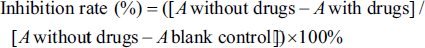Abstract
Background
Gliomas are highly lethal malignancies that develop in the central nervous system. The primary treatment for gliomas involves surgical resection followed by chemoradiotherapy. However, due to the infiltrative growth nature of gliomas, surgical resection is often incomplete. Moreover, the efficacy of chemotherapeutic drugs is constrained by their ability to cross the blood–brain barrier, and the currently utilized agents can lose effectiveness, particularly with prolonged administration. Lentinan, an active compound in Lentinula edodes, exhibits various pharmacological activities.
Purpose
This study aims to investigate the anti-tumor effects of lentinan on glioma U251 cells.
Materials and Methods
Cell proliferation assays, cell fluorescence staining, scratch healing experiments, and transwell chamber experiments were conducted to assess the anti-tumor activity of lentinan on U251 cells. Additionally, quantitative real-time polymerase chain reaction (qPCR) and Western blot experiments were performed to validate the anti-tumor mechanism of lentinan.
Results
The findings revealed that lentinan significantly suppressed the proliferation of U251 cells, induced robust apoptosis, and decreased the cells’ migration and invasion capabilities. Furthermore, lentinan notably influenced the gene and protein expression of p53, Bcl-2, Cyto-c, Bax, Caspases, and MMP-9 in U251 cells.
Conclusion
These findings suggest that lentinan may inhibit glioma cells by activating P53 and caspase-related apoptosis pathways.
Introduction
Gliomas are highly malignant intracranial tumors that present a significant threat to human health (Wang et al., 2023). Traditionally, gliomas are classified into four grades by the WHO, with the prognosis worsening as the grade increases (Gao et al., 2016). Currently, the preferred treatment strategies for gliomas involve extensive surgical resection followed by systematic chemotherapy and/or radiotherapy (Spina et al., 2023). Gliomas typically exhibit low compatibility with radiotherapy (van den Bent et al., 2023), making postoperative chemotherapy essential. However, the development of effective anti-glioma chemotherapeutic agents has been slow due to the presence of the blood–brain barrier. Temozolomide is among the most recommended first-line chemotherapy agents (Kao et al., 2023), but long-term administration often leads to chemotherapy resistance (Rezaee et al., 2023). Despite aggressive treatment approaches, the 5-year survival rate for glioma patients following surgery remains limited to date (Brown et al., 2019; Grimm & Chamberlain, 2013).
The diverse activities and multiple targets of natural compounds offer promising avenues for the prevention and treatment of diseases, including brain tumors (Wang et al., 2021; Zhu et al., 2022). Lentinula edodes, a well-regarded mushroom in both medical and nutritional realms (Breene, 1990), is abundant in various bioactive components such as proteins (Ann et al., 2014), polysaccharides (Xiang et al., 2022), and various small molecules (Ohnuma et al., 2000; Roszczyk et al., 2022). The exploration of the pharmacological properties of L. edodes remains a prominent focus in scientific research (Fau et al., 2016; Górska-Jakubowska et al., 2021; Kaleta et al., 2019, 2021).
Lentinan (LTN), a polysaccharide containing β-(1,3)-
Previously, LTN was reported to inhibit U251 cell proliferation and induce apoptosis, but the potential mechanism of cell apoptosis was not discussed (Yuan et al., 2023). In our study, we investigated the anti-glioma effects of LTN on U251 cells.
Materials and Methods
Materials
LTN (S23110, 96%, Shanghai, China) was purchased from Yuanye Bio-Technology. Cell proliferation was monitored by Cell Counting Kit-8 provided by MedChemExpress (Shanghai, China). Acridine orange/ethidium bromide (AO/EB) and 4′,6-diamidino-2-phenylindole (DAPI) were purchased from KeyGEN BioTECH (Nanjing, China).
Cell Culture
The glioma cell line U251 and DMEM medium were purchased from Procell Life Science (Wuhan, China). The cells were cultured in produced with 10% fetal bovine serum and 100 µg/mL of Penicillin–Streptomycin. The incubator was set to 37°C and filled with 5% CO2. When the cell growth density reaches 85% of the culture dish, cell passage is performed. After the cells reach the logarithmic growth phase, subsequent experiments will be conducted.
Cell Proliferation Assay
Glioma cells (4 × 103 for each well) were cultured in 96-well plates with 200, 400, and 800 µg/mL of LTN for 24–72 h (Zhao et al., 2016). 5 µM Adriamycin hydrochloride (DOX) was selected as positive control culture. Then, the inhibition rate was calculated as following formula:
Colony Formation Unit Assay
Glioma cells (3×102/well) were cultured in a 6-well plate with 200, 400, and 800 µg/mL of LTN for 21 days. When cells are cultured more than six times to form a visible cell community, the culture can be stopped. Wash twice with phosphate-buffered saline (PBS), stain with crystal violet solution for 15 min, wash twice with PBS again, take photos under a microscope, and then count the community for statistical analysis (Wan et al., 2020).
