Abstract
Background
The herbal Poria cocos-Liquorice drug pair (PLDP) possesses the ability to be a diuretic, stimulating the spleen and benefiting the kidney, which plays an important role in the treatment of Henoch–Schönlein purpura nephritis (HSPN). However, the mechanism of action is unknown.
Objectives
Through the method of network pharmacology, this research sought to determine the mechanism of PLDP against HSPN.
Materials and Methods
The screening of active ingredients in PLDP was conducted by Traditional Chinese Medicine Systems Pharmacology (TCMSP) databases, while their targets were obtained from the TCMSP and Swiss Target Prediction (STP) databases. The genes of HSPN were searched by OMIM, DisGeNET, and GeneCards databases. Then, the common targets of active ingredients in PLDP and HSPN were mapped by Venn analysis. To get the main targets, the researchers utilized the STRING database to construct the protein–protein interaction (PPI) network of the common targets. Then, the function of gene ontology (GO) and the enrichment of the Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway of the main targets were examined on the Metascape database to identify the molecular mechanism of PLDP against HSPN. The most relevant signaling pathways with HSPN screened from the top 20 pathways were defined as the key pathways. Finally, the “active ingredient-main target-key pathway” network was built in order to identify the core targets and key active ingredients of PLDP against HSPN.
Results
There were 101 active ingredients and 360 targets for PLDP. One hundred and fifty-seven genes for HSPN and 35 common targets between PLDP and HSPN were identified. Through the “active ingredient-main target-key pathway” network, quercetin, kaempferol, polyporenic acid C, dehydrotumulosic acid, poricoic acid A, and naringenin were identified as key active ingredients; TNF, NOS3, RELA, AKT1, ICAM1, and IFNG were identified as core targets; and the key pathways include TNF signaling pathways, HIF-1 signaling pathways, and IL-17 signaling pathways.
Conclusion
The research initially investigated the pathways, active ingredients, and targets involved in PLDP against HSPN. The mechanism appears to be linked to its immunomodulatory and anti-inflammatory properties, thus establishing a scientific foundation for further investigation.
Keywords
Introduction
Henoch–Schönlein purpura nephritis (HSPN) is a systemic disorder characterized by necrotizing small vasculitis, which is the most prevalent pediatric secondary glomerular condition (Liu et al., 2021). About 30%–50% of children and 49%–83% of adults will progress from HSP to HSPN (Ding et al., 2021). In general, HSPN patients have an excellent prognosis, while 10%–20% of them will develop renal failure (Ding et al., 2019). The common clinical symptoms of HSPN include purpura of the skin, proteinuria, hematuria, and impairment of the kidney (Delbet et al., 2017; Li et al., 2019).
KDIGO guidelines recommend glucocorticoids and cyclophosphamide for individuals on HSPN who have nephrotic-range proteinuria (KDIGO, 2012). However, the quality of the evidence is low (grade 2D of recommendation), which is largely extrapolated from immunoglobulin (Ig)A nephropathy in adults rather than HSPN in children (Ding et al., 2019). It is worth noting that glucocorticoid drugs have obvious negative effects, including hypertension, hyperlipidemia, hyperglycemia, and abnormal osteoporosis. Cyclophosphamide can also cause liver toxicity and bone marrow toxicity (Ferrara et al., 2019; Ponticelli & Glassock, 2019). In this case, traditional Chinese medicine (TCM) is considered a complementary and alternative medication regimen for many kidney diseases. Many shreds of evidence show that TCM is beneficial for treating HSPN, such as Poria cocos, Liquorice, Chinese yam, Herba cirsii, Tripterygium glycoside, Tanshinone IIA, Herba Pteridis Multifidae, Herba Achyranthis Asperae and so on (Ding et al., 2019; Fan et al., 2020; Long, 2019; Liu et al., 2021).
