Abstract
Background
Molecular pathogenesis of obesity is initiated by cellular oxidative stress along with increased adipogenesis. For the search of better alternatives of synthetic molecules in terms of their adverse events, naturally, existing compounds are now a research focus. In this context, Xanthium strumarium (X. strumarium) Linn. exhibited anti-diabetic activities and proved to have efficacy in neutralizing very near pathological targets to obesity.
Purpose
This study is aimed toward bioactivity-guided fraction isolation of methanolic leaves extract, where bioactivity refers to anti-adipogenesis and antioxidant efficacy in in vitro enzymatic and three-day transfer, inoculum 3×105 cells (3T3-L1) adipocyte differentiation assay.
Materials and Methods
Collection and drying of the raw leaves followed by cold extraction in methanol and flash chromatography for bioactive fraction isolation. 3T3-L1 cell line was utilized for adipocyte differentiation assay of those and on the other hand in vitro pancreatic lipase, α-glucosidase, and α-amylase enzymatic assays were analyzed. Based on exhibited activities, selected fractions are further characterized by high-performance thin-layer chromatography-mass spectrometry (HPTLC-MS).
Results
The most active fractions from these assays underwent characterization via HPTLC-MS. Fractions 2 and 3 exhibited potent activities in all enzymatic assays (α-glucosidase IC50: 2.48 ± 0.015, 2.84 ± 0.030 µg/mL; α-amylase IC50: 1.98 ± 0.050, 1.79 ± 0.045 µg/mL; pancreatic lipase IC50: 3.16 ± 0.030, 3.18 ± 0.040 µg/mL) and displayed significant inhibition of adipocyte differentiation in 3T3-L1 cell line. These fractions further identified through HPTLC-MS, contained compounds like β-sitosterol and quercetin, affirming their potential bioactivity.
Conclusion
The study underscores the potential of X. strumarium Linn. fractions as promising natural alternatives for combating obesity-related pathways, highlighting their significance in developing future anti-obesity therapeutics.
Introduction
The escalating global obesity crisis aggravated by the COVID-19 pandemic entails substantial health risks linked to metabolic cardiovascular and psychosocial complications. Its correlation with heightened mortality underscores the imperative for efficacious treatments (Kwok et al., 2020). Although rare monogenic obesity syndromes do exist, multifactorial etiologies overwhelmingly predominate thereby complicating the intricacies of treatment and necessitating a personalized approach (Huvenne et al., 2016). Current interventions encompass lifestyle modifications behavioral therapies and in severe cases anti-obesity medications (AOM) or bariatric surgery. However, the history of AOM is marked by severe adverse effects necessitating stringent regulatory criteria. Consequently, there is a compelling need for innovative safer pharmacotherapies propelling intensive research endeavors in the pursuit of effective obesity medications (Saeed et al., 2014). The shortcomings of synthetic obesity medications are arising from the nuanced nature of obesity. Rimonabant, fenfluramine, and dexfenfluramine faced bans due to adverse effects. Healthcare providers underprescribe anti-obesity drugs (only 2% eligible receive prescriptions) influenced by stigma, misconceptions, and cost barriers, hindering their broader use despite FDA approval. The interplay of genetic predisposition and intricate metabolic processes presents a formidable challenge in crafting universally effective solutions (Cheung et al., 2013). Oxidative stress disrupts insulin secretion, impacting tissues through reactive oxygen species (ROS)-induced cellular damage. Elevated ROS generated during lipolysis, intensify oxidative stress, impair mitochondrial activity, and promote β-oxidation (Tangvarasittichai, 2015). The resulting NADH/NAD+ imbalance activates PKC, AGE, and NOX pathways, amplifying ROS production (Satyajit & Pattnaik, 2021). This surge affects endothelial function via NF-κB and TGF-β pathways, creating a pro-inflammatory environment. AGE accumulation activates NOX/NF-κB worsening cytokine synthesis and contributing to heightened adipogenesis in metabolic syndrome. This intricate molecular cascade involving PKC, AGE, and NOX pathways highlights oxidative stress’s central role in promoting increased adipogenesis (Giacco & Brownlee, 2010).
Xanthium strumarium Linn. (X. strumarium) an annual Asteraceae member is gaining scientific prominence for its diverse phytochemical composition. Its chemical constituents such as steroids, and caffeoylquinic acid derivatives exhibit potent inhibitory effects on aldose reductase a key enzyme in diabetic complications (Fan et al., 2019). Additionally, the plant targets α-glucosidase, PTP1β, and AGEs and demonstrates antioxidant activity suggesting potential therapeutic applications in diabetes mellitus (Lu et al., 2023; Shen et al., 2020). This demonstration of inhibitory effects on aldose reductase is a key player in diabetic complications but also a target factor relevant to obesity. This dual impact suggests that X. strumarium may be a multifaceted role in addressing both diabetes and obesity (Hwang et al., 2016). Research on three-day transfer, inoculum 3×105 cells (3T3-L1) adipocytes a prevalent model for adipogenesis offers crucial insights into obesity mechanisms. These cells offer a valuable platform to investigate the intricate mechanisms involved in obesity development and progression. By exploring processes such as adipocyte differentiation, lipid metabolism, and inflammatory responses in 3T3-L1 cells researchers gain essential insights into potential targets for obesity treatment (Etesami et al., 2020).
