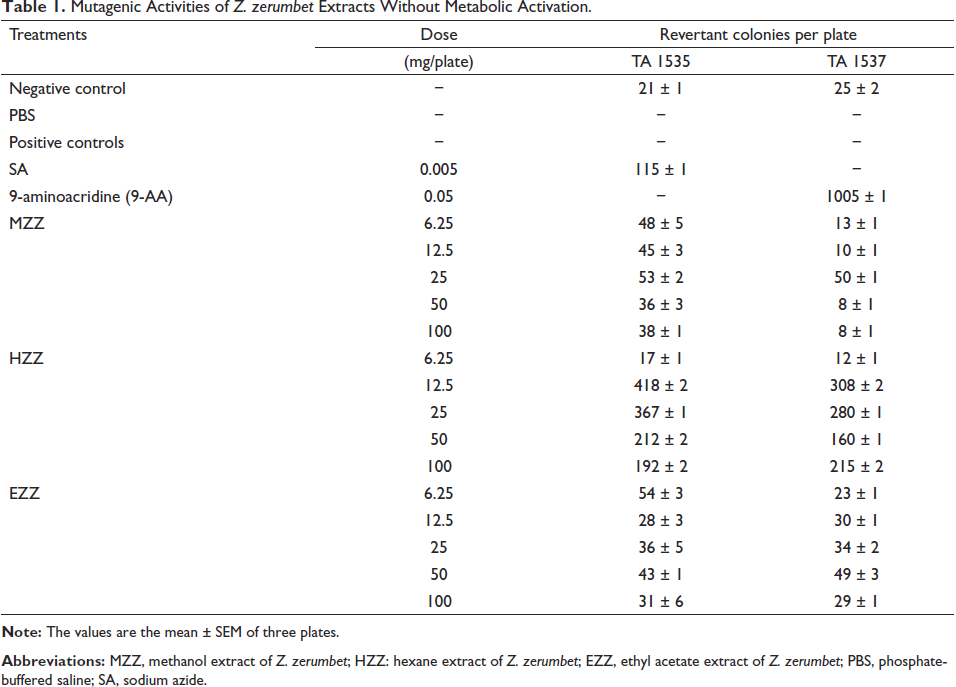
The results of the mutagenicity assay are provided in Table 1. Notably, the number of revertants caused by the methanol extract of Z. zerumbet (MZZ) (6.25-25 mg per plate) was twice as high as that of the negative controls, except for S. typhimurium TA 1537. Similarly, for the two strains, the number of revertants induced by the hexane extract of Z. zerumbet (HZZ) increased to twice that of the negative controls, except for one group with a concentration of 6.25 mg/plate. On the other hand, the number of revertants induced by the ethyl acetate extract of Z. zerumbet (EZZ) (6.25 and 50 mg/plate) was also twofold higher than the negative controls, except for S. typhimurium TA 1537. However, all concentrations still maintained a revertant count above average. Overall, HZZ exhibited the highest mutagenic potential compared to EZZ and MZZ. Thus, these findings indicate that MZZ, HZZ, and EZZ are mutagenic in both S. typhimurium strains TA 101535 and TA1537, independent of metabolic activation.
Mutagenic Activities of Z. zerumbet Extracts Without Metabolic Activation.
