Abstract
Purpose:
Fluconazole is an effective anti-fungal drug. Due to the limitations of fluconazole, such as poor water solubility and consequently low ocular bioavailability, an optimized fluconazole nanoemulsion in-situ gel formulation (temperature-sensitive) was developed.
Methods and Materials:
To verify formulation’s safety for ophthalmic use, preparation was tested for potential ocular toxicity using a cell viability assay on retinal cells. The hen’s egg test-chorioallantoic membrane (HET-CAM), as a borderline test between in vivo and in vitro techniques, was chosen for investigating the irritation potential of the formulation. HET-CAM test was done by adding the formulation directly to the CAM surface and monitoring the vessels visually in terms of irritation reactions. Eye tolerance was determined using the modified Draize test.
Results:
Viability assay on retinal cells displayed that fluconazole nanoemulsion in-situ gel formulation was non-toxic and can be safely used in the eye at concentrations of 0.1% and 0.5%. HET-CAM and Draize tests revealed that optimized formulation of fluconazole did not result in any irritation and was considered non-irritant and well-tolerated for ocular use.
Conclusion:
Regarding to the findings of the three mentioned methods, fluconazole nanoemulsion in-situ gel formulation is harmless and as a proper and safe alternative, can be considered for ocular delivery of fluconazole in the future.
Introduction
In today’s society, various eye infections such as fungal infections can occur despite the development of public health. 1 Fluconazole (FLZ) as an azole prevents the ergosterol synthesis. Among different azoles, fluconazole has less effect on steroid metabolism in humans. 2
Various challenges are existing for topical ocular drug delivery because of the unique structure of the eyes. 3 The hydrophilic and lipophilic properties of different corneal tissue parts can reduce ocular bioavailability. Moreover, the loaded drug can be drained away into the nasolacrimal duct by various structural and physiological phenomenon such as blinking reflex and tear drainage. 4 Low ocular bioavailability has been overcome by different strategies; for instance, nanotechnology can control drug release. Nanoemulsion (NE), a nano-sized system, is a transparent system which has been established to accomplish prolonged ocular drug delivery, reduced drug administration and adverse effects, and high patient adherence. Besides, drug penetration to deeper layers of ocular structure and corneal absorption can be improved by the nano-range size of nanoemulsions. 5 Nanoemulsion in-situ gel systems by a sol-gel transition at a definite temperature, pH, or ionic strength prolong residence time in the target organ. 6 The previous study displayed that pH-triggered nano emulsified in-situ gel (NE-ISG) of fluconazole was more effective for ocular fungal infections because of greater permeation, sustained drug release, and prolonged precorneal residence time along with no signs of tissue injury in corneal toxicity studies. 7
Besides the efficacy, the safety of the ophthalmic products must be considered because of the sensitivity of the eye tissue. Although the effectiveness of nanoemulsion in-situ gel systems in ocular drug delivery has been confirmed, the extreme reactivity of nanomaterials and the high concentration of surfactants and co-surfactants in their structure make them cause irritation and corneal injury. 8 Among several eye toxicity tests, in vitro tests have always been the first choice. 9 Cell culture study can provide a toxicity evaluation in an easy system that minimizes the influence of confounding variables. The irritation potential evaluation can be performed by application the Hen’s Egg Test-Chorioallantoic Membrane (HET-CAM) method. HET-CAM assay, an alternative for in vivo animal assessments, is very beneficial, easy, cheap, sensitive, and well-established to screen possibly toxic, teratogenic, and irritant products. 10
According to the physicochemical properties of fluconazole, such as poor water solubility leading to low ocular bioavailability, an optimized fluconazole nanoemulsion in-situ gel (temperature-sensitive) formulation has been developed. 11 Conversion of solution at room temperature (25°C) to clear gel at corneal surface temperature (30–34°C) was done using a mixture of Poloxamer 188 and Poloronic 407 as thermoresponsive gelling agents. Given the advantages of nanoemulsion in-situ gel systems, the current study was designed to evaluate the toxicity and irritation potential of fluconazole nanoemulsion in-situ gel (FLZ NE-ISG) formulation by MTT, HET-CAM, and Draize tests.