Nuclear Fluorescence Staining
Cells (2×104/well) were cultured in a 6-well plate with 200, 400, and 800 µg/mL LTN. After washing with PBS, 5 mg/mL DAPI was added. The cells were washed with PBS after 10 min incubation. For AO/EB staining, the procedure was similar to the DAPI staining, but the dye solution was 100 µg/mL AO/EB. After staining, clean with PBS and take pictures under an inverted fluorescence microscope. Take six photos per hole, with three parallel holes in each group.
Scratch Wound Healing Assay
U251 cells (2×104/well) were inoculated in a 6-well plate with 200, 400, and 800 µg/mL of LTN. The wound healing was observed for 72 h. After scratching, the control group cells were replaced with a new culture medium, while the treatment group cells were replaced with LTN. Capture the migration of cells toward the scratch site at the same location every day. Using transwell chamber experiments to statistically analyze the number of cells migrating through the membrane and the impact of LTN on cell migration.
Transwell Analysis
Cells (105/well) were seeded in the chambers of a 24-well plate with LTN and cultured for 48 h. Photos were taken as described elsewhere (Wan et al., 2020). In the transwell chamber experiment, matrix gel was added into the chamber, and 104 cells were inoculated using LTN medium. Add culture medium containing LTN and 10% FBS to the lower chamber. After 72 h of cultivation, wash twice with PBS, stain with crystal violet solution for 15 min, and take pictures of the small chamber membrane under an inverted microscope. Take five photos of each room, with three parallel rooms. Finally, calculate the number of cells crossing the chamber and statistical analysis.
Quantitative Real-time Polymerase Chain Reaction (qPCR)
The primers of qPCR are as follows: β-actin: 5′-AGAGCT ACGAGCTGCCTGAC-3′, 5′-AGCACTGTGTTGGCGTACAG-3′; Bax: 5′-CAAACTGGTGCTCAAGGCCC-3′, 5′-GAGAC AGGGACATCAGTCGC-3′; Bcl-2: 5′-CGGTTCAGGTACT CAGTCATC-3′, 5′-CGGTGGGGTCATGTGTGTG-3′; P53: 5′-GTTCCGAGAGCTGAATGAGG-3′, 5′-TTATGGCGGGA GGTAGACTG-3′; Caspase-3: 5′-CAAACTTTTTCAGAGGGGATCG-3′, 5′-GCATACTGTTTCAGCATGGCA-3′; MMP-9: 5′-CAGACCTTTGAGG GCGAACT-3′, 5′-TACCCATCTCCG TGCTCTCT-3′. Reaction conditions are as follows: 95°C for 5 min, 40 cycles of 95°C for 30 s → 55°C for 45 s → 72°C for 30 s, and 72°C for 10 min.
Western Blotting
