Abstract
Background
Cayratia albifolia C. L. Li (Jiao Mei Gu (JMG)) has potential therapeutic effects in inflammatory diseases.
Objectives
The primary goal of the research was to focus on the antioxidant and anti-inflammatory qualities of C. albifolia and the underlying mechanisms that contribute to these benefits.
Materials and Methods
To explore the anti-inflammatory abilities of JMG, we employed enzyme-linked immunosorbent assay and nitric oxide (NO) assay in a lipopolysaccharide (LPS)-induced macrophage inflammation model. Furthermore, flow cytometry was employed to assess the antioxidant effects of C. albifolia, and Western blotting was used to reveal the underlying mechanisms.
Results
Ethyl acetate extract of C. albifolia effectively reduced the release of NO, interleukin-6, tumor necrosis factor-α, and prostaglandin E2 in macrophages. Additionally, it prevented the expression of reactive oxygen species. Mechanistically, the extract may downregulate the expression of inducible nitric oxide synthase, cyclooxygenase-2, the nuclear factor-erythroid 2-related factor 2 (Nrf2), and heme oxygenase-1 (HO-1) protein expression.
Conclusion
The ethyl acetate extract of C. albifolia demonstrated remarkable anti-inflammatory and antioxidant effects by modulating the Nrf2/HO-1 axis.
Introduction
Inflammation is an immune defense response induced by the body in response to infection. Under pathological conditions, pathogens stimulate immune cells, generating inflammatory mediators including interleukin-6 (IL-6), tumor necrosis factor-α (TNF-α), and prostaglandin E2 (PGE2). This process also promotes the release of cyclooxygenase-2 (COX-2) and inducible nitric oxide synthase (iNOS) (Dinarello, 2010; Lopes Fischer et al., 2019). The acute inflammatory response normally protects the host from the threat of pathogenic infection and injury. However, when the inflammatory response develops to a certain stage, it causes damage to the organism (Dalli et al., 2017). Inflammation is closely linked to oxidative stress, often co-occurring with macrophage activation, which increases cellular oxygen consumption and results in an overproduction of ROS. This excessive reactive oxygen species (ROS) production, in turn, stimulates the formation of inflammatory mediators (Mittal et al., 2014). When inflammation and oxidative stress in the body form a vicious circle, if this process cannot be controlled, irreversible damage to the body and tissues can occur (Mittal et al., 2014). Macrophages, as the main coordinators of the inflammatory response, exert pro-inflammatory functions by binding to toll-like receptor 4 (TLR4), activating downstream MAP kinase and κB kinase (IKK) complexes, and producing inflammatory chemicals like IL-6, TNF-α, and nitric oxide (NO) (Oishi & Manabe, 2018; Rossol et al., 2011). Nowadays, the most commonly prescribed anti-inflammatory medications are still nonsteroidal anti-inflammatory drugs; however, the vast majority of anti-inflammatory drugs are susceptible to adverse effects such as gastrointestinal, hepatic, or renal disturbances (Bindu et al., 2020). Therefore, a major issue in treating inflammatory-related disorders today is the creation of more powerful anti-inflammatory medications with fewer harmful side effects (Dinarello, 2010; Eghtedari et al., 2021).
Lipopolysaccharide (LPS) is recognized as a typical inflammatory inducer that activates macrophages (Ru et al., 2016). In this inflammation model, LPS can stimulate downstream signaling pathways of RAW264.7 cells, such as nuclear factor-κB (NF-κB) and nuclear factor-erythroid 2-related factor 2/heme oxygenase-1 (Nrf2/HO-1) pathways, which control the inflammatory response (Li et al., 2016).
Botanists have recognized the vine plant Jiao Mei Gu (Dong medicine; JMG) as Cayratia albifolia C. L. Li. It is primarily grown in the Dong region of southwest China (Figure 1A). In folklore, C. albifolia is mostly used for the treatment of rheumatic lumbago, rheumatoid arthritis, urinary tract infections, septic infections, and viral upper respiratory tract infections (Gong et al., 2009). Pharmacological studies have revealed that C. albifolia contains chemical constituents such as flavonoids, steroids, lipids, terpenoids, and volatile oils (Cui et al., 2012). Earlier studies have demonstrated that aqueous extracts of JMG could inhibit inflammatory responses by modulating NF-κB and inhibiting the expression of cluster of differentiation 86 (CD86) in macrophages and the level of membrane potential (Li et al., 2022; Luo et al., 2020). Therefore, the anti-inflammatory activity of JMG has attracted much attention.
