Abstract
Introduction
Macrophages are capable of exerting both proinflammatory and anti-inflammatory functions in response to distinct environmental stimuli, by polarizing into classically inflammatory state (M1) and anti-inflammatory phenotype (M2), respectively. The Wnt/β-catenin signaling plays an important role in the tissue homeostasis and immune regulations, including the macrophage polarizations. However, the molecular mechanism of Wnt/β-catenin signaling in regulating alveolar macrophage polarization in an inflammatory state remains unclear.
Methods
The Wnt/β-catenin signaling-altered phenotypes of murine macrophage-like RAW264.7 cells
Results
The activation of Wnt/β-catenin signaling inhibited macrophage M1 polarization, but promoted alternative M2 polarization in murine RAW264.7 cells under a naïve state. Interestingly, in an LPS-induced inflammation condition, the enhanced Wnt/β-catenin activity suppressed both M1 and M2 polarizations in RAW264.7 cells
Conclusion
These results suggest a mechanism by which Wnt/β-catenin signaling modulates macrophage polarization in an inflammation state by regulating the Stat signaling pathway.
Introduction
Macrophages exert significant functions in maintenance of tissue immune homeostasis, 1 owning to their plasticity and versatility in response to insults in local microenvironment of tissues. 2 Phenotypically, macrophages can be grouped into classically pro-inflammatory activation of M1 phenotype (M1 polarization), and alternatively anti-inflammatory M2 phenotype (M2 polarization) in response to various stimuli. For instance, lipopolysaccharide (LPS) and/or IFN-γ are able to induce macrophage M1 polarization, while IL-4 promotes M2 polarization.3,4 These macrophage subtypes can be identified by their special cell surface markers and secretory factors. 5 Functionally, M1 macrophages exert their immune defensing roles by producing nitric oxide (NO) via inducible nitric oxide synthase (iNOS) and pro-inflammatory cytokines, including interleukin (IL)-1β, IL-6, IL-12, and tumor necrosis factor-α (TNF-α).6–8 On the contrary, M2 macrophages express arginase1 (Arg1) and CD206, and secrete a mass of anti-inflammatory factors including IL-10, TGF-β1, IL-4, and IL-13.8,9 In addition, M2 macrophages can be further divided into three subtypes, M2a (induced by IL-4 or IL-13), M2b (activated by IL-1 receptor ligands or immune complexes (IC) plus LPS), and M2c (activated by IL-10, TGF-β1, and glucocorticoid hormones), in response to distinct stimuli.9,10 Moreover, macrophage phenotypes can be identified by their expression of specific markers, such as CD80 and CD86 for M1 macrophages, and arginase1 (Arg1) and CD206 for M2 macrophages.
Apart from cytokines and transcript factors, accumulating evidences suggest that developmental signaling pathways, such as the wingless-type MMTV integration site (Wnt) signaling are also involved in the regulation of macrophage polarizations,2,11–13 despite its underlying mechanism remains largely unknown. Based on the dependence of β-catenin, the Wnt signaling pathway can be classified into canonical Wnt/β-catenin signaling and non-canonical Wnt pathway. 14 However, its regulatory role and mechanism in macrophage polarization in response to an inflammatory stimulation remain unknown. A dysregulation of Wnt/β-catenin signaling can alter the microenvironmental inflammations, which ultimately leads to several types of diseases, including cancer, and the idiopathic pulmonary fibrosis (IPF).15,16
In views of above studies, we thus hypothesized that Wnt/β-catenin signaling may play roles in macrophage polarization in response to an inflammatory condition. In this study, we showed that the Wnt signaling ligand Wnt3a activated Wnt/β-catenin signaling, promoted M1 polarization but inhibited M2 polarization in mouse macrophage-like RAW264.7 cells at a naïve state. By contrast, under a lipopolysaccharide (LPS)-induced inflammatory state, Wnt/β-catenin inhibited both M1 and M2 polarization of RAW264.7 cells