Materials and methods
Materials
Triacetin (glycerol triacetate) and Propylene glycol (PG) were obtained from Samchun Chemical Co., Ltd, South Korea. Poloxamer 407, Poloxamer 188, and Cremophor RH40 were purchased from Sigma Aldrich Chemical Company, USA. Fluconazole powder was offered kindly from Iran Daru Pharmaceutical Company, Iran. Other solvents, chemicals, and animals were obtained locally.
Preparation of nanoemulsion in-situ gel formulation
Cold low energy method was employed to prepare FLZ NE-ISG formulation. In this method, the lipid and aqueous phases were prepared separately. The aqueous phase was made by dissolving the desired number of gelling agents (13% of Poloxamer 407, 1.25% of Poloxamer 188) in the deionized water with continuous stirring under a magnetic stirrer at 500 rpm for 5 hours in the ice bath. The lipid phase was prepared by pouring 0.3% of fluconazole powder, 5% of Cremophor RH40 as a surfactant, 5% of PG as co-surfactant, and 5% of Triacetin as oil in the vial with continuous stirring for 10 min. After obtaining a homogenous mixture, the aqueous phase was added to the organic phase and stirred at 500 rpm in the ice bath overnight.
Ocular toxicity tests
Cell viability assay (MTT test)
Human retinal pigment epithelial cells (RPE) were cultured in a 96-well plate (1 × 104 cells per well) in 0.1 ml of DMEM-F12 (1:1 mixture of Dulbecco’s Modified Essential Medium (DMEM) and Ham’s F-12) + fetal bovine serum 10% (FBS) + 2 mM L-Glutamine. Following 24 h incubation at 37°C and 5% CO2, cells were rinsed with PBS and three different concentrations of selected FLZ NE-ISG formulation (0.1, 0.5, 1% v/v), blank NE-ISG formulation, and fluconazole (dispersion of 3 mg fluconazole powder in 1 ml of distilled water) mitigated with fresh medium, was added to the plate and incubated for 1 hour. Untreated cells were used as a reference for cell viability determination (positive control), and DMSO was used as a negative control. After 3 incubation hours with the fresh medium encompassing 50 μg/ml of MTT, cell viability was determined by measuring the absorbance at 550 nm on an ELISA plate reader.12,13 The following equation was used for calculating cell viability:
The investigation was performed in triplicate, and results were stated as mean ± standard deviation.
HET-CAM test
Ten fertilized chicken eggs were selected to evaluate the irritation potential of each concentration. The eggs were incubated at 37 ± 1°C and 65 ± 1% relative humidity for 8 days. Eggs were rotated gently every 12 hours in the equatorial axis. Eggs with the chorioallantoic membrane’s optimal illumination were chosen. A flashlight was used to perceive embryo development in detail. CAM exposing was performed by cutting eggshell and removing the inner membrane without any injury to blood vessels. 200 µL of FLZ NE-ISG formulation, buffer phosphate solution (pH = 7.4) as a negative control and NaOH 0.1 M for coagulation, acetone for hemorrhage, and propylene glycol (PG) for hyperemia as positive controls, were applied on the CAM surface. The irritation effect of each formulation and substance was assessed by visual inspection changes of the blood vessels such as coagulation, hemorrhage, and hyperemia during 300 seconds at 0.5, 2, and 5 min following CAM exposure. Irritation potential was examined based on the HET-CAM irritation score classification systems noted in Table 1.
