Abstract
Background
Glioma is a recurrent form of primary malignant cancer that occurs in the brain and central nervous system. The adults are the major victims of gliomas, and the men are mostly affected. At present, gliomas are treated with surgical resection followed by radiation and the administration of chemodrugs such as temozolomide. The invasive and high recurrence rate of gliomas often reduces the effectiveness of treatment, which leads to poor recovery. Hence, more potent drugs without side effects on long-term treatment need to be discovered to treat gliomas. Morin is one such promising phytochemical that possesses immense pharmacological properties. It is proven to exert anti-inflammatory, antioxidant, anticancer, antibacterial, antidiabetic, and neuroprotective properties. The current research focuses on examining the growth inhibition and apoptosis-inducing potency of morin against glioma C6 cells.
Materials and Methods
Rat C6 glioma cells were treated with different concentrations of morin and analyzed for cytotoxicity using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) assay. The intracellular reactive oxygen species (ROS) production and alteration of mitochondrial membrane potential (MMP) by morin in glioma cells were examined with dichlorodihydrofluorescein diacetate (DCHF-DA) and rhodamine 123 staining. The apoptotic induction in glioma cells was analyzed with dual staining and it was confirmed by quantifying the apoptotic protein using the enzyme-linked immunosorbent assay (ELISA) technique. The anti-inflammatory property of morin was assessed by the inflammatory cytokines tumor necrosis factor-α (TNF-α), nuclear factor-kappa B (NF-κB), cyclooxygenase-2 (COX-2), and interleukin-6 (IL-6) using the ELISA technique.
Results
Our MTT results show that morin treatment significantly reduced the cell viability of rat C6 glioma cells. It also significantly increased ROS generation and decreased MMP in rat C6 glioma cells. Morin treatment also effectively increased caspase-3 and caspase-9, the proapoptotic protein Bax, and decreased the antiapoptotic protein Bcl-2. The induction of apoptosis by morin in glioma cells was evident with our acridine orange/ethidium bromide (AO/EtBr) staining results. Morin also decreased the inflammatory cytokine levels in rat C6 glioma cells.
Conclusion
Our findings have proven that morin significantly induces apoptosis by increasing the levels of apoptotic protein via the generation of ROS. It also effectively reduced inflammatory cytokine levels, thereby exerting anticancer activity against glioma cells. Therefore, morin can be subjected to further research to be designed as a potent anticancer drug to treat gliomas.
Introduction
Gliomas, the primary tumors that originate from the neuroglial stem cells, are responsible for the majority of deaths related to brain tumors. Gliomas are a diffusive infiltrative type of tumor with an increased progressive nature. Gliomas were classified into grades based on histopathology, molecular and genetic markers, and proliferation rate (Jiang et al., 2017). The World Health Organization (WHO) graded gliomas into four categories based on their infiltrative and proliferative nature (Shahcheraghi et al., 2019). Among the glioma types, glioblastoma is the most malignant type of cancer, and the least malignant was to be pilocytic astrocytoma (Mesfin & Al-Dhahir, 2023). In adults, gliomas account for 75% of primary malignant brain tumors, with an incidence among men and especially among the white race (Davis, 2018, Ostrom et al., 2022). The global incidence differs based on epidemiology where China, the United States, and India tend to be in the first three positions. The incidence rate of brain tumors has drastically increased in the past 30 years (GBD, 2016; Jiang et al., 2021).
The prime diagnostic tools for gliomas are neurological examination and histological examination, which were performed to grade the tumor (Kowalczyk et al., 2020; Morgan, 2015). The severity and symptoms of gliomas differ based on the location, size, and type of gliomas. The symptoms were systemic, which causes cognitive decline, motor dysfunction, language disorder, memory deficits, and so on, based on the location of gliomas. About 80% of the patients experience seizures, and these are more often reported in young patients than in old (Posti et al., 2015; Rossetti & Stupp, 2010). The symptoms are not only the cause of the impairment of quality of life in glioma patients but also the treatment provided (Giulioni et al., 2014; Lapointe et al., 2018). Even though surgical resection is a very tedious option since the location is an immensely delicate region of the body, it is the prime option for glioma patients. Additionally, the patients were treated with radiotherapy and chemotherapy drugs such as temozolomide (Nabors et al., 2017). Cognitive impairment, encephalopathy, cerebellar dysfunction, and spinal cord toxicity are the adverse side effects experienced by glioma patients treated with current therapies (Raisa & Marhaendraputro, 2019). Hence, the need for alternatives to treat glioma is urgent in the healthcare field.