Protein samples (50 µg) were added to the well. Anti-β-actin, anti-Bcl-2, anti-cytochrome c, anti-Bax, anti-caspase-3/8/9, anti-MMP-9, and anti-p53 were purchased from ABclonal (Wuhan, China). The working concentrations of these primary antibodies were all 1:1,000. The secondary antibody was diluted to 1:5,000 for use.
Statistical Analyses
All data were expressed as mean ± standard error of the mean (SEM) and analyzed using IBM SPSS17.0 (New York, USA). Statistical analysis was conducted by using one-way analysis of variance (one-way ANOVA) and t-test. Each experiment was performed in at least three biological replicates.
Results
Effect of LTN on the Proliferation Ability of U251 Cells
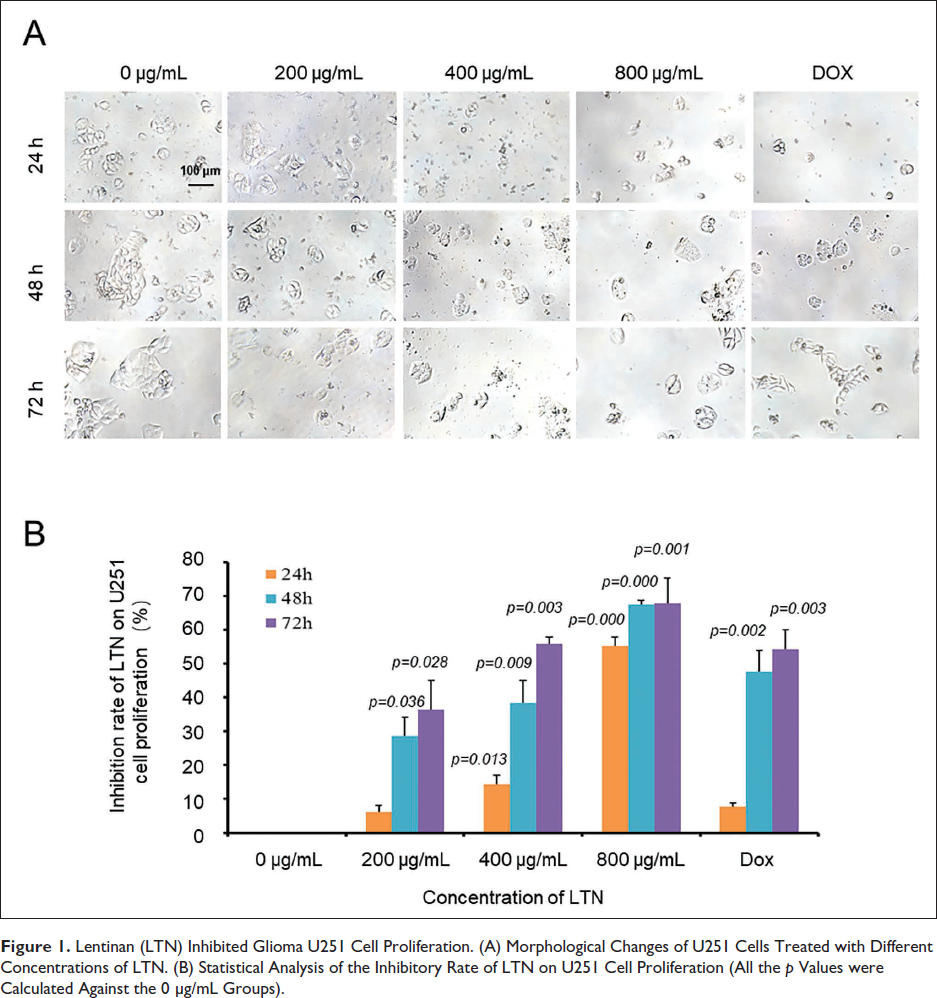
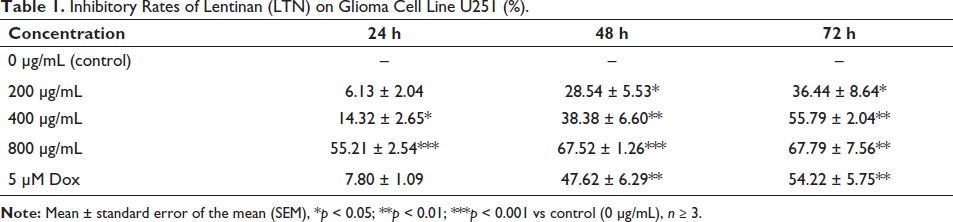
When treated by LTN, U251 cells exhibited significant morphological changes characterized by cell shrinkage and fragmentation in the culture media (Figure 1A). These features were potentiated in line with the increased LTN concentrations and the incubation time, indicating a time- and dose-dependent inhibition property (Figure 1B, Table 1). For 24 h culture, the calculated IC50 was 749.17 ± 31.14 µg/mL. When it came to 48 h culture, the IC50 was as low as 391.30 ± 20.46 µg/mL (Table 2). Under our selected conditions, when the concentration of LTN was 400 µg/mL and the incubation time was 48 h, the inhibitory rate of LTN on U251 cells was nearly comparable with that of the Dox control.
Lentinan (LTN) Inhibited Glioma U251 Cell Proliferation. (A) Morphological Changes of U251 Cells Treated with Different Concentrations of LTN. (B) Statistical Analysis of the Inhibitory Rate of LTN on U251 Cell Proliferation (All the p Values were Calculated Against the 0 µg/mL Groups).
Inhibitory Rates of Lentinan (LTN) on Glioma Cell Line U251 (%).
IC50 of Lentinan (LTN) on Glioma Cell Line U251.

Effect of LTN on the Colony-forming Ability of Glioma Cells
Colony-forming ability can not only be used to investigate the proliferation ability of tumor cells in situ but also indirectly indicate the ability of tumor cells to form new lesions in a new environment. In this study, the colony-forming ability of U251 cells was negatively correlated to the concentrations of LTN (Figure 2). The number of colonies in the absence of LTN was 85.67 ± 4.55 (Table 3). When the LTN was 800 µg/mL, the newly formed cell colonies numbers decreased by nearly 80 folds (Figure 2).
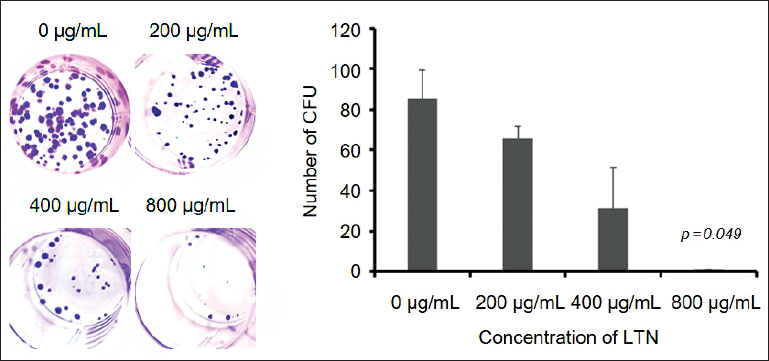
The Colony-forming Unit (CFU) of Glioma Cell Line U251 Under Different Concentrations of Lentinan (LTN).

