Abstract
β-Patchoulene (β-PAE) is a tricyclic sesquiterpene isolated from patchouli oil. According to our previous study, β-PAE has anti-inflammatory activity in vivo; however, its anti-inflammatory response still remains unconfirmed in vitro. Therefore, this study is committed to demonstrate the anti-inflammatory effect of β-PAE on lipopolysaccharide (LPS)-stimulated RAW264.7 macrophages. According to our results, pre-treatment with β-PAE significantly decreased the protein and messenger RNA (mRNA) levels of pro-inflammatory cytokines including tumor necrosis factor-α (TNF-α), interleukin (IL)-6, and IL-1β while increased the expressions of anti-inflammatory cytokines like IL-10 in a dose-dependent manner. In addition, real-time polymerase chain reaction (PCR) also revealed that β-PAE could interrupt the mRNA expressions of inducible nitric oxide synthase (iNOS) and cyclooxygenase-2 (COX-2) and thus decreased the levels of nitric oxide (NO) and prostaglandin E2 (PGE2) in LPS-stimulated RAW264.7 macrophages. In conclusion, these results indicated that β-PAE exerted potent anti-inflammatory activity by maintaining the balance between pro- and anti-inflammatory cytokines as well as suppressing iNOS and COX-2 signaling pathways.
Introduction
Inflammation is the body’s protective response against invading pathogens, characterized by redness, swelling, heat, and pain. 1 As inflammation is a characteristic of innate immune response, the occurrence of inflammatory diseases attributes to the innate immunocyte such as macrophages, dendritic cells, and natural killer cells as well as their secreted products. 2 Macrophages are of particular importance to immunity and immune responses. 3 Once activated by bacterial endotoxin, macrophages will destabilize the balance of pro- and anti-inflammatory cytokines. Besides, activated macrophages also release nitric oxide (NO) and prostaglandin E2 (PGE2), the inflammatory indices. 4 Therefore, inhibitors of these inflammatory mediators may be able to alleviate anti-inflammation.
Pogostemon cablin (Blanco) Benth. (patchouli) is an important medicinal plant in ancient China, which applies to remove dampness, relieve sunstroke, and regulate gastrointestinal functions. 5 Silva-Filho et al. 6 reported that the anti-inflammatory effect of patchouli oil may attribute to a single or synergistic effect of its major components such as patchoulol and α-bulnesene or other minor constituents. And our previous study had demonstrated that β-patchoulene (β-PAE, the chemical structure shown in Figure 1(a)), another major component of patchouli oil, has anti-inflammatory property in vivo. However, whether β-PAE confers a cytoprotective effect by regulating inflammatory response in vitro still remains uncertain. Therefore, this study was conducted to demonstrate whether β-PAE has anti-inflammatory effect in vitro.
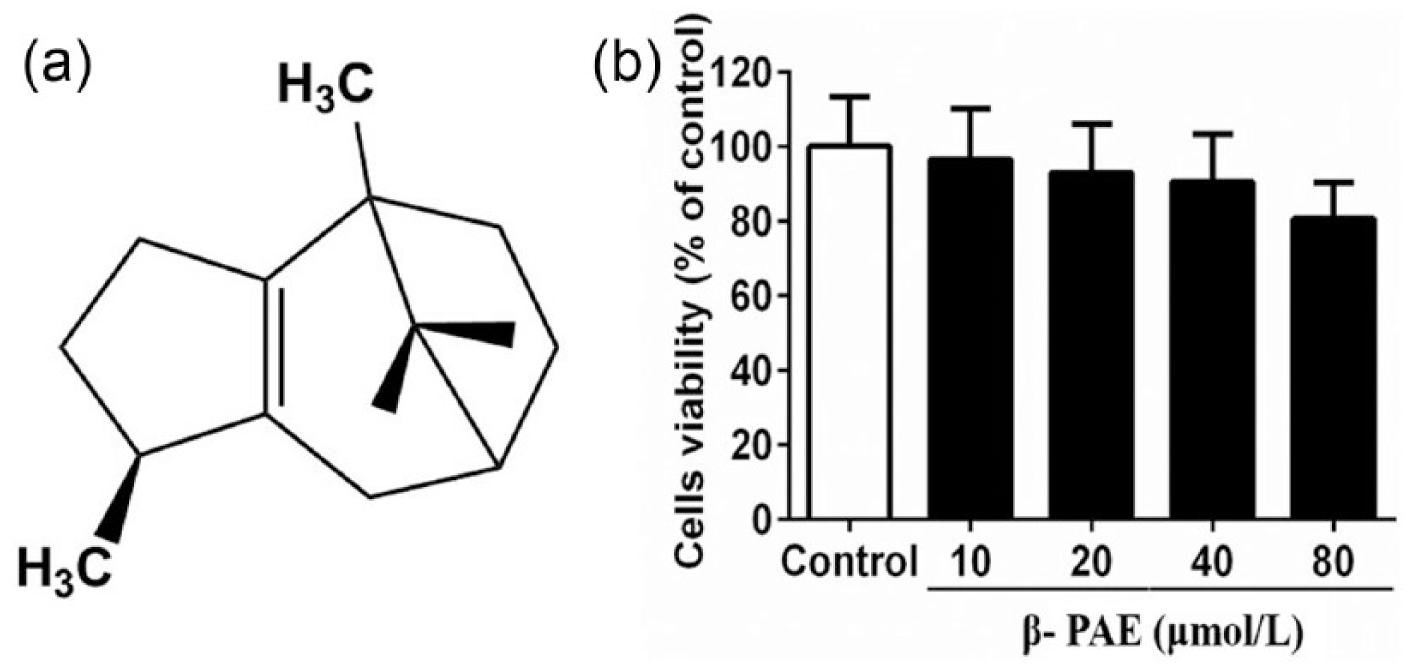
