Abstract
Tumor suppressor p53 and proto-oncogene survivin are challenging targets for anticancer drugs in acute lymphoblastic leukemia (ALL) which are associated with chemoresistance. Yet, no p53 and survivin-modulating drug with low toxicity and high efficacy has been approved for clinical application in ALL. Consequently, the search for novel compounds which target p53 or survivin is needed to further advance ALL treatment. Prodigiosin, a secondary metabolite of Serratia marcescens induces apoptosis in cancer cells with no toxicity on normal cells. However, the possible potential of prodigiosin as p53- and survivin-modulating agent in ALL cells has not been investigated. Wt-p53 Molt-4 cells were treated with 100 to 600 nM prodigiosin, after which, viability, cell proliferation rates, survivin and p53 protein levels, caspase-3 activation, and apoptosis were evaluated. After 24-, 48-, and 72-h treatments with 100 to 600 nM prodigiosin, cell proliferation rates were measured to be 93.7–77.3%, 75.5–58.3%, and 55–23.3%, respectively. Treatment for 48 hours with 100 to 600 nM prodigiosin resulted in 41–19% decrease in survivin protein levels followed by 450–950% increases in caspase-3 activation levels. Prodigiosin induced remarkably p53 accumulation and increased p53/survivin and caspase-3/survivin ratios by 6.1 to 11.3 and 10.3 to 47.5-fold at 100 to 600 nM, respectively. Survivin protein levels were inversely proportional to p53 accumulation levels. Low survivin protein levels combined with high levels of p53 accumulation were correlated to higher apoptotic rates. P53 and survivin as molecular targets of prodigiosin contribute to caspase-3-dependent apoptosis in ALL cells and this compound represents an attractive p53- and survivin-modulating agent in ALL.
Introduction
Chemotherapy is successfully used to treat acute lymphoblastic leukemia (ALL), but a proportion of patients experience treatment failure and/or relapse. 1 Several studies have shown that p53 inactivation and survivin overexpression are associated with chemoresistance and poor prognosis in ALL. 2 –4
P53 is one of the most important tumor suppressor proteins. This protein plays important roles in cell cycle progression, DNA repair, and apoptosis. At initial diagnosis of ALL, the incidence of p53 mutation appears to be low, 5 but in relapsed pediatric ALL, p53 mutations have been detected in 5 to 10% of patients. 6 Thus, most cases of ALL express wt-p53. Consequently, this would make wt-p53 an attractive target in ALL treatment.
Survivin, a member of the family of inhibitor of apoptosis proteins, plays a key role in the regulation of cell cycle progression, drug resistance, and poor prognosis. 4,7 Survivin is overexpressed in ALL cells but rarely in normal cells. 4 In consequence, this would make survivin an attractive therapeutic target for treatment of ALL patients.
With reference to the above-mentioned subjects, targeting p53 or survivin, in ALL cells by a therapeutic agent may provide minimal toxicity to normal cells and may open up avenues to new therapeutic strategies for ALL-directed therapy.
Microorganisms have been studied for their secondary metabolites with anticancer properties. 8 In this regard, prodigiosin (Figure 1; 2-methyl-3-pentyl-6-methoxyprodigiosene), a red pigment derived from cell wall of Serratia marcescens exerts apoptotic effect on different kinds of cancer cells with low toxicity on normal cells. 9,10

Structure of prodigiosin isolated from cell wall of Serratia marcescens.
Prodigiosin targets several vital processes that potentially contribute to its anticancer function. These include acidification of the cytoplasm by acting as H+/Cl− symporters, arrest of cell cycle progression by upregulation of p21WAF/CIP1, induction of DNA damage by inhibiting topoisomerases I and II and reduction in ATP production by uncoupling energy from mitochondrial proton gradient to ATP synthesis. 10 –12 However, little is known regarding the effects of prodigiosin on the wt-p53 and survivin expression as two key important proteins which are associated with chemoresistance in ALL. 2 –4
In the present study, we investigated the capacity of prodigiosin to trigger apoptosis and to modulate p53 as well as survivin in ALL cells.
