Abstract
Background
The rising incidence of obesity and its related health issues demand novel approaches prioritizing safety and effectiveness. In light of concern about the side effects of pharmaceutical therapies, research into alternative medicines has accelerated. Neolamarckia cadamba (NC), a tropical medicinal plant with a diverse phytochemical profile, presents a promising avenue for obesity management.
Objectives
The objective of this research was to evaluate the possible role of isolated fractions in mitigating obesity using antioxidant and enzyme inhibition studies. Additionally, the 3T3-L1 adipogenesis differentiation assay was utilized further to understand the effect of these fractions on adipocyte behavior. Furthermore, we used analytical techniques, high-performance thin-layer chromatography in conjugation with mass spectrometry (HPTLC–MS), to evaluate the chemical characteristics of the bioactive fractions.
Materials and Methods
Cold maceration extraction followed by column chromatography was utilized to isolate the bioactive fraction which was subjected to meticulous phytochemical analysis as well as in vitro antioxidant assays. Enzyme inhibition assays were conducted to evaluate their impact on pancreatic lipase, alpha-glucosidase, and alpha-amylase followed by the 3T3-L1 adipogenesis study to assess adipocyte behavior. Finally, HPTLC–MS was utilized for chemical characterization.
Results
Fraction 1 demonstrated the most potent activity, also exhibited significant antioxidant potential, and effectively inhibited key enzymes involved in adipogenesis. In the 3T3-L1 cell line, the fraction notably reduced lipid accumulation and triglyceride levels. Chemical analysis revealed the presence of compounds including stigmasterol, gallic acid, quercetin, 4-hydroxycoumarin, and naringin.
Conclusion
The study underscores the potency of NC and offers a holistic approach to obesity management, providing a promising approach for the development of effective and safer therapeutic interventions.
Introduction
The prevalence of obesity, which has been referred to as a global health crisis, has reached unprecedented levels, and its complex interconnections have resulted in a cascade of related diseases. With the escalating incidence throughout the world, obesity is an alarming threat to health. Disorders like non-alcoholic fatty liver disease (NAFLD), stroke, venous thromboembolism, diabetes, osteoarthritis, hypertension, and cardiovascular disorders are closely related to the rising prevalence of obesity worldwide (Perdomo et al., 2023). Preadipocyte cells undergo conversion into adipocytes through the consumption of enormous quantities of fatty foods. Adipocytes reserve fat in the triglycerides form and liberate it during situations where there is a decrease in calories. Therefore, the imbalance between appetite-inhibiting hormones like cholecystokinin (CKK), glucagon-like peptide (GLP-1), and appetite-stimulating hormones like ghrelin causes obesity-associated complications (Dickinson et al., 2023). Being a multifaceted metabolic disease, obesity emphasizes the need for novel therapeutic strategies to address the growing health crisis. Several pharmaceutical therapies like phentermine, liraglutide, and orlistat are available for the management of obesity; however, they frequently lead to undesirable side effects like steatorrhea, hypoglycemia, vitamin malabsorption, and elevated blood pressure. Furthermore, several antiobesity medications have been banned because of their associated adverse consequences (Jackson et al., 2015). Therefore, concerns about the potential long-term impact of pharmaceutical drugs have necessitated the exploration of alternative methods that are harmonious with the body’s natural function for disease management (Dragano et al., 2020). Investigating natural plant-based therapies is an intriguing possibility because they can potentially treat obesity-related illnesses without having the detrimental consequences of synthetic drugs. The tropical medicinal plant of the Rubiaceae family Neolamarckia cadamba (NC) (Roxb.) Bosser, which is endemic to Southeast Asia and some parts of Australia, has prompted scientific attention because of its extensive phytochemical profile and long history of traditional uses in various ailments. Various communities employ parts of plants to treat numerous illnesses, including fever, inflammation, uterine complaints, infections, skin diseases, and neurological disorders. However, currently, to date, there is no investigation into its potential role in combating obesity (Pandey & Negi, 2016; Singh & Pattnaik, 2023). Recently, NC leaves have evolved into a particularly intriguing source of bioactive compounds, with the potential for a range of therapeutic uses. In the context of obesity management, the plant presents an intriguing avenue for investigation, given its ability to modulate important key pathways linked to lipid metabolism.
