Abstract
Ischemic stroke is a kind of sudden vascular disease of the brain brought on by ischemic hypoxic necrosis of the brain tissue which is becoming a significant global source of death and long-term impairment. Present common treatment options for cerebral ischemia including thrombolytic therapy, mechanical embolization, and intracranial arterioplasty can only benefit a few patients due to the narrow therapeutic time windows. Traditional Chinese Medicine (TCM) prescriptions possess the qualities of multi-ingredients, multi-targets, synergistic and multi-efficacy advantages as a means of treating ischemic stroke. Taohong Siwu Decoction (THSWD) is a famous TCM prescription that promotes blood flow, removes blood clots, adds nourishment, and tonifies blood. Clinical and laboratory research results show the significant advantages of THSWD in ischemic stroke treatment. This review summarizes THSWD’s natural materials and processing methods, pharmacological effects, and related mechanisms to treat ischemic stroke. Its mechanism of action focuses on improving hemorheology, anti-inflammatory response, reducing excitotoxicity, anti-calcium overload, anti-oxidative stress, etc. Moreover, we systematically summarize THSWD and its use in combination with other treatments. Finally, the research obstacles of THSWD in the treatment of ischemic stroke are summarized, and the application prospects in the medical field are prospected.
Introduction
Strokes are divided into ischemic stroke and hemorrhagic stroke, and over 80% of all acute strokes are ischemic stroke which is the main global cause of death and chronic impairment (Sarvari et al., 2020). Ischemic stroke is a result of an abrupt decrease or loss of blood circulation inside brain tissue, causing an equivalent loss of neurological function and brain harm (Lakhanet al., 2009). Following brain ischemia, cellular and metabolic damages will cascade, including oxygen and glucose deficiency, neuroinflammation (Maidaet al., 2020), oxidative stress damage, alternated autophagy and apoptosis (Duris et al., 2018; Sekerdag et al., 2018), excitotoxicity (Khoshnam et al., 2017), and dysregulated angiogenesis (Hatakeyama et al., 2020), excitatory amino acid toxicity, free radical damage, calcium overload, blood-brain barrier (BBB) damage, and other pathophysiological processes. At present, the main goal of stroke treatments is to open blocked blood arteries immediately using thrombolytic mechanical recanalization. Recombinant tissue plasminogen activator (rt-PA) is the only medication that has currently received FDA approval (Wardlaw et al., 2014). The therapeutic window for rt-PA, however, is severely constrained within 4.5 hours of stroke, and there is a high chance of hemorrhagic transformation, which can have serious repercussions and cause permanent disability. Recently systemic injection combined with localized BBB disruption has gained popularity as a combination approach. However, non-selective BBB opening may allow hazardous chemicals to reach the brain (Jiang et al., 2018). Thus, an appropriate, and reliable therapeutic approach for multiple pathological events occurring in cerebral ischemia is necessary and urgently needed to reduce mortality (Johnson et al., 2016).
For thousands of years, Chinese people have used Traditional Chinese Medicine (TCM), and it has been shown in various clinical and laboratory studies that TCM is beneficial in treating ischemic stroke. TCM prescriptions possess the qualities of multi-ingredients, multi-targets, synergistic and multi-efficacy advantages (Duan et al., 2018; Wang et al., 2023). TCM has been demonstrated to have effective neuroprotective and repairing properties, including preserving BBB function, lowering excitatory amino acid toxicity, controlling energy metabolism, increasing anti-oxidation, anti-inflammatory, and anti-apoptosis, as well as boosting neurogenesis, angiogenesis, and synaptogenesis (Wang, Xiao, et al., 2021). The famous classic TCM prescription Taohong Siwu Decoction (THSWD) has noteworthy adjuvant therapeutic effects for ischemic stroke. Clinical practice shows that treatment of ischemic stroke with THSWD outside the onset time window still has neuroprotective effects (Huang et al., 2014). This review article systematically summarizes THSWD-related therapeutic applications and mechanisms intended to offer direction and fresh concepts for the clinical management of ischemic stroke.
THSWD and Its Applications on Ischemic Stroke
Taohong Siwu Decoction
THSWD is listed in the first batch of classic prescriptions published by the National Administration of TCM and has been applied to treat bloating, discomfort, blood clots in menstrual blood, and irregular menstruation brought on by blood stasis for over 700 years in China. THSWD was first recorded in “Yilei Yuanrong” compiled by Wang Haogu in the Yuan dynasty and recorded in the renowned medical text Yizong Jinjian, which Wu Qian put together in Qing Dynasty. THSWD is frequently used to increase blood flow and remove blood clots. THSWD, for instance, is frequently used to treat disorders in women brought on by scarcity and stasis of postpartum blood. It also plays a vital role in enhancing hemorheology and mediating anticoagulation (Xu, 2011) (Table 1).