Numerical scores of irritation responses in the HET-CAM test. 14

Draize test
Prediction of ocular tolerance against developed FLZ NE-ISG formulation was performed using the modified Draize irritancy test. Three male healthy New Zealand rabbits weighing 3–4 kg were used in each group. Rabbits were maintained in cages at 22–25°C and fed at the same time every day till the day of the experiment. The experiment was accepted by Hamadan University of Medical Sciences Medical Ethics Review Board (Approval NO. IR. UMSHA.REC.1398.169). It was done under the guidelines of the National Institutes of Health for the care and use of Laboratory Animals (NIH Publications No. 8023, revised 1978). The following groups were designed to perform a modified Draize irritancy test: The positive control group (100 µl of sodium dodecyl sulfate (SDS) 1% w/v). The negative control group (100 µl of phosphate buffer, pH = 7.4). The blank group (100 µl of NE formulation without fluconazole). Test group (100 µl of NE formulation of fluconazole).
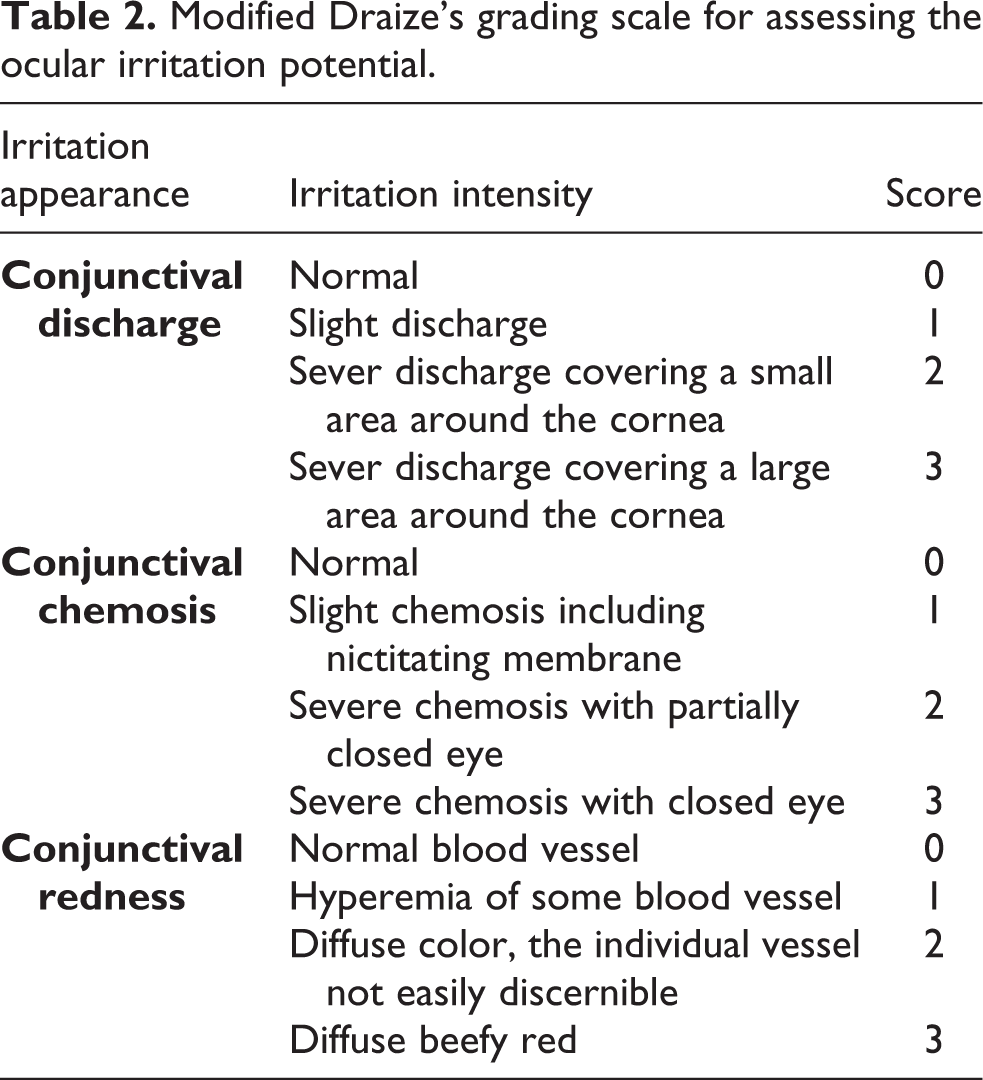
Sterilization of each solution was done using filter 0.22 µm before instillation. Inflammation and irritation reactions such as discharge, chemosis, and redness were examined by observing cornea and conjunctiva tissue after 1, 2, 3, 4, 6, 8, and 24 h of sample instillation. As shown in Table 2, the irritation score of each eye was considered as the sum of the three parts, and the ultimate irritation score was considered as the average of the sum of the scores. A score of more than two was considered a clinically significant irritant. 15
Modified Draize’s grading scale for assessing the ocular irritation potential.