In this study, raw leaves were gathered and dried, then subjected to cold extraction using methanol. Following this, flash chromatography was utilized to separate and isolate bioactive fractions. The adipocyte differentiation assay was conducted on the 3T3-L1 cell line, while in vitro enzymatic assays for pancreatic lipase, α-glucosidase, and α-amylase were performed. Based on the exhibited activities, selected fractions underwent further characterization high-performance thin-layer chromatography-mass spectrometry (HPTLC-MS) was employed to isolate bioactive fractions.
Materials and Methods
Chemicals and Reagents
Methanol (R012J22), p-nitrophenyl butyrate (PNPB) (0055), orlistat (49923033), sulphuric acid (PC11211136), phosphate buffer, sodium buffer, Griess reagent, potassium hexacyanoferrate solution, hydrogen peroxide (7548260123), sodium nitroprusside, Griess reagent, naphthylethylenediamine di-hydrochloride (BKA073145).
Plant Material
Samples of X. strumarium leaves were gathered from the vicinity of Birla Institute of Technology, Mesra, and Ranchi Jharkhand. Authentication of the plant was done by the Scientist K. Karthigeyan in Central National Herbarium Howrah with Specimen No.—CNH/Tech11/2022/15.
Preparation of Extract
In the cold extraction procedure, coarsely powdered leaves of X. strumarium were placed in an airtight conical flask for continuous maceration with the chosen solvent methanol. The methanolic leaves extract was subsequently concentrated using a rotary evaporator (Singh et al., 2023). The concentrated extracts obtained were dried in petri plates and stored at room temperature within a desiccator. The final dried extract, acquired through this meticulous procedure, was carefully preserved for future utilization in in vitro studies (Nurcholis et al., 2021; Srinivas et al., 2011).
Bioactive Fractionations of the Extract
Following the cold maceration process, 5 g of the dried leaves extract underwent flash chromatography using Agela technologies with (Cat No: CN140020-0, Spec: NH2, 20 g with particle size—40–60 µm) to isolate and prepare bioactive fractions. A flash column was utilized with a flow rate set at 10 mL/min and a pressure of 150 psi. The fraction size was adjusted to 50 mL per tube leading to the collection of four bioactive fractions. Subsequently, these samples were stored at 5°C in preparation for in vitro analysis (Keshava et al., 2020).
Phytochemical Analysis
Thin Layer Chromatography (TLC) Analysis
A meticulous TLC was conducted on the collected bioactive fractions derived from X. strumarium. In pursuit of the optimal separation of compounds within these isolated fractions, diverse solvent mixtures were judiciously employed on the TLC plate. This methodological approach aimed to unveil the intricate composition of the bioactive components, providing a nuanced insight into the chemical profile of X. strumarium. The thoughtful selection of solvent mixtures underscores the precision and thoroughness of the analytical process contributing to the refinement of the investigation into the pharmacological attributes of this botanical specimen (Sahoo et al., 2020).
Total Flavonoid Content
The total flavonoid content in fractions FI, F2, F3, and F4 was determined using the aluminum chloride colorimetric method as outlined in the journal by (Singh & Pattnaik, 2023).
Total Phenolic Content
The total phenolic content in each bioactive fraction was determined using the Folin-Ciocalteu method, as described by (Hatami et al., 2014) with gallic acid as the standard. Initially, 5 mg of each fraction was dissolved in 5 mL of ethanol. Then, 2.5 mL of Folin-Ciocalteu’s reagent was added along with 0.1 mL of the samples. Subsequently, 1 mL of the mixture was added, and the sample was incubated at 40°C for 30 minutes before being transferred to a microplate reader for measurement of absorbance at 750 nm.
Total Steroidal Content
The total steroidal content was quantified following the protocol outlined by (Ramya & Dhamotharan, 2015) utilizing a ferric chloride colorimetric assay. A 10 mg portion of the extract was used to which 2 mL of 4N sulfuric acid was added followed by 2 mL of ferric chloride solution (0.5% w/v). Then 0.5 mL of the resulting sample was mixed with 0.5 mL of potassium hexacyanoferrate solution (0.5% w/v) and the mixture was heated in a water bath set at 70°C ± 2°C for 30 minutes with occasional agitation. The final absorbance was measured at 780 nm using a microplate reader.
In Vitro Antioxidant Activity
Trolox Equivalent Antioxidant Capacity Assay (2,2′-Azino-bis3-ethylbenzothiazoline-6-sulfonic acid [ABTS])
The antioxidant capacity was evaluated by measuring the scavenging ability against ABTS following the protocol described by (Suseela et al., 2010).