Numerous research has demonstrated that plant-derived phytoconstituents, such as phenols, saponins, hesperidin, quercetin, and steroids (β-sitosterols) along with many other bioactive substances exhibit potent antiobesity properties so investigations of these phytoconstituents in plants are preliminary strategy. Obesity is associated with elevated oxidative stress as well as reactive oxygen species (ROS) in adipose tissue. Antioxidants are compounds that aid in refraining from oxidative stress by neutralizing ROS formation and inhibiting inflammatory signaling pathways which can treat the advancement of obesity. Therefore, to treat or prevent disorders where oxidative stress is thought to be the reason, medicinal plants having antioxidant properties have been successfully employed. Further, some novel methodologies by inhibiting enzymes targeting obesity have been utilized for obesity treatment. Alpha-amylase and alpha-glucosidase are the two major enzymes involved in the carbohydrate digestion process, where inhibitors of these enzymes limit the intestinal absorption of carbohydrates and aid in weight loss. Pancreatic lipase enzymes help in the conversion of dietary triglycerides into free fatty acids and are responsible for 70% of total lipids hydrolysis. So, inhibition of pancreatic lipase leads to calorie deficit and obesity management. Therefore, for the investigations of the antiobesity activity of plants, one of the most potent screening methods is to search for effective pancreatic lipase inhibitors. Further, researchers extensively utilized 3T3-L1 cell lines for the study of adipogenesis, exploration of metabolic pathways, and investigation of the molecular mechanisms of obesity. This cellular model accurately recapitulates the process of adipogenesis and screening of potential therapeutics which makes it a significant and valuable tool for investigating potential antiobesity agents. Finally, high-performance thin-layer chromatography in conjugation with mass spectrometry (HPTLC–MS) is a cutting-edge analytical technique that helps to identify bioactive constituents found in herbal plants where HPTLC is an analytical method that enables the separation and identification of various components within a complex mixture, while MS is utilized for detection that yields information about the molecular mass of the separated compounds. This analytical approach provides insights into their putative mechanisms of action in addition to facilitating the identification and quantification of constituents in the fraction (Sidharthan & Ashajyothi, 2023).
Central to our investigation, a hypothesis was designed where initially bioactive fractions from NC leaf extract were isolated. Further, to select the most potent fraction, rigorous phytochemical evaluation and an array of in vitro experiments aimed at antioxidant capacity and enzyme inhibition were utilized, Further, to provide mechanistic insights to assess the ability of the bioactive fractions, investigations of enzyme inhibition will be utilized to evaluate the bioactive fractions capacity to control important enzymes involved in the metabolism of lipid and adipogenesis, like alpha-amylase, pancreatic lipase, and alpha-glucosidase linked to obesity. Furthermore, the 3T3-L1 adipogenesis cell differentiation study, which is a well-researched cellular system that mimics adipocyte differentiation, will be employed for further clarification of the effect of the bioactive fractions on adipocyte differentiation and lipid accumulation to gain molecular insights. An additional facet of this research involves the characterization of lead compounds present in the bioactive fraction employing the HPTLC–MS technique. In summary, by utilizing a variety of methodologies, this multidisciplinary study aims to assess the antiobesity potential of NC bioactive fractions thoroughly. The insights gained from this study could be used for the development of innovative therapeutic interventions aimed at combating obesity and its associated complications.
Materials and Methods
Solvents and Reagents
The solvents and reagents used in the experiment were of analytical grade toluene, silica gel G, ethyl acetate (Merck Darmstadt, Germany), β-sitosterol, and 2,2-diphenyl-1-picrylhydrazyl (DPPH) (Sigma Aldrich Company). Other chemicals like petroleum ether, hydrogen peroxide (H2O2), ascorbic acid, gallic acid, alpha-glucosidase, quercetin, Griess reagent, and orlistat used were of Fisher Scientific and Rankem Company.
Plant Material Collection
The leaves of NC were collected from the campus of Birla Institute of Technology, Mesra, Ranchi, Jharkhand, India in June 2023. The leaves were authenticated by the Central National Herbarium, Botanical Survey of India, Howrah, West Bengal, India (Ministry of Environment, Forest and Climate Change) with reference no. CNH/Tech.II/2022/87b.
Extraction Procedure
The leaves were thoroughly washed in running water, dried in the shade for 10 days, and further pulverized into powder for the cold maceration extraction technique. For this, 1 kg of powdered leaves was macerated in 2 l of 85% methanol (methanol–water, 85:15) as a solvent in the conical flask. The flask was kept for 72 hours at room temperature with intermittent shaking. Further, Whatman filter paper no. 1 was utilized for the filtration of powdered leaves and solvent mixture. Finally, a rotary evaporator was used for the concentration of filtrate at 40°C to yield the crude extract of leaves. The resultant product was finally vacuum lyophilized and stored aside at 4°C for further studies (Mohanty & Pattnaik, 2021).
Fractionation of NC Extract
Column chromatography was utilized to isolate the bioactive guided fractions from crude leaf extract of NC obtained through the cold maceration technique. Several solvent combinations were utilized for the optimization of the meticulous separation of different components on the TLC plate where the mobile phase toluene:ethyl acetate (6:4) was the best-developed solvent. When the column was ready, 5 g of crude leaf extract (sample) was placed at the top of the cylinder-shaped glass column. Thereafter, the mobile phase was allowed to pass through the column for the isolation of bioactive fractions of NC extract (Gharpure et al., 2021).