Anti-mutagenicity Assay
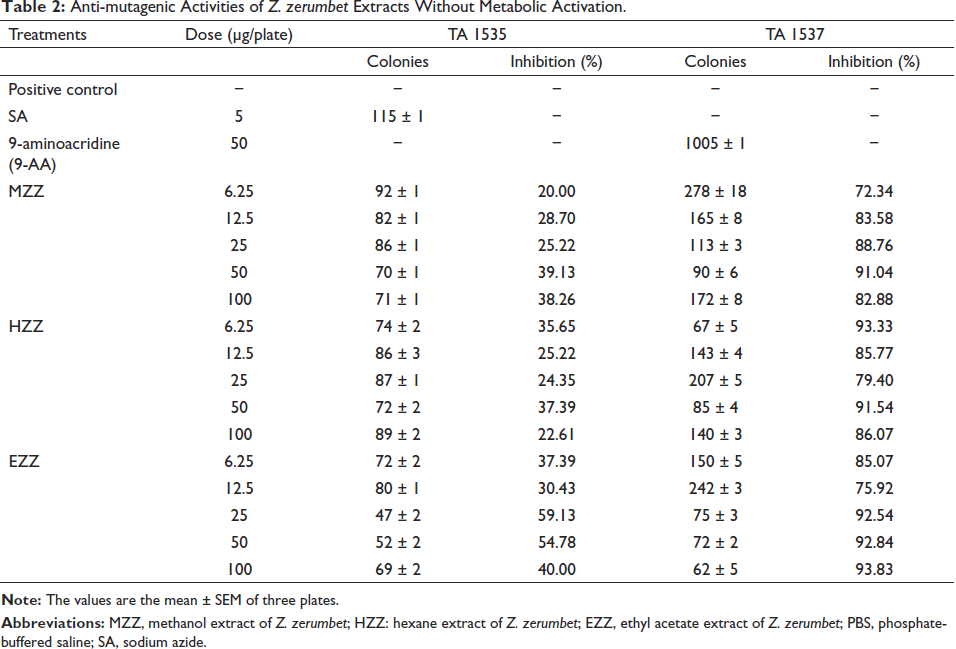
The anti-mutagenic activity of the samples was demonstrated by their ability to retain the mutagenic effects of NaN3 and 9-AA. As presented in Table 2, the MZZ exhibited a lower number of revertants compared to the positive control, with inhibition rates of 39.13% against NaN3 and 91.04% against 9-AA. Both inhibitions were observed at a concentration of 50 mg/plate. HZZ displayed a 37.39% inhibition against NaN3 at a concentration of 50 mg/plate while achieving a 93.33% inhibition against 9-AA at a concentration of 6.25 mg/plate. The most potent anti-mutagenic effect was observed in EZZ, with inhibition rates of 59.13% against NaN3 and 93.83% against 9-AA.
Anti-mutagenic Activities of Z. zerumbet Extracts Without Metabolic Activation.
Cytotoxicity Assay
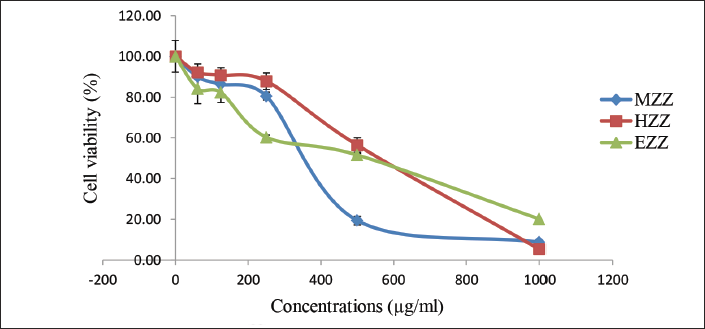
Based on Figure 1, it is evident that among the extracts, MZZ exhibited the highest cytotoxicity against the V79-4 cell line, surpassing HZZ and EZZ. The estimated IC50 value for MZZ was 388.50 ± 135.75 µg/mL. On the other hand, the IC50 values for HZZ and EZZ were 514.17 ± 135.75 and 589.67 ± 241.67 µg/mL, respectively.
Statistical Analysis
The results were presented as the mean ± SEM of three independent assays. The effect was analyzed using a one-way ANOVA test, and a significance level of p < 0.05 was considered statistically significant.