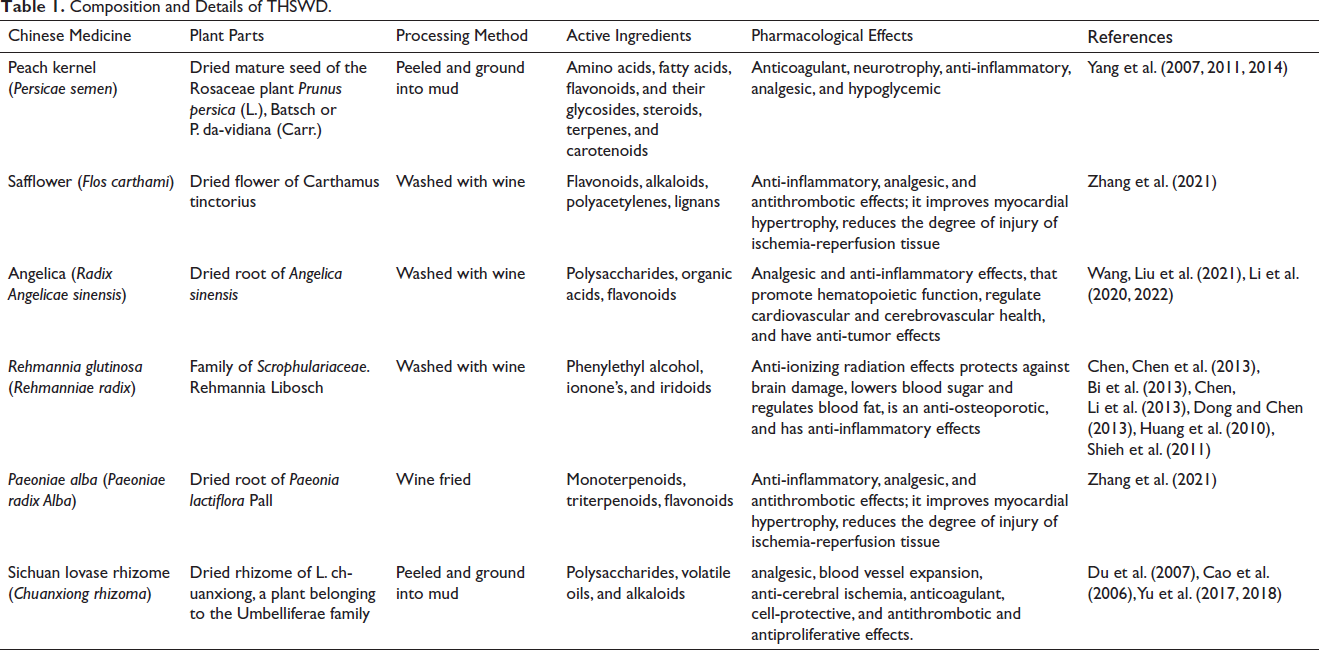
Composition and Details of THSWD.
The Effect of Pharmacological Components of THSWD on Ischemic Stroke
THSWD is composed of six natural medicines: Peach kernel (Persicae semen), Safflower (Flos carthami), Angelica (Radix Angelicae sinensis), Rehmannia glutinosa (Rehmanniae radix), Paeoniae alba (Paeoniae Radix Alba), Sichuan lovase rhizome (Chuanxiong rhizoma). Ferulic acid, kaempferol, quercetin, paeonol, paeoniflorin, ligustilide, and caffeic acid are the quality control indicators of THSWD. Their respective pharmacological actions participate in the management of ischemic stroke. (a) Peach kernel (P. Semen): Amygdalin is the quality control detection index of peach kernel. Modern research has revealed that peach kernels can expand peripheral blood vessels, increase blood flow, reduce platelet aggregation, promote fibrinolysis, reduce blood viscosity, resist thrombosis, and protect against regional cerebral ischemia (Yi, 2012). Peach kernel oil can reduce atherosclerotic lesions and reduce the incidence of ischemic stroke by regulating blood lipid levels (Hao et al., 2019). (b) Safflower (F. carthami): Quercetin and kaempferol are the quality control detection indicators of safflower, and the former is a common component of safflower and peach kernel. Safflower, quercetin, and kaempferol may be the main components of the peach kernel-safflower drug pair synergistically promoting blood circulation and improving hemorheology in rats with acute blood stasis (Yang et al., 2018). The safflower yellow pigment in safflower can promote the formation of new blood vessels in the cerebral infarction area and the reconstruction of collateral vessels. The therapeutic effects of dehydrosafflower yellow B and hydroxysafflor yellow A on cerebral ischemia-reperfusion injury may be related to the targeted regulation of the SIRT1 signaling pathway, the inhibition of platelet aggregation, the inhibition of ferroptosis and parthanatos death, and the attenuation of oxidative stress and apoptosis. Death and other mechanisms of action play a synergistic role (Chen et al., 2022; Fangma et al., 2021). (c) Angelica (Radix Angelicae sinensis): Angelica polysaccharides can impede the formation of platelet aggregation and the apoptosis of rat hippocampal neurons to protect against cerebral ischemic injury (Liet al., 2019). The organic acids and volatile oils contained in angelica can effectively regulate blood lipids and treat cerebrovascular atherosclerosis. Angelica root methanol extract can effectively improve ischemic brain injury by activating MAPK and another brain signaling pathway (Leeet al., 2021). (d) R. glutinosa (R. Radix): Sugars and verbascoside are the quality markers of R. glutinosa. R. glutinosa polysaccharides have significant protective effects on cerebral ischemia and brain injury and can slow down the cerebral edema caused by ischemia and reduce the volume of cerebral infarction in middle cerebral artery occlusion (MCAO) mice. Verbascoside can regulate the cholinergic pathway and play a neuroprotective role in ischemic brain injury (Li et al., 2018). (e) P. alba (P. radix alba): Kaempferol is a common component of safflower and white peony, and its anti-oxidation and anti-inflammatory pharmacological activities have protective effects on brain damage. Paeoniflorin is the quality control detection index of Radix Paeoniae Alba. Paeoniflorin and amygdalin, as the core active ingredients of THSWD, have a synergistic effect on promoting angiogenesis. The mechanism of action may be related to the activation of the EGFR HIF-VEGFA signaling pathway. The synergistic effects of paeoniflorin, chuanxionglide, and paeoniflorin on anti-oxidative stress, anti-inflammation, and anti-apoptosis may be related to the regulation of the SIRT1-FoxO1 signaling pathway. (f) Sichuan lovase rhizome (C. rhizoma): Ligustilide, a common component of Umbelliferae Angelica and Chuanxiong, can play a neuroprotective role in patients with ischemic stroke (Xie et al., 2020). The common component, ferulic acid, can eliminate a large number of oxygen free radicals produced in the later stage of ischemic stroke by inhibiting the production of free radical enzymes and playing an anti-oxidative role. It can regulate blood lipids, improve microcirculation, prevent organ ischemia, and resist platelet aggregation, etc. (Li et al., 2021).
