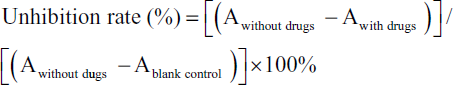Abstract
Background
Gliomas are harmful to human health; they are the most common primary intracranial tumor. Ginsenoside CK (GS-CK) is converted from the diol ginsenoside. This study aimed to explore the effects of GS-CK on glioma cells SHG-44 and U251MG in order to provide clinical value for the treatment of gliomas.
Materials and Methods
Cell proliferation was detected using the CCK-8 and CFU detection experiments. Cell apoptosis was detected by DAPI and acridine orange/ethidium bromide (AO/EB) fluorescence staining. Cell scratch and transwell assay were used to detect the effect of GS-CK on cell migration and invasion ability. Apoptosis-related protein expression was detected in the two cell lines after treatment with GS-CK.
Results
Cell proliferation is obviously inhibited, and cell migration and invasion were also significantly inhibited by GS-CK. It also induced cell apoptosis in a time- and dose-dependent manner. GS-CK induced significant changes in mitochondrial apoptosis pathway-related protein expression of cytochrome C, p53, Bax, Bcl-2, Caspase-3/8/9, Cleaved Caspase-3, and MMP-9 in glioma cells.
Conclusion
GS-CK can inhibit glioma cells by regulating mitochondria-related apoptosis p53-bax-caspases pathway.
Introduction
Glioma, especially glioblastoma, is a type of intracranial tumor that seriously endangers human health (Wu et al., 2012). Currently, surgery and systematic chemoradiotherapy based on pathological findings are the preferred treatments. In recent years, therapies targeting gene changes have rapidly developed and played an increasingly important role in glioma diagnosis and treatment. However, due to the lack of distinct demarcation between glioma and normal brain tissues, complete removal is difficult in cytology, and postoperative tumor recurrence is usually rapid with upgraded transformation. According to statistics, the average patient survival time after surgery is approximately 14 months (Van Meir et al., 2010). Various postoperative treatments, including chemoradiotherapy and electric field therapy, are designed to specifically induce programmed death of tumor cells. The ultimate antitumor mechanism of these measures is to induce apoptosis by acting on related signaling pathways (Bellail et al., 2009).
Ginseng is a traditional Chinese medicine known for its effect of “greatly tssssonifying vitality, tonifying spleen and lungs, generating saliva and relieving thirst, calming nerves and increasing intelligence.” Reported benefits of ginsenosides include antioxidant, antitumor, immunomodulatory, and antibacterial effects (Baik et al., 2021; Chen et al., 2021; Li et al., 2018; Liu et al., 2022; Zhou et al., 2015). Among these is ginsenoside CK (GS-CK), an unnatural ginsenoside that can be converted from other diol ginsenosides, which has shown excellent antitumor effects (Sun et al., 2021). However, there is limited research on the use of GS-CK in glioma, and its regulatory role in this disease has not yet been fully elucidated.
In our study, we investigated the anti-glioma effects of GS-CK on cells, including changes in phenotype such as cell proliferation, apoptosis, migration, and invasion. Additionally, we analyzed the expression of related proteins at a molecular level.
Materials and Methods
Materials
The materials were GS-CK (B21045, Shanghai Yuanye Bio-Technology Co., Ltd.), CCK-8 (MCE, China), acridine orange/ethidium bromide (AO/EB) (Solarbio, China), and DAPI (KGA215, KeyGENEBioTECH, China).
Cell Culture
The cell lines SHG-44 and U251MG were purchased from Procell Life Science. Cells were cultured with DMEM medium. Notably, 10% fetal bovine serum was always maintained in the medium. At the same time, 100 µg/mL of penicillin-streptomycin was maintained. The cells were grown in an incubator at 37°C with 5% CO2.
Cell Proliferation Assay
Notably, 3 × 103 cells in each well were inoculated (96-well plates). And the cells were co-incubated with different concentrations of GS-CK (50−200 µg/mL) for 24−72 h (Liu et al., 2018). For positive control, we chose 5 µM DOX. Then, the experiment was conducted according to the instructions (n ≥ 3). Then, the data were calculated by the following formula:
Colony Formation Unit Assay
Glioma cells (4 × 102 /well) were inoculated in a 6-well plate, they were co-incubated with different concentrations of GS-CK (50−200 µg/mL) for 21 days, and the new medicated medium was replaced six times. Furthermore, crystal violet staining experiments were used, different areas were selected, and photographs of the cells were magnified (100×) with a Nikon microscope (n ≥ 3) (Wan X. et al., 2022).