DPPH (2,2-Diphenyl-1-Picrylhydrazyl) Scavenging Assay
The evaluation of free radical scavenging activity using the DPPH-hydrate assay was carried out following the protocol described by (Polu et al., 2017). First, 2 mL of the test sample dissolved in methanol was mixed with 2 mL of a 0.04 mg/mL DPPH solution containing various concentrations of bioactive fractions (F). These mixtures were then incubated in darkness at room temperature for 30 minutes following which the absorbance at 515 nm was measured using a microplate reader.
Nitric Oxide Radical Scavenging Assay
The methodology for quantifying nitric oxide radicals generated by sodium nitroprusside was conducted following the procedure outlined by (Pandian et al., 2015). In this method, a reaction mixture containing 5.0 mL of sodium nitroprusside (5 mM) in phosphate-buffered saline at pH 7.3 was incubated at 25°C for 3 hours along with varying concentrations of bioactive fractions. During the incubation period, nitrite ions were diazotized and coupled with sulfanilamide and naphthylethylenediamine dihydrochloride to produce a purple azo dye, serving as an indicator of nitric oxide radical production. Absorbance readings were recorded at 546 nm to quantify the generated chromophore.
Hydrogen Peroxide Scavenging Assay
The assessment of the extract’s capacity to neutralize hydrogen peroxide (H2O2) was conducted according to the protocol outlined by (Shabbir et al., 2013). In this process, 0.1 mL aliquots from extract concentrations ranging between 25 and 400 g/mL were placed into Eppendorf tubes. The volume was then adjusted to 0.4 mL using 50 mM phosphate buffer (pH 7.4) following the addition of 0.6 mL of a 2 mM H2O2 solution. After vortexing, the reaction mixture’s absorbance was measured at 230 nm following a 10-minute reaction time. This method enables the quantitative assessment of the extract’s capability to scavenge hydrogen peroxide.
Enzyme Inhibition Assay
α-Glucosidase Inhibition Assay
In the α-glycosidase enzyme inhibition assay following the method described by (Telagari & Hullatti, 2015) microplate wells were prepared with a phosphate buffer. Each well received 50 µL of the buffer and 10 µL of α-glycosidase solution. Various concentrations (0.5, 1, 1.5 mg/mL) of isolated fractions were then added and the mixture was incubated at 37°C for 15 minutes. Thereafter 20 µL of a 5 mM p-nitrophenyl-β-d-glucopyranoside (P-NPG) solution was introduced and the reaction was allowed to continue for an additional 20 minutes at the same temperature. The enzymatic reaction was halted by adding 50 µL of 0.1 M sodium carbonate solution. Absorbance at 405 nm was measured with acarbose used as a standard for comparison. This approach effectively evaluated the α-glycosidase enzyme inhibition potential of the tested fractions across different concentrations offering valuable insights into their inhibitory activity.
α-Amylase Assay
The α-amylase assay was conducted following the methodology outlined by (Venkatachalam & Muthukrishnan, 2012).
Pancreatic Lipase Assay
According to (Zhang et al., 2008) the literature review describes a study in which an enzyme solution was prepared at a concentration of 0.1 mg/mL in 0.1M Tris HCl (pH 8). Test samples were obtained from isolated fractions at concentrations of 0.5, 1, and 1.5 mg/mL. Subsequently, 30 µL of each test sample was mixed with 180 µL of enzyme buffer and incubated at room temperature for 15 minutes. After this initial incubation, the samples underwent an additional 15 minutes of incubation at 37°C following the addition of 5 µL of 10 mM PNPB. The final step involved measuring absorbance at 405 nm using a microplate reader with orlistat serving as the standard drug. This methodically conducted protocol allowed for a thorough investigation of the inhibitory effects of the test samples on enzyme activity enabling comparison with the standard drug, orlistat.
HPTLC-MS Analysis
Subsequent to the execution of in vitro antioxidant and enzyme inhibition assays, the resultant data underwent meticulous statistical scrutiny and comparative evaluations for the identification of the most efficacious bioactive fractions. Selected fractions underwent further analysis through HPTLC-MS to delineate their chemical composition. Utilizing 10 × 10 cm 60 F254 aluminum-backed silica gel plates for compound separation, test samples were applied employing an automatic (TLC) sampler (CAMAG) with specified parameters. The development process employed a toluene: ethyl acetate (6:4) solvent system and the resultant plates run up to 90 mm underwent subsequent drying and derivatization with anisaldehyde solvent. For HPTLC-MS analysis, a parallel procedure was implemented omitting the derivatization step. The developed plates were subjected to molecular mass determination on an MS interface (CAMAG TLC-MS Interface 2) using a Thermo-Scientific LTQ-XL mass spectrometer. This methodological rigor facilitated a comprehensive exploration of the chemical constituents within the bioactive fractions, with two images captured using CAMAG TLC Visualizers for subsequent analytical purposes (Osman et al., 2021).