In vivo studies reviewed the bioactive components and metabolites of THSWD, and the primary metabolic pathways of THSWD in vivo were shown to be hydrolysis, glucuronidation, and sulfation (Duan et al., 2020). Histidine metabolism was discovered to be involved in the way that THSWD treated acute blood stasis. Metabolisms of tyrosine, arachidonic acid, and glycerophospholipid, are also closely related to the metabolites of THSWD (Duan et al., 2020; Ma et al., 2018). Different metabolites are closely related to the pharmacological effects of THSWD. Dai et al. (2023) studied the distribution of THSWD in various tissues of MCAO rats, the findings indicated five major constituents of the THSWD including ligustilide, amygdalin, paeoniflorin, hydroxysafran yellow A, and ferulic acid were widely distributed in brain tissues, and there were drug interactions between different components. Mudanpioside E, paeoniflorin, lactiflorin, benzoylpaeoniflorin, β-benzoyloxypaeoniflorin are the main material basis for THSWD to treat ischemic stroke (Duan, 2019). Wang et al. (2017) and others found that catalpol, ligustilide, ferulic acid, paeoniflorin, paeonifloride glycoside, and amygdalin six blood components may be the direct effective components of THSWD by using LC-MS/MS technology. Li et al. discovered that ferulic acid, amygdalin, paeoniflorin, and other components can repair neuron damage, regulate the level of neurotransmitters in the brain, and improve cerebral vascular circulation, suggesting that they are the material basis for protecting the brain nervous system (Li & Guo, 2016). Duan et al. detected THSWD active ingredients and their metabolites were found in both blood and brain using UHPLC-Q-TOF-MS techniques. They found that flavonoids, aromatic organic acids, and benzoquinones were the notable active ingredients of THSWD, which could improve cerebrovascular circulation and promote blood flow by anti-ADP inducing platelet aggregation and inhibiting thrombin activity (Duan et al., 2020). Safflower, quercetin and kaempferol may be the main components of the peach kernel-safflower pair synergistically promoting blood circulation and improving the hemorheology of rats with acute blood stasis (Yang et al., 2018). Aromatic acids, glycosides and phthalides in THSWD can improve cerebral vascular circulation and promote blood circulation by anti-ADP-induced platelet aggregation and inhibition of thrombin activity.
Clinical Applications of THSWD to Treat Ischemic Stroke
The original or modified THSWD or THSWD combined with other prescriptions have been clinically utilized extensively to treat ischemic stroke and show significant treatment effects (Table 2).
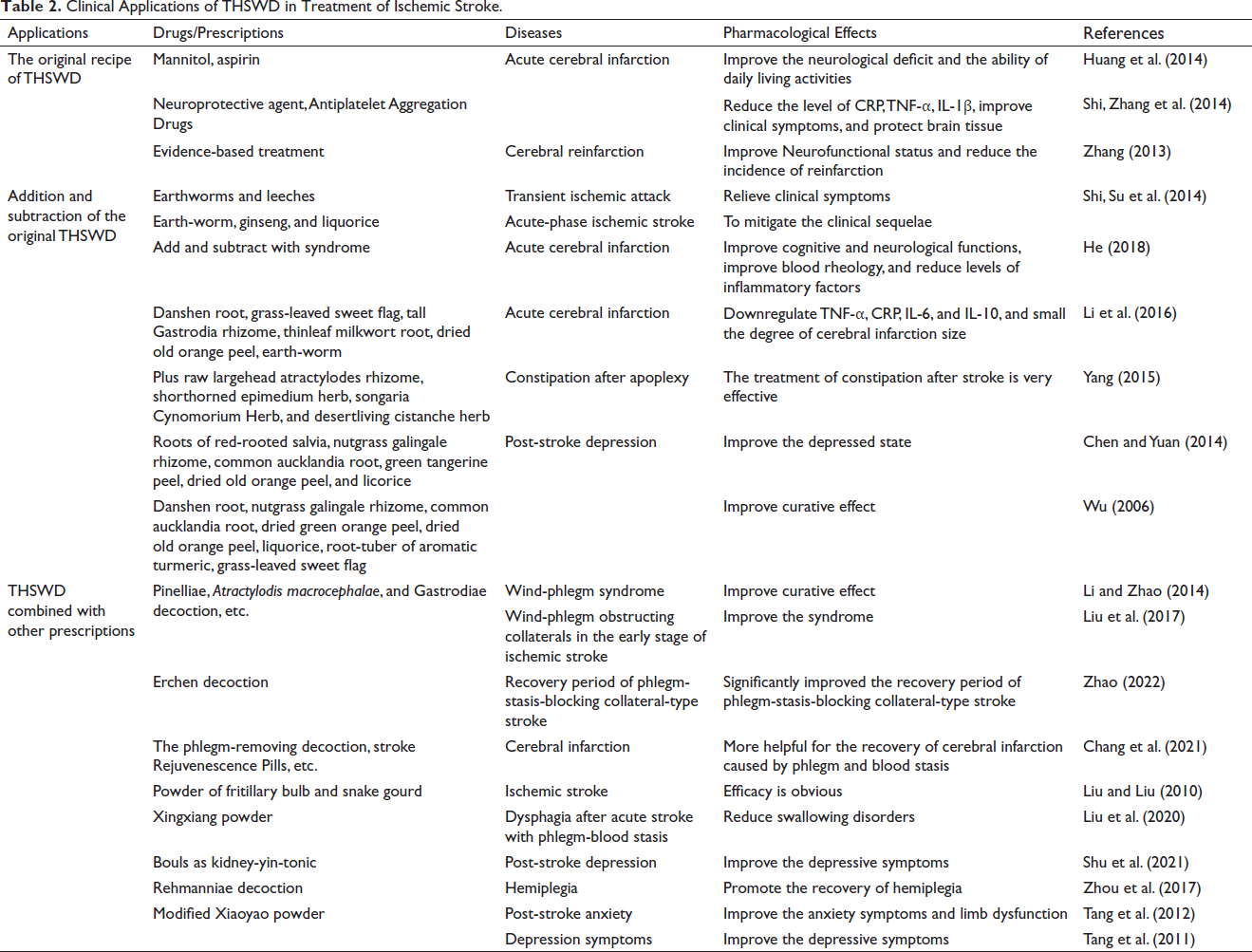
Clinical Applications of THSWD in Treatment of Ischemic Stroke.