Phytochemical Evaluation
Total Phenolic Content (TPC)
The TPC of NC bioactive fractions was carried out with the Folin–Ciocalteu method, where gallic acid was taken as the standard compound (Kupina et al., 2018). First, 1 ml of each bioactive fraction was mixed with 2.5 ml of 10% Folin–Ciocalteu’s reagent. Further, 3 ml of 7.5% sodium carbonate was mixed with the above solution. Finally, the resultant sample was incubated at 40°C for 60 minutes. The absorbance was taken by a microplate reader at 765 nm.
Total Flavonoid Content (TFC)
The TFC of NC bioactive fractions was estimated by aluminum chloride colorimetric assay, where quercetin was taken as the standard compound (Yee et al., 2023). At first, 1 ml of fraction was added to 5 ml of distilled water. Thereafter, 0.5 ml of 10% sodium nitrite solution was prepared and mixed with the above solution. Further, 5% aluminum chloride solution (0.30 ml) was added to create the resulting mixture. After incubating the above mixture at 40°C for 10 minutes, it was further combined with 5 ml of 10% sodium hydroxide solution. After 15 minutes, a microplate reader was used to measure the sample’s absorbance at 510 nm.
Total Steroid Content (TSC)
The steroid content of all the NC bioactive fractions was determined by ferric chloride colorimetric assay with some modifications. β-Sitosterol was used as the standard compound. First, 1 ml of sample was taken and added to 2.5 ml of iron (III) chloride. Then 2.5 ml of sulfuric acid (4N) was measured and mixed with this solution and allowed to stand for 5 minutes. Further, 1 ml of potassium hexacyanoferrate (III) solution was mixed with the resulting mixture and heated in a water bath at 72°C for 40 minutes. Finally, the absorbance was determined with the help of a microplate reader at 780 nm (Nayak & Pattnaik, 2024).
In vitro Antioxidant Assay
DPPH Assay
The free radical scavenging activity of the bioactive fraction of NC was evaluated through DPPH assay with minor modifications (Tsai & Lin, 2019). At first, 1 ml of all the NCF of different concentrations (100, 300, and 500 µg/ml) was added to 2.5 ml of methanol. Thereafter, 3 ml of 0.1 mM of DPPH solution was measured and mixed with the above solution. This solution was kept at room temperature for 40 minutes in a dark condition. Further, the same procedure was followed for the standard solution. Ascorbic acid was taken as the standard compound. Finally, the absorbance of all the samples and standard solution was measured with the help of a microplate reader at 523 nm.
Trolox Equivalent Antioxidant Capacity Assay (2,2’-Azino-bis3-Ethylbenzothiazoline-6-Sulfonic Acid (ABTS))
The ABTS assay is based on the discoloration of ABTS by antioxidant compounds, which reflects the amount of scavenged ABTS radicals. ABTS scavenging activity of the bioactive fraction of NC at various concentrations (100, 300, and 500 µg/ml) was determined for measuring the relative antioxidant potential as described by Flórez et al. (2022). At first, 7 mM ABTS was added to 2.45 mM potassium persulfate in water to form a reaction mixture. The above mixture was kept at room temperature for about 16 hours. Further, 8 ml of methanol was mixed with the above solution mixture. Thereafter, 5 µl of NC bioactive fraction in varying concentrations (100, 300, and 500 µg/ml) was combined with the 4 ml of previously prepared ABTS sample. The final solution was left to stand at room temperature for 40 minutes and absorbance was determined by a microplate reader at 523 nm.
Nitric Oxide (NO) Radical Scavenging Assay
NO radical scavenging assay was measured using the protocol outlined by Seleshe et al. (2022). First, 5 ml of sodium nitroprusside (10 mM) was added to phosphate-buffered saline (0.5 mM) to prepare the solvent mixture. Further, various concentrations (100, 300, and 500 µg/ml) of all the NCF were combined with the previous mixture. The resulting mixture was incubated at 35°C for 150 minutes. Thereafter, 0.5 ml of Griess reagent was accurately measured and mixed properly. The mixture was further incubated at room temperature for 30 minutes. Finally, the absorbance was taken using a microplate reader at 540 nm.
H2O2 Scavenging Assay
The H2O2 scavenging assay was determined according to Lyu et al. (2022). At first, an H2O2 solution (50 mM) was prepared in phosphate buffer. The solution was accurately maintained at pH 7.4. Further various concentrations (100, 300, and 500 µg/ml) of bioactive fractions were prepared and the final volume was adjusted. Finally, 0.6 ml of H2O2 solution was combined with the above mixture and left to stand for 10 minutes. Ascorbic acid was taken as the standard compound. The absorbance was determined at 230 nm in a microplate reader.