3T3-L1 Cell Line Differentiation Assay
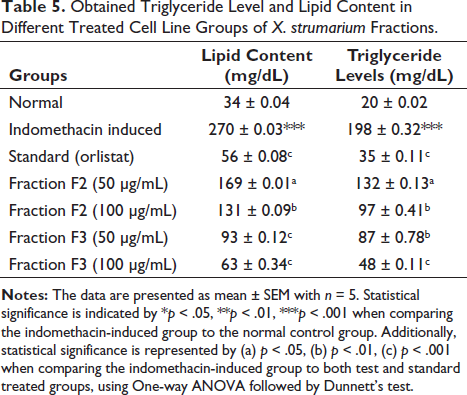
The 3T3-L1 cell line differentiation assay was meticulously conducted using the Hi Media Adipogenesis Kit. 3T3-L1 cells were obtained from NCCS Pune, India (Request No-587/2023-24). Initially, 3T3-L1 preadipocytes were cultured in Dulbecco’s Modified Eagle Medium supplemented with 10% FBS and antibiotics until reaching 70% confluence. After that, the cells were seeded in 24-well plates at a density of 5 × 10^5 cells per well in 200 s of cell suspension. Upon reaching the desired confluence, the growth medium was substituted with differentiation media 1 containing various concentrations (50, 100, 200, 300, and 500 µg/mL) of treatment and standard drugs. Incubation transpired for precisely 48 hours followed by the addition of differentiation medium 2 and another 48 hours incubation period. Control groups remained untreated with differentiation media receiving only growth media throughout the experiments outlined in the kit. Following successful differentiation induction cells underwent treatment with control media over a defined period typically spanning six to eight days with media changes administered at 48 hours intervals. This process was meticulously monitored until distinct differentiation became apparent. Post-differentiation, cells were stained with specific markers Oil Red O to visualize lipid droplet formation a characteristic of adipocyte differentiation. Images were captured using a phase-contrast microscope. Quantification of adipogenesis was conducted using assays provided in the kit, and resulting data were subjected to rigorous analysis to ascertain the extent of 3T3-L1 cell differentiation into adipocytes. Additionally, triglyceride levels were quantified from scraped cells using the triglycerides quantification kit (Sigma Aldrich).
Computational Transformation and Image Analysis
The computational transformation of the 3T3-L1 adipocyte differentiation assay images into color-enhanced diagrams involved several steps. First, the black and white BMP images, representing phase-contrast microscopy of Oil Red stain-treated samples, were loaded using OpenCV. These images were then processed to identify black spots, indicative of adipocyte differentiation. Using Open CV’s thresholding techniques, the grayscale images were converted into binary format to isolate the black spots. Contours were detected around these spots, employing the find Contours function, enabling identification based on their areas. A minimum threshold for spot size (10 pixels, for instance) was set to ensure accuracy in marking the relevant spots. To visually emphasize these spots in the output, a copy of the input grayscale image was converted to a color (BGR) format. Black spots were then marked in red using contour drawing functions, resulting in a color-enhanced diagram showcasing the areas of interest in vibrant red.
Statistical Analysis
All experiments were conducted in triplicate, except for the determination of total phenol, flavonoid, and steroid content. The statistical significance of the data was assessed using a one-way analysis of variance (ANOVA) with Dunnett’s test, performed with GraphPad Prism 8.3.1 software. Results are presented as mean ± standard error mean (SEM). A significance level of p < 0.05 (*), p < 0.01 (**), and p < 0.001 (***) was considered statistically significant, while “ns” indicates non-significance.
Results
Phytochemical Analysis
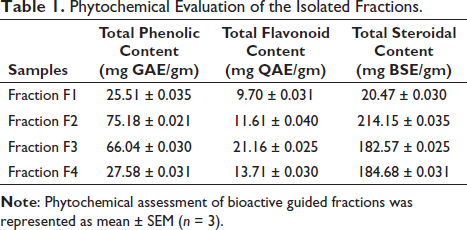
The phytochemical analysis involved assessing four bioactive fractions (FI, F2, F3, and F4) using various quantitative tests. The total phenolic content of these fractions was determined to be 25.51 ± 0.035, 75.18 ± 0.021, 66.04 ± 0.030, and 27.58 ± 0.031 mg gallic acid equivalent/gm, respectively. For total flavonoid content, fractions F1, F2, F3, and F4 exhibited values of 9.70 ± 0.031, 11.61 ± 0.040, 21.16 ± 0.025, and 13.71 ± 0.030 mgQAE/gm, respectively, using Quercetin as the standard. Similarly, the total steroidal content of the bioactive fractions, measured using β-sitosterol equivalent (BSE) as the standard, yielded values of 20.47 ± 0.030, 214.15 ± 0.035, 182.57 ± 0.025, and 184.68 ± 0.031 mg BSE/gm for fractions F1, F2, F3, and F4, respectively, taking BSE as standard as shown in Table 1.