Discussion
The main objective of the current study was to evaluate the mutagenic, anti-mutagenic, and cytotoxic effects of Z. zerumbet rhizome extracts utilizing different solvents, namely methanol, hexane, and ethyl acetate. While Chang et al. (2012) previously investigated the mutagenicity and genotoxicity of Z. zerumbet extracts, their focus was limited to ethanol extracts. A phytochemical investigation conducted by Kader et al. (2011) revealed the presence of various compounds in the ethanol extract of Z. zerumbet, including zerumbone, zerumbone epoxide, diferuloymethane, feruloyl-p-coumaroyl-methane, di-p-coumaroyl-methane sesquiterpenoids, flavonoids, aromatic compounds, and vanillin. In contrast, our study encompassed a broader spectrum by including methanol, hexane, and ethyl acetate extracts.
Our findings suggest that the methanol, hexane, and ethyl acetate extracts of Z. zerumbet demonstrate moderate mutagenic activity in the absence of metabolic activation (S9) across various aspects, indicating potential carcinogenic properties. It is crucial to acknowledge that the context and concentration of the extract play a critical role in determining the potential health implications when considering extracts with moderate mutagenic effects. Nonetheless, it is important to note that all the extracts exhibited greater anti-mutagenic activity than mutagenic activity in the absence of metabolic activation, aligning with the observations made by Taherkhani and Rustaiyan (2016). This suggests the possibility of cancer prevention due to the simultaneous presence of anti-mutagenic activities, even though mutagenic effects may be unavoidable in plant extracts. Furthermore, the mutagenicity of Z. zerumbet can be attributed to histidine and histidine-related precursors present in the plant extracts, which can interfere with the Ames test by increasing the number of spontaneous revertants and potentially leading to false-positive results (Jin et al., 2009).
Apart from its antitumor (Huang et al., 2005; Murakami et al., 2004) properties, ZER, which is the main component found in the extracts of Z. zerumbet rhizome, also possesses sedative (Sulaiman et al., 2010) and antinociceptive (Sulaiman et al., 2009) effects. Notably, the observed mutagenic effects cannot be attributed to the presence of ZER in the extracts, as it was determined to be non-mutagenic regardless of the absence or presence of a metabolic initiation system (Al-Zubairi, 2012). Among the extracts, HZZ exhibited the most potent mutagenic effect, likely due to a combination of flavonoids (Beudot et al., 1998; Fernandes de Sá Ferreira & Ferrão Vargas, 1999) and the compound’s specific structure (Beudot et al., 1998). Chung et al. (2008) identified ZER and 6-methoxy-2E, 9E-humuladien-8-one as the compounds present in the hexane extracts.
There is a scarcity of scientific studies investigating the mutagenic effects of Z. zerumbet extracts. The mutagenic effects of these extracts have not been extensively researched or well documented in scientific literature. In contrast, Zingiber officinale Roscoe, commonly known as ginger, has received recognition for its potential health benefits, including its anti-inflammatory and antioxidant effects. Ginger has been extensively studied for its mutagenic and cytotoxic properties. The pungent compounds responsible for its spicy aroma are gingerols, shogaols, paradols, and zingerone (Khan et al., 2016), with 6-gingerol being the principal compound. In dried ginger, the concentration of gingerols decreases, while the concentration of shogaols, resulting from gingerol dehydration, becomes more abundant (Sharifi-Rad et al., 2017). Several compounds, such as 6-shogaol, 10-gingerol, and an enone-diarylheptanoid analog of curcumin, have been identified as cytotoxic in human tumor cell lines (Peng et al., 2012). Ginger has shown some level of mutagenicity in TA 100, TA 1535 (Nagabhushan et al., 1987; Soudamini et al., 1995), and TA 98 strains (Abudayyak et al., 2015). However, the mutagenic activity of ginger was significantly lower than that of established mutagens like NaN3 or methylnitronitrosoguanidine (MNNG) (Soudamini et al., 1995). This mutagenicity could be attributed to gingerols and shogaols (Soudamini et al., 1995; Yang et al., 2010). Interestingly, the presence of zingerone suppressed the mutagenic activity of gingerols and shogaols in a dose-dependent manner. Although all three compounds share a similar chemical structure, zingerone has a shorter side chain compared to the mutagenic compounds, suggesting that the side chains may be responsible for the mutagenic activity of gingerol and shogaol (Metz & Cupp, 2000). Therefore, the observed mutagenic activity of ginger extracts in vitro is likely the result of the combined action of both pro- and anti-mutagenic compounds present in ginger (Nagabhushan et al., 1987).