The herbal pair of Poria cocos-Liquorice drug pair (PLDP) has the effects of dampness diuretic, invigorating the spleen and benefiting the kidney (Wang et al., 2021), which has a significant impact on immunological regulation and anti-inflammatory properties (Dong et al., 2021; Xie et al., 2015). There are many prescriptions for the combination of Poria cocos and licorice in ancient books, such as the Lingguishugan decoction and Lingguiganzao decoction records in “Shang Han Za Bing Lun”; Si Jun Zi Tang and Shenlingbaizhu records in “Tai Ping Hui Min He Ji Ju Fang”. The compatibility of Poria cocos and licorice is often used to treat kidney disease, respiratory illness, and cardiovascular disease. In the meantime, Zhang et al. explored that Poria cocos and Licorice were the most frequently used Chinese herbal compounds for HSPN therapy based on data mining technology (Zhang et al., 2019). Xu et al. found that Poria cocos and Licorice were the top five most frequently used drugs in the prescriptions for the treatment of HSPN through analysis of the kinds of literature collected by the China National Knowledge Infrastructure (CNKI) and Wan Fang Database from January 1993 to December 2019 (Xu & Liu, 2020). The clinical experiment confirmed that the prescription containing Poria cocos and Licorice can significantly decrease hematuria and proteinuria in HSPN patients (Long, 2019). Unfortunately, the specific mechanism of PLDP therapy for HSPN is not clarified. Network pharmacology, an emerging research concept and method, integrates multidisciplinary technologies and contents to build a drug–ingredient–target–disease network from an overall perspective and explore the association between diseases and drugs (Jin et al., 2020; Li & Zhang, 2013). Therefore, this study takes PLDP as the research object and constructs a “key active ingredient-main target-key pathway” network to investigate the mechanism of action on HSPN therapy and provide new ideas for clinical application (Figure 1).
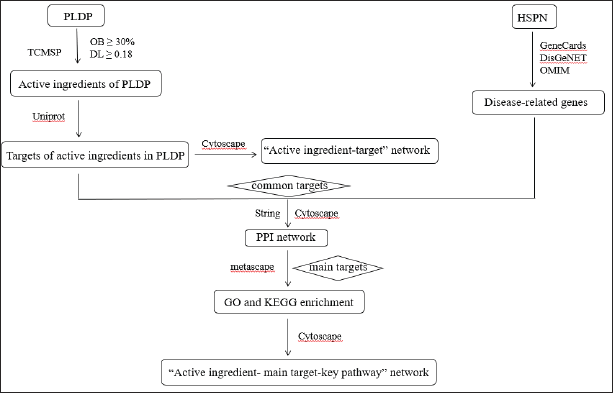
Materials and Methods
Examination of Chemical Ingredients and Active Ingredients of PLDP
The TCMSP database was searched using the keywords “Poria cocos” and “Licorice” to gather the chemical ingredients of PLDP.
Active ingredients of PLDP were selected from the chemical ingredients that conformed to both drug-likeness (DL) ≥0.18 and oral bioavailability (OB) ≥30%. The resemblance between a compound and a recognized medication is referred to as DL. OB represents the amounts of oral drugs digested and absorbed by the gastrointestinal tract, passed through the liver and finally reached the systemic circulation ratio of the oral dose (Jin et al., 2020). TCMSP is a unique herbal medicine system pharmacological platform where we can get the relationship between targets, drugs, and diseases.
Forecasting the Potential Targets of Active Ingredients in PLDP
The TCMSP and STP databases were searched to forecast the targets of active ingredients in PLDP. Calibrate the acquired targets using the UniProt database with “Homo sapiens”. The Swiss Target Prediction database predicts the target of a compound based on its similarity to the configurations of identified compounds. The UniProt (Universal Protein) database is a protein database containing protein sequences, functional information, and research paper indexes.