Phytochemical Evaluation of the Isolated Fractions.
Antioxidant Activities
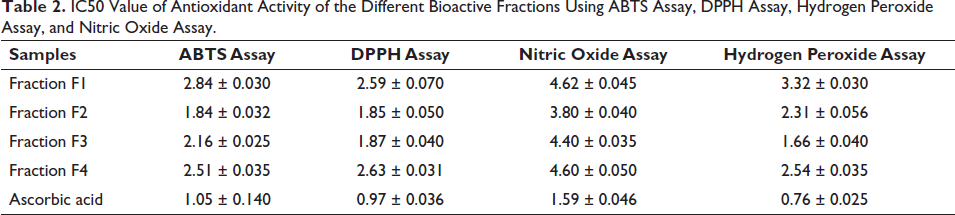
Four different antioxidant assays (ABTS, DPPH, H2O2, and NO2 assays) were utilized to evaluate and compare the antioxidant capacities of specific bioactive fractions and to elucidate their modes of action. Fractions 2 and 3 demonstrated the most potent DPPH scavenging activity with IC50 values of 2.59 ± 0.07, 1.85 ± 0.05, 1.87 ± 0.04, and 2.63 ± 0.031 µg/mL, respectively. In the ABTS assay, fraction F1, F2, F3, and F4 showed IC50 values of 2.84 ± 0.030, 1.84 ± 0.4, 2.16 ± 0.025, and 2.51 ± 0.035 µg/mL, respectively. For the NO2 assay, fractions 2 and 3 exhibited the most potent antioxidant activity with IC50 values compared to other fractions: 4.62 ± 0.042, 3.80 ± 0.04, 4.40 ± 0.035, and 4.60 ± 0.05 µg/mL. Regarding the H2O2 scavenging assay, fractions F2 and F3 showed the most potent scavenging activity among the other fractions, with values of 3.32 ± 0.03, 2.31 ± 0.05, 1.66 ± 0.04, and 2.54 ± 0.035 µg/mL, respectively. Ascorbic acid was utilized as the standard for all antioxidant activities. The overall results revealed that fractions 2 and 3 showed the most potent activity compared to other fractions as shown in Table 2.
IC50 Value of Antioxidant Activity of the Different Bioactive Fractions Using ABTS Assay, DPPH Assay, Hydrogen Peroxide Assay, and Nitric Oxide Assay.
Enzyme Inhibition Assay
The efficacy of individual bioactive fractions was evaluated through α-glucosidase, α-amylase, and pancreatic lipase assays. The percentage inhibition activity of the individual fractions at various concentrations (0.1, 0.3, 0.5, 0.7, 0.9 µg/mL) was determined. In the α-glucosidase assay, fractions F2 and F3 displayed the most potent inhibition activity, with IC50 values of 3.12 ± 0.035, 2.48 ± 0.015, 2.84 ± 0.030, and 3.22 ± 0.040 µg/mL, respectively. Similarly, in the α-amylase assay, fractions F2 and F3 exhibited the highest inhibition activity, with IC50 values of 2.89 ± 0.055, 1.98 ± 0.050, 1.79 ± 0.045, and 2.24 ± 0.025 µg/mL, respectively. The pancreatic lipase inhibition assay results revealed that fractions F2 and F3 had higher potency than other fractions based on their IC50 values of 3.36 ± 0.045, 3.16 ± 0.030, 3.18 ± 0.040, 4.16 ± 0.035 µg/mL, respectively, as shown in Table 3.
Enzyme Inhibition Assay of the Different Bioactive Fractions Using Pancreatic Lipase, α-Glucosidase, and α-Amylase Activity.
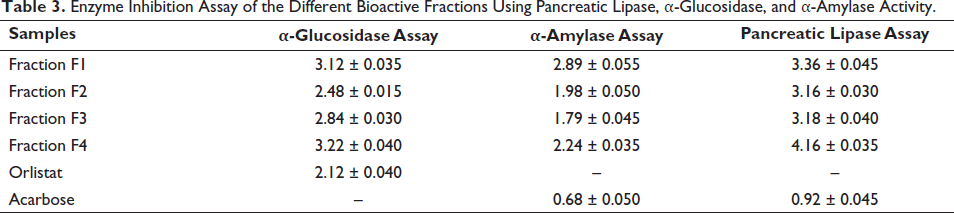
The enzyme inhibition assay results (IC50 µg/mL) of bioactive-guided fractions were presented as mean ± SEM (n = 3). Statistical significance was denoted by *p < 0.05, **p < 0.01, ***p < 0.001, and “ns” for not significant when compared to all other groups using one-way ANOVA followed by post hoc Tukey test.