Chemoprevention is an upcoming and promising treatment in the field of oncology. Treating patients with natural or synthetic agents to prevent, suppress, or reverse carcinogenesis in the initial phase of malignancy is the prime goal of this treatment. At present, the Food and Drug Administration (FDA) has approved about 10 drugs as chemopreventive drugs; among these, natural-based phytochemicals are the most prominent (Ranjan et al., 2019; Vogel et al., 2010). Phytomedicines are widely accepted alternative medicines to treat various acute and chronic diseases (Gong et al., 2020). More than 5000 herbal medicines are sold all around the world, and their demand is increasing due to their minimal or nil toxicity and easier access than chemical drugs (Patwardhan et al., 2005; Rajput et al., 2021).
Morin, 3,5,7,2′,4′-pentahydroxyflavone, is one such phytochemical isolated from the plants of the Moraceae family (Caselli et al., 2016). Morin possesses immense pharmacological properties like anticancer (Nandhakumar et al., 2012; Sivaramakrishnan & Niranjali Devaraj, 2009), antioxidant (Hyun et al., 2015; Lee et al., 2017; Rizvi et al., 2015), anti-inflammatory (Yu et al., 2020), neuroprotective (Bachewal et al., 2018; Komirishetty et al., 2016), nephroprotective (Jonnalagadda et al., 2013), antidiabetic (Paoli et al., 2013), hepatoprotective, cardioprotective, and so on (Verma et al., 2019). Morin effectively protects against LPS-induced bone loss (Park et al., 2022) and type-2 diabetes (Lawal et al., 2022). However, its salutary activity on the glioma has not been addressed yet. Therefore, in this study, we analyzed the antitumorogenic effect of morin against the glioma cell line in vitro.
Materials and Methods
Chemicals
Morin was purchased from Sisco Research Laboratory, Mumbai. The medium and reagents required for culturing glioma cells, such as Dulbecco’s Modified Eagle Medium (DMEM), heat-inactivated fetal bovine serum (FBS), trypsin–ethylenediaminetetraacetic acid (EDTA), and antibiotic/antimycotic solution, were procured from Sigma Aldrich. Staining dyes such as dichloro-dihydro-fluorescein diacetate, rhodamine 123, acridine orange (AO), and ethidium bromide (EtBr) were also obtained from Sigma Aldrich, USA.
Culture Collection
C6 rat glioma cells were purchased from the American Type Culture Collection (ATCC) for the present study. Upon arrival, the cells were examined for contamination and confluency under the microscope. After confirming the cells were not contaminated and were adherent to the cell culture flask, the growth medium was changed and incubated at 37°C in a 5% CO2-supplemented incubator. The growth medium was supplemented with 10% FBS and 1% antibiotic/antimycotic solution. The cells were trypsinized upon attaining 80% confluency using a trypsin–EDTA solution and subcultured for further experiments.
MTT Assay
Briefly, 1 × 104 C6 rat glioma cells per well were seeded in a 96-well culture plate and treated with different concentrations of morin ranging from 5 to 100 µM/mL. The cells were then incubated at 37°C in a CO2-supplemented incubator for 24 h. The medium was changed to DMEM medium without FBS supplementation and incubated for 4 h. Twenty microliters of 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide (MTT), consisting of 5 mg/mL, were added to the cells and then further incubated for 4 h at 37°C. Formazan crystals were further dissolved using 180 µL of DMSO solution. The plates were placed on the rocker for 15 min to complete the dissolution of formazan crystals. Absorbance was read at 570 nm using the microplate reader. The percentage of viable cells was calculated.
Assessment of ROS Generation
The reactive oxygen species (ROS) generated by morin on glioma cells were assessed using DCFH-DA stain. C6 glioma cells (3 × 105)/well were seeded in the six-well culture plate and incubated at 37°C in a 5% CO2-supplemented incubator for 24 h. Morin (25 and 50 µM/mL) was added to the cells and further incubated for 24 h. After incubation, the cells were then treated with 25 µM of DCFH-DA and incubated for 20 min. The fluorescence emission of the cell was read using a fluorescence microplate at an excitation of 485 nm and an emission of 530 nm. The intensity of the fluorescence was measured.