4′, 6-Diamidino-2-phenylindole Fluorescence Staining
Glioma cells (2 × 104/well) were inoculated in a 6-well plate and were co-incubated with different concentrations of GS-CK (50−200 µg/mL). The cells were washed with PBS and fixed with methanol after 24−72 h. Then, DAPI solution (100 µL) was added for 10 min, washed with PBS, and photographed with an inverted microscope.
AO/EB Fluorescent Staining
Glioma cells (2 × 104 /well) were inoculated in a 6-well plate, they were co-incubated with different concentrations of GS-CK (50−200 µg/mL) for 48 h, and 10 µL AO/EB (1:1) solution was added and mixed for 5 min. Then the cells were washed and photographed with a microscope (100×) (Wan et al., 2019).
Scratch Wound Healing Assay
When cells grew in a petri dish, the surface of the cell was scratched and then co-incubated with different concentrations of GS-CK (50−100 µg/mL). Images were taken under a microscope at 0−48 h, and the location of the photo was the same at each action time point under the same drug concentration (n ≥ 3).
Transwell Analysis
Glioma cells (105/well) were added into the chamber in 24-well plates. Medium containing GS-CK (50−100 µg/mL) was used for 48 h (n ≥ 3). Crystal violet stained cells and photos were taken under a microscope (200×) (Lin et al., 2021). Six photos of each concentration were taken (n ≥ 3).
Western Blotting
Protein samples (60 µg) were added to each well. Antibodies in this research were purchased from AB clonal. Primary antibodies were anti-β-actin (AC038), anti-Bcl-2 (A19693), anti-cytochrome c (A13430), anti-Bax (A19684), anti-caspase-3/8/9 (A19654/A11324/A0281), anti-MMP-9 (A11147), and anti-p53 (A3185). The dilute concentration of the primary antibody was 1:1000, and the dilute concentration of the secondary antibody was 1:5000. An ECL kit was used for visualization.
Statistical Analyses
All data were expressed as mean ± SEM, and analyzed using SPSS and Microsoft Excel software. Statistical analysis methods we use t-tests and one-way analysis of variance (ANOVA).
Results
GS-CK Inhibited Cells Proliferation in vitro
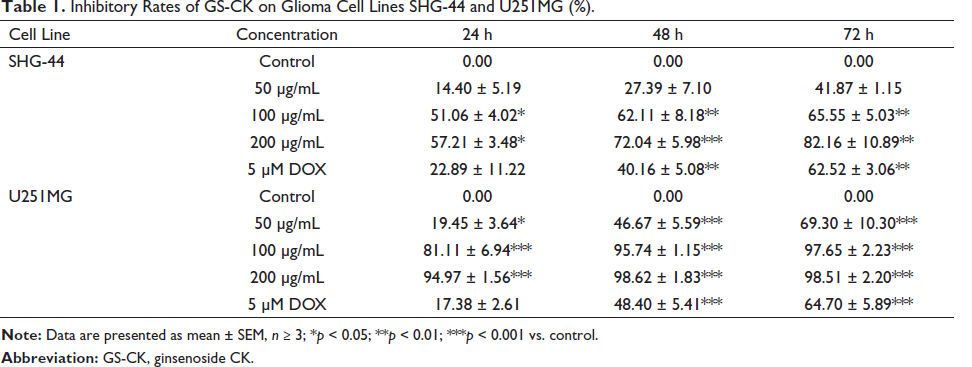
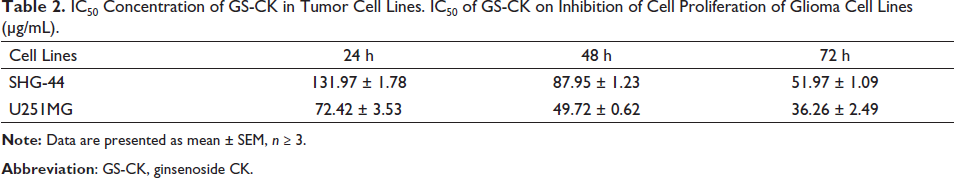