The Leaf, Fruit (A), and Roots (B) of Cayratia albifolia C. L. Li.

However, the active fraction of Dong medicine JMG still needs investigation. Our preliminary study showed that the ethyl acetate extract of JMG exerted greater bioactivity compared to other fractions. The main innovation of this study is to elucidate for the first time the anti-inflammatory and antioxidant capabilities of ethyl acetate alcoholic extract of JMG and to explore the underlying mechanisms. The results might provide a strong basis for additional study and the development of this Dong medicine.
Materials and Methods
Preparations for the Alcoholic Extracts of the Plant Material
The plants of C. albifolia (JMG) were collected at the Key Laboratory of Dong Medicine of Hunan Province. The laboratory verified the veracity of the plants using lot no. 20170923. The roots of JMG (Figure 1B) were macerated in 75% ethanol at 60°C. It was condensed at lower pressure with a rotary evaporator to obtain a dried extract. The extract was then extracted using the organic solvent ethyl acetate to obtain an ethyl acetate extract. It was then frozen into powder form using a freeze dryer at –70°C and kept for subsequent use at 4°C.
Cell Culture
The RAW 264.7 line of cells (cat: CL0190) was acquired from Procell (Wuhan, China). The cells were cultivated at 37°C and 5% CO2 humidity after being inoculated in dishes with media designed specifically for RAW264.7 cells. Before starting each experiment, the culture medium in each well was entirely replaced with fresh medium, with or without ethyl acetate extract of JMG.
Cell Viability Assay
The impact of ethyl acetate extract of JMG on cell viability was estimated by the MTT kit (Beijing Solarbio, Beijing, China). The cells were seeded at a density of 5 × 103 cells/well in 96-well plates, and the cells were grown adherent to the wall overnight. Cells were incubated with ethyl acetate extract of JMG at different concentrations (270, 90, 30, 10, 3.3 µg/mL) for 24 h. The supernatant was cautiously aspirated following incubation. Each well received 110 µL of Formazan liquid, and the cells were swung for 10 min at a slow speed on a shaking table. Ultimately in the zymography instrument, the absorbance levels were detected at 490 nm.
Cell Morphology Observation
Cells were planted onto 12-well plates at 4 × 105 cells/well, followed by 1-h pretreatment with dexamethasone (DEX, 10 µg/mL) or ethyl acetate extract of JMG, and LPS (1 µg/mL) was added for 18 h. Then, the morphological change in the shape of the cells was noticed and photographed by Axio Vert.A1 inverted fluorescence microscope.
Nitrite Assay
After plating RAW264.7 cells on 12-well plates, they were exposed to ethyl acetate extract of JMG (3.3, 10, 30 µg/mL) for 1 h, and then stimulated for 24 h with LPS (1 µg/mL) to induce an inflammation model. NO concentration was determined using a Griess kit from Shanghai Beyotime (Shanghai, China). Absorbance values were detected with an enzyme meter at 540 nm, and NO concentrations were calculated from the standards.
Enzyme-linked Immunosorbent Assay (ELISA)
Cells were inoculated in 24-well plates, and ethyl acetate extract of JMG (3.3, 10, 30 µg/mL) or DEX (10 µg/mL) was pretreated for 1 h. After activation with LPS (1 µg/mL) for 24 h, supernatants from cells were gathered, and PGE2, IL-6, and TNF-α levels in cell supernatant fluid were detected through the appropriate ELISA kits in accordance with the suggestions (MultiSciences Biotech, Huangzhou, China).
Flow Cytometry
Cells were cultivated in 12-well plates at 4 × 105 cells/well and incubated for 24 h. Cells were first treated with ethyl acetate extract of JMG (3.3, 10, 30 µg/mL) or DEX (10 µg/mL) for 1 h and subsequently cultured for 6 h with 1 µg/mL LPS. Cells were cultured with 10 µM of 2′,7′-dichlorodihydrofluorescein diacetate (DCFH-DA) for 30 min in an incubator at 37°C. Serum-free media for cell culture was used three times to wash the cells. Ultimately, the cells were collected after resuspension with phosphate-buffered saline (PBS), and dihydrodichlorofluorescein (DCF) fluorescence was detected on a CytoFlex flow cytometer (USA). The software FlowJo was used to analyze the data.