Materials and Methods
Cell culture and Wnt3a conditional medium
The murine macrophage-like cell line RAW264.7 cells (ATCC# TIB-71) were purchased from American Type Culture Collection (ATCC) (Manassas, VA, USA) and cultured in DMEM supplemented with in 10% newborn calf serum (NBCS, Thermofisher, Shanghai, China) and 1% penicillin/streptomycin at 37°C in a humidified atmosphere of 5% CO2. Both the Wnt3a producing cell line, L-cell Wnt3a (overexpressing mouse Wnt3a, ATCC# CRL-2814) and its parent L cells (ATCC# CRL-2648) were purchased from ATCC and cultured in DMEM supplemented with 10% heat-inactivated fetal bovine serum (FBS, Biological industries, Cromwell, CT, USA) and 1% penicillin/streptomycin at 37°C in a humidified atmosphere of 5% CO2. Wnt3a and control L cells (80%–90% confluency) were refreshed with 5% NBCS DMEM and cultured for additional 24 h. The culture supernatant of both Wnt3a and L cell lines were collected for the preparation of Wnt3a-conditional media (Wnt3a-CM or Wnt3a) and control media (Ctrl-CM), respectively. 17
Treatments of RAW264.7 cells
The RAW264.7 cells (1 × 106 cells/well) were seeded in 6-Well plates and cultured overnight prior being treated with following conditions: Ctrl-CM, Wnt3a-CM, Wnt3a-CM plus 1 μ
Immunoblotting analysis
The total protein of whole cells was extracted lysis buffer containing protease and phosphatase inhibitors (KeyGen Biotech. Co. Ltd, Nanjing, China) for 40 min on ice. Total protein lysates of mouse lung tissues were homogenized using Enhanced RIPA Lysis Buffer (Keagene Biotech Ltd, Beijing, China). The lysates were then centrifuged at 12,000 rpm for 20 min at 4°C, and the resulted supernatant was collected as whole cell extracts. The Pierce BCA Protein Assay Kit (Keygen Biotech, Nanjing, China) was employed to detect the concentration of total proteins. 40 μg of total proteins were resolved and separated in 8–10% sodium dodecyl sulfate-(SDS-) polyacrylamide gel (SDS-PAGE), followed by being transferred to a polyvinylidene fluoride (PVDF) membranes (Millipore, Billerica, MA, USA). After being blocked with 5% fat-free milk in 1× Tris-buffered saline (TBS) buffer for 1 h at RT, the membrane was probed with primary antibodies to protein of interest overnight at 4°C. Following washing four times with 0.1% Tween-20 in TBS, the membrane was incubated with appropriate horseradish peroxidase (HRP)-conjugated secondary antibodies for 2 h at RT. Finally, immune blots were developed to enhance chemiluminescence (ECL) reagent (Advansta, Menlo Park, CA, USA). Relative expression of proteins was calculated using Image J Software version 1.46 (https://rsb.info.nih.gov/ij/). The net intensity of each blot was measured and normalized by the β-actin internal control, and was marked as densitometric arbitrary units (A.U.). Fold changes of proteins of interest were calculated using the intensity of blot of protein of interest in experimental group over the controls. 17 All of the experiments were performed at least three times. The primary antibodies used for immunoblotting analysis were listed in Supplementary Table 1.
Quantitative reverse transcription PCR (qRT-PCR)
The cells were lysed directly with TRIzol Reagent (Invitrogen, USA), the total RNAs were then reversed transcription (RT) into cDNA with the PrimeScript RT kit (DRR047 A, Takara, Japan) per manufacturers’ instructions. The assay was performed using SYBR® Premix Ex Taq TM (Takara Bio, Japan) in a LightCycler 480 II PCR system (Roche Diagnostics, Switzerland).