We tested the effect of GS-CK on the proliferation of glioma cells. As shown in Figures 1A, B and Table 1, GS-CK showed the highest inhibitory effect on SHG-44 glioma cells (82.16 ± 10.89%) and U251MG (98.51 ± 2.20%) at 200 µg/mL and after 72 h. The results showed that the inhibitory effect of GS-CK on glioma cells was significantly greater than that of the positive control 5 µM DOX. Then, the IC50 value was calculated (Table 2), and the inhibition rate of GS-CK on glioma cells ranged from 36.26 ± 2.49 to 131.97 ± 1.78 µg/mL. By comparison, GS-CK showed a stronger inhibitory effect on U251 cells than SHG-44. However, in glioma cells treated with GS-CK after 48 h, their IC50 values were significantly less than 100 µg/mL of the two glioma cells. Therefore, GS-CK showed excellent anti-tumor effects in glioma cells. We systematically monitored the cytopathological changes under different drug and time conditions. In Figure 1C, the changes in cell morphology and density after GS-CK intervention can also indicate its significant inhibitory effect. GS-CK inhibited CFU of glioma cells in vitro.
Inhibitory Rates of GS-CK on Glioma Cell Lines SHG-44 and U251MG (%).
IC50 Concentration of GS-CK in Tumor Cell Lines. IC50 of GS-CK on Inhibition of Cell Proliferation of Glioma Cell Lines (µg/mL).
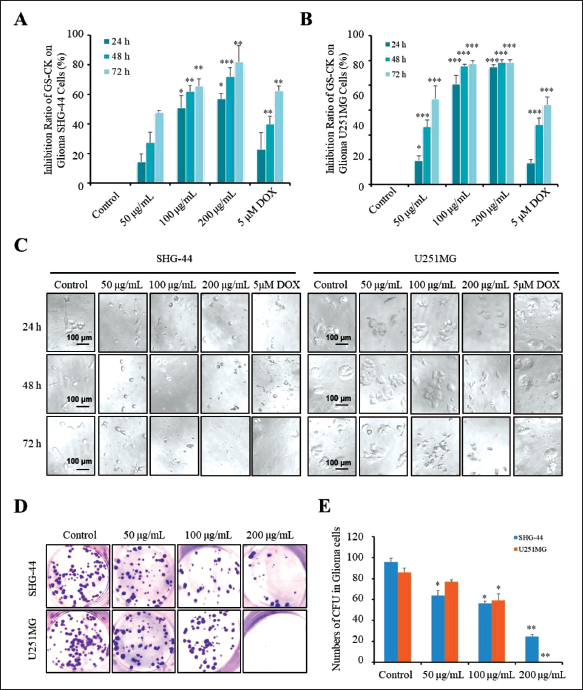
A colony formation unit assay was performed to further verify the inhibitory effect of GS-CK on the proliferation of glioma cells. Glioma cells were treated with different concentrations of GS-CK. Compared with the control group, GS-CK showed a stronger inhibitory effect on the colony formation ability of U251MG cells (Figure 1D and E). As shown in Table 3, the number of the two cell colonies in the control group was 95.80 ± 3.62 and 85.67 ± 4.55. When the concentration of GS-CK was 200 µg/mL, the number of SHG-44 and U251MG cell colonies decreased significantly to 24.76 ± 1.45 and 0.33 ± 0.19, respectively.
The CFU of Glioma Cell Lines SHG-44 and U251MG Under Different Concentrations of GS-CK (%).

GS-CK-induced Glioma Cells Apoptosis
In the experiment exploring the morphology of GS-CK-treated glioma cells, it was observed that the number of suspended cells in the medium increased significantly after GS-CK was treated, indicating that GS-CK not only affects cell viability but also has a high potential to induce cell apoptosis. Based on the IC50 values of GS-CK on glioma cells, a concentration of 100 µg/mL exhibited a significantly higher inhibition rate of more than 50%. In consideration of the data obtained from cell morphology and cell density analyses, a concentration of 200 µg/mL resulted in very few viable cells. For the remaining experiments, the maximum concentration used was 100 µg/mL.