Western Blot
The cells were inoculated in 6-well plates at 10 × 105 cells/well and grown for 24 h. After 1 h of pretreatment with ethyl acetate extract of JMG (3.3, 10, 30 µg/mL), LPS (1 µg/mL) was given for 18 h of stimulation. Cells received washing using PBS, cells were lysed in RIPA lysate, and cell supernatants containing total protein were pipetted into new tubes after centrifuging for 20 min at 12,000 rpm and 4°C. The BCA approach was used to quantify the level of protein. The protein (30 µg) of each sample was segregated by 8% or 12% sodium dodecyl sulfate-polyacrylamide gel electrophoresis and then moved to polyvinylidene difluoride membrane. Nonspecific binding was reduced by sealing with milk containing 5% skimmed milk for 1 h. After the addition of COX-2, iNOS, HO-1, Nrf2, glyceraldehyde-3-phosphate dehydrogenase (GAPDH), and β-actin primary antibodies, the membranes were incubated at 4°C for a whole night. The following day, the membrane was cleaned three times and cultured with the corresponding secondary antibodies for 1 h at room temperature. Finally, a ChemiDoc XRS+ enhanced chemiluminescence detection system was employed to identify the immunoreactive bands. Additionally, ImageJ software was utilized to quantify the target protein expression degree.
Statistical Analysis
All the data were statistically analyzed by GraphPad Prism 9 software (La Jolla, CA, USA), and all experimental data were described as mean ± standard error (SEM). Comparisons between several groups were evaluated by one-way analysis of variance (ANOVA), with p < 0.05 regarded as statistically significant.
Results
Effect of Ethyl Acetate Extract of JMG on Cell Viability
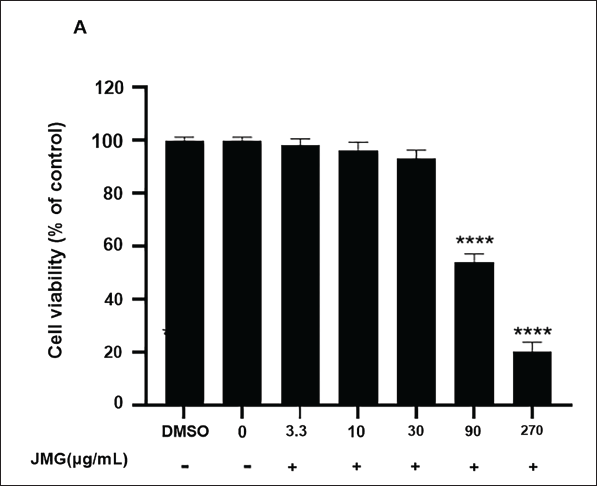
MTT results indicated that dimethyl sulfoxide (DMSO) had no influence on cell viability (p > 0.05). Ethyl acetate extract of JMG concentration below 30 µg/mL showed no toxic effect on cells in contrast to the 0 µg/mL JMG group (p > 0.05, Figure 2A). When the concentration of ethyl acetate extract of JMG reached 90 µg/mL, there was a significant inhibitory effect on cell viability (p < 0.0001). Therefore, ethyl acetate extract of JMG at concentrations of 3.3–30 µg/mL was selected for the anti-inflammatory activity test on RAW264.7 cells activated with LPS.