ELISA for cytokine assay
To determine the concentration of TNF-a, IL-6, IL-10, and TGF-β, the culture media of RAW264.7 cells were collected and centrifuged at 1000
LPS challenge and adenoviral vector delivery in mouse lungs
Animal experiments comply with the ARRIVE guideline. Experiments involved in mice were approved by the Institutional Animal Care and Use Committee of Ningxia University (NXULS20180123-3), and the ethics committee for the use of animals of Ningxia University. C57/BL6 mice with 6–8 weeks old and body weights of 18–20 g were used in this study (Vital River company, Beijing, China) and bred in a specific pathogen-free (SPF) environment conditions under 12 h light/12 dark cycles with
To investigate the role of Wnt signaling in regulation of inflammation-induced alveolar macrophages
Immunofluorescent (IF) staining
To assess the polarization of primary alveolar macrophages in mice, cells collected from the BALF were mounted on cover slides for determining M1 and M2 macrophage phenotypes by immunofluorescent co-localization staining. The slides were fixed with 4% paraformaldehyde (PFA) at RT for 20 min. Then the fixed tissue on slides was washed with PBS and treated with 0.2% Triton X-100 in PBS for 30 min at RT to permeabilize cell membranes, followed by being incubated with 5% normal donkey serum in Blocking Buffer (Thermos scientific, Germany) in PBS for 30 min to block non-specific binding. The slides were then probed with primary antibody to macrophages marker F4/80 (1:200, Caltag laboratories), M1 marker iNOS (1:100, #13120, Cell Signaling Technology), or M2 marker CD206 (1:200, 18,704-1-AP, Proteintech) in 5% normal donkey serum in Blocking Buffer at 4°C overnight. The slides were washed twice with PBS, subsequently incubated with a fluorescent-labeled secondary antibody at RT for 2 h. Nuclei were counterstained with DAPI (H-1200, Vector Laboratories, Burlingame, CA). Images were examined and photographed with Leica TCS SP2 A0BS Confocal System (Leica, Germany) or Olympus IX73(Olympus, Japan). The image was analyzed using Leica Confocal Software v.2.6.1 or Olympus cellSens Entry 2.3.
Flow cytometry assay
In order to detect the frequency of macrophage phenotype in RAW264.7 cells treated in various conditions, the cells were fixed with fixation buffer (BioLegend, Cat#420801) in the dark for 30 min at 4°C and washed twice with pre-cold PBS. The cellular membranes were permeabilized using Intracellular Staining Permeabilization Wash Buffer (BioLegend, Cat#421002) for 20 min at 4°C. Then, the cells were washed twice with pre-cold PBS, followed by being incubated with TruStain FcX™ (BioLegend, Cat#101319) at 1.0 μg per 1 × 106 cells in a volume of 100 μL for 10 min at 4°C to prevent non-specific binding of immunoglobulin to Fc receptors. The cells were subsequentially probed with fluorescently labeled antibodies, PE anti-mouse CD86 (1:200, BioLegend, Cat#105007) and FITC anti-mouse CD-206 antibody (1:1000, BioLegend, Cat#141703) for overnight at 4°C, followed by washing with cold PBS. Flow cytometry (BD LSR, USA) assay was used to assess the frequency of polarization of RAW264.7 cells toward the M1 and M2 phenotypes. Cytometric data were analyzed using FlowJo software (TreeStar, Inc., USA).
Statistical analysis
In this study, all experiments were performed for at least three independent biological repeats. Data are presented as mean ± standard deviation (SD) and were assessed using PRISM 6 (GraphPad Software, La Jolla, CA, USA). One-way ANOVA and
Results
LPS enhances Wnt/β-catenin signaling activity in RAW264.7 cells
Several lines of study suggested that Wnt/β-catenin signaling was able to regulate macrophage polarizations.