Ideally, the use of any therapeutic intervention should involve a thorough risk-to-benefit evaluation. Therefore, it is important for all treatments to either carry no risks or have minimal risks. Herbal medicines, similar to chemically defined therapies, can potentially lead to adverse events. Consequently, a risk-benefit assessment should be conducted for these therapies, including ongoing toxicological testing and rigorous quality assurance measures such as pharmacovigilance (Holtmann et al., 2020). To evaluate the potential therapeutic use of Z. zerumbet extracts, the risk–benefit ratio should be established by assessing the selective toxicity of methanolic, hexane, and ethyl acetate extracts on normal and cancer cells. The cytotoxicity effects of the extracts on both cell types can be investigated using the trypan blue assay. The cells can be treated with various concentrations of Z. zerumbet extracts for 24 h, and the IC50 and EC50 values can be determined for each extract. The therapeutic index (TI) can then be calculated to assess whether the cytotoxic effects of the extracts are selective for cancer cells in comparison to non-cancer cells. A higher therapeutic index indicates the potential for future development of a chemotherapeutic agent (Omar & Zain, 2018). TI is defined as the ratio between the half-inhibitory concentration (IC50) of the extract that becomes toxic in normal cells and the half-effective concentration (EC50) of the extract that is effective in cancer cells (Cha et al., 2005). This index provides a simple measure for assessing the safety and efficacy of target drugs. Extracts with a high therapeutic index are capable of effectively killing cancer cells at lower concentrations (i.e., lower EC50 concentrations in cancer cells compared to higher IC50 concentrations in normal cells) compared to extracts with a low therapeutic index (Deepa et al., 2012).
Scholars strive to find anti-mutagens, a class of chemicals that can reduce the occurrence of mutations, to combat the harmful impact of mutagenicity (Gautam et al., 2016; Słoczyńska et al., 2014). Anti-mutagens can be classified into the following categories according to their respective mechanisms of action: those with antioxidant properties, those that hinder the activation of mutagens, those that function as blocking agents, and those with multiple modes of action as anti-mutagens (Słoczyńska et al., 2014). Z. zerumbet extracts are likely to contain bioactive compounds that possess both cytotoxic and anti-mutagenic properties. These compounds exert their effects through various mechanisms, such as apoptosis induction, inhibition of cell proliferation, and anti-inflammatory actions. They exhibit selective cytotoxicity by targeting cancer cells while sparing normal cells. Additionally, these compounds hinder crucial signaling pathways involved in cell growth, thereby slowing down or halting the proliferation of cancer cells.
In the anti-mutagenicity assay, EZZ exhibited the most robust anti-mutagenic activity against 9-AA without metabolic activation, although it was less effective against NaN3-induced mutagenicity. This suggests that EZZ can effectively prevent the non-covalent binding of 9-AA to DNA (Ferguson & Denny, 2007), but it does not inhibit the interaction between NaN3 and DNA, which may result in selective modifications to the genome (Gulluce et al., 2010). Although tannins, as phenolic compounds, are known to potentially impact anti-mutagenic activity, our study found that tannins were not responsible for the observed anti-mutagenic effect, as no metabolic initiators were present (Tanaka et al., 1998).