Cytotoxic effect of β-PAE on RAW 264.7 macrophages. (a) Chemical structure of β-patchoulene (β-PAE). (b) The viability of RAW264.7 macrophages treated with different concentrations of β-PAE was evaluated by the MTT assay. The values shown represent the mean ± SEM.
Materials and methods
Plant materials and reagents
Patchouli oil (Lot. 150305) was obtained from Guangzhou Baihua Flavours and Fragrances Company Ltd. (Guangzhou, China). β-patchoulene (β-PAE) was isolated from this patchouli oil according to our previous study. 7
Lipopolysaccharide (LPS) was purchased from Sigma-Aldrich (St. Louis, MO, USA). RPMI-1640 culture medium, fetal bovine serum (FBS), and penicillin-streptomycin were purchased from Gibco (Grand island, NY, USA). Phosphate-buffered saline (PBS, 0.067 mol/L, pH 7.2) was purchased from Hyclone (Beijing, China). Phosphoric acid, sulfanilamide, and N-(1-naphthyl)ethylenediamine dihydrochloride were purchased from Macklin (Shanghai, China). Cell Titer 96® AQueous One Solution Cell Proliferation Assay was purchased from Promega (Madison, WI, USA). RNAiso Plus, Prime Script™ RT reagent kit with genomic DNA (gDNA) eraser (Perfect real time) and SYBR® Premix Ex TaqTM II (Tli RNaseH Plus) were purchased from TaKaRa (Shiga, Japan). Oligonucleotide primers were purchased from Bioneer (Seoul, Korea). Enzyme-linked immunosorbent assays (ELISA) kits were purchased from eBioscience (San Diego, CA, USA). All other chemicals were of analytical grade available unless stated specifically.
Cell culture and drug treatment
The RAW264.7 cell line was obtained from the American Type Culture Collection (ATCC; Rockville, MD, USA). These cells were grown at 37°C in RPMI 1640 medium supplemented with 10% FBS and 1% penicillin-streptomycin in a humidified atmosphere of 5% CO2. β-PAE was dissolved in dimethyl sulfoxide (DMSO) and LPS was dissolved in phosphate buffer (pH 7.2). For treatment, murine RAW264.7 macrophages were seeded in 96-well sterile disposable culture plates at 5 × 104 cells/well for 24 h, unless stated specifically. Afterward, cells were cultured in serum-free medium and incubated with various concentrations of β-PAE (10, 20, and 40 µM) for 2 h and then with LPS (final concentration, 100 ng/mL) for an additional 24 h incubation. In addition, cells in control group were only treated with serum-free medium containing <0.1% DMSO.
Cell viability assay
Cell viability was measured by Celltiter 96® AQueous One Solution Cell Proliferation Assay according to the manufacturer’s instruction. Briefly, after β-PAE treatment for 24 h incubation, the medium was removed and replaced with fresh medium. A total of 20 µL of Cell Titer 96® AQueous One Solution was then added to each well and further incubated at 37°C for 4 h. Upon termination, the supernatant was removed and replaced with 150 µL of DMSO so as to dissolve the formazan product formed by metabolically viable cells. The plates were scanned at 490 nm with a Thermo Scientific Microplate Reader. Cell viability was expressed as a percentage of the control group without LPS treatment.