The Original Recipe of the THSWD
THSWD promotes blood circulation without harming the normal climate and has been used extensively to treat ischemic stroke. Clinical application of the original THSWD has shown that THSWD can protect brain tissue, reduce the incidence of reinfarction, reduce serum inflammatory factors (Shi, Zhang, et al., 2014), improve clinical symptoms and improve the neurological deficit score National Institutes of Health Stroke Scale (NIHSS) of ischemic stroke patients with acute cerebral infarction (Huang et al., 2014).
Addition and Subtraction of the Original THSWD
Modified THSWD by adding and subtracting Chinese herbs in THSWD according to distinct patient syndrome could greatly improve the treatment effects. THSWD plus earthworm, ginseng, and liquorice to treat acute ischemic stroke with an aura of stroke syndromes achieved significant healing effect (Shi, Su, et al., 2014). Yang et al. reported that THSWD plus raw large head atractylodes rhizome, short-horned epimedium herb to treat constipation after apoplexy according to abdominal distension syndromes, while plus milkvetch root and pilose asiabell root according to spontaneous sweating (Yang, 2015). The root of red-rooted salvia promotes blood flow to remove blood clots, licorice nourishes blood and astringing yin, and softens the liver. THSWD-added roots of red-rooted salvia, and nutgrass galingale rhizome showed improved effects on patients with post-stroke depression (Chen & Yuan, 2014). THSWD combined with injection Mannitou, deproteinized calf blood extractives injection, alprostadil, and edaravone can downregulate Tumor Necrosis Factor-α (TNF-α) and other inflammatory factors, small the degree of cerebral infarction size in acute cerebral infarction patients (Li et al., 2016).
THSWD Combined with Other Prescriptions
THSWD combined with other TCM prescriptions is also widely employed to treat ischemic stroke according to patients with distinct syndrome. THSWD plus the Pinelliae, Atractylodis macrocephalae, and Gastrodiae decoction can alleviate the ischemic stroke dysfunction condition (Li & Zhao, 2014; Liu et al., 2017). THSWD combined with Erchen decoction to treat ischemic stroke with lower NIHSS scores and higher Barthel index significantly improved the recovery period of stroke (Zhao, 2022). THSWD combined with phlegm-removing decoction improved the scores of neurological deficits and the recovery of cerebral infarction (Chang et al., 2021). THSWD plus fritillary bulb powder and snake gourd improved the total effective rate (the rate of basically healing, effective, and progressive cases according to “Stroke diagnosis and efficacy evaluation criteria” in 1996) (Liu & Liu, 2010). THSWD plus Xingxiang powder-treated patients with dysphagia after stroke showed an improvement in neurological function (Liu et al., 2020). Post-stroke anxiety and depression is a common sequela of ischemic stroke. Modified Xiaoyao powder together with THSWD improved patients’ post-stroke anxiety and depression symptoms (Tang et al., 2012; Tang et al., 2011). THSWD combined with acupuncture to treat patients with acute cerebral infarction and achieved a curative effect (Zhou et al., 2017).
Conventional Western medicine combined with THSWD can improve the clinical manifestations of ischemic stroke-related diseases to a certain extent, which may be better than monotherapy. The curative effect of the original prescription of THSWD is related to improving the clinical symptoms of patients with ischemic stroke, reducing the neurological deficit, and reducing the incidence of re-infarction. The addition and subtraction of other drugs, combined with other prescriptions, have a wider range of applications, involving the improvement of anxiety symptoms and limb dysfunction, depression symptoms, swallowing disorders, post-stroke constipation, and other sequelae. The addition and subtraction versions or combined use with other prescriptions are based on THSWD, which can be used for different syndrome types and help patients recover. However, these fields need additional study group types due to the small number of research, lack of standardization of studies, analysis of large clinical samples, and simultaneous samples. Doctors have different preferences regarding medication; therefore, there is no consensus on the use of specific drugs. THSWD treats strokes with different syndrome types. The literature shows that although there are syndrome types, the objectiveness of syndrome evaluation is poor, resulting in inaccurate syndrome evaluation. Additionally, the unequal quality of TCM in the literature study and the irregular amount of various THSWD TCM constituents might have a negative impact on the study’s findings. Finally, due to limited observation time and lack of long-term follow-up trials, definitive systematic reviews are still lacking.