Effect of Ethyl Acetate Extract of JMG on Morphological Changes in Cells
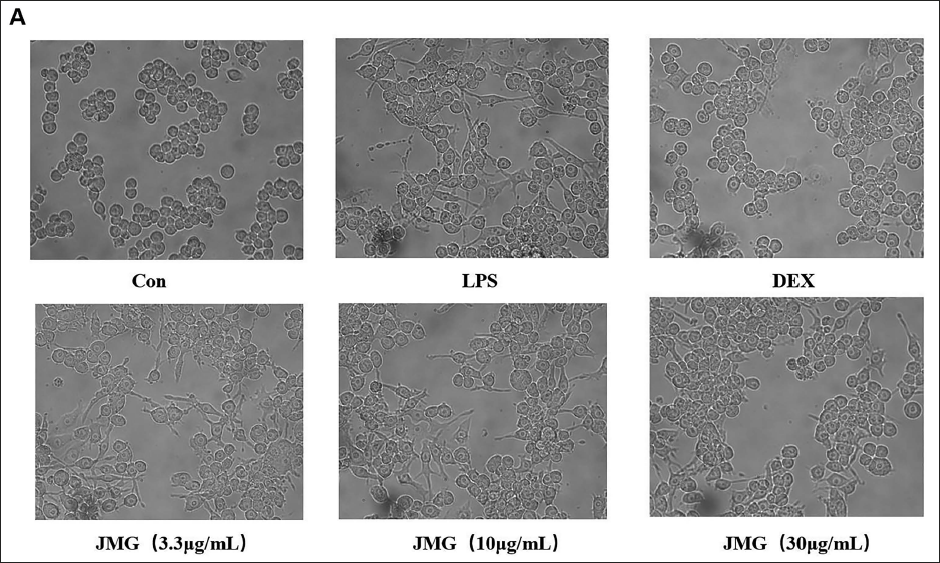
As shown in Figure 3A, the normal control cells were round with clearly visible nuclei and cytoplasm. After 18 h of LPS stimulation, the cells appeared to be differentiated, with many tentacles and blurred nuclei and cytoplasm. However, cells pretreated with different concentrations of ethyl acetate extract of JMG (3.3, 10, 30 µg/mL) and DEX (10 µg/mL) showed a decrease in the level of cell differentiation. Among them, the degree of cell differentiation was significantly reduced with 30 µg/mL JMG to treat, which was close to the DEX control group (Figure 3A).
Ethyl Acetate Extract of JMG Suppressed LPS-induced NO and PGE2 Production in RAW264.7 Cells
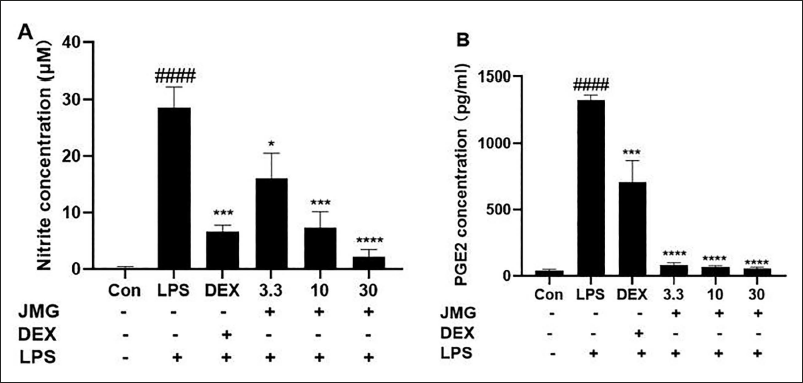
The level of nitrite (a long-lasting oxidation result of NO) in the cell culture medium was considerably increased after LPS stimulation for 24 h (p < 0.0001, Figure 4A). Pretreatment with ethyl acetate extract of JMG considerably decreased LPS-stimulated NO production in a dose-dependent approach (p < 0.05, p < 0.001, p < 0.0001, Figure 4A). The level of PGE2 in the cells supernatant was also extensively increased (p < 0.0001, Figure 4B) after LPS stimulation for 24 h. The JMG-treated reduced the PGE2 levels in contrast to the LPS-induced model group (p < 0.0001, Figure 4B).