Materials and methods
Cell line and culture condition
Wt-p53Molt-4 cells (T-ALL) that serve as a model for human T-lymphoblastic leukemia cells were purchased from the National Cell Bank of Iran (Pasture Institute of Iran, Tehran) and were grown in RPMI-1640 medium containing 10% (v/v) fetal bovine serum, penicillin (100 U/ml)/streptomycin (100 µg/ml) all from (PAA, Austria), 20 mM HEPES (4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid) and 2 mM L-glutamine (Roche, Germany) at 37 °C in a humidified incubator with 5% carbon dioxide (CO2).
Chemicals, drugs, and kits
Pure 2-methyl-3-pentyl-6-methoxyprodigiosene (prodigiosin; Figure 1) isolated from Serratia marcescens and trypan blue was purchased from Sigma Chemical Company (Bengaluru, Karnataka, India). Caspase-3 activation, cell proliferation, and Annexin-V Fluos kits were purchased from Roche, Germany. Human survivin and p53 ELISA kits were purchased from Abcam (Cambridge, Massachusetts, USA). Other general chemicals were purchased from Merck Chemical Company (Darmstadt, Hesse, Germany).
Preparation of prodigiosin and cell treatments
Prodigiosin was dissolved in absolute methanol to prepare a 100-mM stock solution and stored in the dark at −80 °C until use. For each experiment, the prodigiosin was freshly prepared from the stock solution at the concentrations ranging from 100 nM to 600 nM by serially diluting in culture medium. Control cells were cultured in the medium containing same concentration of absolute methanol (v/v) as the prodigiosin-containing medium. The final methanol concentration never exceeded 0.5% (v/v).
Cell viability assays
Molt-4 cells were seeded at a density of 5 × 105 cells per well in 1.5 ml of complete medium in six-well plates and incubated at 37 °C for 24 h. Thereafter, cells were treated with the indicated concentrations of prodigiosin for 48 h. Following 48-h treatments, the cells were harvested and stained with trypan blue dye after which the numbers of viable cells were counted under an inverted microscope. The viable cells with an intact cellular membrane do not take up the blue dye and maintain a clear appearance, whereas the damaged nonviable cells take up the dye and stained blue as their membrane damages. Cell viabilities for each treatment were converted to a percentage of the untreated control cells.
Metabolic activity and proliferation assays
The 104 cells per well were plated in 96-well plates. After 24-h incubation, Molt-4 cells were treated for 24 to 72 h with different concentrations of prodigiosin and metabolic activities as well as proliferation rates were evaluated by performing water-soluble tetrazolium salt (WST) proliferation assay. Following each incubation period, the WST-1 solution (10 µl) was added to each well and the cells were incubated for an additional 4 h at 37 °C and 5% CO2. The absorbance in each well was then measured with a microplate reader (Biotek, Winooski, Vermont, USA) at 490 nm and a reference wavelength of 620 nm. Appropriate controls including untreated cells and controls lacking cells were considered in experiments. Thereafter, proliferation rates were calculated as (Asample−Ablank)/(Acontrol − Ablank) × 100%. The results were expressed as the percentages of untreated control cells.
To measure the total growth inhibition and concentration causing 25%, 50%, and 75% of growth inhibition values of prodigiosin, the percentages of the proliferation rates on the y-axis were plotted against the concentrations of prodigiosin on the x-axis. Finally, all calculations were performed using regression analysis.
Enzyme-linked immunosorbent assay
P53 protein
Intracellular p53 protein level was assayed by the sandwich ELISA following the procedure provided by the manufacturer. Briefly, 5 × 105 Molt-4 cells per well were cultured in the absence or in the presence of prodigiosin (100, 200, 400, and 600 nM) for 48 h. Next, the cells were washed twice with ice-cold phosphate buffered saline (PBS), and suspended in ready-to-use cold lysis buffer for 30 min followed by centrifugation at 12,000 × g for 15 min at 4 °C. Supernatants were then collected and used for p53 protein assay. Briefly, microtiter ELISA was plate coated with the human monoclonal antibody specific for p53. Following sample application and washing, a biotinylated monoclonal antibody specific for p53 was added and incubation continued for 1 h at room temperature. Next, the wells were washed and the enzyme streptavidin-HRP (Horseradish peroxidase) that binds the biotinylated antibody was added, and incubation continued for 30 min followed by washing. Thereafter, tetramethyl benzidine (TMB) substrate solution was finally added which acts on the bound enzyme to induce a colored reaction product. The optical density values of the samples were recorded at 450 nm using an ELISA reader.