Mechanisms of THSWD to Treat Ischemic Stroke
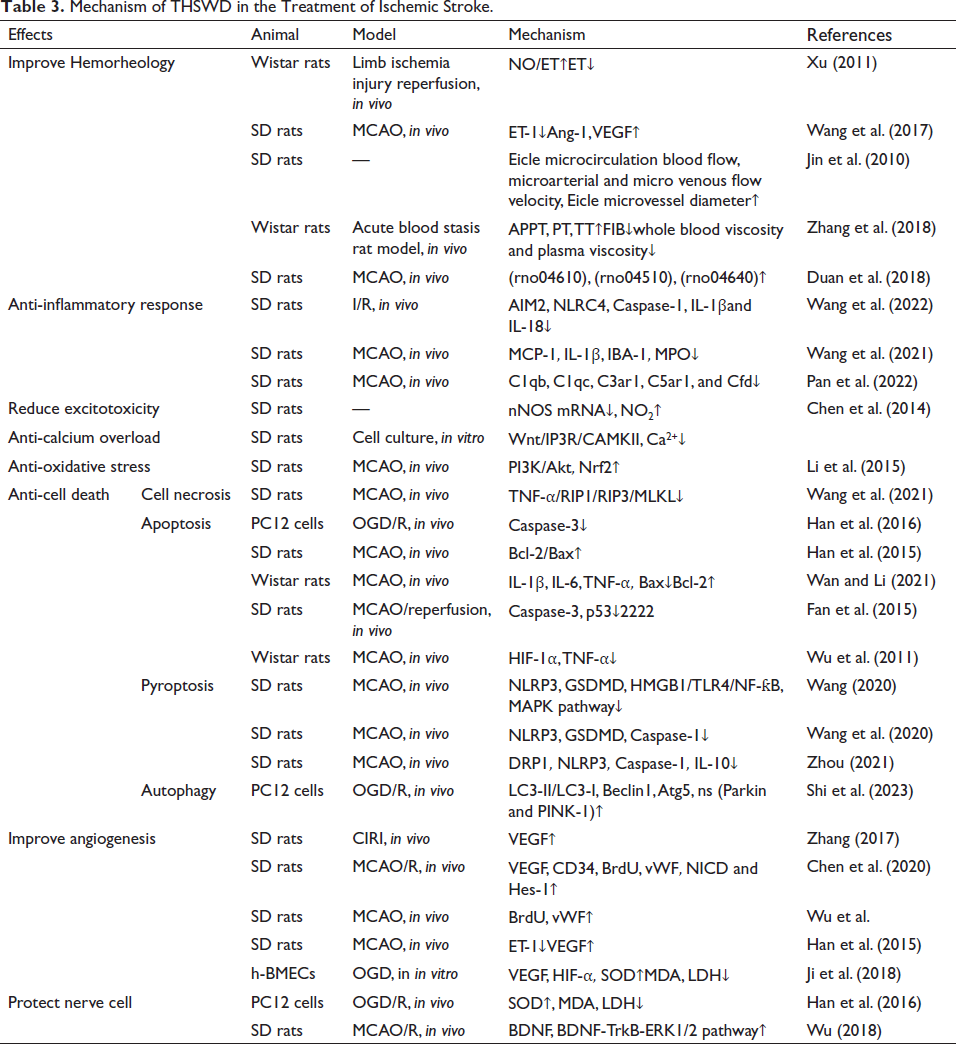
Reduced oxygen, ATP, and glucose levels in brain tissue following cerebral ischemia result in cell death. Numerous processes, such as apoptosis, necrosis, autophagy, and pyroptosis, are involved in cell death (Zhang et al., 1998). Recent studies have demonstrated that multiple factors including inflammatory response, excitotoxicity, calcium overload, free radicals release, and oxidative stress, are essential for the development of cell death following an ischemic stroke (George & Steinberg, 2015). The mechanisms of THSWD in the treatment of ischemic stroke include improved hemorheology, anti-inflammatory response, anti-oxidative stress, anti-calcium overload, promote angiogenesis, and anti-cell death (Figure 1 and Table 3).
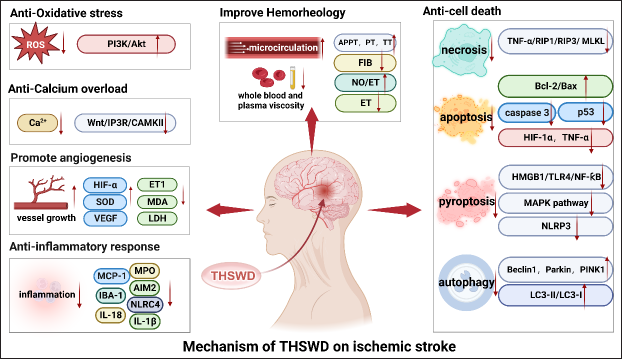
Mechanism of THSWD in the Treatment of Ischemic Stroke.
Improve Hemorheology and Microcirculation
An animal study demonstrated that THSWD significantly improved the condition of rats’ cerebral infarction, and dose-dependently reduced the region of cerebral infarction of rats (Xu, 2011; Wang et al., 2017). Microcirculation can release different vasoactive substances by regulating vascular tension to meet the metabolic needs of all cells in the body (Tibiriçá et al, 2018). Microcirculation disorders, organ tissue ischemia, and hypoxia are secondary to a series of pathophysiological processes that damage the body. THSWD increases microcirculation blood flow in blood deficiency and blood stasis animal models (Jin et al., 2010). THSWD significantly reduce blood pressure, whole blood viscosity, and plasma viscosity, and can significantly prolong partial thrombin enzyme time prothrombin time, thrombin time, and fibrinogen content (Zhang et al., 2018). Duan et al. identified the potential therapeutic targets of THSWD including complement and coagulation cascade reaction (rno04610), focal adhesion (rno04510), and hematopoietic cell pedigree (rno04640) using high-throughput mRNA sequencing in the treatment of experimental MCAO model. The results suggested that THSWD reverse MCAO by inhibiting the coagulation cascade (Duan et al., 2018).