Ethyl Acetate Extract of JMG Decreases the Level of Inflammatory Elements in LPS-induced RAW264.7 Cells
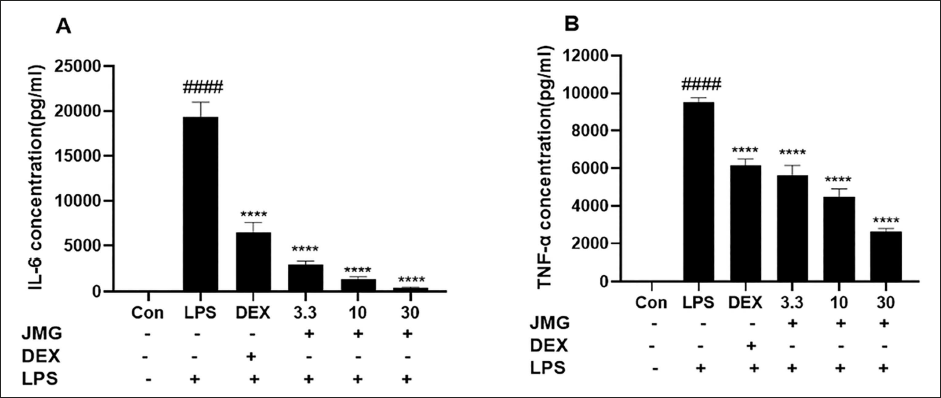
Next, we examined the impact of ethyl acetate extract of JMG on the release levels of IL-6 and TNF-α in LPS-stimulated RAW264.7 cells. After being stimulated by LPS for 24 h, the release of IL-6 and TNF-α in the RAW264.7 cell culture medium was measured by ELISA in the presence or absence of JMG pretreatment. The levels of IL-6 (p < 0.0001) and TNF-α (p < 0.0001) in the culture medium were significantly higher than the blank control group (Figure 5A, B). In contrast to the LPS group, the level of IL-6 and TNF-α in the cell supernatants of 30 µg/mL JMG treatment group decreased by 97.8% and 72.4%, respectively (Figure 5A and B).
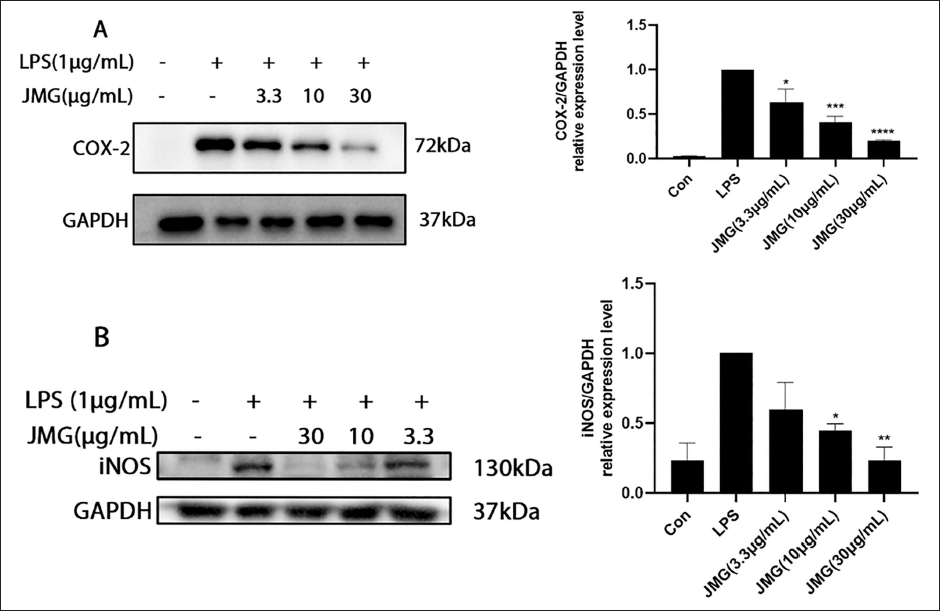
Ethyl Acetate Extract of JMG Reduced the Production of iNOS and COX-2 in LPS-induced Macrophages
The production of the COX-2 and iNOS proteins in the RAW264.7 cells was markedly elevated after LPS (1 µg/mL) stimulation for 18 h. In contrast to the LPS group, the 3.3, 10, 30 µg/mL JMG group downregulated COX-2 (p < 0.05, p < 0.001, p < 0.0001) and iNOS (p > 0.05, p < 0.05, p < 0.01) protein degrees of production in LPS-induced macrophages in a concentration-dependent approach. Under the same conditions, compared with the LPS group, the release of COX-2 in the 3.3, 10, and 30 µg/mL ethyl acetate extract of JMG-treated group decreased by 36.4%, 59%, and 80.3%, respectively, and the level of iNOS in the 3.3, 10, and 30 µg/mL ethyl acetate extract of JMG-treated group decreased by 40.6%, 55.1%, and 76.6%, respectively (Figure 6A and B).