Survivin protein
Intracellular survivin protein level was assayed by the sandwich ELISA following the procedure provided by the manufacturer. As mentioned above, prodigiosin-treated cell lysates were prepared and supernatants were then collected and used for survivin protein assays. Briefly, microtiter ELISA plate coated with the mouse antihuman survivin monoclonal antibody. Following sample application, a biotinylated detection polyclonal antibody from goat specific for human survivin is then added followed by washing with PBS buffer. Thereafter, avidin–biotin–peroxidase complex is added and unbound conjugates were washed away with PBS buffer. TMB substrate solution was finally added and reactions were stopped after 15 min. The optical density values of the samples were recorded at 450 nm using an ELISA reader.
Caspase-3 activity assay
Caspase-3 activity was measured using the caspase-3 colorimetric assay kit following the procedure provided by the manufacturer. Briefly, treated cells were collected after 48-h treatment and resuspended in ready-to-use chilled lysis buffer for 10 min. Next, high-speed centrifugation was performed and the supernatants were collected and used for caspase-3 activation assay. Briefly, reaction buffer, DTT (Dithiothreitol) and DEVD-p-NA substrate were added to samples and plates were incubated at 37 °C for 2 h. The principle was that caspase-3 derived from cellular lysate recognizes the sequence Asp-Glu-Val-Asp (DEVD). The assay is based on spectrophotometric detection of the chromophore p-nitroaniline (p-NA) after cleavage from the labeled substrate (DEVD-p-NA). The p-NA light emission can be quantified using a microtiter plate reader at 405 nm. Comparison of the absorbance of p-NA from an apoptotic sample with an untreated control sample allows determination of the fold increase in caspase-3 activity. Procedure measures only the functionally relevant cleaved caspase-3.
Flow cytometric apoptosis assay
Apoptosis was analyzed by a double-staining method using Annexin-V FLOUS/propidium iodide (PI) labeling solution according to the manufacturer’s instructions. In apoptotic cells, the membrane phospholipid phosphatidyl serine, which is normally found in the internal portion of the cell membrane, becomes translocated to the outer leaflet of the plasma membrane, thereby exposing phosphatidyl serine to the external environment. Annexin-V is a calcium-dependent phospholipid binding protein, has an affinity for phosphatidyl serine, and is useful in identifying apoptotic cells. PI binds to cellular DNA helps in identifying necrotic cells.
Molt-4 cells were treated with different concentrations of prodigiosin for 48 h. Thereafter, the cells were washed twice with sterile cold PBS buffer and after centrifugation, cell pellets were then resuspended in100 µL of 1× binding buffer at a density of 5 × 105 cells/ml with FITC (Fluorescein isothiocyanate)-Annexin V. The cells were gently mixed and incubated in the dark at room temperature for 20 min. To differentiate cells with membrane damage, PI solution was added to the cell suspension prior to the flow cytometric analysis using a fluorescence-activated cell sorter (FACScan, USA, Franklin Lakes, NewJersy). Early apoptosis was defined as cells positive for Annexin V-FITC only. Late apoptosis was defined as cells positive for Annexin V-FITC and PI and necrotic cells were defined as cells positive for PI only.
Statistical analyses
All experiments were repeated twice using triplicate assays and the results were presented as mean ± standard deviation (SD). All calculations were performed using the SPSS version 20 for Windows (SPSS Inc., Chicago, IL, USA). Analysis of variance (ANOVA) followed by a Tukey post-hoc was used for multiple comparisons. A value of p < 0.05 was considered to be statistically significant.
Results
Effect of prodigiosin on cellular morphology
Morphology of treated Molt-4 cells provides invaluable information about the prodigiosin-induced toxicity. In this regard, the treated cells appeared less uniform with loss of membrane integrity. Furthermore, prodigiosin decreased significantly the number of viable cells in a dose-dependent manner (Figure 2).

Morphological features of prodigiosin-treated Molt-4 cells 48-h posttreatment. Magnification: 20×, C: untreated control cells.