Anti-inflammatory Response
The inflammatory response is a crucial stage in the pathophysiology of cerebral ischemia and directly linked to cell death. Neutrophils and monocytes, which are circulating immune cells, are infiltrated during the induction of inflammatory events, and immune cells that have been activated can express and produce cytokines that are both pro- and anti-inflammatory (Liu et al., 1994; Muir et al., 2007). The activation of inflammatory cells is associated with the toxic effects of ROS, necrotic cells, damaged tissue, proteolytic enzymes, and pro-inflammatory cytokines such as TNF, IL-1, and IL-6 (Khoshnam et al., 2017). Inflammation further aggravates brain tissue damage. Therefore, reducing the inflammatory response is of significance in the treatment of post-stroke injuries. THSWD reduce the manifestation of inflammatory substances such as MCP-1 and IL-1β, improve the inflammatory response caused by cerebral ischemia, and thus treat ischemic stroke (Pan et al., 2022; Wang et al., 2022; Wang, Fei, et al., 2021).
Reduce Excitotoxicity
Excitotoxicity is mainly due to an increase in glutamate (Glu) caused by cerebral ischemia, and a series of biochemical cascade reactions lead to nerve cell necrosis (Jiang et al., 2022). The brain needs to transfer energy through the electron transport chain, which leads to oxidative phosphorylation and the reduction in ATP synthesis, ATP depletion secondary to calcium overload, and activation of death signaling proteins, leading to cell death. At the same time, a high level of Glu exists inside brain tissue, and a large amount of excitatory neurotransmitters accumulate extracellularly, causing excitotoxicity (Duris et al., 2018) and leading to neuronal degeneration. Glutamate receptor (NMDAR)-mediated activation of n-nitric oxide synthase (NOS) is key to neuronal excitotoxicity factor.
THSWD reduces the coupling between NMDAR and nNOS and the excitotoxicity of cerebral ischemia. Chen et al. (2014) established the MCAO model after intragastric administration of different doses of THSWD to rats and detected that the expression of nNOS mRNA in the cerebral infarction area was down-regulated, the concentration of NO2 in the serum increased and returned to the level before cerebral ischemia.
Anti-calcium Overload
Calcium overload is a leading factor in cerebral ischemia/reperfusion injury. The Na+/K+ pump and the plasma membrane Ca2+/ATP pump malfunction following cerebral ischemia, the intracellular calcium concentration increases, and the extracellular accumulation of Glu stimulates AMPA and NADA receptors, enhancing the influx of calcium and sodium ions. Protease, lipase, and nuclease-mediated catabolic activities are triggered by calcium influx (Ankarcrona et al., 1995). As additional cell death triggers, nitric oxide (NO), arachidonic acid metabolites, and superoxide are produced as a result of the activation of nNOS, PLA2, and other Ca2+-dependent enzymes (Dirnagl et al., 1999). The combination of calcium ions and calmodulin increases the secretion of 5-hydroxytryptophan and norepinephrine and aggravates cerebral ischemic injury (Liu et al., 2012).
Glu keeps the Na+ channels open and continues to excite receptors after excitotoxicity has occurred. As sodium channels remain open, the intracellular osmotic pressure increases, leading to acute neuronal death. When Glu binds to the NMDAR, Mg2+ is removed, calcium channels are blocked by magnesium ions at rest, and cellular Ca2+ increases, activating the PSD95-NR2B-NOS ternary structure (Christopherson et al., 1999). NOS is released by the activated ternary, which causes the production of reactive nitrogen species (RNS), which are free radicals., activating the proteins transient receptor potential melastatin subfamily members 7 (TRPM7) and 2 (TRPM2), which causes cell death and worsens calcium excess (Tymianski, 2011). THSWD alleviated I/R injury by inhibiting the Wnt/IP3R/CAMKII pathway, reduced TNF-α and IL-1β secretion, and reduced calcium overload.
Anti-oxidative Stress
Oxidative stress results from a discrepancy between the amount of free radicals cells create and their capacity to combat them (Radak et al., 2014). When free radicals like reactive oxygen species and nitrogen (ROS/RNS) develop, surpass the antioxidant defense system’s endogenous scavenging capacity, oxidative and nitrative stress is caused (Khoshnam et al., 2017), which is one of the mechanisms leading to neuronal damage (Radak et al., 2014). Excessive ROS can result in the cellular macromolecules involved in signaling systems being destroyed, leading to apoptotic cell death (Sugawara & Chan, 2003). Oxidative stress produces lipid peroxides, such as malondialdehyde (MDA). Oxidative and nitrification stress act together in the pathophysiological process of cerebral ischemia (Turko & Murad, 2002); ROS and RNS molecules are mediators of acute ischemic stroke injury, causing cranial nerve death.
Transcription factor Nrf2 can regulate a series of anti-oxidation and detoxification phase II proteases. It can scavenge ROS/RNS, expel cytotoxicity, and is the main defense mechanism against oxidative stress (Zhang et al., 2010). HO-1 is a ubiquitous protein in the body that can scavenge free radicals. The expression levels of Nrf2 and HO-1 increase after cerebral artery occlusion in rats (Ishii et al., 2000).