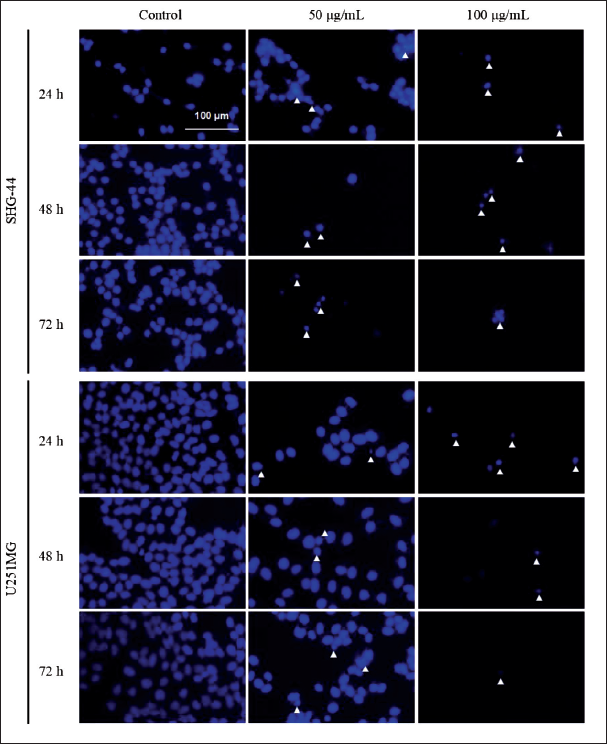
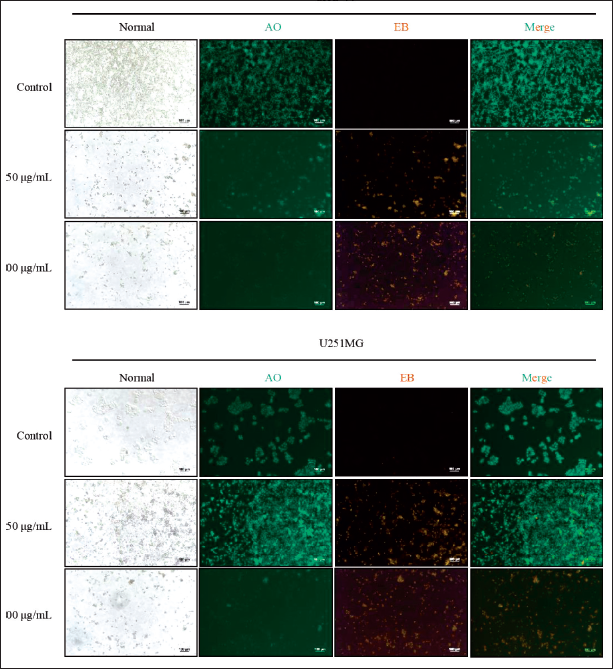
Fluorescent cell staining with DAPI and AO/EB was used to investigate the effect of GS-CK glioma cells, and the results are shown in Figures 2 and 3. With a progressive increase in the GS-CK concentration and administration duration, a large number of densely stained nuclei were observed in glioma cells stained with DAPI, along with the appearance of apoptotic bodies. This indicates an increased proportion of apoptotic cells (Figure 2). Figure 3 shows four types of cells captured under a fluorescence microscope: green and normal (living cells); green pyknosis and bead (early apoptotic cells); orange and normal (non-apoptotic dead cells); and orange pyknosis or bead (late apoptotic cells). As compared with the control group, a significant increase in the proportion of apoptotic cells was observed in the GS-CK-treated group. GS-CK limited cell migration and invasion of glioma cells.