Statistical analysis
One-way analysis of variance (ANOVA) followed by a post-hoc test (Tukey’s test) was carried out for statistical analysis using GraphPad Prism 7. All tests were done triplicate, and the results expressed as mean ± standard deviation (SD). P value <0.05 was selected as a significant level.
Results
Preparation of FLZ NE-ISG formulation
Prepared nanoemulsion formulation had a transparent appearance and was soluble at room temperature (25°C). Increasing the temperature up to the eye temperature range (30–34°C) can convert it into the gel form.
Cell viability assay (MTT test)
At the concentrations of 0.1% and 0.5%, cell viability was more than 75%, and no significant induction of cell death was detected for FLZ NE-ISG formulation, blank NE-ISG formulation, and fluconazole. This data verifies that the safety of FLZ NE-ISG formulation in both concentrations of 0.1% and 0.5% is acceptable. As it is demonstrated in Figure 1, By increasing the concentration up to 1%, the cell viability of FLZ NE-ISG formulation and blank NE-ISG formulation decreased below 75% in RPE cells. Based on the observed results, after 24 h of incubation, FLZ NE-ISG formulation at 0.1% and 0.5% concentrations were considered non-toxic for RPE cells.

PRE cell viability of FLZ NE-ISG formulation, blank NE-ISG formulation, and fluconazole after 24 h according to the MTT test.
HET-CAM test (Ocular irritation test)
Instantly after adding NaOH 0.1 M to the CAM surface, bleeding was observed in the vessels. Thirty seconds later, vessels started to coagulation, which became severe over time. The average cumulative score of NaOH 0.1 M (score = 19.66), can confirm high irritation potential of NaOH to the ocular tissue. Thirty seconds after exposure of acetone to the CAM surface, mild injury to the blood vessels happened. It should be noted that bleeding after 2 minutes, and coagulation spots after 5 minutes, were detected on the CAM surface (score = 13.33). CAM capillaries revealed slight hyperemia 30 seconds after the instillation of propylene glycol (PG) and further increased over time (score = 4.33). Phosphate buffer administration as a negative control didn’t show any obvious vascular injury. The toxicity effect of the developed FLZ NE-ISG formulation was determined by applying the optimized formulation on the CAM surface and monitoring for 5 minutes. Applying nanoemulsion formulation of fluconazole on the CAM surface didn’t show any sign of irritant response or vascular damage (Figure 2). Our chosen formulation can be considered non-irritant and safe for ophthalmic administration because of the great accurateness of the HET-CAM test. Table 3 represents the average of the cumulative score of stimulation potential of each formulation.

Vascular responses of CAM surface at each time point following application of different samples: (a) NaOH solution (0.1 M); (b) acetone; (c) PG; (d) PBS; (e) FLZ NE-ISG formulation.
The average of the cumulative score in the HET-CAM test.