Development of the Network
The correlation between active ingredients and their respective targets of PLDP was constructed using Microsoft Excel. Then, the “active ingredient-target” network of PLDP was visualized by Cytoscape 3.7.2. Cytoscape is capable of combining basic data into a visual network.
Searching for HSPN-related Genes
GeneCard, DisGeNET, and the OMIM databases were selected to retrieve HSPN-related genes. As a keyword, “Henoch–Schönlein purpura nephritis” was used in all databases. The genes were preserved after integration and deduplication. GeneCards, DisGeNET, and OMIM have gathered a vast array of genes linked to human illnesses.
Venn Analysis of Active Ingredient Targets and Disease Genes
Venn analysis was performed on the targets in PLDP and the genes in HSPN to visualize the common targets. Venn analysis is a graph that can visually show the intersection of multiple things.
Construction of the PPI
Based on the “Homo sapiens” species, the common targets were transferred to the STRING database with a threshold of confidence score ≥0.4 to generate a visual map. After downloading the obtained interactive files in TSV format, Cytoscape was utilized for visualizing the PPI in order to obtain the main targets within the network. The STRING database is capable of studying the interaction network between proteins.
Enrichment Analysis of Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG)
GO and KEGG were carried out utilizing the Metascape database with the predicted main targets. The bioinformatics website was utilized to display the findings. The most relevant signaling pathways with HSPN screened from the top 20 pathways were defined as the key pathways, which were obtained from PubMed with the keywords “(the top 20 pathways [All Fields]) AND (Henoch–Schönlein purpura nephritis) OR (renal damage) OR (nephritis) OR (kidney) [All Fields]. The Metascape database incorporates a variety of authoritative data resources, such as GO and KEGG, to facilitate relevant analyses.
Construction of the “Active Ingredient-Main Target-Key Pathway” Network
It was determined which main targets comprise the key pathways in HSPN and which active ingredients act on these main targets. Then, an “active ingredient-main target-key pathway” network is constructed. Based on the numerical values of the degrees, the top six main targets and active ingredients were defined as the core targets and key active ingredients, respectively.
Results
Chemical Ingredients and Active Ingredients of PLDP
Three hundred and fourteen chemical ingredients (34 chemical ingredients of Poria cocos and 280 chemical ingredients of Licorice) were obtained. Finally, there were 101 active ingredients (13 active ingredients of Poria cocos and 88 active ingredients of licorice) of PLDP were obtained.
Targets Corresponding to Active Ingredients in PLDP
Four hundred and seven targets (including 191 targets of Poria cocos and 216 targets of Licorice corresponding to all 101 active ingredients) were obtained. In the end, 360 corresponding targets of PLDP can be obtained after deleting 47 duplicate targets.
Development of Network
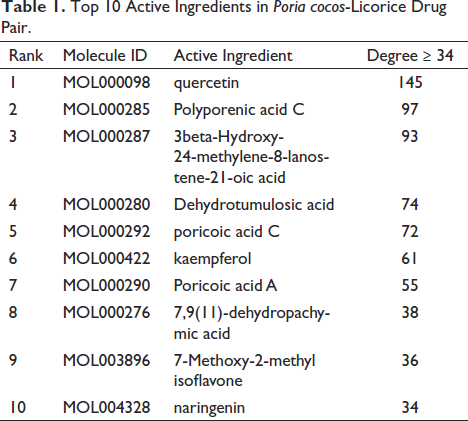
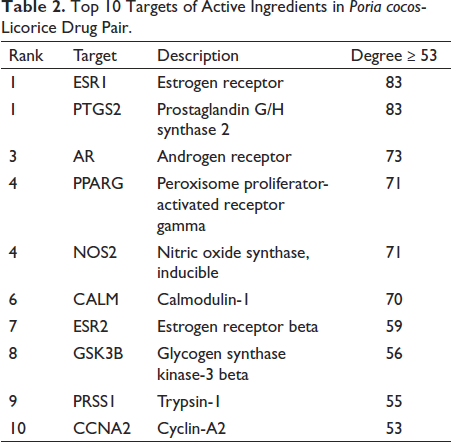
The “active ingredient-target” network of PLDP including 101 active ingredients and 360 targets of PLDP is illustrated in Figure 2. A total of 116 nodes are greater than the average degree value (8.994). Tables 1 and 2 show a screening result based on the degree value for the top 10 active ingredients (degree ≥34) and targets (degree ≥53).