EZZ is believed to contain abundant amounts of kaempferol, which contributes to its sedative effects. Some examples of kaempferol derivatives found in EZZ include kaempferol-3-O-rhamnoside, kaempferol-3-O-(2’- or 3’-acetyl)-rhamnoside, kaempferol-3-O-(4’-acetyl)-rhamnoside, kaempferol-3-O-(3’,4’-diacetyl)-rhamnoside, and kaempferol-3-O-(2’,4’-diacetyl)-rhamnoside (Ruslay et al., 2007). Kaempferol, a flavonoid compound known for its cell-enhancing properties, is highly beneficial when included in the diet through the consumption of leafy foods. Such foods, rich in cell-enhancing compounds, have been associated with a reduced risk of malignancies, hypertension, cardiovascular disease, and stroke. Furthermore, kaempferol is the most active compound found in the ethyl acetate extract of Z. zerumbet (Ruslay et al., 2007).
Besides, the ethyl acetate extract of Z. zerumbet has been found to exhibit protective effects against ethanol-induced brain damage, mediated through its antioxidant properties (Hamid et al., 2018). Antioxidants could neutralize reactive oxygen species (ROS) and free radicals generated by mutagens. By scavenging these harmful molecules, antioxidants can protect cells from oxidative damage and potentially reduce the occurrence of mutagenesis. The anti-mutagenic activity of plant extracts is attributed to their antioxidant compounds. Notably, the study by Makhafola et al. (2016) establishes a clear correlation between the antioxidant activity and anti-mutagenicity of the tested extracts.
Therefore, the anti-mutagenic effect of EZZ can be mainly attributed to the presence of the kaempferol mixture within the extract. The combination of kaempferol with other compounds, such as flavonoids, likely further enhances the anti-mutagenic response of EZZ. Additionally, both HZZ and MZZ demonstrated significant anti-mutagenic activity, indicating potential mechanisms involving radical scavenging, antioxidant activities (Budin et al., 2013), and inhibition of lipid peroxidation. Moreover, the anti-mutagenic effects of EZZ and MZZ may also be closely linked to their ability to inhibit the proliferation of cancer cells, with EZZ exhibiting superior anti-proliferative effects compared to MZZ (Abd Rashid & Pihie, 2005).
Researchers widely prefer to utilize ZER in their studies, particularly those investigating mutagenic and anti-mutagenic properties, due to its plentiful presence in Z. zerumbet rhizome extracts and essential oils. The essential oil alone accounts for approximately 37% of ZER, and significant quantities of alpha-humulene and camphene are also found (Chane-Ming et al., 2003). ZER possesses numerous biological activities that contribute to its anti-mutagenic properties (Kumar et al., 2013). The unsaturated moiety of ZER, characterized by a single isolated double bond, is considered vital for its anti-cancer effects, which exhibit a broad spectrum of suppressive activities, including antiproliferative and apoptosis-inducing effects (Abdel Wahab et al., 2009; Adbul et al., 2008). It has been shown that ZER inhibits the proliferation of leukemia cells by inducing G2/M cell cycle arrest, followed by apoptosis (Xian et al., 2007). Numerous studies have reported the antitumor activity of ZER in both in vitro and in vivo models, including breast, colorectal, cervical, lung, renal cell carcinoma, and skin cancers (Girisa et al., 2019).
NaN3 and 9-AA are well-known direct-acting mutagens commonly used in scientific research. At the molecular level, plant extracts with anti-mutagenic properties may interact with mutagens in various ways. For example, the anti-mutagenic activity of luteolin derivatives isolated from Mentha longifolia (L.) on TA1537 could be attributed to their ability to inhibit the binding of 9-AA to DNA (Orhan et al., 2012). Similar interactions with 9-AA were observed in the study by Gulluce et al. (2010). The specific mechanisms underlying the anti-mutagenic action of Z. zerumbet extracts and their interactions with NaN3 and 9-AA may vary depending on the active compounds present in the extracts and the experimental conditions. According to the anti-mutagenicity results, the potential mechanism of anti-mutagenicity for Z. zerumbet extracts could involve the inhibition of an organic metabolite of azide called L-azidoalanine (Sadiq & Owais, 2000). Certain anti-mutagens may also enhance DNA repair mechanisms within cells. DNA repair processes play a crucial role in identifying and correcting errors or damage in the DNA sequence caused by mutagens, thus preventing the propagation of mutations.