Enzyme Inhibitory Assays
Alpha-amylase Inhibition Assay
Alpha-amylase inhibition assay was estimated using the 3,5-dinitrosalicylic acid (DNSA) method (Zafer et al., 2021). At first, varying concentrations (1, 1.5, and 2 mg/ml) of NCF (test sample) were prepared. Thereafter, 200 µl of the test sample was taken and mixed with 200 µl of alpha-amylase and 300 µl of the phosphate buffer. The previously prepared mixture was incubated at 30°C for 15 minutes. Further, 200 µl of the starch solution was prepared and combined with the above mixture and again incubated for an additional 10 minutes. Finally, 200 ml of DNSA reagent was mixed with the above mixture and boiled in a water bath at 90°C for 15 minutes. The resulting mixture was diluted in an ice bath with 10 ml of distilled water. Acarbose was taken as the standard. Absorbance was determined at 540 nm by a microplate reader.
Alpha-glucosidase Inhibition Assay
Alpha-glucosidase assay was carried out by taking acarbose as the standard compound using the method of Yang et al. (2022). At first, 10 µl of alpha-glucosidase and 50 µl of phosphate buffer solution were taken and accurately measured. Thereafter, 10 µl of alpha-glucosidase was added to the previously prepared phosphate buffer solution and stirred continuously for 5 minutes. Then, various concentrations (1, 1.5, and 2 mg/ml) of NCF were prepared and combined with the above mixture. The resulting mixture was subjected to incubation at 37°C for 40 minutes. Further, 30 µl of 5 mM of p-nitrophenyl-beta-D- glucopyranoside (P-NPG) solution was prepared and mixed with the resulting mixture. At last, the resulting mixture was subjected to additional incubation at 37°C for 10 minutes. Finally, 2 ml of 0.5 M sodium carbonate solution was combined with the above solution and the absorbance was determined at 400 nm by a microplate reader.
Pancreatic Lipase Inhibition Assay
Pancreatic lipase assay was estimated and determined by the techniques outlined by Sosnowska et al. (2022) by measurement of the hydrolysis of p-nitrophenyl butyrate (P-NPB) to p-nitrophenol. First, 0.1 M of Tris-HCl buffer was prepared by accurately measuring 1.5 g of Tris mixed in 100 ml of double distilled water. The solution was precisely maintained at pH 8.0. Thereafter, 1 mg/ml stock solution of pancreatic lipase enzyme was prepared using previously prepared Tris-HCl buffer. Further, various concentrations (1, 1.5, and 2 mg/ml) of NCF were prepared and 20 µl of fraction was combined with 200 µl of enzyme buffer. The resulting mixture was subjected to incubation at 37°C for 30 minutes. There, 10 µl of 10 mM PNPB was prepared and added to the resulting mixture and provided with additional incubation at 40°C for 10 minutes. In this assay, orlistat was used as a standard compound. Finally, the absorbance was determined at 400 nm by a microplate reader.
3T3-L1 Cell Line Differentiation Assay
A rigorous 3T3-L1 cell line differentiation assay was meticulously designed with four separate groups for the cell line study employing a HiMedia adipogenesis kit procured from the National Centre for Cell Science (NCCS), Pune, India. Group 1: cells were exposed to growth media only (control). Group 2: indomethacin-induced differentiation (30 µg/ml). Group 3: orlistat-induced standard treatment (20 µg/ml). Group 4: NCF1 treatment (50 and 100 µg/ml). For the experiment, 3T3-L1 cell lines were procured from NCCS (Pune, India with request no. 587/2023-24). First, the study was initiated utilizing 3T3-L1 preadipocytes which were cultured in DMEM supplemented with 10% fetal bovine serum along with glucose and antibiotics until 70% confluency of the cell was achieved. Further, in a 24-well plate, cells were seeded with 5 × 105 cells per well at 37°C. As soon as the required cell confluency was achieved, the growth medium was substituted with differentiation media. Thereafter, standard and treatment drugs were incorporated at concentrations of 50, 100, 200, 300, and 500 µg/ml. Further, differentiation medium 2 was also added and incubated for 48 hours. Throughout the study, the control group received only the required growth media. After the differentiation of cells for 15 days, oil red O staining was employed for the identification of lipid droplets in the cells. Additionally, triglyceride levels were measured by a triglyceride quantification kit (Sigma Aldrich). Finally, images were captured by a phase contrast microscope for further analysis (Liu et al., 2023).
High-performance Thin-layer Chromatography Mass spectroscopy
After the in vitro antioxidant and enzyme inhibition assays, the obtained results were subjected to rigorous statistical analysis and comparative assessments to determine the most potent bioactive fractions of NC. Further, bioactive fractions were then subjected to additional analysis using HPTLC–MS to reveal their chemical composition.