Active Ingredient-Target Interaction Network. Poria cocos and Licorice Were Represented With Pink Circles. The Active Ingredients of Poria cocos and Licorice Were Represented With Orange and Purple Hexagons, Respectively. The Predicted Targets Were Represented With Blue Diamonds. As the Node Grows Bigger and the Color of the Transparency Darkens, the More Valuable the Node is.

Top 10 Active Ingredients in Poria cocos-Licorice Drug Pair.
Top 10 Targets of Active Ingredients in Poria cocos-Licorice Drug Pair.
Searching for HSPN Genes
A total of 63, 29, and 84 genes were searched and retrieved from three disease databases (OMIM, DisGeNET, and GeneCards databases) using the keyword “Henoch–Schönlein purpura nephritis”. Finally, collation and duplication removal yielded 157 HSPN-related genes.
Venn Analysis of Targets Between Active Ingredients in PLDP and Disease
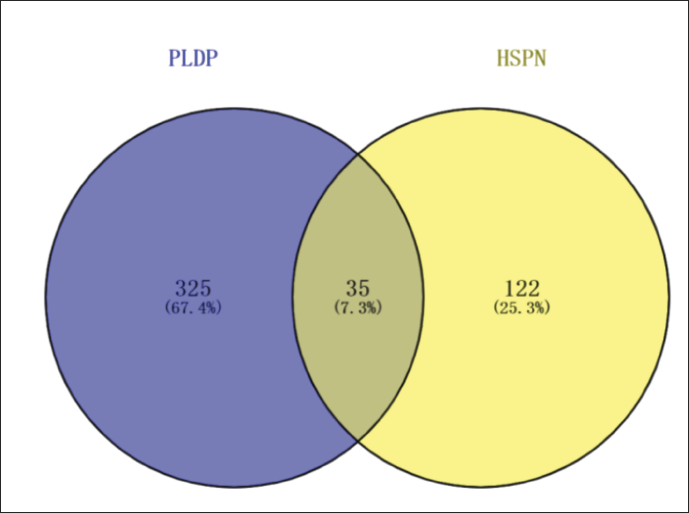
As shown in Figure 3, Venn analysis was conducted of 360 active ingredient targets of PLDP and 157 disease genes in HSPN, 35 common targets were obtained.
PPI Network
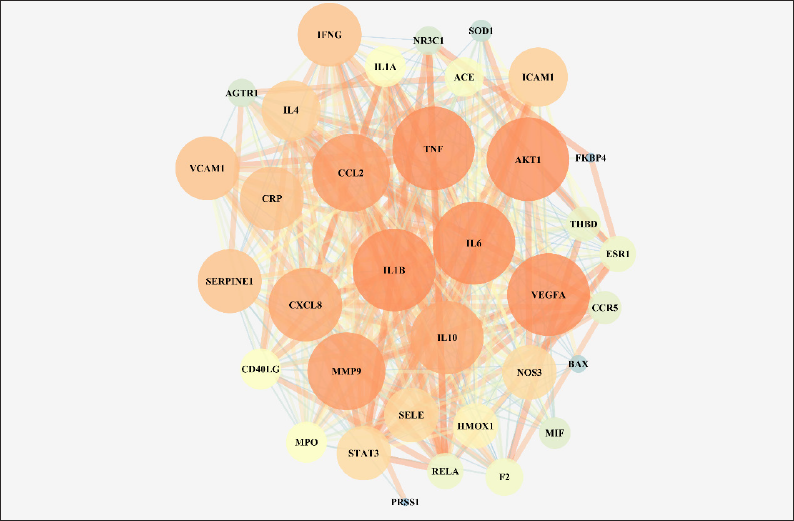
The PPI network underwent enrichment analysis for 35 main targets, which consisted of 405 edges and 35 nodes (Figure 4).