Assessment of MMP Level
The depolarization of mitochondrial membrane potential (MMP) in glioma cells upon treatment with various concentrations of morin was examined using the rhodamine 123 staining technique. The 25 and 50 µM/mL morin-treated and untreated control glioma cells were stained with the lipophilic cationic dye rhodamine 123 and incubated in the dark for 30 min. The fluorescence emission of the cells was read using a fluorescence microplate reader at an excitation of 485 nm and an emission of 530 nm. The intensity of the fluorescence was measured.
Assessment of Apoptosis Induction
The induction of apoptosis by morin on C6 rat glioma cells was examined using the dual staining technique. AO and EtBr stains were used for this staining to examine the apoptotic morphological changes induced by morin on glioma cells. C6 glioma cells (3 × 105)/well were seeded in the six-well culture plate and incubated at 37°C in a 5% CO2-supplemented incubator for 24 h. Morin (25 and 50 µM/mL) were added to the cells and incubated for 24 h. The control, untreated glioma cells, and morin-treated cells were fixed using a mixture consisting of methanol and glacial acetic acid in a 3:1 ratio for 20 min at 4°C. The fixed cells were then treated with a stain solution consisting of AO and EtBr in a 1:1 ratio. The stained cells were incubated in the dark for 30 min and then rinsed with saline. The cells were then viewed under a fluorescent microscope, and the percentage of live and apoptotic cells was calculated.
Quantification of Apoptotic Proteins
Briefly, 3 × 105 C6 rat glioma cells were cultured in a six-well culture plate and treated with 25 and 50 µM doses of morin. The cells were then incubated at 37°C supplemented with 5% CO2 for 24 h. The cells were then trypsinized, and the collected cells were carefully sonicated with intermittent cooling to avoid protein damage due to heat. The sonicated cells were then centrifuged at 15,000 rpm for 5 min at 4°C. The supernatant was collected in sterile tubes and utilized for further quantification of apoptotic proteins using enzyme-linked immunosorbent assay (ELISA) kits (Abcam, USA). Caspase-3, caspase-9, the proapoptotic protein Bax, and the antiapoptotic protein Bcl-2 were quantified using the ELISA kits. The experiment was performed as per the protocol of the manufacturer, and the reagents and standards were freshly prepared before the initiation of the test. The experiment was performed in triplicate; 100 µL of standards and samples were added to the wells of ELISA plates and incubated for 45 min at 37°C. After further incubation, the contents were carefully emptied, 100 µL of the working solution of the biotin-coated antibody was added and incubated for 60 min at 37°C, and then subjected to washing with 1X wash buffer. To the wells working streptavidin–biotin complex solution, 100 µL was added and incubated for 30 min. Then, 90 µL of 3,3′, 5,5′-tetramethylbenzidine substrate was added to the wells after washing the wells with 1X wash buffer thrice. The plates were incubated in the dark for 20 min and 50 µL of stop solution to each well. The plates were then subjected to reading at an absorbance of 450 nm using an ELISA microplate reader. The concentration of apoptotic proteins in control and morin-treated glioma cells was calculated using the standard curve plot.
Assessment of Inflammatory Cytokines
Inflammatory cytokines tumor necrosis factor-alpha (TNF-α), nuclear factor-kappa B (NF-кB), cyclooxygenase-2 (COX-2), and interleukin-6 (IL-6) levels in control and morin-treated glioma cells were evaluated using the commercially available ELISA kits (Abcam, USA). The test was performed as per the guidelines of the manufacturer. The final absorbance of the solutions was read at 450 nm using an ELISA microplate reader, and the concentrations were calculated using the standard curve plot.
Statistical Analysis
The results were statistically analyzed using a one-way ANOVA for intergroup comparison, and a post hoc Tukey’s test was done to evaluate the significance of intragroup sampling. The significant statistical difference was considered to be at a probability level of p < 0.05.