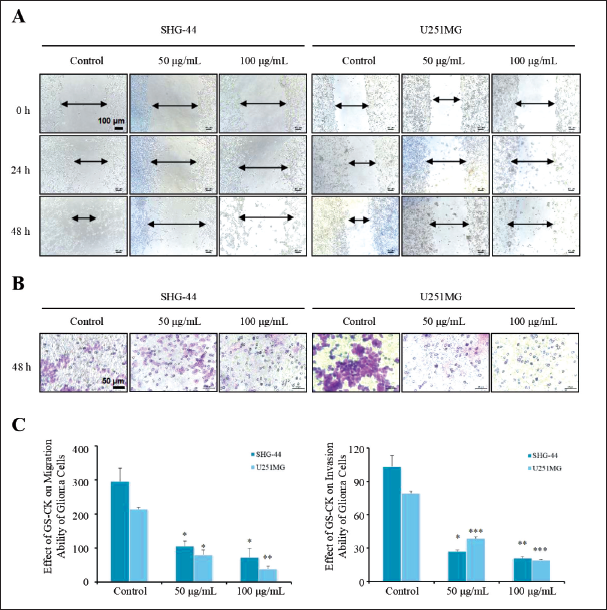
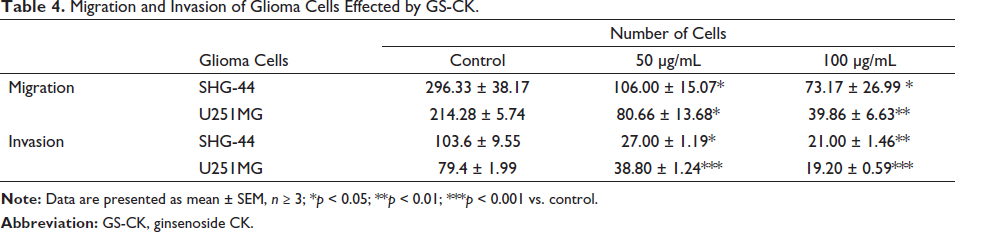
In order to investigate the result of GS-CK on the migration and invasion of glioma cells, we performed scratch wound healing and transwell assays. The results showed that glioma cells grew toward the middle after 24 and 48 h, and the scratch area did not heal in the GS-CK treated group (Figure 4A). The area of the blank space was calculated in each group of pictures, and statistical analysis was performed as shown in Figure 4C. When GS-CK concentration was 50 and 100 µg/mL, the cell migration ability was significantly inhibited. Under the same culture conditions, the cells were monitored again to investigate invasion ability in the matrigel chamber. After 24 h of drug treatment, the migration of glioma cells was inhibited, but not significantly. Cell migration and invasion were significantly inhibited as shown in Figure 4B and C. The data are shown in Table 4. GS-CK regulates the expression of proteins in glioma cell lines
Migration and Invasion of Glioma Cells Effected by GS-CK.
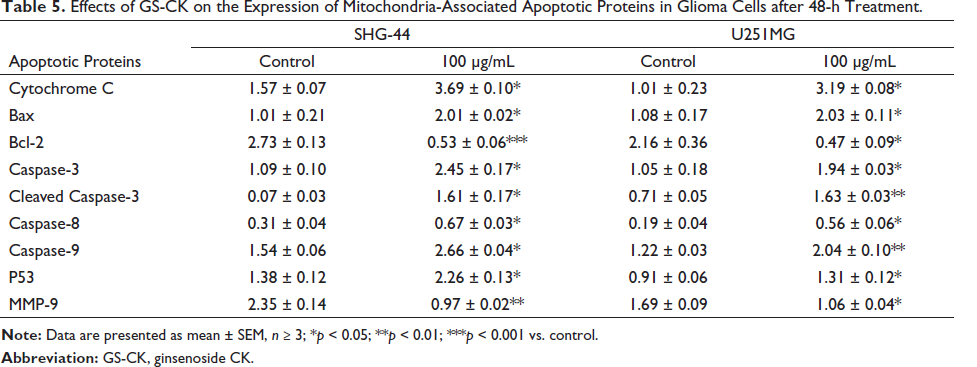
Furthermore, western blotting was performed to detect the expression levels of proteins related to cell apoptosis and migration including the mitochondrial apoptosis proteins Bax, Bcl-2, cytochrome C, and p53, as well as caspase-3/8/9. The protein expression level of MMP-9 was also evaluated to examine the effect of GS-CK on proteins associated with glioma cell migration and invasion. The results are shown in Figure 5A. After analysis, significant changes were observed in the protein expression levels after treatment with GS-CK at concentrations of 50 and 100 µg/mL (shown in Figures 5B, 6A, and Table 5). This suggests that GS-CK exerts an anti-glioma effect through the potential pathway involving p53-bax-caspase.