13
In order to evaluate the effect of Wnt/β-catenin activation on M2 polarization of RAW264.7 cells upon an inflammatory stimulation, the cells were treated with Wnt3a-CM and XAV939 (which directly inhibit Wnt/β-catenin signing by inhibiting tankyrase (TNKS1/2)) in the presence of LPS for 24 h. The cells were collected for assessing the expression of Wnt/β-catenin-associated proteins by immunoblotting assay. Since Wnt3a was one of the classic ligands to activate β-catenin-mediated canonical Wnt signaling pathway, Wnt3a-CM was employed for Wnt signaling activation LPS activates Wnt/β-catenin signaling in RAW264.7 cells. The RAW264.7 cells were treated with 100 ng/mL of LPS in medium of Wnt3a-CM and/or TNK1/2 inhibitor XAV939 (1 μ
Wnt/β-catenin signaling inhibits LPS-induced macrophage M2 phenotype in RAW274.7 cells
To investigate the underlying mechanism of Wnt/β-catenin signaling in macrophage polarization upon an LPS stimulation, the macrophage polarization markers in RAW264.7 cells were evaluated. The results showed that Wnt3a-CM alone could induce the expression of Arg1 (an M2 marker) and inhibit iNOS (an M1 marker), in comparison with the Ctrl-CM group. Whereas the addition of LPS (Wnt3a-CM plus LPS) led an inhibition of the expression of both Arg1 and iNOS in cells cultured in Wnt3a-CM, in comparison with that of Ctrl-CM-treated cells (Ctrl-CM plus LPS). By contrast, the LPS significantly increased Arg1 expression, but only moderately increased the iNOS expression in cells treated with XAV939 (Figure 2(a) and (b)). Of note, a margin reduction of Wnt/β-catenin signaling inhibits LPS-induced macrophage M2 polarization in RAW264.7 cells. RAW264.7 cells were treated with Wnt3a-CM and/or TNK1/2 inhibitor XAV939 (1 μ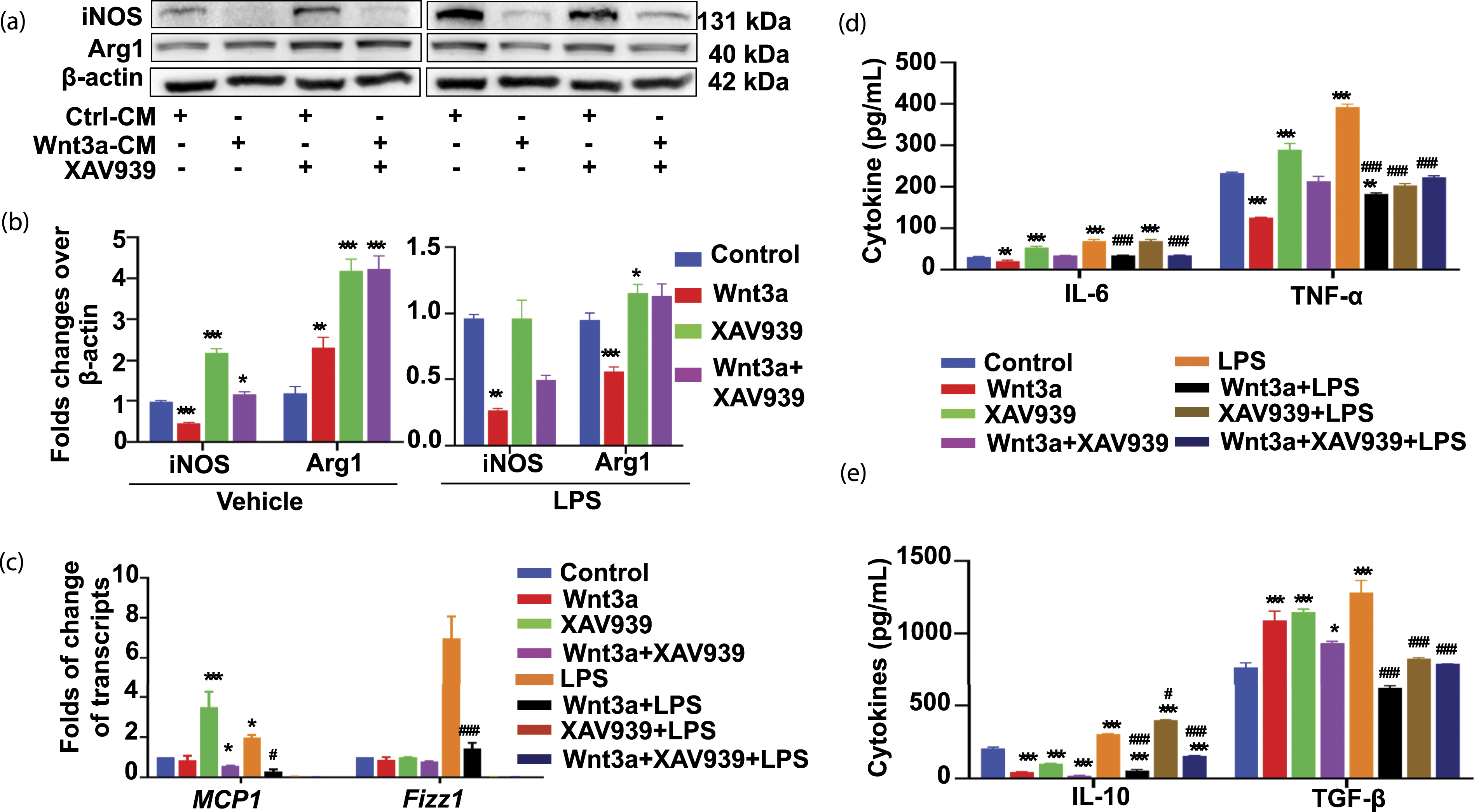
Coincident with the changes in phenotypic surface markers, a significantly reduced IL-6 and TNF-α, cardinal M1 macrophage cytokines, were observed in RAW264.7 cells cultured in Wnt3a-CM alone, compared with the Ctrl-CM-cultured cells, as determined by an ELISA assay (Figure 2(d)). As expected, the Wnt inhibitor XAV939 showed an opposite effect to Wnt3a-CM, which increased the secretion of both IL-6 and TNF-α, particularly the TNF-α, compared to the Ctrl-CM treated cells (Figure 2(d)). Whereas, the addition of LPS in Wnt3a-CM significantly inhibited the secretion of IL-6 and TNF-α as compared to Ctrl-CM plus the inclusion of LPS (Figure 2(d)). Unexpectedly, the production IL-10, a hallmark of cytokine produced by M2 macrophages, was also decreased in cells cultured Wnt3a-CM and XAV939 alone or in combination, in comparison with the Ctrl-CM group (Figure 2(e)). Interestingly, the production of another M2 macrophage-related molecule TGF-β was increased in cells cultured with Wnt3a-CM and XAV939 alone, but had no change in a combination treatment (Figure 2(e)). As expected, however the secretion of IL-10 and TGF-β was remarkably decreased in cells stimulated in Wnt3a-CM plus the inclusion of LPS, compared to that of Ctrl-CM plus LPS (Figure 2(e)).