Draize test
After instillation of phosphate buffer, SDS 1% w/v, Blank NE-ISG formulation, and FLZ NE-ISG formulation, cornea, iris, and conjunctiva tissues of each rabbit were observed for any manifestation of irritation during 1, 2, 3, 4, 6, 8, and 24 h. The score of irritation potential was considered for each sample as shown in Table 4. Our findings were as follows: Phosphate buffer group didn’t show any sign of irritation in the cornea, iris, and conjunctiva tissue. SDS 1% w/v group showed severe ocular irritation such as discharge, redness, and chemosis during 1 hour of administration (Figure 3). Over time sign of discharge was removed, and then chemosis and redness were slightly diminished during 8 hours. After the instillation of blank NE-ISG formulation and FLZ NE-ISG formulation to the lower cul-de-sac of the conjunctiva, a local redness was detected in one rabbit removed during the following hours. Our data indicated that the designed FLZ NE-ISG formulation is non-irritant and well-tolerated.
Modified Draize irritation scores of NE formulation of fluconazole.


Irritation reaction of rabbit eyes to various samples: (a) PBS; (b) SDS; (c) Blank NE-ISG formulation; (d) FLZ NE-ISG formulation.
Discussion
Based on our results from MTT, HET-CAM, and Draize tests, FLZ NE-ISG formulation had not toxic effects on RPE cells and irritant effects on the ophthalmic tissue.
Various restrictions are associated with ocular drug delivery, such as low drug penetration, limited area of absorption, and elimination of instilled dose by blinking, lacrimation, and enzymatic metabolism. The mentioned limitations may result in poor ocular bioavailability, which can be managed by novel drug delivery vehicles. Nanoemulsions can be utilized as drug carriers in different formulations. Based on the droplet size of 200–600 nm, they can simply pass through the cornea. Specific biological stimulus such as ions, pH, or temperature are used to engineer in-situ gel systems with physicochemical assets of the transition of sol to gel. It should be noted that beside advantages such as improving ocular penetration, providing sustained release, decreasing systemic absorption, confining discomfort and blurred vision, and increasing residence time and bioavailability, these systems have disadvantages, including tissue irritation and toxicity induced by nanodroplet size and different excipients (surfactants and gelling agents).16–19 In a study, 20 it was shown that the use of fluconazole-loaded chitin nanogels (Flu-CNGs) could improve the bioavailability of this anti-fungal drug. The controlled release pattern of the prepared Flu-CNGs made it ideal for continuous drug delivery over time. Flu-CNGs showed no signs of toxicity or inflammation to the corneal cells.
Because the eye tissue is so sensitive, it is necessitated to evaluate the irritation and toxicity potential of new-designed formulations. Among various experimental tests for investigating the irritation and toxicity potential of optimized FLZ NE-ISG formulation, MTT, HET-CAM, and modified Draize test were used.
In this study, assessment of cell viability was performed by MTT test on RPE cells treated for 24 hours with the concentrations of 0.1, 0.5, and 1% of the selected FLZ NE-ISG formulation. It should be noticed that the MTT assay by reducing the tetrazolium to the formazan salt via mitochondrial activity, is a repeatable, rapid and reliable technique for the determination of cytotoxicity of various chemical substances. 21 Despite the positive points of this method, there are some limitations related to the MTT technique, including the cytotoxic potential of MTT reagents and affection of MTT reduction rate by culture conditions. 22 Our results confirmed that FLZ NE-ISG formulation in both concentrations of 0.1% and 0.5% is safe.