HPTLC and HPTLC-MS
The study utilized HPTLC-MS data to intricately characterize and identify compounds leveraging molecular mass information acquired from TLC MS interface 2 and the identified compounds are described in Table 4 and the MS data has been provided in the Supplement File Figures S1–S6. The elegant application of HPTLC delineated the separation of six distinct compounds within both the fractions F2 and F3 as provided in the Supplement File Figure S7. A discerning eye was cast upon the realm of bioactivity culminating in the selection of the most potent bioactive fraction. This decision was judiciously made by evaluating the fractions through the lens of in vitro anti-oxidant and enzymatic assays. Within the tapestry of the selected potent fractions (fractions 2 and 3), HPTLC-MS unveiled the presence of a total of six major compounds. Notably amid this chemical symphony, a harmonious similarity resonated between the fractions β-sitosterol, stigmasterol, and quercetin akin to celestial constants having both fractions in their presence. This orchestration of analytical prowess not only unveiled the molecular tapestry of compounds but also spotlighted the shared presence of key elements within the rich composition of the potent fractions.
(m/z) Values of Compounds Derived from Fractions 2 and 3.
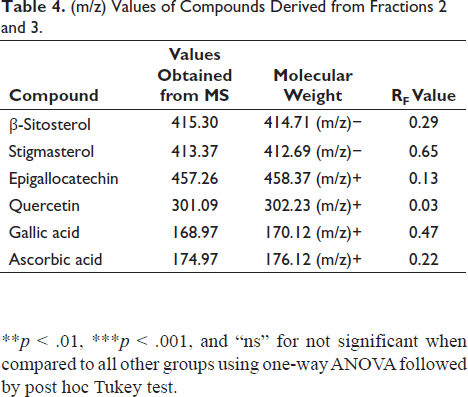
The antioxidant assay results (IC50 µg/mL) of bioactive-guided fractions were presented as mean ± SEM (n = 3). Statistical significance was indicated by *p < 0.05, **p < 0.01, ***p < 0.001, and “ns” for not significant when compared to all other groups using one-way ANOVA followed by post hoc Tukey test.
Dose Dependent 3T3-L1 Toxicity Study
Based on the comprehensive insights gained from in vitro phytochemical analyses, antioxidant assays, and enzyme inhibition investigations, it was apparent that fractions F2 and F3 exhibit promising potential for anti-obesity activity. Encouraged by these compelling results, we proceeded to conduct an in vitro cell line study using the 3T3-L1 differentiation assay. In the initial stages of the investigation, a thorough evaluation of the fractions’ toxicity was essential. The cytotoxicity assessment of the bioactive fractions, administered at various concentrations (50, 100, 200, 300, and 500 µg/mL) in the 3T3-L1 adipogenesis differentiation assay, revealed dose-dependent effects on cell viability. Notably, fractions administered at concentrations exceeding 100 µg/mL demonstrated significant cytotoxicity, leading to a notable decrease in cell viability and eventual cell death. These findings unequivocally emphasize a substantial and evident toxic effect associated with the tested fractions when used at concentrations higher than 100 µg/mL. Consequently, exercising caution is imperative when considering the utilization of these fractions at elevated concentrations. Moreover, further investigations are warranted to unravel the underlying mechanisms contributing to this observed toxicity. This nuanced toxicity profile underscores the critical importance of dose optimization and safety considerations in the potential therapeutic application of these fractions.
Adipocytes Differentiation Assay
The 3T3-L1 differentiation study unraveled a captivating outcome in both the fractions F2 and F3 exhibiting a striking inhibitory effect on cell line differentiation when compared to both the control and indomethacin-induced groups. Notably, the presence of fractions F2 and F3 at a dosage of 100 µg/mL demonstrated a significant dose-dependent inhibition of lipid accumulation within the 3T3-L1 adipocytes. Remarkably this effect was coupled with an impeccable safety profile as evidenced by the absence of any discernible toxicological implications throughout the comprehensive experimental investigations as shown in Figure 1. Under microscopic scrutiny, fractions F2 and F3 treated with 100 µg/mL revealed minimal and diminutive lipid droplets as indicated by the black arrows, while the yellow arrowheads pointed to the adipocytes providing a visual testament to the reduced lipid content. However, even at a dose of 50 µg/mL fractions F2 and F3 exhibited a noticeable reduction in adipocyte differentiation. Furthermore, the efficacy of fractions F2 and F3 extended beyond mere visual cues as reflected in the marked decrease in lipid counts and triglyceride levels observed throughout the study, as elucidated in Table 5 and Figure 1. This comprehensive set of findings solidifies fractions F2 and F3 status as a potent candidate in the realm of interventions against obesity. Computational transformation of diagrams also supported the interpretation in Figure 2.
Images of 3T3L1 Cells Where Images were Captured at a Magnification of 40× Using a Phase Contrast Microscope (Scale Bar of 400 µm) (Oil Red O Staining). Confluent Cells were Treated with (A) Control Group Exposed to Growth Media Only and (B) Indomethacin Induced Differentiation Group (30 µg/mL). (C) Orlistat (STD) Treatment Group (20 µg/mL). (D–G) Test Fractions F2 and F3 Treatment at 50 and 100 µg/mL.