Effect of prodigiosin on cell viability (trypan blue dye exclusion)
Prodigiosin decreased significantly cell viability in a dose-dependent manner and cell viabilities were measured to be 87 ± 1.5%, 81 ± 1.5%, 75 ± 2.1%, and 65 ± 1.4% for 100, 200, 400, and 600 nM prodigiosin, respectively, compared to untreated cells.
Effect of prodigiosin on the metabolic activity and cell proliferation
Treatment of cells with increasing concentrations of prodigiosin diminished metabolic activity and proliferation of Molt-4 cells in a dose- and time-dependent manner (Figure 3(a) to (c)), specifically, after a 72-h treatment compared to untreated cells. After treatment with prodigiosin at concentrations ranging from 100 to 600 nM, cell proliferation rates were measured to be 93.7–77.3%, 75.3–58.3%, and 55–3.3% for 24, 48, and 72 h, respectively (Figure 3(b)). Furthermore, after 24- to 72-h treatment with prodigiosin, cell proliferation rates were measured to be 93.7–55%, 84.7–37.3%, 80.7–28.3%, and 77.3–23.3% for 100, 200, 400, and 600 nM, respectively (Figure 3(c)). We also found that IC25, IC50, IC75, and total growth inhibition values decreased in a time-dependent manner (Table 1).

Effect of prodigiosin on the metabolic activity and cell proliferation. (a) Metabolic activity. (b) and (c) Cell proliferation on a dose response and time course basis. Data represent means of two independent experiments with triplicate assays.*p < 0.05, **p < 0.01, and ***p < 0.001 versus untreated control cells.
Evaluation of total growth inhibition and IC25,50, 75 values in prodigiosin-treated cells.a

a104 Molt-4 cells per well were treated with prodigiosin at concentrations 0, 100, 200, 400, and 600 nM for 24 to 72 h after which IC25, 50, 75 and total growth inhibition values were calculated from the dose-response curves. The results represent mean ± standard deviation of two independent experiments with triplicate assays.
Effect of prodigiosin on the p53 protein accumulation
Since the effect of prodigiosin on the p53 protein accumulation has so far not been addressed in ALL cells, we set out to evaluate the effect of prodigiosin treatment on the accumulation of p53 protein level. We found that after a 48-h treatment with prodigiosin, the relatively low level of the p53 protein in untreated Molt-4 cells increased significantly with increasing prodigiosin concentrations. Specifically, treatment with 400-nM prodigiosin resulted in 3.2-fold increase in the p53 protein accumulation level compared to untreated cells (Figure 4).