Effect of LTN on the Cell Apoptosis of U251
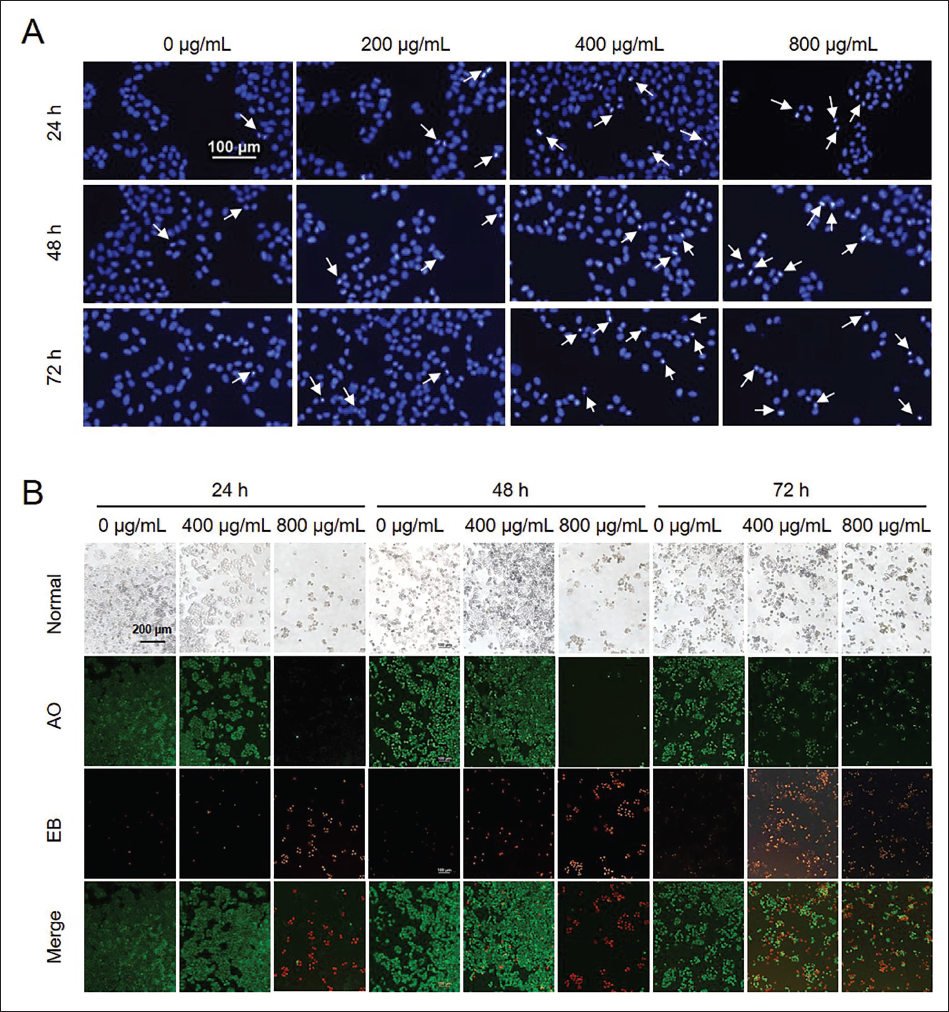
Previously, it was reported that LTN could induce apoptosis of glioma cells (Yuan et al., 2023). In this study, we used nuclear fluorescence staining to reconfirm the effect of LTN on the cell apoptosis of U251 (Figure 3A). A large number of cells with green pyknotic nuclear chromatin were detected, indicating the conspicuous proportion of cells in the early stage of apoptosis (Figure 3B). At the same time, there were also many cells with round bead-like orange chromatin, indicating the presence of cells in the late apoptotic stage. Meanwhile, with the elongation of incubation time and the increase of LTN concentrations, both the two stages of apoptotic cells increased. Thus, the U251 cells showed a time- and dose-dependent apoptotic feature as challenged by LTN.
Nuclear Fluorescence Staining of Glioma Cell Line U251 Treated with Lentinan (LTN). (A) 4′,6-Diamidino-2-Phenylindole (DAPI) Staining Results. (B) Acridine Orange/Ethidium Bromide (AO/EB) Staining Results. Arrows in (A) Indicate the Apoptotic Bodies.