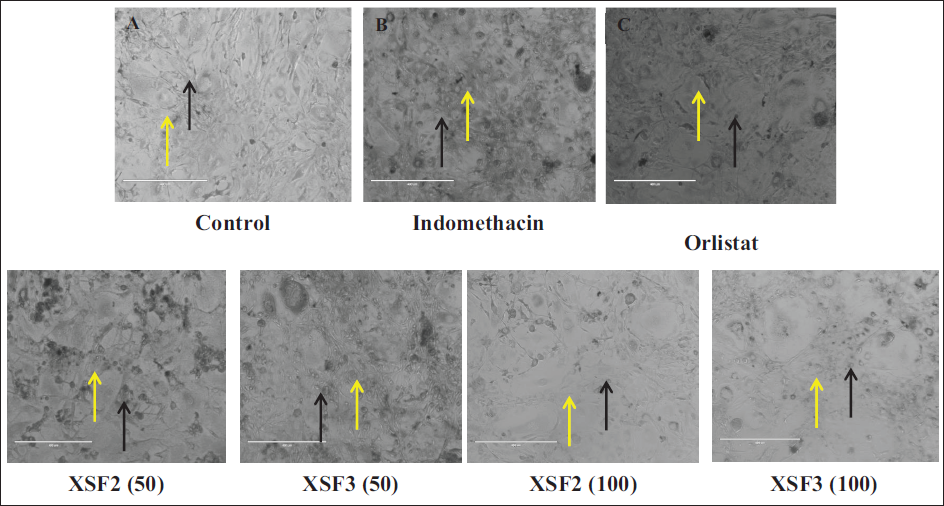
Images of 3T3L1 Cells Where Images were Captured at a Magnification of 40× Using a Phase Contrast Microscope (Scale Bar of 400 µm) (Oil Red O Staining). Confluent Cells were Treated with (A) Control Group Exposed to Growth Media Only (B). Indomethacin Induced Differentiation Group (30 µg/mL) (C). Orlistat (STD) Treatment Group (20 µg/mL). (D–G) Test Fractions F2 and F3 Treatment at 50 and 100 µg/mL.
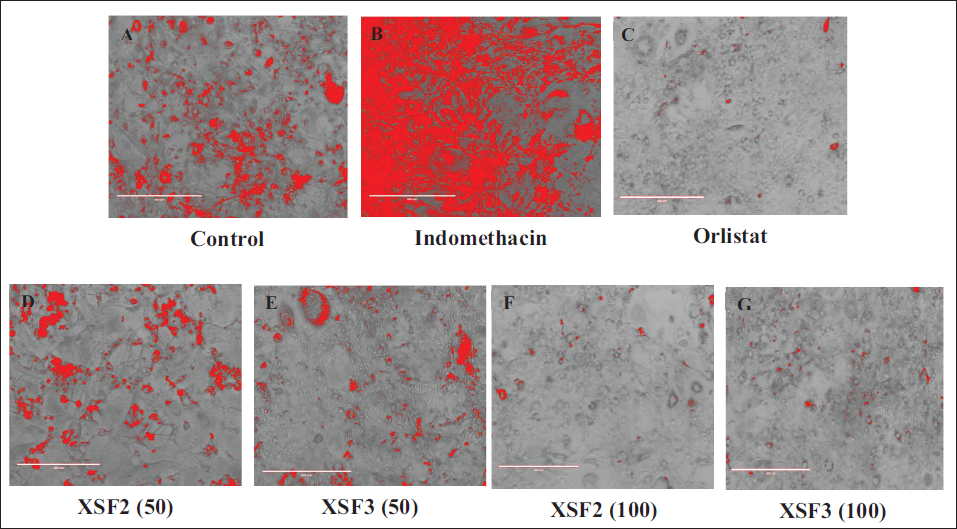
Obtained Triglyceride Level and Lipid Content in Different Treated Cell Line Groups of X. strumarium Fractions.