Nitrite determination
Nitrite concentration in culture media was measured according to the Griess reaction method 8 and presumed to reflect NO levels. Briefly, the supernatants aspirated from 96-well plates were collected after LPS treatment. In all, 100 µL of each supernatant was mixed with the same volume of Griess reagent (equal volumes of 1% (w/v) sulfanilamide in 5% (v/v) phosphoric acid and 0.1% (w/v) N-(1-naphthyl)ethylenediamine-HCl) and incubated for 10 min at room temperature. The absorbance of nitrite was determined at 540 nm. The amount of NO in the samples was expressed as nanomoles per milliliter.
Cytokine determination by ELISA
The LPS-treated supernatants were used to assay for TNF-α, interleukin (IL)-6, IL-1β, IL-10, and PGE2 with commercially available ELISA kit according to the manufacturer’s instructions. The contents of TNF-α, IL-6, IL-1β, IL-10, and PGE2 in the samples were determined from standard curves which were expressed as picograms per milliliter.
Real-time polymerase chain reaction
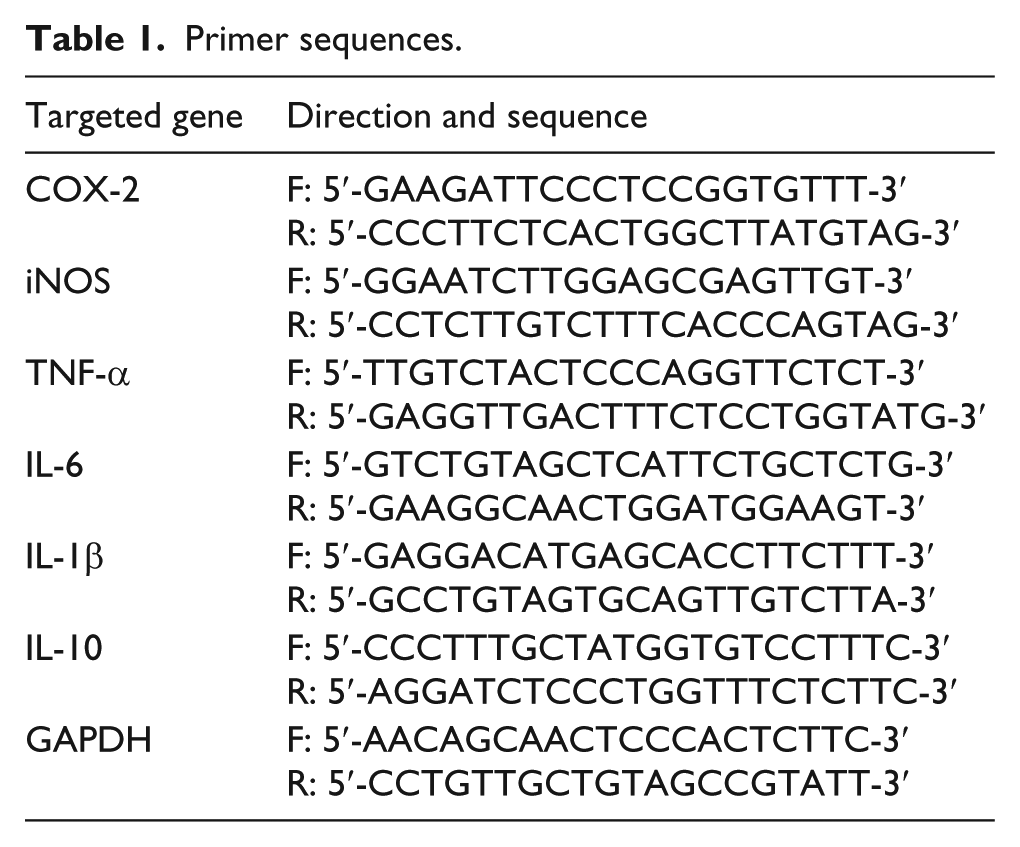
For real-time polymerase chain reaction (PCR), macrophages were seeded in six-well culture plates at 2 × 106 cells/well for 24 h incubation. Afterward, cells were treated with β-PAE and LPS in the same way as that above. The total RNA was isolated from the cells with RNAiso Plus, monitored by the ratio of absorbance at 260 and 280 nm. Then, 1 µg of total RNA was reverse-transcribed as complementary DNA (cDNA) using Prime Script™ RT reagent kit with gDNA eraser (Perfect real time). The cDNA was applied to be a template for real-time PCR with SYBR® Premix Ex Taq™ II (Tli RNaseH Plus). The PCR primers used in this study are listed in Table 1. The amplification sequence protocol was carried out with two steps on the CFX96™ Real-Time PCR Detection System: first step for initial denaturation: cycle 1, 95°C for 30 s; second step for PCR amplification: cycle 40, 95°C for 5 s, 60°C for 30 s. Mean Ct values of target genes were calculated from triplicate measurements and normalized by that of glyceraldehyde-3-phosphate dehydrogenase (GAPDH).
Primer sequences.