Effect of LTN on the Cell Migration and Invasion of U251
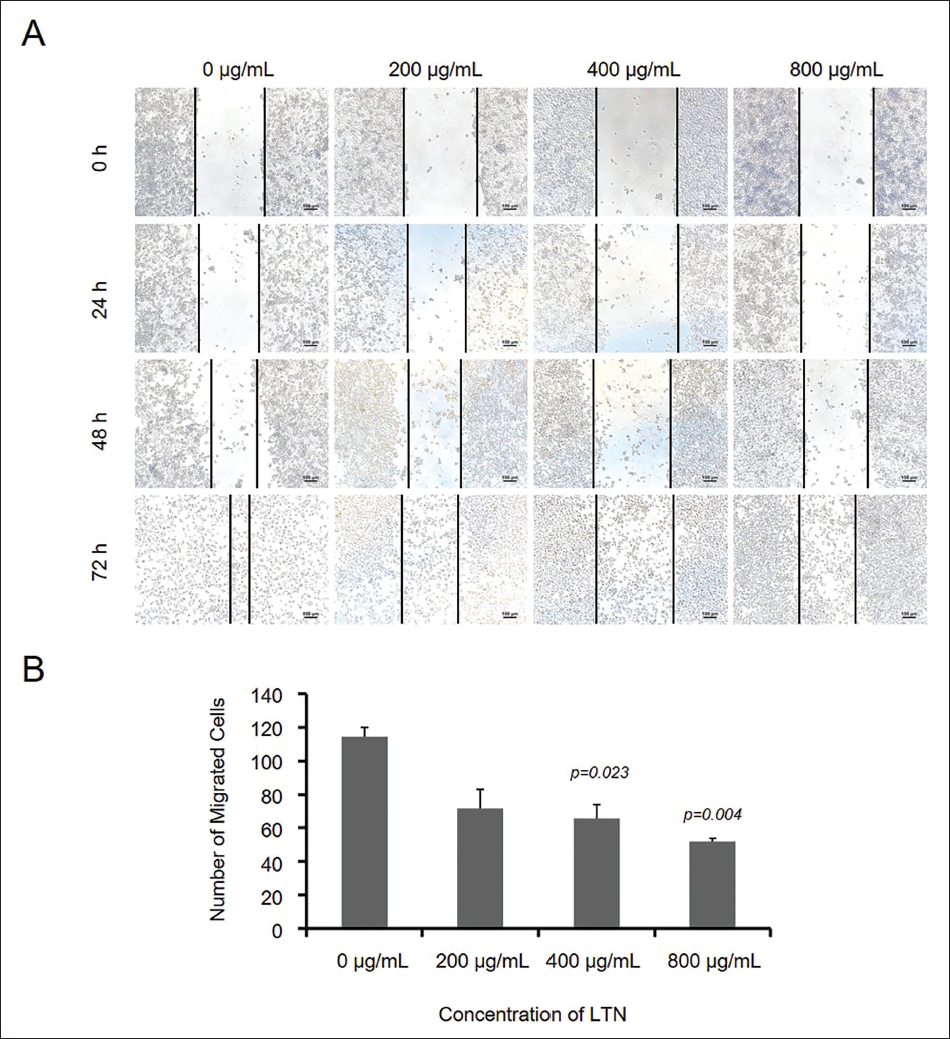
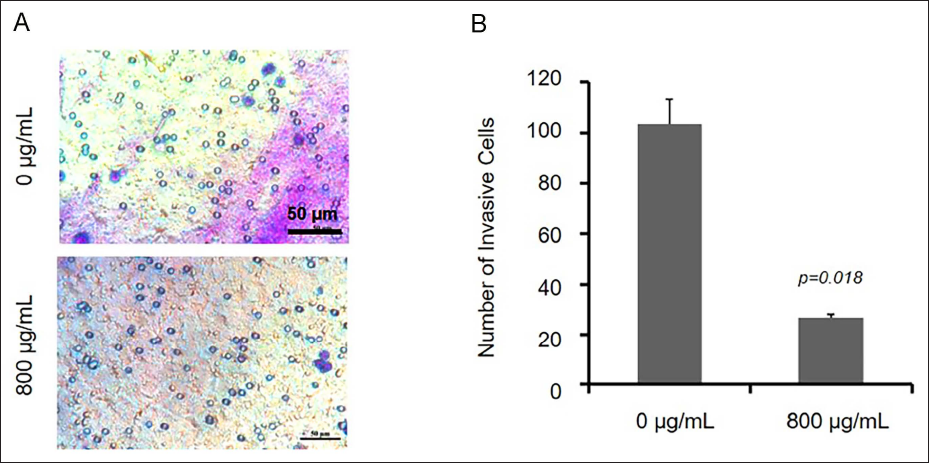
The cell migration ability is utilized to evaluate whether a tumor cell has metastatic ability. In the presence of LTN, U251 cells grew toward the middle zones to different extents after 24–72 h (Figure 4, Table 4). Under the presence of LTN, the invasion ability of U251 cells was monitored in the Matrigel chambers. The invasion of U251 cells was inhibited significantly at 48 h (Figure 5, Table 5). It could be concluded that LTN inhibited the migration and invasion ability of glioma cells (Figure 5).
Effect of Lentinan (LTN) on Migration Ability of U251 Cells Treated with LNT (0–800 µg/mL). (A) The Wound Healing Photos Taken at Different Time. (B) Statistic Analysis of the Migration Cell Numbers (All the p Values were Calculated Against the 0 µg/mL Groups).
Migration of Glioma Cells Affected by Lentinan (LTN) at 48 h.
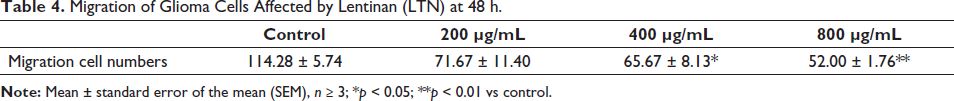
Effect of Lentinan (LTN) on Invasion Ability of U251 Cells Treated with 0 and 800 µg/mL LNT. (A) The Photos of Transwell Results. (B) Statistical Analysis of the Results of (A).
Migration and Invasion of Glioma Cells Affected by Lentinan (LTN) (48 h).