Discussion
This study explores the pharmacologically active compounds present in X. strumarium leaves focusing on their potential anti-obesity properties. The investigation involves isolating and evaluating bioactive fractions from the leaf extract, and assessing their phytochemical composition, antioxidant capabilities, and enzyme inhibition activities (Gligor et al., 2023). Phytochemical analysis revealed the presence of phenolic compounds, flavonoids, and steroidal content in X. strumarium leaves with notably high concentrations observed in fractions 2 and 3. Phenolic and flavonoid compounds are known for their ability to scavenge free radicals, suggesting their potential in combating oxidative stress. In vitro antioxidant assays, including DPPH, ABTS, nitric oxide, and H2O2 radical scavenging activities, showed significant antioxidant effects of fractions 2 and 3 compared to other bioactive fractions. The elevated antioxidant potential correlates with the high phenolic and steroidal content in these fractions, indicating potential health benefits. Examination of enzymatic inhibition activities, particularly against pancreatic lipase, α-amylase, and α-glucosidase, revealed potent inhibitory effects of fractions 2 and 3. This is relevant in terms of triglyceride digestion and carbohydrate hydrolysis, suggesting a potential role in addressing processes associated with obesity. HPTLC-MS analysis identified and characterized six major compounds, with three compounds common to both potent fractions. Noteworthy compounds such as ascorbic acid, epigallocatechin, gallic acid, quercetin, β-sitosterol, and stigmasterol were confirmed to be present in X. strumarium leaves. The efficacy of bioactive fractions 2 and 3 were further demonstrated in antioxidant assays, with IC50 values ranging from 1.85 ± 0.24 to 2.18 ± 0.34 µg/mL for DPPH, 1.87 ± 0.32 to 2.63 ± 0.35 µg/mL for ABTS, and 2.35 ± 0.34 µg/mL to 1.68 ± 0.44 µg/mL for nitric oxide and H2O2 assays. These fractions also exhibited substantial phenolic, flavonoid, and steroidal content, along with significant inhibitory activity against key enzymes involved in obesity. Further investigation using the 3T3-LI adipocyte differentiation assay evaluated fractions 2 and 3 at concentrations of 50 and 100 µg/mL. Comparative analysis against control, indomethacin, and Orlistat treatment groups provided insights into the potential influence of bioactive components on adipogenesis. These findings contribute to a deeper understanding of X. strumarium anti-obesity potential, laying a foundation for future therapeutic strategies targeting metabolic disorders.
Conclusion
This research sheds light on the crucial role of oxidative stress in the complex development of obesity, highlighting its correlation with the generation of ROS. The hypothesis proposing the reduction of oxidative stress and the inhibition of key enzymes (α-glucosidase, α-amylase, and pancreatic lipase) as potential strategies for combating obesity was thoroughly examined. Particularly, fractions 2 and 3 of X. strumarium emerged as promising candidates, displaying exceptional effectiveness in phytochemical composition, antioxidant scavenging, and enzyme inhibition assays. However, careful consideration is necessary as concentrations exceeding 100 µg/mL exhibited significant cytotoxicity in the 3T3-L1 adipogenesis differentiation assay. This toxicity profile underscores the essential requirement for dose optimization and safety evaluations in harnessing the therapeutic potential of these fractions. Moreover, further investigations are imperative to uncover the underlying mechanisms contributing to the observed toxicity.
Summary
Amidst the global obesity crisis, driven by lifestyle shifts and environmental factors, attention turns to X. strumarium leaves. Employing a meticulous cold maceration extraction process a promising product was obtained, initiating an exploratory journey. Preliminary screenings and TLC optimization identified Toluene: Ethyl acetate (6:4) as the optimal solvent system. Guided by these insights flash chromatography isolated bioactive fractions, with fractions 1 and 2 excelling in subsequent analyses of phytochemical content, antioxidant activities, and enzymatic assays. HPTLC identification highlighted β-sitosterol and stigmasterol, emphasizing the plant’s pharmacological potential. This compelling venture beckons further investigation, positioning X. strumarium as a noteworthy contender in the pursuit of obesity mitigation. Notably, fractions F2 and F3 at 100 µg/mL, exhibited a significant dose-dependent inhibition of lipid accumulation in 3T3-L1 adipocytes without toxicity confirming their potency against obesity.
Abbreviations
3T3-L1: Three-day transfer, inoculum 3 × 105 cells; ABTS: 2,2′-Azino-bis3-ethylbenzothiazoline-6-sulfonic acid; ANOVA: Analysis of variance; AOM: Anti-obesity medications; BSE: β-sitosterol equivalent; DPPH: 2,2-Diphenyl-1-picrylhydrazyl; GAE: Gallic acid equivalent; H2O2: Hydrogen peroxide scavenging assay; HPTLC: High-performance thin-layer chromatography; PNPB: p-Nitrophenyl butyrate; SEM: Standard error mean; STD: Standard; X. strumarium: Xanthium strumarium.
Footnotes
Acknowledgments
The authors would like to thank the Department of Pharmaceutical Sciences and Technology and CIF (Central Instrumentation Facility) BIT Mesra, Ranchi for providing the Research facilities for this Research article. I am thankful to Satyajit Mohanty and Tuhin Mukherjee for their valuable suggestions.
Author’s Contributions
The collaborative effort of all authors enriched this work. NN conducted material preparation, tests, data collection, and analysis. The initial draft was crafted by NN with valuable input from AP, who played a pivotal role in conceptualization, extensive editing, and critical revision of the manuscript. Each author meticulously reviewed and endorsed the final version.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Statement of Consent
Not applicable.
Funding
This work was supported by the National Fellowship for Tribal Affairs (NFST) with award no: 202122-NFST-JHA-00489.
Supplementary Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