Results
Cytotoxic effect of β-PAE on RAW 264.7 macrophages
As shown in Figure 1(b), treatment with β-PAE did not show significant signs of cytotoxicity up to 40 µM in RAW 264.7 macrophages, and thus, the nontoxic concentrations of β-PAE were used in this study.
Effect of β-PAE on NO, PGE2 production and inducible nitric oxide synthase, and cyclooxygenase-2 messenger RNA expressions in LPS-stimulated RAW264.7 macrophages
As shown in Figure 2(a) and (b), compared with the control group, LPS stimulation induced significant (P < 0.01) increase in the production of NO and PGE2, which were dramatically (P < 0.05) reversed by pre-treatment with β-PAE in a dose-dependent manner.

Anti-inflammatory effects of β-PAE on the production of (a) NO, (b) PGE2, (c) the transcriptional levels of iNOS, and (d) COX-2 in LPS-stimulated RAW264.7 macrophages. Data are expressed as mean ± SEM. ##P < 0.01 versus the control group. *P < 0.05, **P < 0.01 versus the LPS group.
As shown in Figure 2(c) and (d), upon exposure to 100 ng/mL LPS, the messenger RNA (mRNA) expression of inducible nitric oxide synthase (iNOS) and cyclooxygenase (COX)-2 was significantly increased (P < 0.05) as compared with the control group. β-PAE could significantly (P < 0.01) decrease the transcription levels of iNOS in a dose-dependent manner when compared with the LPS group. As for COX-2, only at the dose of 40 µM, β-PAE exhibited potent (P < 0.05) decline as compared with the LPS group. Although pre-treatment with 10 and 20 µM β-PAE also decreased in COX-2 mRNA expression, there were no significant difference when compared with the LPS group.
Effect of β-PAE on pro- and anti-inflammatory cytokine expressions in LPS-stimulated RAW264.7 macrophages
To further characterize the effect of β-PAE on LPS-stimulated RAW264.7 macrophages, the production and mRNA expressions of TNF-α, IL-6, IL-1β, and IL-10 were quantified. As shown in Figure 3, LPS treatment significantly (P < 0.01) increased the productions of TNF-α, IL-6, and IL-1β to 5.73-, 6.57-, and 9.35-fold, respectively, while decreased IL-10 to 2.37-fold when compared to the control group. β-PAE could significantly (P < 0.05) decrease the protein levels of TNF-α, IL-6, and IL-1β while increase the IL-10 level in a dose-dependent manner as compared with the LPS group. Consistent with the results of these protein levels, real-time PCR also revealed that β-PAE exerted similar effects on the transcriptional levels of these pro- and anti-inflammatory cytokines.

Anti-inflammatory effects of β-PAE on the protein and mRNA expressions of (a) TNF-α, (b) IL-6, (c) IL-1β, and (d) IL-10 in LPS-stimulated RAW264.7 macrophages. The protein levels of TNF-α, IL-6, IL-1β, and IL-10 were measured with ELISA kit, and the mRNA expressions of these inflammatory cytokines were measured with RT-PCR. Data are expressed as mean ± SEM. ##P < 0.01 versus the control group. *P < 0.05, **P < 0.01 versus the LPS group.
Discussion
LPS is a major component with high antigenicity of the outer membrane of gram-negative bacteria, responsible for eliciting macrophage-mediated inflammation.
9
It involves the secretion of many effector molecules such as NO and PGE2. NO, catalyzed by iNOS from
TNF-α, IL-6, and IL-1β are recognized as the most representative pro-inflammatory cytokines, responsible for the initiation of iNOS and COX-2 signaling pathway. 1 In this study, we demonstrated that β-PAE significantly reduced the protein and mRNA levels of TNF-α, IL-6, and IL-1β in LPS-stimulated RAW264.7 macrophages while increased the expression of IL-10, the anti-inflammatory cytokine. Therefore, the above results effectively indicated that the anti-inflammatory effect of β-PAE might be related to maintain the balance between pro- and anti-inflammatory cytokines.
Combined with our previous study, it is believed that β-PAE possesses potential anti-inflammatory activity, and it is worthwhile to exploit β-PAE to be a useful therapeutic agent for inflammation.
Footnotes
Acknowledgements
The authors are grateful to the researches who participated in the study. W.-H.Y. and Y.-H.L. contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by grants from Hong Kong, Macao and Taiwan Science & Technology Cooperation Program of China (No. 2014DFH30010), Science and Technology Planning Project of Guangdong Province, China (Nos 2013B090600007 and 2013B090800052), Guangdong International Cooperation Project (No. 2013508102016), and Higher Vocational Education Brand Specialty Construction Project of Guangdong Province (No. 16061).
