Abstract
Background:
Anti-tumor necrosis factor (TNF) agents are increasingly used for rheumatic diseases and inflammatory bowel disease (IBD), but are associated with the development of anti-TNF-induced lupus (ATIL). Nonetheless, few ATIL studies on non-Caucasian IBD patients exist. Here, we investigated the incidence, clinical features, and risk factors of ATIL in Korea.
Methods:
We retrospectively reviewed the medical records of IBD patients undergoing anti-TNF therapy at our tertiary IBD center between 2008 and 2020. ATIL was diagnosed as a temporal association between symptoms and anti-TNF agents, and the presence of at least one serologic and non-serologic American College of Rheumatology criterion. The risk factors for ATIL occurrence were assessed using multivariate Cox regression analysis.
Results:
Of 1362 IBD patients treated with anti-TNF agents, 50 (3.7%) ATIL cases were suspected, of which 14 (1.0%) received a definitive diagnosis. Arthritis and mucocutaneous symptoms were observed in 13 and 4 patients, respectively. All ATIL cases were positive for anti-nuclear and anti-dsDNA antibodies. Four patients (30.8%) improved while continuing anti-TNF therapy. At the final follow up, the ATIL group (
Conclusion:
Although rare, ATIL is associated with poor treatment response to IBD in Korean patients. ATIL should be considered if arthritis and mucocutaneous symptoms develop during anti-TNF therapy for IBD.
Introduction
Inflammatory bowel disease (IBD), including Crohn’s disease (CD) and ulcerative colitis (UC), is a chronic inflammatory disorder of the gastrointestinal tract. IBD has a multifactorial pathogenesis, which has led to attempts to develop a variety of biological agents. Anti-tumor necrosis factor (TNF) agents target TNF-alpha, which is associated with the pathogenesis of various autoimmune diseases, and are effective and widely used for the treatment of IBD. 1 These agents are generally tolerated in patients, but there are concerns about adverse events such as infection and malignancy. 2 Further, autoimmune-mediated phenomena have been identified during anti-TNF treatments. 3
Anti-TNF agents induce the production of autoantibodies in some patients, including anti-nuclear antibody (ANA) and anti-dsDNA antibody (anti-dsDNA). 4 Some of these cases may develop anti-TNF-induced lupus (ATIL), which is a rare autoimmune disease characterized by clinical and immunological findings similar to systemic lupus erythematosus (SLE). The main clinical and serological features of ATIL include skin rash, arthralgia, serositis, fever and anti-dsDNA positivity, which are somewhat different from the characteristics of other drug-induced lupus.5,6 These features of ATIL have a temporal association with anti-TNF agent exposure, and usually resolve after discontinuation of the anti-TNF agents. The pathogenesis of ATIL remains unknown, but is possibly related to an imbalance between interferon-alpha and TNF-alpha, an increase in apoptotic particles, a cytokine shift to a Th2 response, and a predisposition to bacterial infection. 7 As the use of anti-TNF agents increases, a greater interest in ATIL is also emerging. Notably however, few studies to date have reported on the characteristics of ATIL in IBD patients. In our present study, we aimed to evaluate the incidence, risk factors, and clinical features of ATIL in a single IBD cohort treated with anti-TNF agents.
Methods
Study population
This study was conducted on adult IBD patients who received treatment with anti-TNF agents at a tertiary referral center in South Korea between March 2008 and February 2020. ATIL was diagnosed if the following criteria were met: (1) a temporal association was found between symptoms and anti-TNF agents; (2) at least one serologic American College of Rheumatology (ACR) criterion was noted; and (3) at least one non-serologic ACR criterion was satisfied. 8
IBD patients who were referred to our rheumatology clinic due to a suspicion of rheumatic disease, and for whom
This study was conducted in accordance with the Declaration of Helsinki principles and was approved by the Institutional Review Board of Asan Medical Center (IRB number: 2019-0700). Informed consent was waived because of the retrospective nature of the study.