Results
Morin Treatment Decreased Cell Viability in C6 Rat Glioma Cells
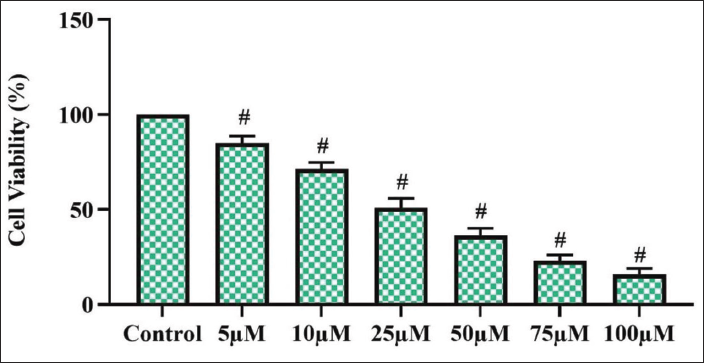
Figure 1 represents the results of the MTT cytotoxicity assay performed in control, untreated gliomas, and at different concentrations of morin-treated glioma cells. The lowest dose of 5 µM/mL of morin treatment decreased the viability of glioma cells up to 3 ± 0.02%, whereas a subsequent increased dose of 25 and 50 µM morin treatment significantly decreased the cell viability to 77 ± 1.2% and 64 ± 1.8%, respectively. The half-maximal inhibitory concentration value was obtained at a dose of 25 µM of morin treatment, which showed only 50% of viable cells. The highest dose of 100 µM morin-treated glioma cells showed only 22 ± 1.6 viable glioma cells.
Morin Treatment Significantly Generated ROS in C6 Rat Glioma Cells
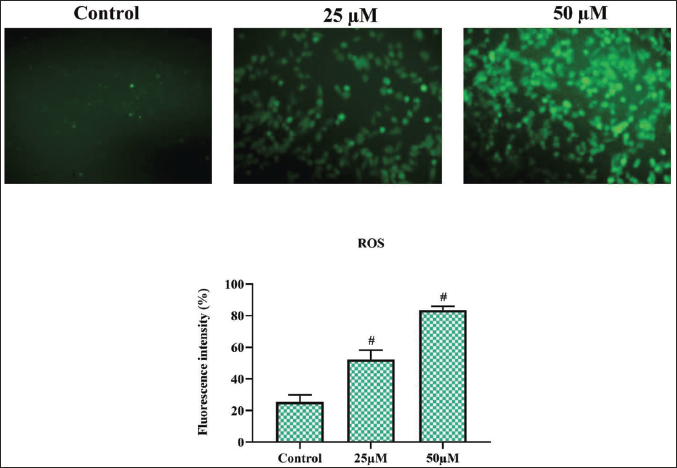
ROS plays a vital role in inducing apoptosis in cancer cells; hence, we quantified the ROS generated in morin-treated and untreated glioma cells using dichlorodihydrofluorescein diacetate (DCHF-DA) staining, and the results are illustrated in Figure 2. Morin-treated glioma cells exhibited increased green fluorescence, indicating the presence of ROS and the intensity of green fluorescence increased in a dose-dependent manner, confirming the morin-induced ROS generation in C6 rat glioma cells. The percentage of fluorescence intensity was 32 ± 2.2 in 25 µM of morin-treated glioma cells, whereas it drastically increased to 56 ± 1.8 in 25 µM morin-treated glioma cells.
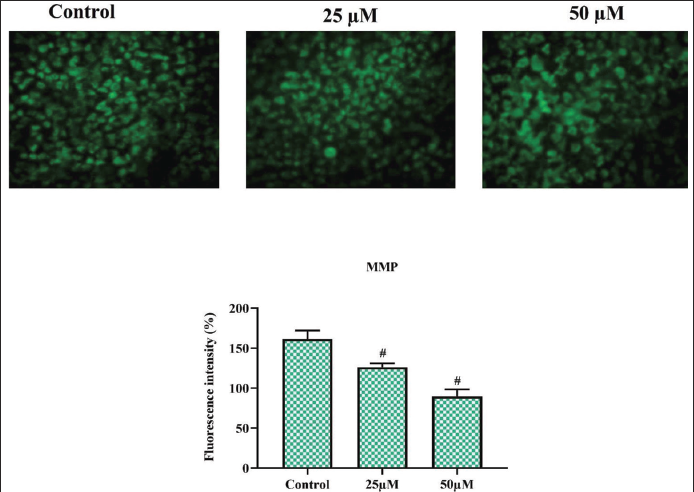
The ATP production of a cell depends on its MMP; the loss of MMP causes bioenergetic stress, leading to apoptosis. In Figure 3, we illustrate the levels of MMP in control and morin-treated C6 rat glioma cells quantified with the fluorescent dye rhodamine 123. Morin treatment significantly depolarized the MMP of glioma cells in a dose-dependent manner. The intensity of rhodamine 123 dye fluorescence was significantly decreased in 25 and 50 µM/mL morin-treated glioma cells. The percentage of fluorescence intensity was 68 ± 1.4 in 25 µM of morin-treated glioma cells, whereas it significantly decreased to 54 ± 0.8 in 50 µM morin-treated glioma cells.