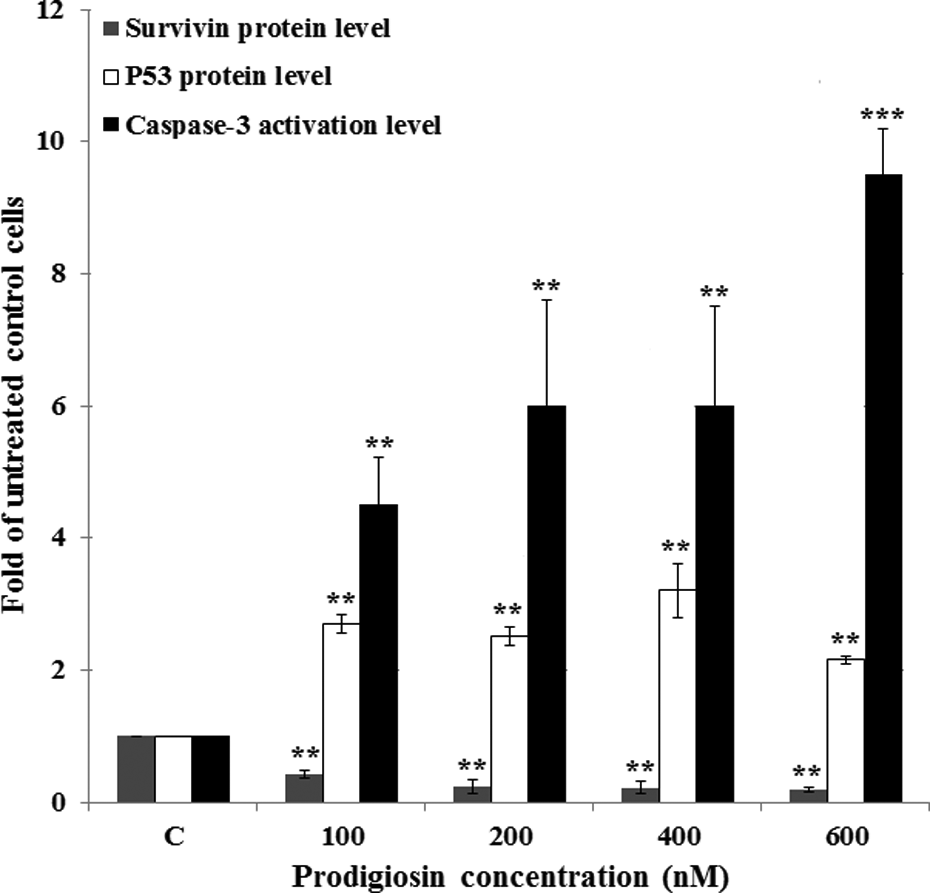
Effect of prodigiosin on the intracellular p53, survivin, and caspase-3 activation levels 48-h posttreatment. Data represent means of two independent experiments with duplicate assays. **p < 0.01, ***p < 0.001 versus untreated control cells (C).
Effect of prodigiosin on the survivin protein level
The effect of prodigiosin on the survivin expression has never been evaluated in ALL cells. Therefore, we set out to evaluate the effect of prodigiosin treatment on the expression of survivin protein. We found that after a 48-h treatment with prodigiosin, the high level of survivin protein in untreated Molt-4 cells decreased significantly with increasing prodigiosin concentrations. Specifically, treatment with 600 nM prodigiosin resulted in 81% decrease in survivin protein level as compared to untreated cells (Figure 4).
Effect of prodigiosin on the caspase-3 activation
Caspase-3 is a well-known marker for apoptotic cell death. As shown in Figure 4, prodigiosin compound induced remarkably caspase-3 activation in a dose-dependent manner and resulted in 4.5- to 9.5-fold increases in caspase-3 activation for 100 to 600 nM prodigiosin as compared to untreated cells.
Effect of prodigiosin on apoptosis induction
To evaluate whether the growth-inhibitory effect observed upon treatment of Molt-4 cells with prodigiosin was due to the induction of apoptosis, prodigiosin-treated Molt-4 cells were stained with Annexin V/PI and analyzed by means of flow cytometer. As depicted in Figure 5(a) and (b), a significant increase in the number of apoptotic cells (early + late) ranging from 37.9% to 96.3% was observed with increasing prodigiosin concentrations.
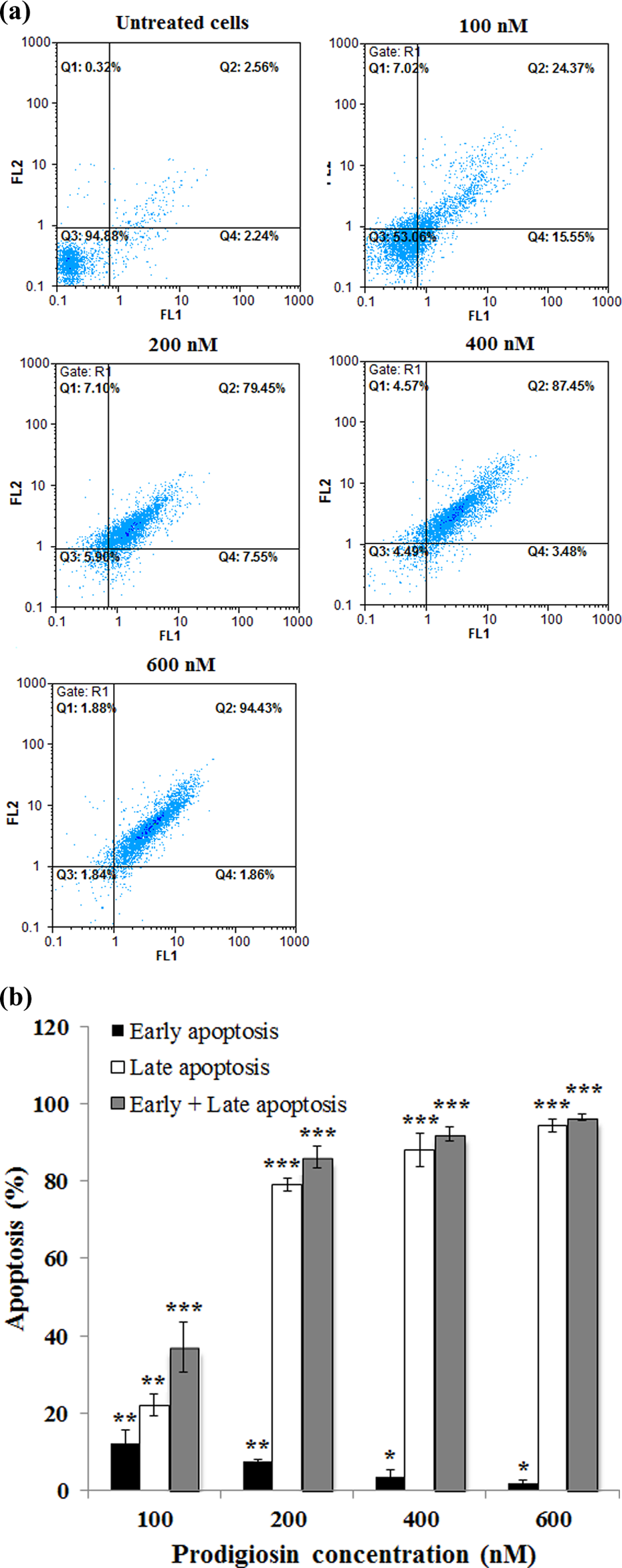
Effect of prodigiosin on apoptosis induction 48-h posttreatment. (a) FL1: Annexin V-FITC (horizontal axis) and FL2: PI (vertical axis). (Q1) prenecrotic cells, (Q2) late apoptosis, (Q3) living cells, and (Q4) early apoptotic cells. (b) Histogram presentation of apoptotic cells percentages. *p < 0.05, **p < 0.01, and ***p < 0.001 versus untreated control cells. PI: propidium iodide.