Statistical analysis
Variables were represented as medians with an interquartile range (IQR) for continuous variables and as numbers (%) for categorical variables. The Mann–Whitney
Results
Incidence of ATIL and comparison of the clinical features of ATIL and non-ATIL cases
Among the total population of 1362 IBD patients (965 CD patients, 397 UC patients) treated with anti-TNF agents at our IBD center, 50 cases were referred to our hospital’s rheumatology clinic due to a suspicion of rheumatic disease. The main reasons for this referral were arthralgia in 23 (46%), mucocutaneous lesion in 7 (14%), fever in 4 (8%), cytopenia in 4 (8%), serositis in 2 (4%), proteinuria and/or hematuria in 2 (4%), abnormal liver function test in 1 (2%), cerebral venous thrombosis in 1 (2%), chest pain in 1 (2%), and others in 5 (10%) patients. ATIL was definitively diagnosed in 14 (1.0%) patients, comprising 10 CD and 4 UC cases.
The clinical characteristics of 50 patients are summarized in Table 1. The median age at the commencement of the anti-TNF therapy was 30 years (IQR 25–45); 39 (78%) patients had CD, and 11 (22%) patients had UC. The median IBD activity score at baseline was a CDAI score of 256.5 (IQR 224.4–284.6) in 35 CD patients and a partial Mayo score of six (IQR 5–7) in 11 UC patients. ANA and anti-dsDNA were both positive in 25 (50%) patients. The ATIL group cases (
Clinical characteristics of the inflammatory bowel disease patients treated with anti-TNF agents.

Data are expressed as a median value (interquartile range) or as a number of patients (%).
Duration of treatment prior to ATIL onset.
Four patients at baseline and five cases at the final follow-up date were excluded due to the presence of an ileostomy or colostomy.
5-ASA, 5-aminosalicylic acid; 6-MP, 6-mercaptopurine; ATIL, anti-TNF-induced lupus; AZA, azathioprine; CDAI, Crohn’s disease activity index; IBD, inflammatory bowel disease; TNF, tumor necrosis factor.
Clinical characteristics and outcomes of ATIL
The clinical characteristics and outcomes among the ATIL patients are detailed in Table 2. The median age at the diagnosis of ATIL was 38 years (IQR 35–47). The clinical features of ATIL arose after a median of 19 months (range, 9–64) of anti-TNF treatment. The most prevalent clinical feature was arthritis in 13 (92.9%) patients, followed by mucocutaneous lesions (i.e., malar rash, photosensitivity, and oral ulcer) in 4 (28.6%) patients and myalgia in 3 (21.4%) patients. Fatigue and cytopenia were found in two (14.3%) cases, and fever and serositis in one (7.1%) patient. One of the ATIL patients presented with acute kidney injury and hematuria after 6 years of anti-TNF treatment without concomitant medication, and tubulointerstitial disease was identified from a renal biopsy in this case. All of the ATIL patients had positive results for ANA (titers, 1:1280 in 10; 1:640 in two; and 1:320 and 1:160 in one patient each) and anti-dsDNA [titers showing a median of 54.4 IU/ml (IQR 21.0–82.4)]. Among the 12 patients who performed anti-ENA antibody tests, 2 also showed anti-Ro positivity. Rheumatoid factor was positive in 2 of the 11 ATIL patients who were assessed.
Clinical features and outcomes in the anti-TNF-induced lupus patients.