Effects of LTN on Apoptosis-related Genes and Protein Expression
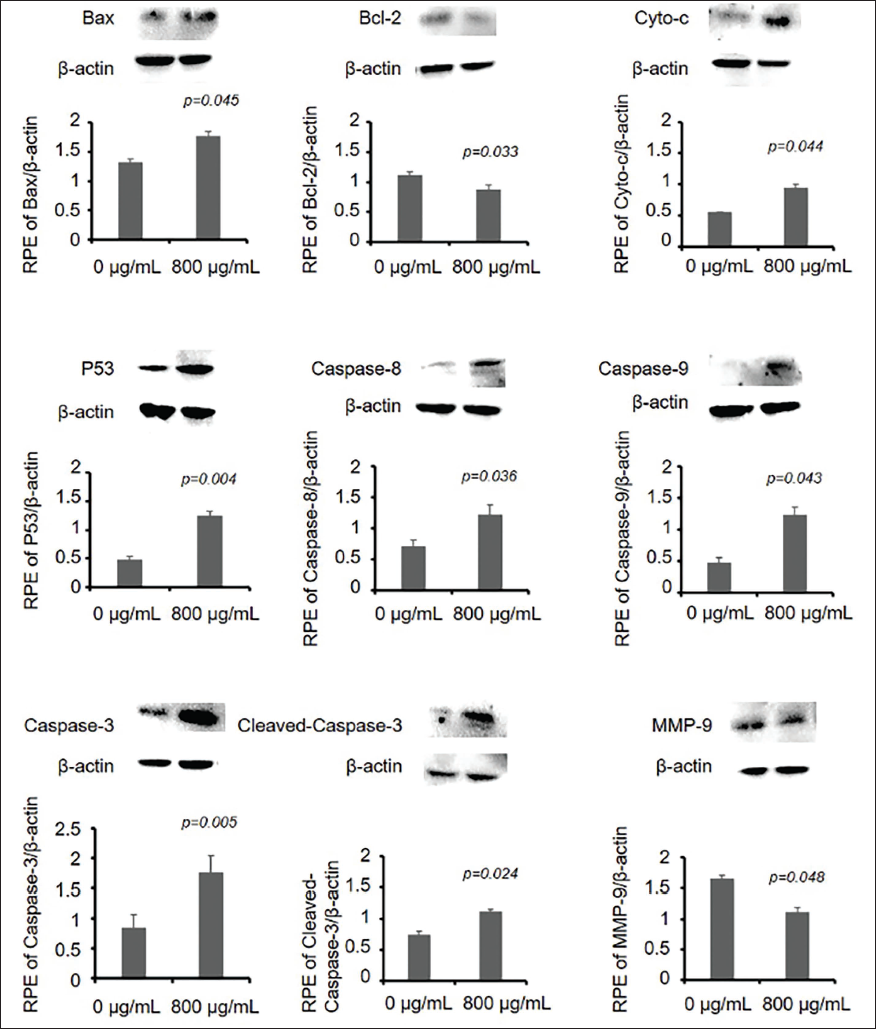
In order to analyze the mechanism of effect of LTN on glioma cells, we first screened several key genes (Bax, Bcl-2, Caspase-3, MMP-9, and P53) for phenotypes with significant changes and tested the regulatory effect of LTN on gene expression in glioma cells. The results are shown in Table 6, where p53, Bax, and Caspase-3 genes are upregulated, while Bcl-2 and MMP-9 genes are downregulated. It can be seen that LTN is involved in the expression of these key genes, thereby regulating the protein expression of corresponding genes.
Effect of Lentinan (LTN) Treatment on Genes of U251 Cells.
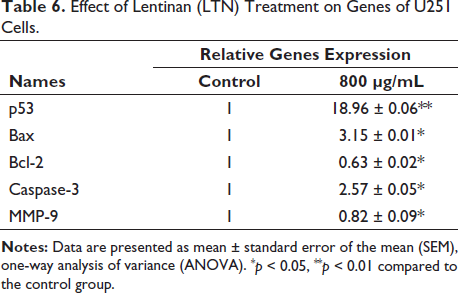
The mitochondrial proteins related to apoptosis, such as Bax, Cyto-c, Bcl-2, as well as p53 and caspase family members (Caspase-3/8/9), were detected for U251 cells after 800 µg/mL LTN treatment. A significant expression difference of these apoptosis-related proteins was observed compared with control group. Bcl-2 was recognized as an antagonist complementary to c-Myc to block P53-dependent apoptosis (Bruckheimer et al., 1998). Its expression was greatly inhibited by LNT, indicating the alleviating of P53 inhibition (Figure 5). MMP-9, a member of the mechanism metalloproteinase family (MMP) that dominates cell migration and invasion, was found to be significantly downregulated, indicating the contribution of LTN on inhibiting cell migration and invasion by affecting the expression of MMP-9 (Figure 5) potentially. Except for Bcl-2 and MMP-9, the other pro-apoptosis proteins were all upregulated by LTN. Collectively, these findings suggested that LTN exerted anti-glioma effects through the pathway involving caspases and p53 in potential.