Developing optimal ocular formulations can be achieved by assessment of any irritation potential of formulation components on ophthalmic tissues. HET-CAM is one of the most applicable and simple tests for ocular irritation potential assessment of the pharmaceutical formulations. CAM includes blood vessels that branch out from the allantoic arteries and veins and acts as a respiratory membrane that covers the chick embryo. Conjunctival damages and toxicity potential can be predicted by changes in blood vessels.23,24 HET-CAM method is an alternative technique for animal-based methods because of rapid and visible responses to irritant chemicals. 25 Besides the advantages of similarity between the CAM blood vessels and vascularized mucosal tissue of the human and rabbit eyes, and the expansive area of the CAM surface, the irritancy response can be affected by particular factors such as the age of the eggs, the volume of the test substance, and exposure time of the CAM surface to the test substance.26,27 According to this method, the effect of the FLZ NE-ISG formulation on the corneal tolerability was evaluated. PBS (pH = 7.4) was engaged as a negative control, and PG, acetone, and NaOH 0.1 M were employed as positive controls, which can lead to Hyperemia, Hemorrhage, and clotting respectively on the blood vessels of CAM surface. Following using PBS, no irritation was detected for 5 minutes. In this study, a zero score of irritancy for the FLZ NE-ISG formulation was detected. In a recent study in 2019, the transparent in-situ gel microemulsion formulation of Sparfloxacin was optimized using 15% of Poloxamer 407 as a gelling agent. Based on previous data, ocular irritation induced by Transcutol P in the HET-CAM test indicated a slight irritation effect in the concentration of 0.05%. 28 The irritation study, based on the HET-CAM test didn’t show any sign of irritation for 3 minutes and a mild visible change after 5 minutes because of the alkaline pH of the formulation. 29
The Draize test can study the safety and ocular tolerability of the topical formulation of FLZ NE-ISG. Despite the negative points of the Draize test, such as the visual rating scale and affection by environmental factors, it is still the certified model to evaluate the irritation potential of pharmaceutical products. Draize was performed using rabbits because of rapid and clear reaction to external substances, wide-eye surface, and low cost. 30 As revealed in Figure 3, instillation of FLZ NE-ISG formulation in three rabbits’ eyes didn’t show conjunctival chemosis or discharge but slightly redness in one rabbit eye, at the first time of application. An irritation score of FLZ NE-ISG formulation didn’t exceed 2 in rabbits’ eyes at any time of the examination according to the modified Draize scores. Conjunctival redness slightly diminished and totally disappeared following 1 hour of instillation of FLZ NE-ISG formulation. According to our findings, and because the rabbit eye is more susceptible to irritation compared to human eyes, 31 it was concluded that FLZ NE-ISG formulation had not irritant effects on the ophthalmic tissue. The results are similar to our previous studies, which show that although a large quantity of surfactant mixture has been used to prepare FLZ NE-ISG formulation, this formulation is reasonably non-stimulant and can be administered for ocular drug delivery.32,33 Similar outcomes have been accomplished in earlier studies using Transcutol P and Poloxamers.34,35 In a study in 2020 with the aim of developing the in-situ gel nanoemulsion of acyclovir, assessment of the irritation potential was performed by HET-CAM and Draize test. In the Draize method, the average total score of 0.33 confirmed the safety of the formulation. 33 In 2011, the ocular irritation potential of in-situ gel nanoemulsion of Dorzolamide was assessed by the Draize method, which showed no irritant effects of the mentioned formulation on the rabbit eye tissue (score = 0.33). 36
In various studies in recent years, different fluconazole nanoparticles have been developed and optimized for eye disease. In one study, it was observed that the use of fluconazole niosomes revealed boosted corneal permeation, prolonged action, and enhanced anti-fungal activity. 37 Also, novel hyalugel-integrated liposomes of fluconazole meaningfully improved corneal permeability in comparison with conventional liposomes and fluconazole suspension. 38
Conclusion
Results obtained from three different methods verify that the developed FLZ NE-ISG formulation is non-toxic on RPE cells and non-irritant on the ophthalmic tissue. It appears that by developing an effective and safe formulation, FLZ NE-ISG formulation can be an applicable candidate to use topically for the treatment of fungal ophthalmic disease and may serve as an appropriate platform for improving ophthalmic drug delivery. However, further in vivo studies as well as therapeutic investigations, should be performed.
Footnotes
Acknowledgment
We would like to thank Mr Mohammad Tavakoli for his support in preparing figures.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The current study was financially supported (Grant No. 9803212158) by the Hamadan University of Medical Science, Hamadan, I.R. Iran.