In agreement with the functional results, cytometric analysis further demonstrated that the frequency of CD86-positive cells (M1 macrophages) (∼24.1%) were significantly decreased, whereas the percentage of CD206-positive cells (M2 macrophages) (∼14.1%) were moderately enhanced in cells cultured in Wnt3a-CM, in comparison with that of Ctrl-CM (Figure 3). While both CD206- and CD86-positive cells were significantly decreased in cultures with LPS, regardless of the presence of Wnt3a (Wnt3a-CM plus LPS or Ctrl-CM plus LPS) (Figure 3). By contrast, both CD206- and CD86-positive cells were significantly increased in the cells cultured with the Wnt inhibitor XAV939 compared to Ctrl-CM plus the inclusion of LPS (Figure 3). These results suggested that the Wnt/β-catenin signaling activation could induce M2 macrophage polarization but inhibit M1 polarization in a naïve state. While in an LPS-induced inflammatory state, the activation of Wnt/β-catenin signaling impaired both macrophage M2 and M1 polarizations. Activation of Wnt/β-catenin signaling inhibits LPS-induced RAW264.7 cell macrophage M2 phenotype. RAW264.7 cells were exposed to Wnt3a-CM and/orTNK1/2 inhibitor XAV939 (1 μ
Involvement of Stats signaling in Wnt/β-catenin-altered RAW264.7 cell polarizations upon LPS stimulation
It has been well documented that the Stats signaling pathway played key parts in the regulation of macrophage phenotype.20,21 To further study whether the Wnt3a-activated Wnt signaling inhibited LPS-induced macrophage M2 polarization was through a mechanism by activating the Stats signaling pathway, the expression of key molecules in Stats signaling cascade was evaluated by an immunoblotting assay in RAW264.7 cells. Indeed, a moderately increased expression of p-Stat3, coinciding with a significantly inhibited expression of p-Stat1, Stat3, and negative feedback inhibitor Socs3 was observed in cells cultured with Wnt3a-CM alone. In consistence, the Wnt3a could inhibit the LPS-induced p-Stat3, p-Stat1, and Stat3 expression (Figure 4(a) and (b)). In addition, the Wnt3a-induced p-Stat3 was further corroborated by immunofluorescent staining assay. More intensive specific immunofluorescence was observed in cells treated with Wnt3a-CM in comparison with other groups (Figure 4(c)). These results demonstrated that Wnt/β-catenin signaling could inhibit LPS-induced macrophage M2 polarization by activating Stats signaling pathway. Wnt/β-catenin signaling inhibits LPS-induced macrophage M2 polarization of RAW264.7 cells by Stats signaling pathway 
Wnt3a suppresses LPS-induced M2 polarization of mouse alveolar macrophages in vivo
In order to further investigate whether Wnt/β-catenin signaling was able to regulate LPS-induced M2 macrophages polarization Wnt/β-catenin signaling inhibits primary alveolar macrophage M2 polarization in LPS-challenged mice through the Stats signaling. (a) A schematic showing workflow of the experimental design. 