THSWD has the effect of reducing oxidative stress after cerebral ischemia, thereby reducing neuronal damage. Li et al. discovered that in contrast to the control group, rats in the THSWD group had decreased infarct volume and improved neurological deficits. Also, THSWD can activate the Nrf2 and PI3K/Akt signaling pathway, and at the same time significantly induce the expression of HO-1 which is controlled by Nrf2 (Li et al., 2015).
Anti-cell Death
Anti-cell Necrosis
THSWD could inhibit brain cell necrosis and decrease brain damage by blocking the TNF-/RIP1/RIP3/MLKL pathway in rats (Wang, Fei, et al., 2021). MCAO rats treated with THSWD showed decreased levels of TNF-α, RIP1, RIP3, and MLKL mRNA expression compared to the control group (Wang, Fei, et al., 2021).
Anti-apoptosis
Under conditions of cerebral ischemia, apoptosis is activated, and TNF-α, Fas (CD95/APO1), and TRAIL (TNF-related apoptosis-inducing ligand) receptors as examples of cell surface death receptors, are triggered. Extrinsic stimuli activate cell surface death receptors, whereas intrinsic apoptotic stimuli are triggered via mitochondrial signaling pathways (Culmsee et al., 2005; Kroemer et al., 2007). The anti-apoptotic protein Bcl-2 is truncated to Bid (Nikoletopoulou et al., 2013), which opens the mitochondrial transition pore and promotes mitochondrial cytochrome c or apoptosis-inducing factor release (Culmsee et al., 2005). The Bcl-2 protein family is not only involved in apoptosis but is also involved in cell necrosis (Nikoletopoulou et al., 2013). In cerebral ischemia injury, the expression of Bcl-2 decreased, while transgenic rats with high expression of Bcl-2 could protect neurons.
Many studies have shown that THSWD has the effect of alleviating cell apoptosis. Hanlan et al. used Hoechst staining to observe that oxygen-glucose deprivation (OGD)-injured PC12 cells treated with serum containing THSWD relieved nuclear pyknosis and reduced cell apoptosis (Han et al., 2016). THSWD upregulated the B-cell lymphoma-2 (Bcl-2)/Bcl-2-related X protein (Bax) ratio to raise the amount of Bcl-2 and decrease apoptosis by decreasing the production of TNF, IL-1, and other inflammatory factors in the cortex (Han et al., 2015). Wan et al. found that Bcl-2 expression was increased by THSWD, but TNF– expression was decreased, and other proteins in the injured hippocampus and cortex of rats (Wan & Li, 2021). Cerebral ischemia activates caspase-3, which eventually leads to cell apoptosis, and p53 plays a crucial part in cell apoptosis. THSWD specifically inhibited pro-apoptotic genes, such as p53 and caspase-3, reduced the number of apoptotic neurons, and thus treated ischemic cerebrovascular diseases (Fan et al., 2015). By blocking HIF-1 and TNF-α, downregulating active caspase-3, and inhibiting apoptosis, THSWD may lessen the infarct volume in ischemia-reperfusion brain damage, downregulating active caspase-3, and inhibiting apoptosis (Wu et al., 2011).
Anti-pyroptosis
THSWD may downregulate GSDMD and inhibit pyroptosis (Wang, 2020). THSWD also reduces the levels of inflammatory elements, such as TNF and IL-2, relieves proteins, such as NLRP3 and caspase-1, in the penumbra, downregulates GSDMD, and inhibits pyroptosis (Wang et al., 2020). Zhou et al. showed that pyroptosis occurs at the site of ischemic injury after acute cerebral infarction. Rats treatment with THSWD reduced the levels of caspase-1, downregulated GSDMD and inhibited pyroptosis, thereby improving cerebral ischemia regeneration (Zhou, 2021).
Anti-autophagy
A particular type of autophagy called mitophagy cleaves damaged or unhealthy mitochondria. Shi et al. discovered that Parkin and PINK-1, as well as the autophagy marker proteins LC3-II/LC3-I, Beclin1, and Atg5, were all upregulated by THSWD and reduced damage to the mitochondrial structure. The agent Mdivi-1 inhibited the above-mentioned beneficial effects of THSWD, suggesting that THSWD can reduce cerebral infarction and neurological deficits by enhancing mitochondrial phagocytosis (Shi et al., 2023).
Improve Angiogenesis
Angiogenesis increases the oxygen and nutrient supply to ischemic brain tissue, promotes neurogenesis and synaptogenesis, and thus promotes neurological recovery (Shen et al., 2014). Vascular endothelial cells secrete various biologically active substances, such as vascular endothelial growth factor (VEGF), endothelin (ET), NO, and endothelium-derived relaxing factor (Li, 2016). Under hypoxic conditions, HIF-1α plays a crucial role in controlling tissue neovascularization through pro-VEGF gene transcription (Zhu et al., 2016). THSWD has an angiogenic effect on ischemic-reperfusion harm to the brain. In cerebral ischemia model rats, by stimulating the PI3K/AKT pathway, THSWD sped up microangiogenesis and enhanced the production of MVD and VEGF in cerebral ischemia-reperfusion cortical regions (Zhang, 2017). THSWD promotes PMPS to carry angiogenesis-related signal molecules, activates the Notch signaling pathway, and has a good angiogenesis effect on I/R injury (Chen et al., 2020). The platelet exosomes produced by THSWD intervening in MCAO model rats can increase the cerebral blood flow on the ischemic side of the rats, increase the expression levels of BrdU and vWF, and promote the proliferation of newborn cells (Wu et al.). Microvascular density in the hippocampus of rats increased after THSWD treatment, and decreased ET-1 levels and induced hippocampal VEGF activity, showed that THSWD could induce cerebral angiogenesis (Han et al., 2015). THSWD can promote the expression of VEGF and HIF-1 while defending brain microvascular endothelial cells (Ji et al., 2018).