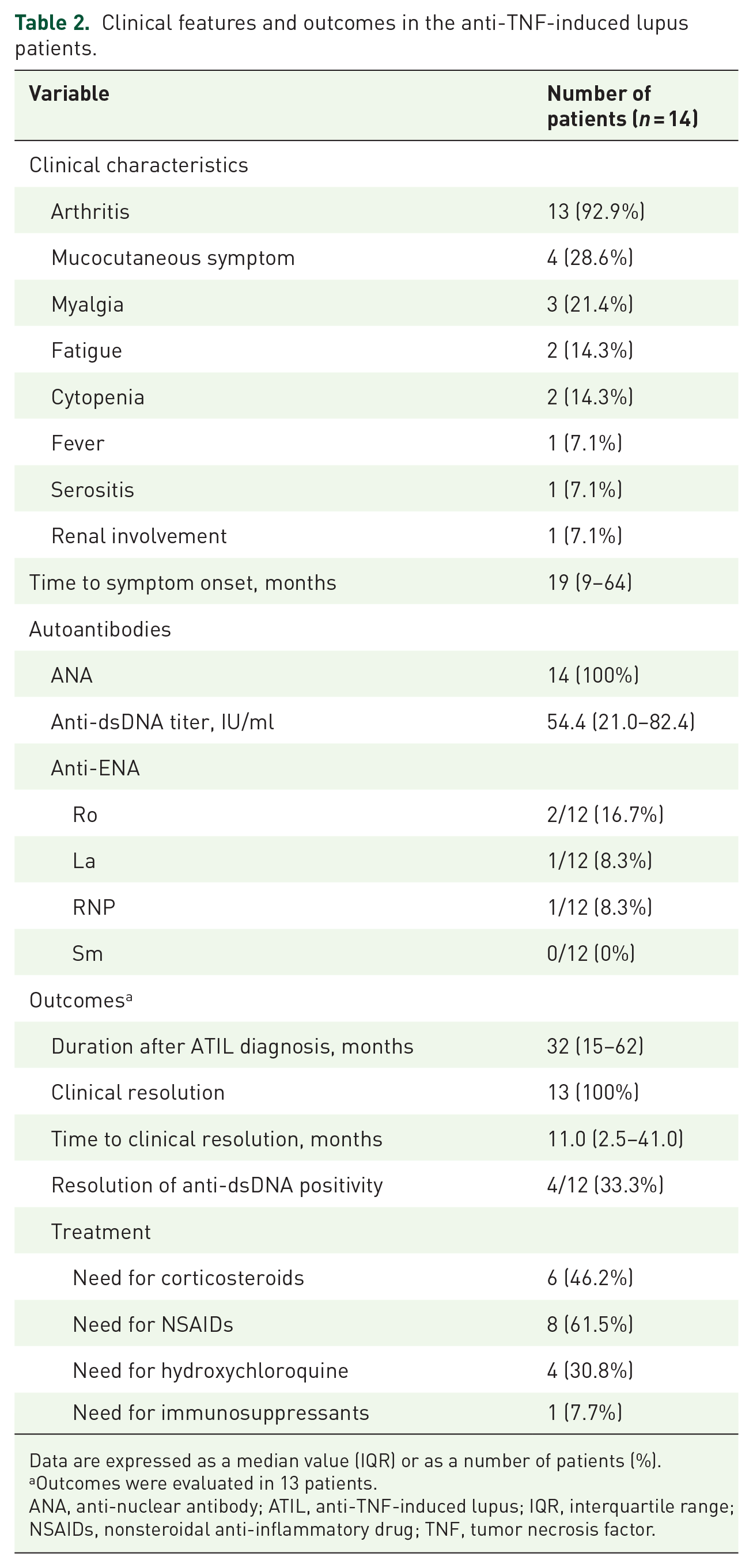
Data are expressed as a median value (IQR) or as a number of patients (%).
Outcomes were evaluated in 13 patients.
ANA, anti-nuclear antibody; ATIL, anti-TNF-induced lupus; IQR, interquartile range; NSAIDs, nonsteroidal anti-inflammatory drug; TNF, tumor necrosis factor.
A total of 13 ATIL patients were followed up for a median of 32 months (IQR 15–62) after their diagnosis. All 13 ATIL patients had a clinical resolution after a median of 11.0 months (IQR 2.5–41.0). Anti-dsDNA titers were normalized in 4 (33.3%) of the 12 patients who were positive for this antibody and performed follow-up tests. Six ATIL patients (46.2%) required corticosteroid treatments (a medium dose in two patients and low dose in four patients). Oral cyclophosphamide was administered to an ATIL patient with tubulointerstitial disease, and the hematuria and acute kidney injury improved within 6 months of this treatment.