Discussion
Glioma is currently an extremely serious type of brain disease worldwide (Yang et al., 2022). At present, complete removal of the tumor tissue is still a challenge, and postoperative interventions are indispensable (Hervey-Jumper & Berger, 2019). However, the efficacy of chemotherapy and biological modulators is still debatable (Chen et al., 2018; Taal et al., 2015). Recently, exploring natural compounds for tumor treatment attracted much attention (Wang et al., 2021). Research has found that medicinal fungi, as nutritional supplements, were beneficial to patients with gliomas (Gargano et al., 2021).
The most important anticancer and immunomodulatory components in fungi are polysaccharides. Some studies have reported that polysaccharides from fungi have a notable inhibitory effect on gliomas. It was found that polysaccharide peptides from Ganoderma lucidum could result in cell cycle arrest and promote cell apoptosis of U251 in vitro (Wang et al., 2018). Grifola frondosa polysaccharide peptides were proved to synergistically contribute to cyclophosphamide and exerted an inhibitory effect on C6 cells (Chan et al., 2011). Polysaccharides of Inonotus obliquus could also effectively inhibit the proliferation of U251 cells by downregulation of Bcl-2 and upregulation of caspases (Ning et al., 2014). These results indicate that polysaccharides are a valuable fungal active ingredient with development value as an adjuvant anti-cancer agent.
In recent years, research has found that the anti-tumor effect of LTN is very significant, especially in terms of inhibiting the migration and invasion ability of human tumor cells, such as colon cancer (Zhang et al., 2021), lung cancer (Qi et al., 2021), liver cancer (Wang et al., 2017), gastric cancer, and ovarian cancer (Jiang et al., 2021; Ina et al., 2013). The possible mechanisms included mediating tumor cell death and apoptosis through immune modulation and oxidative stress. Of note, LTN was demonstrated to be harmless to the body without perceivable toxic effects (Huang et al., 2023). In this light, commercial injection products have been developed and administrated to some kinds of tumor patients (Dong & Wang, 2022). Our study found that the highest inhibition rate of LTN approached 75.53% (72 h), which was significantly higher than the broad-spectrum chemotherapy drug doxorubicin hydrochloride (Gu et al., 2015; Lu et al., 2017). At the same time, the IC50 of LTN on U251 cells was only 391.30 ± 20.46 µg/mL (48 h), which was also superior to the commonly studied polysaccharides (Chan et al., 2011; Ning et al., 2014; Wang et al., 2018).
The p53 gene is an extremely important tumor suppressor gene in the eukaryotes. It also plays an important role in regulating apoptosis. LTN significantly upregulated the expression of p53 after LTN stimulation. The downstream apoptotic proteins of p53 also showed overexpression changes (Figure 6). p53-mediated apoptosis could be blocked at multiple death checkpoints (Shen & White, 2001). The most notable mechanisms were releasing of cytochrome c and the activation of the Caspase cascades (Wang et al., 2014).
Bax and Bcl-2 belong to the B-cell lymphoma/leukemia two families. They not only serve as upstream regulators of Caspase-3 and participate in its activity regulation but also act as direct substrates of Caspase-3. Most members of the Caspases family are involved in programmed cell death. Several studies have confirmed that the inhibitory effect of natural polysaccharides on glioma cells is linked to programmed cell death mechanisms associated with Bcl-2 and Bax. Therefore, the apoptosis of glioma cells following LTN treatment can be attributed to the p53-mediated mitochondrial apoptosis pathway (Bruckheimer et al., 1998; Gao et al., 2023; Hao et al., 2023). In future research, it is necessary to further explore the direct target(s) of LTN.
Effect of Lentinan (LTN) on Some Apoptosis-related Proteins’ Expression in U251 Cells at 48 h.
According to the analysis of research results, it can be understood that LTN can significantly inhibit the proliferation of glioma U251 cells. Morphological observation revealed that cells treated with LTN exhibited morphological abnormalities, including cell shrinkage and inability to spread and grow at the bottom of the culture dish. Otherwise, CCK8 was used to quantitatively analyze the number of cells. As the concentration and duration of LTN increased, the inhibitory effect on cell proliferation became more significant. Further analysis of nuclear fluorescence staining revealed that cell apoptosis became more pronounced. It can be seen that LTN inhibits cell proliferation by inducing tumor cell apoptosis. Subsequently, this study showed that LTN can inhibit cell migration and invasion significantly. Finally, the key targets of LTN on these phenotypes were validated at the gene and protein levels through qPCR and Western blot (WB) experiments. Among them, p53 is a key gene for cell apoptosis, and significant changes were detected in the key proteins of mitochondrial apoptosis, Cyto-c, Bax, and Bcl-2. The expression of downstream caspase family members, Caspases, was significantly increased, indicating that LTN activates the process of cell apoptosis by inducing the expression of caspase. However, the mechanism of action of LTN still needs further in-depth research. It was found that LTN-induced U251 cell apoptosis was through a p53-dependent Caspase pathway.
LTN is the earliest fungal polysaccharide component used in clinical practice. It is made into shiitake mushroom polysaccharide injection and is studied and applied as an anticancer adjuvant in some diseases and postoperative treatment of tumors. However, the treatment of glioma has not yet been carried out. Through our research, we have demonstrated the inhibitory effect of LTN on glioblastoma.