6–8 weeks old C57/BL6 mice were intratracheally instilled with Ad.Wnt3a, Ad.Dkk1, or Ad.Bgl II vectors, followed by intratracheally administrated 0.9 mg/Kg of LPS in 50 μL saline at day 0 and day 7 post the infection. The lung tissues and bronchoalveolar lavage fluids (BALFs) collected at day14 s post the infection were used for analysis. (b) Representative images of immunofluorescent staining for co-localization of F4/80 (green) and CD206 (red) or iNOS (red) in primary alveolar macrophages isolated from BALFs. (c) Representative images of immunofluorescent staining for p-Start3 (green) expression in primary alveolar macrophages collected from BALFs. (d) Quantification p-Start3 (green) cell in primary alveolar macrophages in (c). (e) Representative blots of molecules of Stats signaling and Wnt/β-catenin signaling, including p-Stat3, Stat3, Socs3, p-β-catenin, and p-GSK3β in lung tissues of mice infected with indicated viruses detected by immunoblotting assay. (f) The relative level of proteins of interest in (e) by densitometry analysis. Data were the mean ± SD from three independent t experiments, as analyzed by one-way ANOVA. *, 
Furthermore, we also detected the abundance of p-state3 in primary alveolar macrophages by immunofluorescent staining. A significant less frequency p-Stat3 positive cells was found in mice infected with Ad.Wnt3a, as compared with mice infected with Ad.Bgl II or Ad.Dkk1 (Figure 5(c) to (d)). Molecular analysis of the alteration of Stats signaling pathway in lung tissues by Western blotting assay further demonstrated a markedly decreased p-Stat3 in LPS-challenged mice infected with Ad.Wnt3a, compared to mice infected with Ad.BglII or Ad.Dkk1 (Figure 5(e) and (f)). In consistence, the abundance of negative regulator factor Socs3 was also decreased in LPS-challenged mice infected with Ad.Wnt3a, along with an increase of p-GSK3β, a negative regulator of Wnt/β-catenin signaling, and a decrease of p-β-catenin, a degradable form of β-catenin, were observed in LPS-challenged mice infected with Ad.Wnt3a (Figure 5(d) and (e)). These results suggested that the activation Wnt/β-catenin signaling impeded M2 macrophages polarization in mouse lung
Discussion
In this report, we showed that Wnt/β-catenin signaling could suppress M1 but induced M2 polarization in mouse macrophage-like RAW264.7 cells in a naïve state, while impairs both M1 and M2 polarization of RAW264.7 cells
The Wnt signaling is an evolutionarily conserved cellular pathway that plays key roles in embryonic development 22 and tissue homeostasis.23,24 It normally is tightly regulated, and a dysregulation of this signaling leads the pathogenesis of many human diseases. 25 In addition, a growing body of evidences demonstrated that Wnt signaling was able to modulate macrophage M1/M2 polarizations.11–13
Several studies have reported that Wnt/β-catenin signaling modulated the monocytes differentiation into macrophages.26,27 Indeed, we found that the Wnt/β-catenin activation suppressed the macrophage M1 polarization but induced macrophage M2 state in RAW264.7 cells under a naïve state. This result confirms an important role of Wnt/β-catenin in regulating monocyte-macrophage polarization.26,27 However, Wnt/β-catenin displayed an ability to impair both M1 and M2 macrophages polarizations under an LPS-induced inflammatory condition
A study by Feng and colleagues showed that Wnt3a could aggravate IL-4- or TGFβ1-induced M2 macrophage polarization, while an inhibition of Wnt signaling by blocking β-catenin led to M1 polarization in macrophages.