The experiment was performed at a relative humidity of 60% and 24 ± 2°C. The separation of the compound from fractions was done by utilizing 60 F254 aluminum-coated silica gel plates (10 × 10 cm). Stock solution (1 mg/ml) of fractions F1, F2, F3, and F4 of NC was prepared accurately by dissolving 5 mg of each fraction in 5 ml of methanol. The obtained solution was sonicated for 15 minutes and filtered. Further, the solution was applied by employing an automatic TLC sampler (CAMAG) which was previously fixed to preoutlined specifications by maintaining a band length of 6 mm, 10 mm from the bottom edge, and 15 mm distance from the side. The resulting solvent system was prepared in appropriate quantities utilizing toluene:ethyl acetate in the ratio (6:4 v/v) after trying several solvent combinations. Following sample application, the plates were developed in a presaturated CAMAG twin-trough with 20 ml of the solvent system up to 7 cm (70% height of the plate). The developed TLC plates were allowed to dry for 10 minutes in a heater for 5 minutes. Then, anisaldehyde sulfuric acid reagent was employed for derivatization of the TLC plate and again heated in the heater for 5 minutes. The CAMAG TLC Visualizers 2-win CATS software was implemented for assessing the retardation factor (Rf) values of the separated compounds. A similar methodology was used for HPTLC–MS analysis, although the derivatization step was skipped. After the isolated spots were developed in mobile phase solutions, they were immediately identified on the underivatized HPTLC plate. The isolated spots were then further examined utilizing TLC MS interface 2 (CAMAG). Each spot was meticulously kept under the TLC MS interface and subsequently employed to acquire the corresponding spectral data by mass spectrometry (Thermo Scientific LTQ-XL). The m/z ion peak of the spectrum information obtained from mass spectrometry was utilized for the individual compound identification. This meticulous approach resulted in a thorough investigation of the chemical components found in the NC bioactive fractions (Anokwuru et al., 2022; Menon et al., 2021).
Statistical Analysis
All the data were analyzed in triplicates as Means ± Standard Error Mean (SEM) except for phytochemical analysis. A one-way analysis of variance (ANOVA) followed by Dunnet’s “t” test was utilized for statistical analysis of data where probability (p) values of <0.05 were considered significant: *p < 0.05, **p < 0.01, and ***p < 0.001; ns, not significant. All the results were determined in GraphPad Prism 8.3.1 software.
Results and Discussion
Extraction and Fractionation
After the extraction of NC leaves by cold maceration method, four bioactive guided fractions from NC extract were isolated and stored at 4°C in the refrigerator for further experiments.
Phytochemical Evaluation
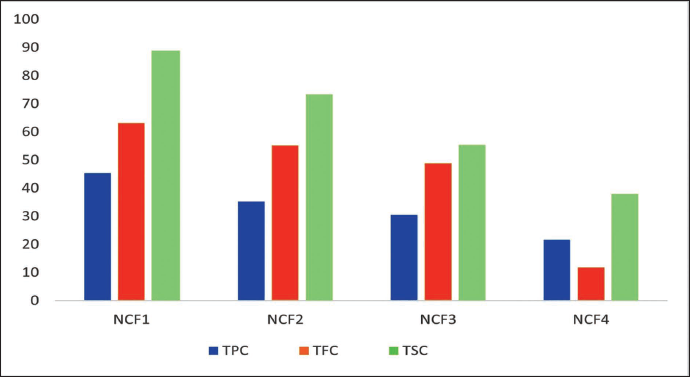
In the phytochemical analysis, all four bioactive fractions of NC were evaluated utilizing total phenolic, flavonoid, and steroidal content. The TPC of bioactive fractions NCF1, NCF2, NCF3, and NCF4 was found to be 45.33, 35.28, 30.57, and 21.67 mg GAE/g (gallic acid equivalent), respectively. The TFC of fractions NCF1, NCF2, NCF3, and NCF4 was found to be 63.23, 55.28, 48.87, and 11.86 mg QE/g (quercetin equivalent), respectively, whereas the total steroidal content of bioactive fractions NCF1, NCF2, NCF3, and NCF4 was found to be 88.91, 73.34, 55.42, and 37.87 mg BSE/g (β-sitosterol equivalent), respectively, as shown in Figure 1. After the successful completion of the phytochemical evaluation, it can be concluded that the NC bioactive fraction has an abundant quantity of phytoconstituents like phenols, flavonoids, and steroids, where each of these constituents is cited as the source of antiobesity agents. These bioactive fractions can be employed for antiobesity treatment strategies.