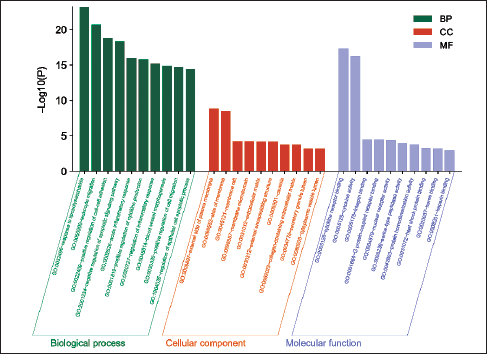
GO and KEGG Analysis
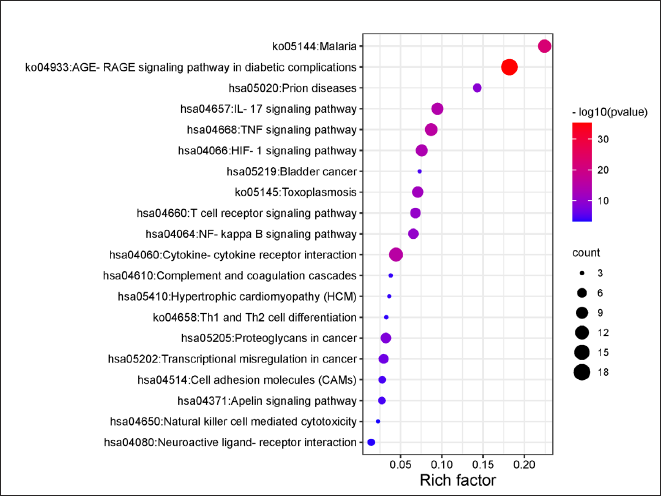
Thirty-five main targets were analyzed for GO and KEGG enrichment. The GO result is shown in Figure 5. A bubble map-visualized analysis of the top 20 pathways is illustrated in Figure 6. The key pathways associated with HSPN include the tumor necrosis factor (TNF) signaling pathways, nuclear factor-kappa B (NF-kappa B) signaling pathways, interleukin-17 (IL-17) signaling pathways, T cell receptor signaling pathways, HIF-1 signaling pathways, natural killer cell-mediated cytotoxicity, cell adhesion molecules (CAMs), Th1 and Th2 cell differentiation, Cytokine–cytokine receptor interaction and the AGE-RAGE signaling pathway in diabetic complications. Accordingly, PLDP appears to regulate these 10 pathways during the management of HSPN.