According to the findings of Norfazlina et al. (2013), variations in solvents, cancer cell lines, extraction methods, and plant species can result in different cytotoxic effects. Consequently, the mutagenic and anti-mutagenic activities of extracts may also vary accordingly. In their study, the hexane extract of Z. zerumbet demonstrated slightly higher activity compared to the ethanol extract. This can be attributed to the polarity of hexane, which tends to attract more ZER, leading to increased cytotoxic activity. The study also revealed that hexane proved to be a more effective solvent for extracting Z. zerumbet compared to ethanol. In contrast, Javanmardi et al. (2003) found that the aqueous extract of pomegranate peel exhibited the highest anti-mutagenic activity while displaying the lowest antioxidant activity compared to ethyl acetate, acetone, and methanol extracts.
Previously, the traditionally prepared crude extract of Z. zerumbet exhibited inhibitory properties on the growth of different cancer cell lines when tested in laboratory conditions. While pressurized liquid extraction (PLE) has emerged as a potential alternative to traditional organic solvent extraction due to its reduced extraction time and solvent volume (Saleh & Sanagi, 2009), the hexane extract of Z. zerumbet obtained through the PLE method still demonstrates comparable cytotoxic effects on both human myeloid leukemia (HL60) and V79-4 cells when compared to hexane extracts produced using the maceration technique.
Kumar et al. (2013) utilized the processing method of ZnCl2-catalyzed Beckmann rearrangement of zerumbone oxime. This approach enabled the successful synthesis of two innovative compounds, namely azazerumbone 1 and azazerumbone 2. Notably, azazerumbone 2 demonstrated superior activity compared to ZER. These newly synthesized compounds exhibited potent protection against NaN3-induced mutagenicity in S. typhimurium strains TA 98 and TA 1531. Furthermore, it was observed that azazerumbone 2 displayed superior anti-bacterial and anti-mutagenic activity compared to azazerumbone 1. Another recent study has reported the synthesis of new zerumbone-bicarbonyl analogues through SeO2 (selenium dioxide) oxidation. The compounds obtained from this synthesis also demonstrated significantly greater anti-mutagenic activity against Salmonella tester strains compared to ZER (Kumar et al., 2017).
No side effects or toxicity were observed at the concentrations utilized in this study. The selection of Z. zerumbet extract concentrations was based on our previous Ames and cytotoxicity studies, ensuring their appropriateness for this investigation (Florinsiah et al., 2014; Ghazali et al., 2011; Norfazlina et al., 2013). The IC50 values revealed that MZZ exhibited the highest cytotoxicity against the V79-4 cell line compared to HZZ and EZZ, with an estimated IC50 of 388.50 ± 135.75 µg/mL. MZZ primarily contains compounds such as ZER, zerumbonoxime, and diferuloylmethane, which have demonstrated cytotoxic effects on mouse cell lines (Matthes et al., 1980). Furthermore, Al-Zubairi et al. (2010a, b) provided additional evidence supporting the cytotoxic effect of ZER.
In a more recent study, Ali et al. (2022) conducted GC–MS analysis, revealing that the main constituents of the MZZ extract were 1,2-benzenedicarboxylic acid (38.4%), ZER (36.9%), and α-cariophyllene (10.6%). The polyphenol-rich extract exhibited significant activity in inhibiting nitric oxide radicals and lipid peroxidation when tested against Ehrlich ascites carcinoma (EAC) cells, surpassing the effects of the standard catechin. In vivo experiments further showed that MZZ effectively inhibited EAC cell growth and restored abnormal hematological characteristics in mice bearing EAC. Additionally, MZZ induced nuclear condensation and fragmentation, both prominent features of apoptosis (Ali et al., 2022).