Morin Treatment Altered the Cell Morphology of C6 Rat Glioma Cells
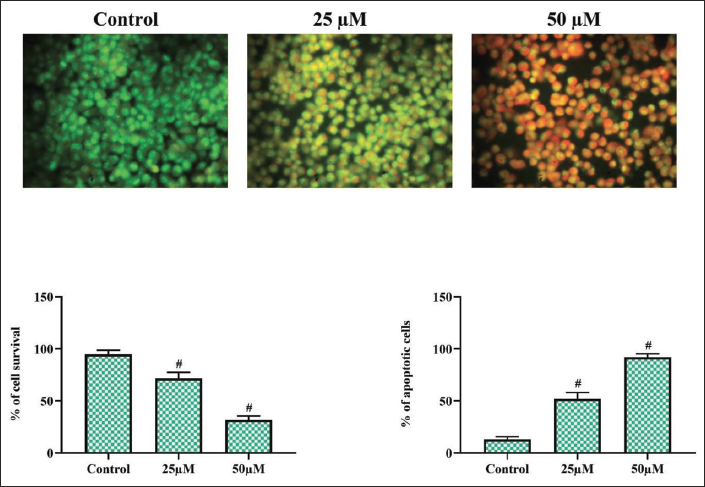
The induction of apoptosis by morin in rat C6 glioma cells was analyzed with the dual staining technique, and the results are depicted in Figure 4. AO was stained in the control cells and stained the nucleus with bright green fluorescence, whereas the morin-treated glioma cells showed increased orange and reddish orange fluorescence emitted due to the staining of EtBr. Morin-treated glioma cells (25 µM/mL) exhibited orange fluorescence, indicating the initial stage of apoptosis, which showed 74 ± 2.2% of viable and 28 ± 2.8% of apoptotic cells, whereas 50 µM/mL morin-treated glioma cells exhibited reddish-orange fluorescence, which showed 32 ± 2.6% of viable and 78 ± 2.8% of apoptotic cells.
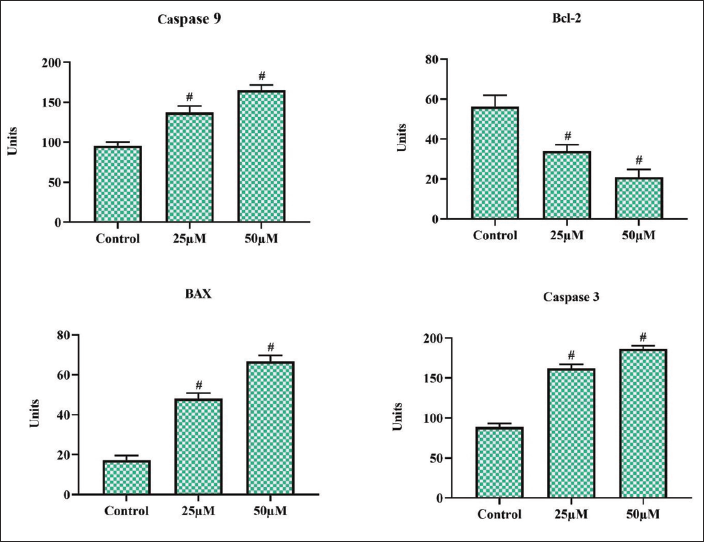
Morin Treatment Triggered Caspases and Inhibited Antiapoptotic Protein in Rat C6 Glioma Cells
Figure 5 depicts apoptotic protein caspases, Bax, and antiapoptotic protein Bcl-2 levels in morin-treated and untreated rat glioma cells. Initiator and executor caspases were significantly elevated in the morin glioma cells. The proapoptotic protein Bax was significantly amplified in morin-treated glioma cells. Morin treatment significantly declined the levels of antiapoptotic protein Bcl-2 in glioma cells, which confirms the induction of apoptosis by morin in glioma cells.