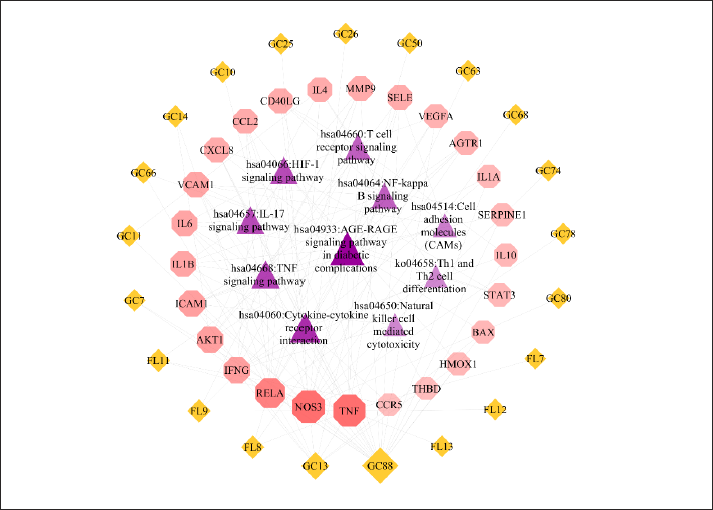
Construction of “Active Ingredient-Main Target-Key Pathway” Network
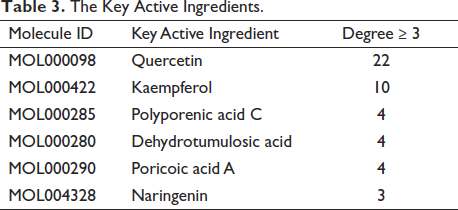
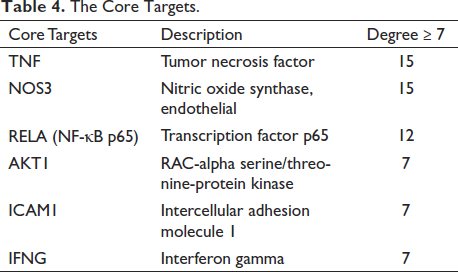
Depending on the key pathways, the network was constructed (Figure 7). According to the degree values, quercetin, kaempferol, polyporenic acid C, dehydrotumulosic acid, poricoic acid A and naringenin were the key active ingredients of PLDP therapy for HSPN (Table 3). TNF, NOS3, RELA (NF-κB p65), AKT1, ICAM1, and IFNG were the core targets of PLDP therapy for HSPN (Table 4).
The Key Active Ingredients.
The Core Targets.
Discussion
HSPN can cause autoimmune reactions, and the recurrent accumulation of immune complexes containing IgA in the glomerulus can cause mesangial hyperplasia and renal damage, resulting in non-specific inflammatory reactions (Pohl, 2015). Its pathogenesis includes disturbance of coagulation, chemotaxis of proinflammatory cytokines, such as IL-17, vascular endothelial growth factor (VEGF), and TNF-α, and disorders of humoral immunity dominated by IgA and cellular immunity dominated by T lymphocytes (Long, 2019; Liu et al., 2021). Clinical treatment of HSPN includes inhibition of mesangial hyperplasia, control of the inflammatory response, prevention of renal fibrosis, and elimination of proteinuria and hematuria (Xu et al., 2018; Zhang et al., 2018). Modern pharmacological investigations have confirmed that Poria cocos can inhibit NOS expression and T cell proliferation, reduce creatinine and urea levels, attenuate renal fibrosis, and play biological functions consisting of anti-inflammation, immunomodulation, and kidney protection (Dong et al., 2021; Nie et al., 2020). Licorice has the effects of antioxidation, anti-inflammatory, and immunomodulation; it delays the pathological process of HSPN by downregulating inflammatory factors such as TNF-α (Xie et al., 2015; Yang et al., 2017).
Six active ingredients of PLDP, namely quercetin, kaempferol, and naringenin (contained in Licorice); polyporenic acid C; dehydrotumulosic acid; and poricoic acid A (contained in Poria cocos), were key active ingredients of PLDP against HSPN. Quercetin, one of the most commonly taken flavonoids in the diet, can decrease the expression of HIF-1 and VEGF to protect the kidney from hypoxic injury and vascular endothelial injury (Alshanwani et al., 2020). Vascular endothelial injury is the pathological basis of HSPN, which increases vascular permeability, leads to increased blood viscosity and slow blood flow, and aggravates renal ischemia and hypoxia (Yuan et al., 2012; Li & Cao, 2020). Meanwhile, quercetin can mitigate HSPN by reducing the level of ICAM-1 and keeping inflammatory cells from infiltrating the kidney (Tong et al., 2017).