The blood–brain barrier is a protective barrier that separates the blood plasma from the brain cells, created by the capillary walls of the brain and glial cells. Additionally, there is a barrier between the blood plasma and cerebrospinal fluid formed by the choroid plexus. These barriers serve to block the entry of certain substances, primarily harmful ones, from reaching the brain tissue from the bloodstream. In the realm of glioma treatment, the crucial focus lies in the capacity to effectively target and breach the blood–brain barrier in order to combat brain tumors. Our research has revealed promising results regarding the efficacy of LTN in combating gliomas, presenting a significant breakthrough in the realm of glioma prevention and treatment. In future studies, we can use animal experiments to demonstrate whether LTN can successfully pass through the blood–brain barrier and the molecular mechanisms by which it acts through the blood–brain barrier.
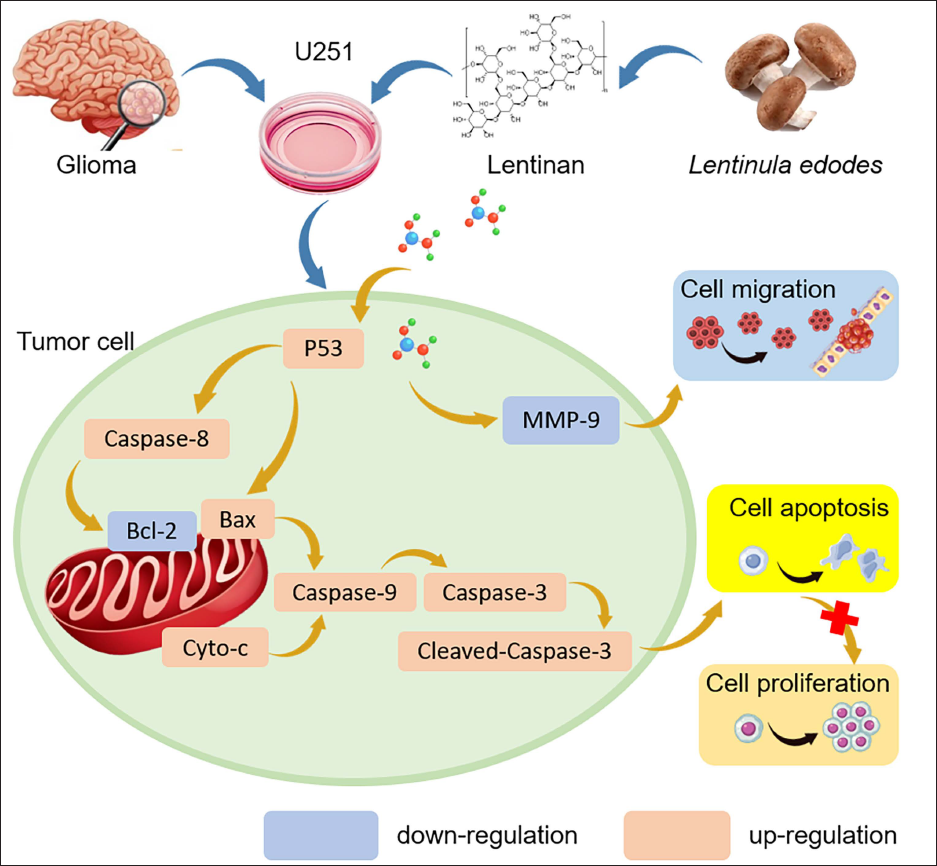
For high molecular weight drugs, the transportation potential across the blood–brain barrier is an important concern. Unfortunately, we had no direct evidence that LTN could easily diffuse or be transported across the blood–brain barrier. It was reported that intraperitoneal administration of LNT could alleviate the symptoms of an inflammatory demyelinating disease of the CNS (Zhang et al., 2022). This might imply that LNT had the potential to enter the cerebrospinal fluid. It was highly warranted that the question could be solved in the near future. The potential mechanism of LNT against gliomas is summarized in Figure 7.
The Potential Mechanisms of Lentinan (LTN) Antagonizing Glioma Cells.
Conclusion
In this study, we have shown that LTN can effectively suppress the proliferation of U251 cells. It can induce varying levels of cell apoptosis through pathways related to p53 and Caspase. These results could pave a new way to treat gliomas by using natural compounds.
Abbreviation
LTN: Lentinan.
Footnotes
Acknowledgments
We would like to express our gratitude to the persons in Xu’s lab.
Author Contributions
Conceptualization, F.X. and J.W.; software, X.L.; validation, Y.S. and P.G.; formal analysis, X.W.; methodology, investigation and resources, data curation, project administration, X.W.; writing—original draft preparation, Y.S.; writing—review and editing, F.X.; visualization, J.W.; supervision, P.G. and F.X.; funding acquisition, F.X. and X.W. All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Science and Technology Development Plan Project of Jilin Province (20230508166RC) and the Program of Science and Technology Research Project of Education Department of Jilin Province (JJKH20241091KJ).