Protect Nerve Cells
THSWD has the effect of protecting nerve cells after ischemia and hypoxia. Han et al. (2016) established the OGD cell model and found that the SOD activity increased in the OGD cell model treated with serum containing THSWD, while the levels of MDA and LDH decreased reduced, and the morphological damage of PC12 cells was alleviated. Wu et al. found that THSWD upregulates BDNF expression, activates the BDNF-TrkB-ERK1/2 signaling pathway, regulates neuronal survival, and restores cerebral ischemic injury. Experiments also show that upregulating the expression of BDNF affects synapses, suggesting that haptophysin and the synapse-associated protein, PSD-9,5 are involved in axonal growth and synaptic plasticity (Wu, 2018).
Conclusion
THSWD is a well-known prescription of TCM, THSWD has the functions of improving the overall state of ischemic stroke patients, relieving patients’ pain, reducing complications, and shortening recovery time. Studies have shown that THSWD can reduce the harm of ischemic stroke to human health by improving hemorheology, anti-inflammatory response, reduced excitotoxicity, anti-calcium overload, anti-oxidative stress, etc. It provides a research direction for the academic community and also brings hope for the rehabilitation of patients with ischemic stroke. However, the compatibility of prescriptions or the principle of increasing or decreasing the dose of TCM is still unclear. Proteomics, metabolomics, and other systems biology methods are used too rarely, resulting in insufficient research on the comprehensive screening mechanisms and targets of THSWD. The pathogenesis of ischemic stroke is complex. Studies have been done on how THSWD impacts the factors that lead to ischemic stroke, such as inflammatory response, calcium overload, cell apoptosis, etc., but only a few pathways have been studied for each physiological process. The study of a certain pathway is not comprehensive enough and needs to be further explored by designing experiments. To completely clarify the clinical effectiveness and safety of THSWD in treating ischemic strokes, clinical researchers should conduct multi-center, large-sample prospective cohort studies. These studies would also offer an adequate and trustworthy practical foundation for its clinical use. In summary, THSWD has shown great promise in the prevention and treatment of ischemic stroke, but the clinical value and medication rules, efficacy, and mechanism of THSWD to treat ischemic stroke still need to be comprehensively, systematically, and thoroughly researched.
Abbreviations
AIM2: absent in melanoma 2; Akt: protein kinase B; APPT: activated partial thromboplastin time; Bax: Bcl-2-associated X protein; BBB: blood-brain barrier; Bcl-2: B lymphocyte tumor-2; Bcl-2/Bax: B-cell lymphoma-2/Bcl-2-related X protein ratio; CAMKII: Calmodulin-dependent protein kinase-Ⅱ; Caspase-3: cysteine aspartate protease-3; ET: endothelin; FDA: food and drug administration; FIB: fibrinogen. Glu: Glutamate; HIF-α: hypoxia inducible factor-1; HIF-1α: hypoxia inducible factor-1α; HMGB1: high mobility group box 1; IBA-1: ionized calcium binding adaptor molecule-1; IL-1β: interleukin-1β; IL-18: interleukin-18; IP3R: inositol 1,4,5-triphosphate receptor; Lc3I: MAP1LC3-Ⅰ; Lc3I: MAP1LC3-Ⅱ; LC/Q-TOF-MS: liquid chromatography and quadrupole time-of-flight mass spectrometry; LDH: lactate dehydrogenase; MAPK: mitogen-activated protein kinase; MCAO: middle cerebral artery occlusion; MCP-1: monocyte chemoattractant protein-1; MDA: malondialdehyde; MLKL: mixed lineage kinase domain-like protein; MPO: myeloperoxidase; NF-κB: nuclear factor-κβ; NIHSS: national institutes of health stroke scale; NLRP3: NOD-like receptor protein 3 inflammasome; NMDAR: glutamate receptor; NO: nitric oxide; NOS: nitric oxide synthase; OGD: oxygen-glucose deprivation; p53: cellular tumor antigen p53; PI3K: phosphatidylinositol 3-kinase; Pink1: PTEN induced putative kinase 1; PLA2: phospholipaseA2; PSD95-NR2B-NOS: ternary structure; PT: prothrombin time; RIP1: receptor interacting protein-1; RIP3: receptor interacting protein-3; RNS: radical reactive nitrogen species; ROS: reactive oxygen species; ROS/RNS: reactive oxygen species/nitrogen; rt-PA: recombinant tissue plasminogen activator; SOD: superoxide dismutase; TCM: traditional Chinese medicine; THSWD: Taohong Siwu Decoction; TLR4: toll-like Receptor 4; TNF-α: tumor necrosis factor-α; TRAIL: TNF-related apoptosis-inducing ligand; TRPM2: transient receptor potential melastatin subfamily member 2; TRPM7: transient receptor potential melastatin subfamily member 7; TT: thrombin time; UPLC/MS: ultra performance liquid chromatography-mass spectrometry; VEGF: vascular endothelial growth factor.
Footnotes
Availability of Data and Materials
All the data used to support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
This work was supported by the Natural Science Foundation of Shandong Province (ZR2020KH003), the Innovation Team Project of Colleges and Universities in Jinan (2020GXRC012), and College Student Innovation and Entrepreneurship Training Program of Shandong Province (2022083).