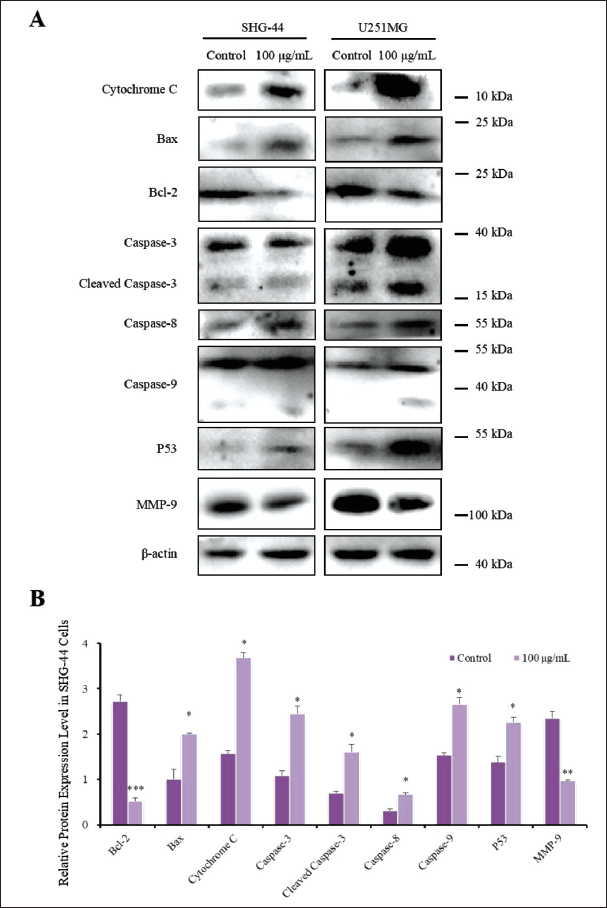
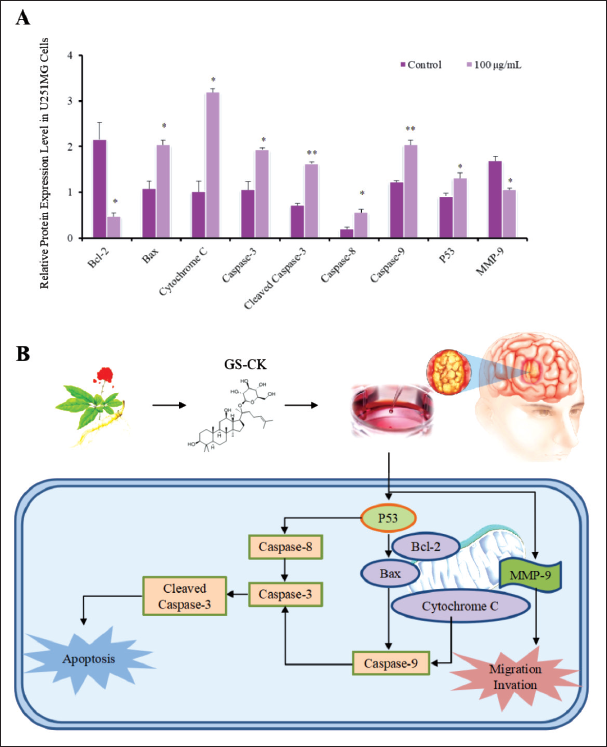
Effects of GS-CK on the Expression of Mitochondria-Associated Apoptotic Proteins in Glioma Cells after 48-h Treatment.
Discussion
Glioma is the most common intracranial tumor worldwide with high mortality and disability rates (Abi et al., 2021). Recent molecular pathologic studies of gliomas have revealed that molecular drivers have a significant impact on disease treatment and overall prognosis. This has changed the traditional paradigm of glioma classification and treatment, redefined our understanding of gliomas, and revealed the potential feasibility of intervening with relevant factors to tackle this intractable and deadly disease (Ruff et al., 2020). In our study, we found that GS-CK inhibited the proliferation and colony-forming ability of glioma cells, as well as their migration and invasion while promoting apoptosis in these cells.