Effect of prodigiosin on the p53/survivin protein and caspase-3/survivin protein ratios
The balance between tumor suppressor and proto-oncogene genes expression is a critical key point to determine if a cell undergoes apoptosis. With this in mind, we determined the relative ratios of tumor suppressor p53 to proto-oncogene survivin protein after 48-h treatment with prodigiosin. In this regard, treatment with different concentrations of prodigiosin resulted in 6.1- to 12.9-fold increases in p53/survivin ratios compared to untreated cells (Figure 6).

Evaluation of the p53/survivin and caspase-3/survivin protein ratios in prodigiosin-treated cells. 5 × 105 Molt-4 cells per well were treated with prodigiosin at concentrations 100, 200, 400, and 600 nM for 48 h and p53, survivin, and caspase-3 protein levels were measured. Thereafter, p53/survivin and caspase-3/survivin protein ratios were calculated. The results represent mean ± SD of two independent experiments with duplicate assays. **p < 0.01 and ***p < 0.001 versus untreated control cells.
The level of proapoptotic marker such as caspase-3 versus the level of antiapoptotic marker such as survivin could be important in determining the resistance of cells to apoptosis. As shown in Figure 6, the caspase-3/survivin protein ratios were increased dramatically with increasing prodigiosin concentrations (100 to 600 nM) and resulted in 10.3- to 47.5-fold increases compared to untreated cells.
Associations between the p53/survivin protein and caspase-3/survivin protein ratios and apoptosis
We found that there were significant correlations between the p53/survivin protein, and caspase-3/survivin protein ratios with total apoptotic rates (early + late) and were measured to be 0.92 and 0.8, respectively.
Discussion
Prodigiosin could successfully target the wt-p53 and survivin expression and showed an attractive antileukemic activity in Molt-4 cell line as a model for ALL cells. This ability of prodigiosin is particularly interesting, because p53 is a challenging target for drug discovery due to the lack of accessibility of a receptor–ligand interaction or an enzyme active site.