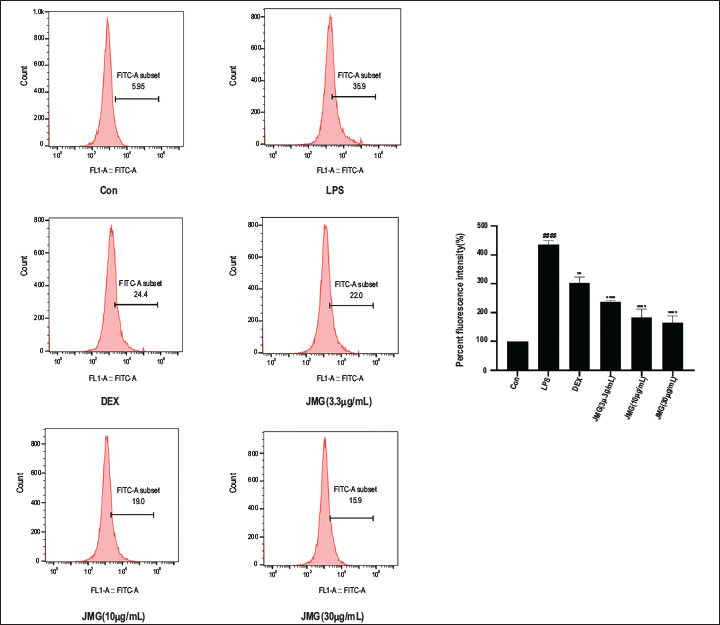
Ethyl Acetate Extract of JMG Inhibited LPS-stimulated ROS Expression in RAW264.7 Cells
Flow cytometry was employed to verify the inhibitory function of ethyl acetate extract of JMG on LPS-induced oxidative stress. The LPS group showed considerably higher ROS levels in contrast to the blank control (p < 0.0001, Figure 7A). The ROS levels of 3.3, 10, 30 µg/mL JMG groups and DEX control group were greatly lower than the LPS group and the inhibitory effect was most pronounced in the high-dose JMG group (p < 0.0001, Figure 7A). In summary, ethyl acetate extract of JMG dose-dependently inhibited the production of ROS and showed excellent antioxidant effects.
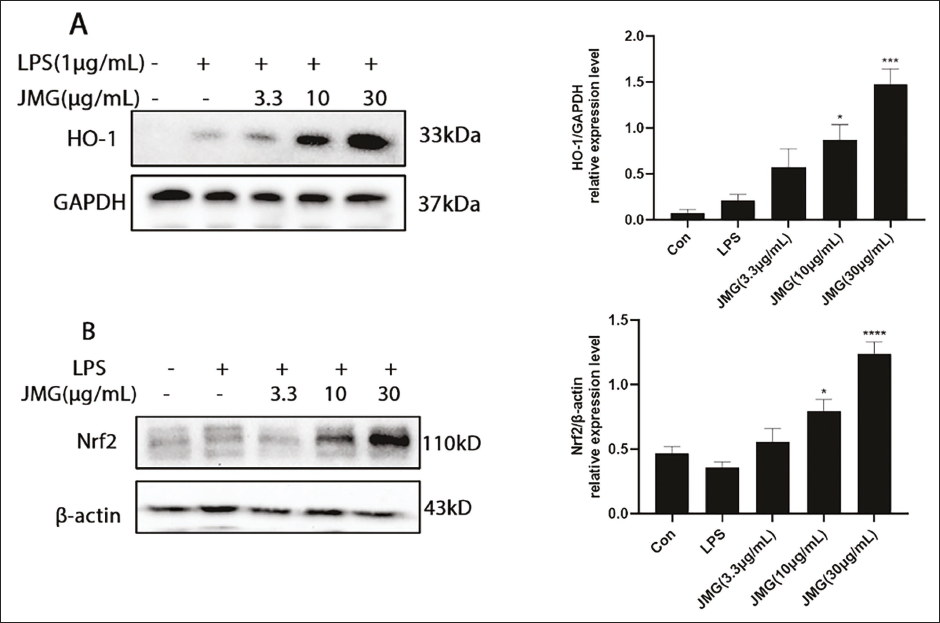
Effect of Ethyl Acetate Extract of JMG on Nrf2/HO-1 Protein Expression
As essential proteins against oxidative stress, Nrf2 and HO-1 are crucial for cell protection. In Figure 8A and B, in contrast to the LPS group, 3.3, 10, and 30 µg/mL ethyl acetate extract of JMG treatment increased HO-1 expression by 167.5%, 308.5%, and 593.2%, respectively, and increased Nrf2 expression by 55.3%, 122.5%, and 247.8%, respectively. As shown in Figure 8A and B, the 30 µg/mL JMG group significantly upregulated the expression of HO-1 (p < 0.001) and Nrf2 (p < 0.0001) in contrast to the LPS group.