In vitro Antioxidant Assay
After the phytochemical evaluation, bioactive fractions underwent four different antioxidant assays such as DPPH, ABTS, H2O2, and NO2 assay for the rigorous assessment of their antioxidant capacity. In the DPPH assay, NCF1 showed the most potent antioxidant activity based on IC50 values, where IC50 of NCF1, NCF2, NCF3, and NCF4 were 1.34 ± 0.61, 1.75 ± 0.71, 2.23 ± 0.37, and 2.65 ± 0.43 µg/ml, respectively. Further, in the ABTS assay, IC50 values of NCF1, NCF2, NCF3, and NCF4 were found to be 2.13 ± 0.32, 2.42 ± 0.31, 2.69 ± 0.28, and 2.86 ± 0.67 µg/ml, respectively, with fractions NCF1 and NCF2 showing better potency than other fractions. Then, again NO2, assay revealed that NCF1 showed better scavenging activity than other fractions with IC50 values of NCF1, NCF2, NCF3, and NCF4 as 1.34 ± 0.34, 1.79 ± 0.55, 1.68 ± 0.29, and 2.33 ± 0.28 µg/ml, respectively. Finally, in the H2O2 assay, IC50 values of NCF1, NCF2, NCF3, and NCF4 were found to be 2.05 ± 0.33, 2.81 ± 0.46, 2.76 ± 0.18, and 2.98 ± 0.59 µg/ml, respectively, where NCF1 showed most potent activity than other fractions as shown in Table 1. After the successful execution of an array of in vitro antioxidant assays, it can be concluded that NC bioactive fractions have significant antioxidant capacity to counteract ROS and oxidative stress by scavenging free radicals in the cell. Therefore, these bioactive fractions can aid in the treatment of oxidative stress-related disorders like obesity.
In vitro Antioxidant Activities of Bioactive Fractions Using 2,2-Diphenyl-1-Picrylhydrazyl (DPPH) Assay, 2,2’-Azino-bis3-Ethylbenzothiazoline-6-Sulfonic Acid (ABTS) Assay, Nitric Oxide Assay, and Hydrogen Peroxide Assay.
Enzyme Inhibition Assay
After the successful completion of in vitro antioxidant activities, the bioactive fraction was evaluated based on enzyme inhibition assays like alpha-amylase, alpha-glucosidase, and pancreatic lipase. The alpha-amylase assay revealed that NCF1 showed the best enzymatic inhibitory activity in all the fractions, with IC50 values of NCF1, NCF2, NCF3, and NCF4 as 1.19 ± 0.74, 1.34 ± 0.43, 2.21 ± 0.71, and 2.88 ± 0.28 µg/ml, respectively. In the alpha-glucosidase assay, NCF1, NCF2, NCF3, and NCF4 showed IC50 values of 1.33 ± 0.29, 1.48 ± 0.16, 2.21 ± 0.83, and 2.42 ± 0.71 µg/ml, respectively, with NCF1 showing most potent inhibition activity. Finally, the results of pancreatic lipase demonstrated that NCF1 showed better activity than other bioactive fractions with IC50 values of NCF1, NCF2, NCF3, and NCF4 as 2.35 ± 0.59, 3.58 ± 0.35, 2.44 ± 0.61, and 4.11 ± 0.33 µg/ml, respectively (Table 2). After the implementation of an enzyme inhibition assay targeting obesity, it can be concluded that NC bioactive fractions have significant alpha-amylase, alpha-glucosidase, and pancreatic lipase inhibition potential. Therefore, these bioactive fractions will effectively decrease the carbohydrate as well as fat absorption in the gastrointestinal tract leading to regulation of appetite and obesity management.
Enzyme Inhibition Assays of the Bioactive Fractions Using Alpha-amylase, Alpha-glucosidase, and Pancreatic Lipase Assays.
3T3-L1 Adipocyte Differentiation Assay
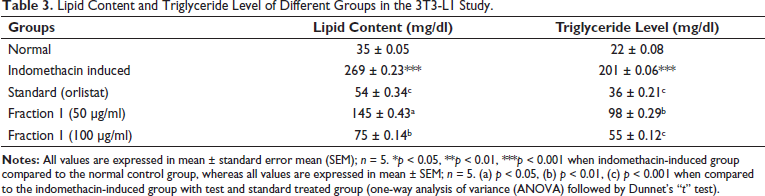
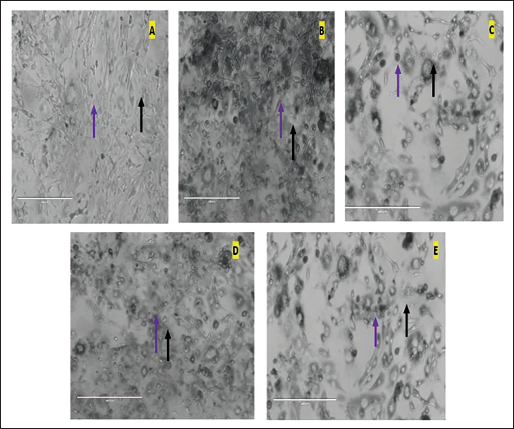
After the completion of the in vitro antioxidant and enzyme inhibition study, utilization of the 3T3-L1 differentiation assay established certain remarkable findings in NCF1 which substantially inhibited the differentiation of cells as compared to the indomethacin. Additionally, the fraction NCF1 at 100 µg/ml concentration demonstrated a noticeable inhibition of the accumulation of lipids in 3T3-L1 adipocytes. Interestingly, along with this observation, there were no obvious toxicological ramifications during the extensive experimental studies. Lipid droplets were seen in extremely small amounts after treatment with NCF1 at 100 µg/ml (Figure 2). The arrows in red and yellow, correspondingly, represent adipocytes and lipid droplets. Moreover, NCF1 additionally exhibited a significant decrease in adipocyte differentiation at a dosage of 50 µg/ml. The toxicological assessment showed that this antiadipogenic effect was accomplished at a dosage level that did not affect the cell viability. Based on these results, the bioactive fraction, NCF1 successfully inhibited adipocyte development in 3T3-L1 preadipocytes (Table 3), and it also significantly reduced lipid content and triglyceride levels throughout the investigation. Therefore, by employing a 3T3-L1 differentiation assay, the bioactive fraction revealed important insights into the intricate biology of adipose tissue which can aid in the development of interventions against obesity.