ZER demonstrates significant cytotoxic activity against various types of cancers, including lung, colon, leukemia, ovarian, skin, liver, and breast cancers. It exhibits selective cytotoxicity against human tumor cell lines (Deng et al., 2022; Girisa et al., 2019). Additionally, ZER also demonstrated cytotoxic and genotoxic effects on murine thymoma cells (Hamid et al., 2017). Therefore, ZER plays a pivotal role in the cytotoxic activity of MZZ. It should be noted that MZZ exhibited cytotoxicity against normal cells at exceptionally high doses, which may enhance its potential as an anti-cancer agent.
Previously, our laboratory conducted an assessment of the cytotoxicity of Z. zerumbet extracts, specifically focusing on the hexane extract, using HL60 (with an IC50 value of 63.72 ± 5.363 µg/mL) and V79-4 (with an IC50 value of 40 µg/mL) cell lines. It was observed that the hexane extract may target distinct cellular features present in various cell types, leading to its cytotoxic effects. Additionally, the hexane extract was found to induce apoptosis as the primary mode of cell death (Norfazlina et al., 2013). Furthermore, the cytotoxicity of Z. zerumbet extracts was also evaluated using different cell lines, such as the human dermal fibroblast adult cell line (HDF-a). The extract was determined to be non-toxic at concentrations up to 1000 µg/mL and even exhibited an increase in cell proliferation at a concentration of 750 µg/mL. Interestingly, treatment with Z. zerumbet extract resulted in an accelerated rate of cell migration after 48 h (Latif et al., 2019).
Additionally, in a previous study focusing on nephrotoxicity, we discovered that the ethyl acetate extract of Z. zerumbet exhibits nephroprotective properties against paracetamol-induced nephrotoxicity (PCM) (Abdul Hamid et al., 2012). This protection is likely attributed to the extract’s antioxidant properties. When given intraperitoneally at doses of 200 mg/kg and 400 mg/kg along with oral administration of PCM, the extract effectively prevented PCM-induced nephrotoxicity and oxidative damage in the kidneys. The effectiveness of the extract was supported by significant reductions in plasma creatinine levels, as well as decreased levels of plasma and renal malondialdehyde (MDA), plasma protein carbonyl, and renal advanced oxidation protein product (AOPP). Both doses of the extract also resulted in notable increases in plasma and renal glutathione (GSH) levels, along with enhanced plasma superoxide dismutase (SOD) activity. The histological examination further revealed a reduction in the severity of renal cellular damage. Moreover, the dose of 400 mg/kg exhibited greater efficacy in providing protection compared to the 200 mg/kg dose.
The use of chemotherapy with anti-cancer drugs is the typical therapeutic approach for cancer. However, this approach is often accompanied by severe side effects and complications, which pose significant challenges in clinical settings. For instance, the side effects of current therapeutic treatments like cisplatin have been a concern (Al-Zubairi et al., 2010a). Studies have indicated that Z. zerumbet extracts possess potential anticancer properties and have shown cytotoxic effects against various cancer cell lines in laboratory experiments. In the comparison of cytotoxicity between Z. zerumbet extracts (hexane and ethanol) and current chemotherapeutic agents, the anti-cancer drug Doxorubicin displayed greater potency on HL60 cells, exhibiting an IC50 of 1.231 ± 0.3689 µM. Conversely, the hexane extract revealed an IC50 of 63.72 ± 5.363 µM, while the ethanol extract demonstrated an IC50 of 345.3 ± 34.69 µM (Norfazlina et al., 2013).