GS-CK is an active metabolite of ginsenoside Rb1, which is an effective anti-inflammatory substance (Wang et al., 2021). In recent years, several studies have reported the significant antitumor activity of GS-CK, particularly its inhibition of proliferation and invasion in various human tumors, such as osteosarcoma, hepatocellular carcinoma, lung cancer, cervical cancer, breast cancer, and esophageal cancer (Chen et al., 2020; Xue et al., 2021; Yin et al., 2021; Zhang et al., 2021, 2022). GS-CK interferes with tumor cells in several ways, including inhibiting tumor growth-regulating autophagy and endoplasmic reticulum stress, inducing tumor cell apoptosis, targeting glutamine metabolism, hypoxia-induced epithelial–mesenchymal transformation (EMT), and suppressing proliferation and invasion of tumor cells. With the disclosure of more anti-tumor Chinese medicine, the systematic anti-tumor measures of Chinese medicine and Western medicine can achieve a better therapeutic effect (Xu et al., 2011).
Recent research has revealed that GS-CK inhibits the expressions of the anti-apoptosis-related protein Bcl-2 and apoptosis-related pathway PI3K/AKT/NF-κB, while promoting the expression of pro-apoptosis proteins Bax and caspase 3 in gastric cancer HGC-27 cells, whereas ginsenoside Rb1 had no effect. EMT plays a vital role in the progression and prognosis of HCC, and GS-CK has been shown to inhibit the HIF-1α/NF-κB signaling pathway and expression levels of EMT-related proteins and cytokines in hypoxia-induced or TNF-α-stimulated hepatoma cell lines (Wan Y. et al., 2022). These studies demonstrated that GS-CK exerted very strong antitumor effects with different mechanisms of action on different tumor cells.
Previous research has shown that GS-CK shows obvious anticancer activity by inhibiting cell proliferation, inducing cell cycle arrest, and promoting cell apoptosis. The results of this study demonstrated that GS-CK can effectively inhibit the proliferation and colony-forming units of glioma SHG-44 and U251MG cells, with the maximum inhibition rates being 82.16 ± 10.89% and 98.51 ± 2.20%, respectively. Additionally, nuclear fluorescence staining results confirmed that GS-CK induced apoptosis of glioma cells. Moreover, GS-CK demonstrated a significant inhibitory effect on the migration and invasion abilities of glioma cells, particularly U251MG cells. The study also found that GS-CK affects the protein expression levels of apoptosis and migration/invasion-related proteins in glioma cells. These findings suggest that GS-CK can significantly inhibit multiple phenotypes of glioma cells. However, further research is needed to explore the mechanism and molecular targets of GS-CK in inhibiting glioma cells.
Conclusion
In this study, it was found that GS-CK effectively inhibited the proliferation and CFU of glioma cell lines SHG-44 and U251MG. Additionally, GS-CK induced varying degrees of cell apoptosis in the fluorescent staining experiment. Moreover, the cell scratch experiment and transwell assay demonstrated the ability of GS-CK to limit cancer cell migration and invasion. Western blot analysis suggested the involvement of GS-CK in regulating the “p53-bax-caspase pathway” related to apoptosis and migration in cells, contributing to the inhibitory effect on glioma cells. These results suggest that GS-CK could be a potential drug candidate for the future treatment of glioma.
Summary
Ginsenoside-CK inhibits the proliferation of glioma cell lines SHG-44 and U251MG.
Ginsenoside-CK promotes apoptosis of glioma cell lines SHG-44 and U251MG.
Ginsenoside-CK limits the migration and invasion ability of glioma cell lines SHG-44 and U251MG.
Ginsenoside-CK regulates glioma cells through matrix metalloproteinase family members and mitochondrial apoptosis-related proteins.
Footnotes
Acknowledgments
The authors are grateful to all those who helped in this study.
Abbreviations
GS-CK: Ginsenoside CK.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval and Informed Consent Statement
This study was approved by the First Hospital of Jilin University on March 4, 2021. Informed consent doesn’t apply because the study did not involve animal or human experiments.
Funding
This work was financially supported by the Science and Technology Development Plan Project of Jilin Province (20210204074YY and 20230508166RC) and the Program of Science and Technology Research Project of the Education Department of Jilin Province (JJKH20220876KJ).