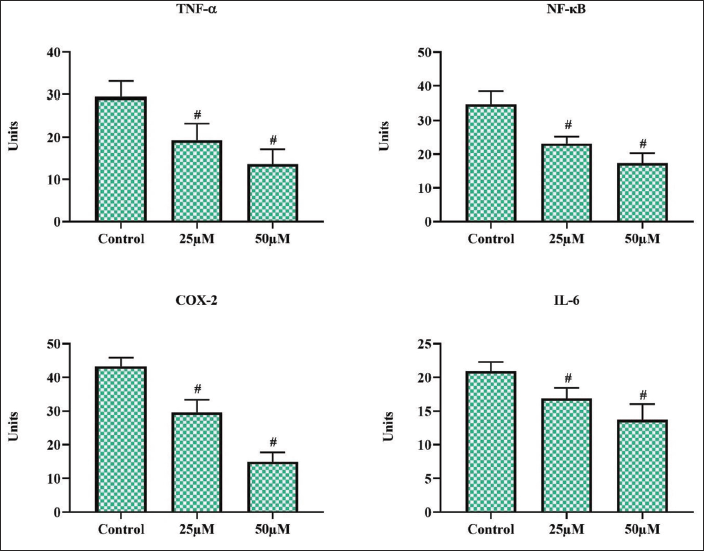
Morin Treatment Reduced the Levels of Inflammatory Cytokines in Rat C6 Glioma Cells
Anti-inflammatory agents are proven to be effective in treating cancer; hence, we assessed the effect of morin on inflammatory cytokines and depicted the results in Figure 6. Morin treatment significantly decreased the key inflammatory initiator proteins, TNF-α and NF-κB, in a dose-dependent manner. It also decreased the levels of COX-2 and the IL-6 protein, which further confirms the anti-inflammatory properties of morin.
Discussion
Glioma, a primary malignant cancer with a significant annual incidence rate of six cases per 1 lakh of the global population, is a great task for healthcare professionals to treat. The incidence of gliomas is reported more in the male population compared to the female population (GBD, 2016). Gliomas are classified based on the size and location of the tumor; pilocytic astrocytomas are the least malignant type of brain tumor, whereas glioblastomas are the most infiltrative, diffusive type of glioma (Mesfin & Al-Dhahir, 2023). Diagnosing glioma at an early stage is very rare since most of the patients experience common non-specific symptoms such as fatigue, headache, and malaise during the initial stage of tumors. Whereas at the advanced stage, 80% of patients were reported to have seizures (Peeters et al., 2020; Posti et al., 2015; Rasmussen et al., 2017). Neuroimaging is the prime diagnostic tool for screening gliomas, which is done mostly during the advanced stage of malignancy (Rice et al., 2016). Despite the delicate nature of the brain, the primary treatment prescribed for gliomas is surgical resection, which causes even worse side effects with supportive subsequent chemo and radiation therapies. Therefore, discovering a drug that can cure and prevent glioma without causing any adverse effects is the need of the day.
In this study, we analyzed the anticancer effect of phytochemical flavanol morin against glioma cells in vitro. Glioblastoma multiforme is the most aggressive type of glioma, with a minimal survival rate of 1–2 years. These types of gliomas possess a high vascularization, angiogenic, and proliferative rate with the least prognosis (Louis et al., 2016). C6 glioma cells are rat glioma cells that exhibit the same invasive pattern as human glioma cells. It is also a reliable in vitro model for testing the pharmacological properties of various newly designed anticancer drugs. C6 glioma cells were considered the gold standard model in glioma research (Giakoumettis et al., 2018); hence, in our study, we tested the potency of our drug morin against C6 glioma cells.
Natural phytochemicals are the most promising candidates for anticancer treatment studies; they have proven to possess antiproliferative properties against cancer without causing any changes to normal cells (Jang et al., 2020, Tavana et al., 2020). Among phytochemicals, flavonoids are a well-studied molecule, and they have incredible effects against various degenerative, inflammatory, and cancer diseases. Flavones, a type of flavonoid, have been proven to cross the blood–brain barrier and also render minimal side effects on normal cells (Michaud-Levesque et al., 2012; Zamin et al., 2009). Morin is one such flavone compound with numerous pharmacological properties, such as anti-inflammatory, antioxidant, oxidative DNA damage, lipid peroxidation, and so on (Caselli et al., 2016; Rajput et al., 2021). Morin treatment significantly prevented the proliferation of C6 glioma cells in a dose-dependent manner. This confirms the antiproliferative property of morin against gliomas, which is the prime property of an anticancer drug.