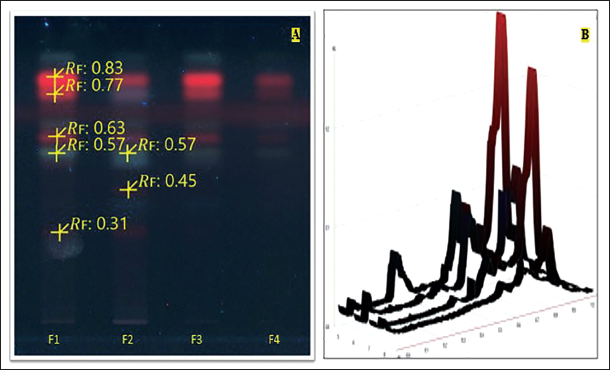
(A) Chromatograms of Bioactive Fractions F1, F2, F3, and F4 were Obtained on High-performance Thin-layer Chromatography (HPTLC) F254 Plates Developed at 254 nm. (B) HPTLC Densitogram of the Chromatograms F1, F2, F3, and F4.
Lipid Content and Triglyceride Level of Different Groups in the 3T3-L1 Study.
HPTLC–MS
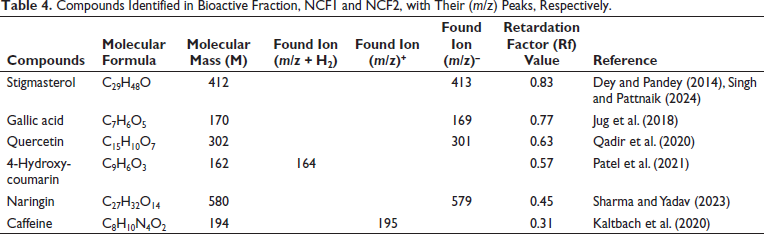
After the in vitro antioxidant, enzyme inhibition, and 3T3-L1 adipocyte differentiation studies, the NC bioactive fractions underwent rigorous HPTLC–MS analytical technique. The research employed HPTLC–MS to precisely identify and characterize compounds by employing molecular mass information obtained from mass spectroscopy. The results of this meticulous approach demonstrated the separation of compounds with major compounds found in NCF1. Five and two compounds were identified in NCF1 and NCF2, respectively, with one compound common in both fractions. The characteristic Rf of the compounds was also obtained as shown in Figure 3. After that, HPTLC–MS was utilized to identify the compounds as stigmasterol (found m/z-579 with Rf 0.83), gallic acid (found m/z-160 with Rf 0.77), quercetin (found m/z-301 with Rf 0.63), 4-hydroxycoumarin (found m/z-164 with Rf 0.57), naringin (found m/z-579 with Rf 0.45), caffeine (found m/z-195 with Rf 0.31). Additionally, HPTLC densitograms of compounds were obtained utilizing the CAMAG HPTLC visualizer (Table 4). Further, the mass spectrum and associated peaks of all the compounds were obtained, which were authenticated from the NIST mass spectrum webbook (
Effect of Fraction F1 on the Different 3T3-L1 Cell Line Groups, Where Images were Captured at a Magnification of 40× Using a Phase Contrast Microscope (Scale Bar of 400 µm). (A) Control Group Exposed to Growth Media Only. (B) Indomethacin-induced Differentiation Group (30 µg/ml). (C) Standard Orlistat Treatment Group (20 µg/ml). (D) Test Fraction F1 Treatment at 50 µg/ml. (E) Test Fraction F1 Treatment at 100 µg/ml. Adipocytes and Lipid Droplets are Indicated by the Violet and Black Arrows, Respectively.