The key active ingredient, kaempferol, alleviates the inflammatory response of HSPN because of its ability to reduce oxidative stress and inflammation (Ashrafizadeh et al., 2020; Calderón-Montaño et al., 2011). Moreover, kaempferol treats inflammatory diseases by reducing IL-1 and TNF-α, while inhibiting the transfer of NF-κB p65 mediated by TNF-α to the nucleus (Ren et al., 2019). Peng et al. found that kaempferol could attenuate the inflammatory response and renal injury in HSPN because of its inhibitory effect on renal androgen receptor expression and inhibition of renal inflammation and oxidative stress (Yuan et al., 2021).
It has been indicated that key active ingredients, polyporenic acid C, dehydrotumulosic acid, and poricoic acid A, effectively attenuated renal fibrosis by blocking the renin-angiotensin system (RAS) (Wang, 2019). Polyporenic acid C and dehydroturmoic acid can significantly promote natural killer cell activity and IFNG secretion to enhance non-specific immunity, exerting beneficial effects in immunomodulatory activity (Chao et al., 2021). Cai et al. found that polyporenic acid C inhibited iNOS and reduced the vascular active marker NO to achieve an anti-inflammatory effect (Cai & Cai, 2011). Meanwhile, poricoic acid A could inhibit inflammation and oxidative stress through the IκB/ NF-κB signaling pathway, significantly decrease urea and creatinine levels, and improve podocyte injury and renal fibrosis (Nie et al., 2020). Key active ingredients, such as naringin, can effectively reduce the expression of signal factors related to the inflammatory response, such as iNOS, and TNF, playing an anti-inflammatory role (Chen et al., 2016). It has been confirmed that naringin suppresses the expression of inflammatory factors through the TGF-β/Smads pathway and plays an anti-renal fibrosis role (Wang R. et al., 2021). Importantly, inhibition of TGF-β overexpression is the focus to prevent and treat HSPN (Zhang et al., 2020).
As part of the innate immune response, the NF-κB signaling pathway is of significant importance in signal transmission (Wang et al., 2012). RELA (NF-κB p65) a master regulator of inflammation and innate immunity can regulate the induction and elimination of inflammation in the context of renal damage (Sanz et al., 2010). Other cells in the kidney, such as tubular epithelial and mesangial cells, could promote inflammation via activation of NF-κB signaling pathways (Song et al., 2019). The serum levels of NF-κB in children with HSPN are abnormally high, and the expression level increases with the patient’s condition (Tan et al., 2021). Both two key active ingredients quercetin and kaempferol, could suppress the progression of inflammatory diseases by inhibiting the NF-κB signaling pathway (Jantan et al., 2021).
Aside from anti-inflammatory, immunomodulatory is also the key to the treatment of HSPN. It has also been identified that pediatric patients diagnosed with HSPN experience disruptions in their humoral immune response and dysfunction of T cell subsets, consisting of Th17, Th1, and Th2 cells (Ordonez et al., 2009; Shuiai et al., 2017). Abnormal T cell receptor signaling (key pathway) is associated with immune cell dysfunction and autoimmunity, which causes autoantibody deposition and inflammatory cell infiltration in the kidney, leading to complications of HSPN (Moulton & Tsokos, 2015). The natural killer cell has played an important role in immune-mediated glomerulonephritis (Hu et al., 2017; Uchida et al., 2019). Activated natural killer cells are not only involved in the development of HSPN but also have the potential to serve as innovative biomarkers for HSPN (Imai et al., 2020). Interferon-γ (IFNG), a cytokine released by natural killer cells, is involved in purpura nephritis and renal fibrosis (Law et al., 2017). According to the report, the T-cell subset Th1/Th2 balance in HSPN patients with proteinuria may be transformed into Th1 dominance, and the imbalance of Th1 and Th2 cell differentiation may influence the development and progression of nephritis (Tsuruga et al., 2011). T cell subsets were infiltrated by regulating CAMs (Yoshizaki et al., 2010).