21
Mechanistically, the Jak/Stat singling has been well recognized as one of the key cellular pathways in controlling macrophage polarization.13,20,21 It has been demonstrated that Wnt/β-catenin signaling could promote the IL-4- or TGFβ1-induced M2 macrophage polarization by activation of Stat3 in kidney fibrosis.
21
In line with this finding, we also found Wnt signaling activated by Wnt3a ligand could induce Stat3 (Tyr705) phosphorylation and activate Stat signaling in naïve RAW264.7 cells
Of note, there were several limitations in this study. First, a Wnt3a conditional medium (Wnt3a-CM) produced from cell line rather than purified Wnt3a protein was employed for Wnt/β-catenin activation in this study. Undefined factors secreted from the cell line might introduce signaling noises and impact the interpretation of results at some extent. Second, XAV939, a small molecule inhibitor of tankyrase (TNKS) was used for inhibition of Wnt/β-catenin signaling, which might possess undefined activities other than the inhibition of Wnt signaling. For example, XAV939 unexpectedly failed to downregulate Arg1 in RAW264.7 cells (Figure 3(a)), we currently could not rule out the reason causing the conflict result. Therefore, using a secreted inhibitor of Wnt/β-catenin signaling, such as Dickkopf-1 (Dkk1) would be a better antagonist to match with Wnt3a, a secreted Wnt agonist in future study. Third, the size of experimental animals was not statistically calculated and justified in this study, a well-designed animal experiment will improve the quality of data in future.
Conclusions
Collectively, this study demonstrated that the Wnt/β-catenin signaling could induce macrophage M2 polarization but inhibit M1 polarization of RAW264.7 macrophage-like cells in a naïve state
Supplemental Material
sj-pdf-1-eji-10.1177_20587392211059362 – Supplemental Material for Wnt/β-catenin signaling regulates lipopolysaccharide-altered polarizations of RAW264.7 cells and alveolar macrophages in mouse lungs
Supplemental Material, sj-pdf-1-eji-10.1177_20587392211059362 for Wnt/β-catenin signaling regulates lipopolysaccharide-altered polarizations of RAW264.7 cells and alveolar macrophages in mouse lungs by Jiali Yang, Ying Wang, Dandan Yang, Jia Ma, Shuang Wu, Qian Cai, Jing Xue, Chao Yuan, Jing Wang and Xiaoming Liu in European Journal of Inflammation
Footnotes
Acknowledgments
The authors thank Mr Fuyang Song for his technical supports in cytometric analysis.
Authors' contributions
XL conceived and designed the experiments; JY and YW conducted the experiments, analyzed data and drafted the manuscript; DY, JM, SW, QC, JX, CY and JW performed experiments, acquired data and analyzed data; XL interpreted data and critically revised the manuscript. All authors read and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by grants from the National Natural Science Foundation of China (Nos. 31860318 and 32070863), Natural Science Foundation of Ningxia (No. 2018AAC03171), and Ningxia Youth Talents Supporting Program (TJGC2019080). The funders played no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Ethics approval and consent to participate
Experiments involved in mice were approved by the Institutional Animal Care and Use Committee of Ningxia University (NXULS20180123-3) and the ethics committee for the use of animals of Ningxia University.
Data Availability
All the data supporting our findings are contained within the article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