Compounds Identified in Bioactive Fraction, NCF1 and NCF2, with Their (m/z) Peaks, Respectively.
Conclusion
In the quest to overcome the escalating rate of obesity and its associated ailments, innovative interventions are imperative that demand a multidisciplinary therapeutic strategy that addresses the complexities of the ailment but also prioritizes safety concerns. While various pharmaceutical interventions exist, concerns regarding their adverse effects have spurred the exploration of natural remedies. One of them, a tropical medicinal plant NC, has provoked scientific curiosity due to its traditional medicinal uses and its rich phytochemical profile although its role in combating obesity remains unexplored until now (Mishra et al., 2023).
First, our study delves into the exploration of NC leaf fractions, harnessing their active compounds to combat obesity. Through a rigorous meticulous array of phytochemical analysis and in vitro antioxidant experiments utilizing DPPH, ABTS, NO2, and H2O2 assay, unveil the antioxidant potential of NCF, which is crucial in oxidative stress mitigation and one of the important hallmarks of obesity. Furthermore, our investigations extend to the enzyme inhibition assay for where selected fractions successfully inhibited alpha-glucosidase, pancreatic lipase, and alpha-amylase based on their IC50 values. These enzymes are crucial parameters of obesity pathophysiology specifically modulating adipogenesis and carbohydrate metabolism so inhibitors of these enzymes aid in the management of obesity (Pratap Singh & Pattnaik, 2024).
A further pivotal aspect of our research involves the 3T3-L1 adipogenesis differentiation model, which is a well-established model for adipocyte differentiation therefore specifically used in obesity research. The results demonstrated the excellent potential of potent fraction (NCF1) which exhibited antiobesity activity by significantly decreasing the accumulation of lipids and triglyceride levels in 3T3-L1 adipocytes as compared to other fractions, scrutinizing the impact of NCF on adipocyte behavior (Wang et al., 2023; Zhao et al., 2022).
Additionally, this study utilized sophisticated analytical techniques, including HPTLC–MS, to provide unprecedented insights into the pharmacological properties and chemical composition of NCF. This analytical technique facilitates the identification of lead compounds like stigmasterol, gallic acid, quercetin, 4-hydroxycoumarin, and naringin in bioactive fractions. Overall, the potent fraction NCF1 showed significant antiobesity activity in all the rigorous methodologies. Crucially, the research study extends beyond laboratory confines, encompassing the implications of our findings. Utilizing the natural remedy offered a holistic, therapeutic approach for the management of obesity interventions. The implications of our study utilizing NCF provide promising tangible benefits for obesity treatment. Based on the initial findings demonstrating the antiobesity potential of NC leaf bioactive fractions through in vitro studies targeting obesity-related pathways, further investigations will utilize in vivo animal models to explore molecular mechanistic pathways. Employing numerous assays assessing antioxidant activity, enzyme inhibition, cell line studies, and HPTLC–MS analysis, these in vitro studies have provided ample insights into the efficacy of NC bioactive fractions. The subsequent in vivo research will elucidate adipogenesis, carbohydrate metabolism, and other crucial aspects of obesity pathophysiology, aiming to validate and expand our understanding of NC’s antiobesity effects at a molecular level, thus evaluating its therapeutic potential for obesity management. Bridging the gap between the modern scientific approach and traditional wisdom paved the way for the formulation of innovative pharmaceutical products contributing to natural and safer therapy in obesity management.
Supplemental Material
Supplemental material is available for this article online.
Supplemental Material for Exploring Obesity Treatment Potential of Neolamarckia cadamba Bioactive Fraction: A Multifaceted Investigation Combining In Vitro, 3T3-L1 Adipogenesis Differentiation, and HPTLC–MS Techniques to Identify Lead Compounds by Shivangi Kumari and Ashok Kumar Pattnaik, in Pharmacognosy Magazine
Footnotes
Abbreviations
NC: Neolamarckia cadamba; NCF: Neolamarckia cadamba fractions; HPTLC: High-performance thin-layer chromatography; TPC: Total phenolic content; ABTS: 2,2’-Azino-bis3-ethylbenzothiazoline-6-sulfonic acid; QE: Quercetin equivalent; GAE: Gallic acid equivalent; BSE: β-Sitosterol equivalent; SEM: Standard error mean.
Acknowledgments
The authors are thankful to the Department of Pharmaceutical Sciences and Technology and Central Instrument Facility of Birla Institute of Technology, Mesra, Ranchi for providing all the necessary chemicals, equipment, and analytical tools for the study. The authors are also thankful to NCCS, Pune for providing the 3T3-L1 cell line and Eurofins Advinus Lab, Bangalore for providing their lab to conduct cell line studies.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
NA.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