Discussion
JMG has been used for an extremely long time to treat inflammatory conditions as a traditional ethnomedicine grown in the Dong ethnic area. It was found that the main active components separated from the ethyl acetate extract of JMG were β-sitosterol, 3,4-dihydroxybenzoic acid, and resveratrol (Peng et al., 2022). Among them, β-sitosterol, a plant-derived nutrient, has been revealed in modern pharmacology to have anti-inflammatory and significant antioxidant activity (Khan et al., 2022). It has been found that β-sitosterol reduced the percentage of foot swelling in rats, decreased the number of neutrophils in pleurisy as well as the volume of pleural effusion in rats, and inhibited the inflammatory response to ear edema in mice (Paniagua-Pérez et al., 2017). Second, β-sitosterol induced an anti-inflammatory phenotype by affecting macrophage polarization and has been shown to attenuate rheumatoid inflammation in rats (Liu et al., 2019). In addition to this, some studies have reported that β-sitosterol showed strong oxygen radical scavenging ability and exhibited significant antioxidant potential (Bhat et al., 2019). In addition, it has been reported that 3,4-dihydroxybenzoic acid reduced adrenaline-stimulated production of ROS for the treatment of heart failure (Bai et al., 2023). Resveratrol, as a polyphenol compound, can effectively scavenge oxygen free radicals by regulating mitochondrial dysfunction (Huo et al., 2021). Therefore, based on previous studies, it is quite meaningful to research the anti-inflammatory and antioxidant activities of JMG as a herbal medicine containing a variety of anti-inflammatory and antioxidant compounds. In addition, our study is consistent with previous studies. In the current research, it was also illustrated that the alcoholic extract of JMG ethyl acetate has significant anti-inflammatory as well as antioxidant effects by decreasing the expression of oxygen free radicals.
The main features of inflammation are the extravascular fluid and leukocyte exudation and the production of inflammatory mediators, resulting in symptoms such as redness, swelling, pain, and fever (Arulselvan et al., 2016). Currently, research has demonstrated that the development of inflammation is tightly associated with cancer and that cellular mutations lead to sustained high expression of inflammatory mediators in normal tissues, thereby inducing cancer (Guo et al., 2017). Macrophages are extensively dispersed in human tissues and have a crucial role in coping with inflammation-related diseases as well as cancer (Shapouri-Moghaddam et al., 2018). In the inflammatory response, the inflammatory proteins iNOS and COX-2 can induce NO and PGE2 production (Lee et al., 2012, 2017). NO plays an essential part in inflammation by expressing iNOS and enhancing the production of inflammatory molecules such as IL-6, TNF-α, and IL-1β from macrophages, and also mediates cellular senescence together with ROS (Kim et al., 2020; Zhao et al., 2021). TNF-α and IL-6, as important pro-inflammatory factors, could promote the release of inflammatory mediators from inflammatory cells and are positively correlated with the inflammatory response (Liang et al., 2018). Therefore, inhibiting the expression of these inflammatory factors may alleviate the inflammatory reaction. To verify the anti-inflammatory activity of the ethyl acetate extract of JMG in this research, we assayed multiple inflammatory mediators. Our study confirmed the inhibitory effect of the above inflammatory factors. The outcomes indicated that the ethyl acetate extract of JMG strongly inhibited LPS-induced expression of inflammatory factors including NO, TNF-α, PGE2, and IL-6. Additionally, the ethyl acetate extract of JMG significantly inhibited the degree of expressiveness of COX-2 and iNOS protein production in RAW264.7 cells. In particular, high-dose ethyl acetate extract of JMG significantly reduced the release of COX-2 and iNOS by 80.3% and 76.6%, respectively.