Both anti-inflammatory and immunomodulatory targets and pathways play a part in HSPN therapy. The role of CAMs, such as ICAM, has been studied to be responsible for recruiting leukocytes to inflammation sites (Kobayashi et al., 2007). ICAM is related to inflammatory diseases and immune function and is involved in a variety of vasculitis diseases, which is highly correlated with the pathogenesis of HSPN (Li & Cao, 2020). Its release is altered during HSPN crescent formation and accelerates inflammatory mediators release and the proliferation of mesangial cells (Cheng et al., 2020). TNF is the signature inflammatory factor of acute inflammation, which affects the immune environment and participates in renal injury caused by various factors (Li et al., 2017; Milanesi et al., 2019; Wang et al., 2018; Xiao et al., 2020). TNF-α is an important type of TNF, which is mainly secreted by mononuclear macrophages.
One research suggested a link between serum TNF-α and HSPN, and the seriousness of the illness depended on the level of TNF-α in the serum (Yuan et al., 2018). It contributes to the pathogenesis of HSPN by regulating immune response and inducing inflammation through the TNF signaling pathway (Tang et al., 2021). IL-17 signaling pathway is linked to autoimmune diseases and is activated in HSPN to promote inflammatory cytokines leading to renal tissue damage (Amatya et al., 2017; Wang G. et al., 2021). IL-17 overexpression in renal tissue contributes to renal pathological damage in HSPN children and may be involved in the pathogenesis and development of disease (Shao et al., 2014). Meanwhile, IL-17 produces a proinflammatory condition by stimulating endogenous immune cells and renal cells to synthesize inflammatory cytokines (Paquissi & Abensur, 2021). The receptor action of the IL-17 protein sets off a chain reaction of pathways linked to inflammatory responses (such as NF-κB signaling pathways), which are involved in HSPN immunity and inflammatory pathogenesis (Rayego-Mateos et al., 2020). Licorice exerts anti-inflammatory effects through cytokines for instance TNF-α, IFNG, and IL-17 (Richard, 2021).
In addition to being anti-inflammatory and immuno modulatory, meliorating ischemic hypoxia can also reduce renal damage in HSPN patients. HIF is the main regulator of the hypoxia response, composed of HIF-α and HIF-β. HIF-1α is a subunit of HIF-α, which is very important in the HIF signaling pathway (Corrado & Fontana, 2020). In HSPN patients, increased blood viscosity leads to renal ischemia and hypoxia, resulting in persistently elevated levels of HIF-1α expression in renal tissue. High levels of HIF-1α lead to increased glomerular filtration membrane permeability, increased plasma protein filtration, and even urinary protein, which further exacerbates the deterioration of HSPN (Hiden et al., 2018). Quercetin can downregulate the HIF-1α signaling pathway in the kidneys of rats subjected to hypoxic stress (Alshanwani et al., 2020).
Therefore, the key pathways, core targets, and key active ingredients significantly contribute to the management of HSPN by exerting anti-inflammatory, immunomodulatory, and meliorating effects on ischemic hypoxia.
Conclusion
In summary, this study screened multiple key active ingredients with potential application value from PLDP, which can realize the control and treatment of HSPN by inhibiting mesangial hyperplasia, controlling the inflammatory response, preventing renal fibrosis, and eliminating proteinuria and hematuria through multiple targets and pathways. The research results preliminarily revealed the pharmacological effects of PLDP on the management of HSPN, which laid a good foundation for further investigation of its potential mechanism and provided new ideas for HSPN treatment. Additionally, there are still some deficiencies present in the paper. This study was basically completed through multiple network data platforms, and the data lacked completeness, accuracy, and timeliness. Further validation through animal and clinical experiments is required to confirm the forecast of targets and pathways.
Footnotes
Declaration of Conflicting Interests
The authors declare no conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research received funding from the Hospital level project of Xi’an Children’s Hospital (No. 2021H09).
Statement of Informed Consent and Ethical Approval
Not applicable.