P53 mutations in primary leukemia are relatively rare, 13 –15 but about 30% of samples from patients with relapsed T-ALL have p53 mutations suggesting a deficient p53 signaling pathway in T-ALL cells. 16 Additionally, in relapsed pediatric ALL, p53 mutations have been detected in 5 to 10% of patients. 6 Thus, most cases of T-ALL and pediatric ALL express wt-p53. Consequently, this would make wt-p53 an attractive target in ALL treatment.
In our study, prodigiosin induced apoptosis in Molt-4 cell line which is T-ALL cells with wt-p53. In line with our study, Hong et al. 17 found that prodigiosin restores p53 signaling in colorectal cancer cells harboring hotspot p53 mutations. These evidences could mean an advantage of prodigiosin over other chemotherapeutics that need functional p53 to provoke its cytotoxic effect.
Survivin has been found to be upregulated in ALL but rarely in normal cells. Therefore, it may provide a potential target for selective therapy in ALL. 4 Survivin mediates the suppression of apoptosis by direct inhibition of caspase-3. 18 In our study, induction of caspase-3 activation through inhibition of the survivin expression seems to be one of the molecular pathways leading to prodigiosin apoptotic effect in Molt-4 cells. In agreement with our data, different researchers showed that prodigiosin can dose-dependently induce downregulation of survivin and trigger caspase-3 activation and apoptosis in different kinds of cancer cells. 19 –21
Prodigiosin decreased metabolic activity of Molt-4 cells, particularly after a 72-h treatment, suggesting that this compound may exhibit its cytotoxic effects against mitochondria. In this regard, it has been shown that prodigiosin is accumulated in mitochondria and impairs the mitochondrial function. 10
Late apoptotic and necrotic cells are Annexin-V and PI positive. To distinguish late apoptotic cells from necrotic ones, the results obtained from caspase-3 activation assay may provide better interpretation. Our data showed that prodigiosin dose-dependently induces caspase-3 activation, suggesting that most dead cells seen in Q2 of flow cytometry graphs are apoptotic cells not necrotic ones. In this context, based on the flow cytometry data, the decreases in cell viability and proliferation could be attributed to apoptosis induction by prodigiosin.
Molt-4 cells showed high degree of sensitization to prodigiosin compound and these cells became more sensitive to prodigiosin cytotoxicity with increasing the drug exposure time. In agreement with our data, different investigators have shown that prodigiosin and its derivative cycloprodigiosin hydrochloride induce apoptosis in malignant cells with no marked toxicity in normal cells. 10,17,22,23
Based on our data, prodigiosin induced dramatically wt-p53 accumulation. To confirm, the activity of induced wt-p53 accumulation, evaluation of downstream p53-target genes expression is highly valued. It has been shown that survivin is a p53 target gene. Survivin gene promoter contains a p53 response element. Increased expression of p53 represses survivin promoter activity, resulting in decreased survivin protein expression. 24 –26 In our study, the level of survivin protein was inversely proportional to p53 accumulation level. Low protein levels of survivin combined with high levels of p53 accumulation were correlated to higher caspase-3 activation level and apoptosis rate, suggesting activity of induced wt-p53 accumulation in prodigiosin-treated cells.
Doxorubicin is commonly used to treat ALL and functions through a p53-dependent mechanism. 27,28 However, resistance to doxorubicin is observed not only in cancer cells having p53 mutations, but also in wt-p53 cancer cells. 29 Additionally, doxorubicin upregulates survivin without inducing apoptosis in mutant-p53ALL cells. 26 Therefore, prodigiosin could be introduced as a new ALL-sensitizing agent to chemotherapeutic drugs due to its effect on decreasing survivin expression and induction of apoptosis in ALL cells with or without wt-p53. This hypothesis should be further explored.
Conclusions
Our findings provided the first evidence that wt-p53 and survivin as molecular targets of prodigiosin contribute to caspase-3-dependent apoptosis in ALL cells and this compound with high proapoptotic capacity represents an attractive p53- and survivin-modulating drug in ALL.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors express their great appreciation for the financial support received for this work from Iran National Science Foundation (INSF; Grant no.: 95820654).