Recently, ROS, once considered to be tumor initiators, are now known to be a bright side of the moon to anticancer drug researchers. ROS activates various signaling pathways in cells and also the signals from the cytoplasm to the nucleus, which regulate the expression of a wide range of signaling proteins in normal cells. Cancer cells hinder the excessive synthesis of ROS, thereby protecting the cancer cells from ROS-induced apoptosis (Perillo et al., 2020). Various phytochemicals such as piperlongumine (Dhillon et al., 2014), curcumin (Thani et al., 2012, Gersey et al., 2017), quercetin (Kusaczuk et al., 2022), capsaicin (Oyagbemi et al., 2010) have also been proven to increase the intracellular ROS in cancer cells, thereby depolarizing the mitochondria and also causing cell cycle arrest. In our study, morin treatment significantly amplified the levels of ROS in C6 glioma cells and also declined the MMP of the cells. This may be the reason for the decreased number of viable cells reported in our MTT assay.
Apoptosis, a programmed cell death, was classified into three types: Type I, the classical apoptotic cell death; Type II, which occurs due to the involvement of lysosomal cathepsins, and Type III, the necrotic type of cell death, which is classified by cytoplasmic changes. Type I programmed cell death was characterized by cell shrinkage, condensation of chromatin, and the presence of degraded DNA (Clarke, 1990; Gozuacik & Kimchi, 2004; Wang et al., 2012). In the present study, the morin-treated C6 glioma cells showed the characterized features of apoptotic cells, which was evidenced by our AO/EtBr staining results. This proves the induction of apoptosis by morin in C6 glioma cells. Further, to confirm the activation of apoptotic signaling proteins by morin in C6 glioma cells, we quantified the caspases, which play a major role in apoptosis. Morin treatment significantly increased both the initiator and executor caspases, confirming the induction of apoptosis. It also significantly decreased the antiapoptotic protein Bcl-2 expression and amplified the proapoptotic protein Bax expression.
The immune response of the central nervous system is greatly influenced by the microglial cells. During malignancy, the M2 polarization of microglia leads to immunosuppressive and tumor-promoting nature. Cytokines play a vital role in all stages of tumor development, such as proliferation, growth, invasion, and angiogenesis of the cancer cells (Borisov & Sakaeva, 2015; Gabrusiewicz et al., 2011). Morin treatment significantly decreased the levels of inflammatory cytokines, TNF-α, NF-κB, COX-2, and IL-6. This may be due to the anti-inflammatory properties of morin. The generation of increased levels of ROS, which triggered the proapoptotic proteins and inhibited the inflammatory cytokines, maybe the reason for increased apoptotic cells in the morin-treated C6 glioma cell line, as evidenced in our microscopic staining studies.
Conclusion
In conclusion, our results suggest that the morin treatment has proven to inhibit cell proliferation and induce apoptosis in glioma C6 cells. The morin treatment effectively induced the production of ROS and thereby facilitated oxidative stress. The expression of apoptotic genes was also augmented by the morin treatment in the C6 cells. Hence, it was clear that morin is effective in inhibiting cell growth and promoting apoptosis in C6 glioma cells. Overall, our findings confirm that morin is a potent anticancer drug that can be subjected to further research to treat glioma patients.
Summary
Glioma, primary malignant brain tumor with poor prognosis at initial stage reported to have high mortality rate. Morin is proven to exert anti-inflammatory, antioxidant, anticancer, antibacterial, antidiabetic, and neuroprotective properties. Morin treatment also effectively increased the caspase-3 and caspase-9, and proapoptotic protein Bax, and decreased the antiapoptotic protein Bcl-2. The induction of apoptosis by morin in glioma cells was evident with our AO/EtBr staining results. Morin also decreased the inflammatory cytokine levels in rat C6 glioma cells.
Abbreviations
MMP: Mitochondrial membrane potential; TNF-α: Tumor necrosis factor α; NFκB: Nuclear factor-κB; IL-6: Interleukin-6; ELISA: Enzyme-linked immunoassay; ROS: Reactive oxygen species; MTT: (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide).
Footnotes
Acknowledgments
This work was supported by Fourth Military Medical University, China. This project was supported by Researchers Supporting Project number (RSP2024R283) King Saud University, Riyadh, Saudi Arabia.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by Fourth Military Medical University, China. This project was supported by Researchers Supporting Project number (RSP2024R283) King Saud University, Riyadh, Saudi Arabia.
Statement of Ethical Approval and Informed Consent
None