The potential of combining Z. zerumbet extracts with other substances or drugs to mitigate their mutagenic effects while enhancing their anti-mutagenic properties should be further investigated through thorough experimental studies. In our previous study, we investigated the effects of Nigella sativa (Ns) and Z. zerumbet (Zz) on the HL60 cell line using varying combination doses. The results revealed antagonistic interactions between Ns and Zz at different combination dose settings. When administered alone, both Ns and Zz exhibited stronger anti-proliferative properties compared to their co-administration. Ns (petroleum ether extract) had an IC50 value of 654.9 µg/mL, indicating lower cytotoxicity compared to Zz (hexane extract), which inhibited 50% of HL60 viable cells at a concentration of 63.72 µg/mL. These findings suggest that combining Ns and Zz may not be a safe approach for developing a drug (Norfazlina et al., 2014). However, the combination of ZER with vinblastine and paclitaxel demonstrated strong synergistic effects in inhibiting the proliferation of HeLa cells, indicating a similar mechanism of action to clinically used chemotherapeutic drugs (Ashraf et al., 2019). Furthermore, the combination of ZER with cisplatin exhibited a dose-dependent induction of cytotoxicity and apoptosis in head and neck squamous cell carcinoma (Schnoell et al., 2022). Considering these results, it is advised to exercise caution when contemplating the combination of Z. zerumbet extracts with other substances or drugs. This careful approach is crucial due to the potential for increased mutagenic effects and cytotoxicity associated with such combinations.
The investigation of Z. zerumbet extracts, in conjunction with other treatments such as anti-leptospiral agents, is noteworthy for the control of leptospirosis through chemoprophylaxis. This is particularly due to the extracts’ ability to cause DNA damage. The anti-leptospiral activity of the hexane extract of Z. zerumbet has been demonstrated against various serovars of Leptospira spp., including Canicola, Australis, and Batavie, as well as serovar Patoc of Leptospira biflexa. However, no significant anti-leptospiral activity was observed for the ethyl acetate and methanol extracts. These findings suggest that the hexane extract effectively inhibits the growth of L. biflexa serovar Patoc and L. interrogans serovar Australis through its DNA-damaging properties (Ibrahim et al., 2021).
However, the identification of the key compound responsible for the mutagenic, anti-mutagenic, and cytotoxic effects of Z. zerumbet remains unexplored among various extract types. Further studies are warranted to investigate the mechanisms of action and the specific compound mixtures present in Z. zerumbet. Genotoxicity evaluations are crucial nonclinical safety studies necessary for the registration and approval of pharmaceutical products on the market. Additionally, it is essential to conduct studies on the genotoxicity of medicinal plants widely used by the general population to identify any ingredients that may pose mutagenic and carcinogenic risks (Chang et al., 2012). However, there are certain limitations to our study. We were unable to identify the specific components within the rhizome of Z. zerumbet responsible for its mutagenic, anti-mutagenic, and cytotoxic properties. Furthermore, we did not establish the precise molecular mechanism through which all the extracts inhibit V79-4. To address these gaps, further investigations are planned, which involve isolating the active constituents from the raw extract and examining their potential anti-cancer properties.
Conclusion
Based on these findings, it is apparent that all Z. zerumbet (L.) Smith extracts possess mutagenic, anti-mutagenic, and cytotoxic properties against the tested bacterial strains. Moreover, these extracts exhibit promising anti-mutagenic activity, suggesting their potential utility as chemopreventive agents and in anti-cancer treatments.
Summary
Despite possessing mutagenic and cytotoxic properties, extracts of Z. zerumbet (L.) Smith demonstrate potential as an anti-mutagenic agent, suggesting their usefulness in chemoprevention and anticancer treatments.
Abbreviations
MZZ: Methanol extract of Z. zerumbet; HZZ: Hexane extract of Z. zerumbet; EZZ: Ethyl acetate extract of Z. zerumbet; S.typhimurium: Salmonella typhimurium; MTT: Methyl thiazolyl tetrazolium; PBS: Sodium phosphate buffer; FBS: Fetal bovine serum; DMSO: Dimethyl sulfoxide; NaN3: Sodium azide; 9-AA: 9-aminoacridine; G6PD: Glucose-6-phosphate dehydrogenase; OD: Absorbance; CO2: Carbon dioxide; DNA: Deoxyribonucleic acid; ATCC: American Type Culture Collection.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Fundamental Research Grant Scheme (FRGS/1/2013/SKK01/UKM/02/1) provided financial backing for this study. This grant was awarded by the Ministry of Higher Education (MOE), Government of Malaysia.