Oxidative stress is caused by a disequilibrium between the amount of ROS and antioxidants produced by the body. When the body is under prolonged stress, it triggers inflammation by activating signaling cascades and related transcription factors (Dharshini et al., 2022). ROS, as the most potent inflammatory mediator, play a crucial role in oxidative stress (El-Benna et al., 2016). The release of ROS is a result of increased biological demand for oxygen in the damaged areas of inflammation (Arulselvan et al., 2016). Oxidative stress-related inflammation has also been demonstrated to be impacted by high expression of COX-2 and iNOS, and inflammatory markers IL-6 and TNF-α (Federico et al., 2007). Therefore, the detection of the expression of inflammatory factors, inflammatory mediators, and ROS is of great interest for the comprehensive evaluation of the anti-inflammatory function of ethyl acetate extract of JMG. Therefore, in this study, we first used flow cytometry to verify that LPS induced RAW264.7 cells to express a large amount of ROS, while ethyl acetate extract of JMG reduced LPS-induced ROS expression in the cells. In addition, HO-1 and Nrf2 suppress the generation of inflammatory proteins in the inflammatory process and are regarded as essential anti-inflammation and antioxidant regulatory factors (Loboda et al., 2016). When ROS is overexpressed, it stimulates the dissociation of Nrf2/Keap 1 heterodimer and promotes the nuclear translocation of Nrf2 while activating the downstream production of HO-1 to exert antioxidant effects (Yu et al., 2017). Thus, the Nrf2/HO-1 pathway performs a crucial part in limiting ROS expression, macrophage activation, and the release of inflammatory factors (Mansouri et al., 2022; Sauler et al., 2015; Zhang et al., 2019). In this study, after treating the cells with different concentrations of ethyl acetate extract of JMG for 18 h, JMG increased the release of Nrf2/HO-1 proteins and even exhibited antioxidant and anti-inflammatory effects. Taken together, the results demonstrated that the alcoholic extract of JMG ethyl acetate inhibited ROS release, activated HO-1 and Nrf2 protein expression, and possessed significant antioxidant effects.
Although the ethyl acetate extract of JMG inhibited inflammatory response by activation of the Nrf2/HO-1 axis, the current research involved only in vitro experiments and could not provide a comprehensive response to the overall role of ethyl acetate extract of JMG in the inflammatory system. Because ethyl acetate extract of JMG is a mixture, its anti-inflammatory mechanisms are complex.
Conclusion
In conclusion, the present research is the initial one to describe the anti-inflammatory and antioxidant properties of ethyl acetate extracts of Dong medicine JMG on LPS-stimulated RAW264.7 cells. The study verified a significant anti-inflammatory effect of ethyl acetate extract of JMG via regulating the levels of expressiveness of Nrf2 and HO-1. Our results may provide a solid basis to continue the study and advancement of C. albifolia.
Abbreviations
C. albifolia: Cayratia albifolia C. L Li; LPS: Lipopolysaccharide; NO: Nitric oxide; IL-6: Interleukin-6; TNF-α: Tumor necrosis factor-α; PGE2: Prostaglandin E2; ROS: Reactive oxygen species; DCFH-DA: 2′,7′-Dichlorodihydrofluorescein diacetate; iNOS: Inducible nitric oxide synthase; COX-2: Cyclooxygenase-2; Nrf2: Nuclear factor-erythroid 2-related factor 2; HO-1: Heme oxygenase-1; ELISA: Enzyme-linked immunosorbent assay; TLR4: Toll-like receptor 4; NF-κB: Nuclear factor-κB; DEX: Dexamethasone; DMSO: Dimethyl sulfoxide.
Author Contributions
Yi Tan completed the experiments and wrote the manuscript; Yi Tan, Liang Cao, Dan Zhou, Biao Qin, Yanbin Zhang, Jianxin Liu, Kai He, Bin Li, and Zaiqi Zhang developed and scheduled the research; Liang Cao was responsible for providing JMG plants; Zaiqi Zhang was in charge of providing financial assistance. Every author has reviewed and approved the final paper before submission.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This research was approved by the Research Ethics Committee (REC) at Hunan University of Medicine (Approval number: 2024 A03027).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was funded by the Open Project of the Hunan Provincial Key Laboratory of Dong Medicine Research (Grant Number YS2019YFE012440).