During the follow-up period for ATIL, the anti-TNF therapy was discontinued at diagnosis in one patient with serositis and one further case with fever, myalgia, and arthritis, both of which achieved clinical resolution without relapse (Figure 1). Among 11 patients who were maintained on anti-TNF agents, five patients with arthritis or skin rash showed improved clinical features with low doses of corticosteroid, nonsteroidal anti-inflammatory drugs, or hydroxychloroquine. However, the remaining six patients had no improvement. These six cases experienced resolution after discontinuation of the anti-TNF agents (in two patients) or a switch to vedolizumab (in three patients). The remaining ATIL patient was switched to a second anti-TNF agent (from adalimumab to infliximab) but did not achieve improvement. A clinical resolution was eventually achieved in this case after the discontinuation of infliximab.

Clinical course of anti-TNF-induced lupus.
Risk factors associated with the occurrence of ATIL
We identified the risk factors for ATIL occurrence in our current cohort of IBD patients treated with anti-TNF agents (Table 3). Univariate Cox regression analysis revealed that the age at anti-TNF agent commencement (HR 1.055, 95% CI 1.005–1.108,
Risk factors for the occurrence of anti-TNF-induced lupus.

Data are expressed as a median value (interquartile range) or number of patients (%).
Variables with a
Four patients were excluded due to the presence of an ileostomy or colostomy.
5-ASA, 5-aminosalicylic acid; CD, Crohn’s disease; 6-MP, 6-mercaptopurine; AZA, azathioprine; CDAI, Crohn’s disease activity index; CI, confidence interval; HR, hazard ratio; IBD, inflammatory bowel disease; TNF, tumor necrosis factor; UC, ulcerative colitis.
Discussion
We found from our current analyses that ATIL is a rare clinical disease in IBD patients treated with anti-TNF agents, occurring more frequently in cases of UC and a long disease duration. IBD activity was also poorly controlled in patients with ATIL despite anti-TNF therapy. Newly developed arthritis and mucocutaneous lesions were found to be common symptoms of ATIL. However, we found also that the prognosis of ATIL was favorable, even in patients who were maintained on anti-TNF agents.
The induction of autoantibodies is common in patients receiving anti-TNF therapies. In the reported safety data for one of the commonly used anti-TNF agents, infliximab, ANA develops in 63.8% of rheumatoid arthritis (RA) patients and 49.1% of CD patients, and anti-dsDNA positivity develops in 13% of RA patients and 21.6% of CD patients, after treatment with this drug. 12 It must be noted, however, that the incidence of ATIL is low. Post-marketing studies on anti-TNF treatment have reported an ATIL occurrence rate of only 0.1–0.22%.13,14 Prior IBD studies have indicated incidence rates ranging from 0.6% to 10%.15–17 In our present study, ATIL was observed in 1% of the IBD patients at our hospital treated with anti-TNF agents, consistent with the findings of other studies. Notably in this regard, the incidence of ATIL is difficult to assess due to many confounding factors, including a lack of suspicion, various study designs, and difficulty in distinguishing ATIL from the extra-intestinal symptoms of IBD.
ATIL occurred in our current cases after 1–5 years of anti-TNF therapy, as also reported in previous studies.18,19 Most of our current cases developed arthritis, and mucocutaneous lesions and myalgia were also recorded frequently in this population. We also observed one ATIL patient with tubulointerstitial nephritis with acute kidney injury and hematuria. Picardo
With regard to the outcomes in our current ATIL cohort, all of the patients eventually had clinical resolution. However, the effects of maintenance, discontinuation, or switching of anti-TNF agents on the disease course of ATIL remains unclear. There have been several reports that ATIL improves after anti-TNF discontinuation.20–23 On the other hand, we found from our current investigation that four ATIL patients with arthritis and skin rash were maintained on anti-TNF agents without relapse. A study by Beigel
Our current ATIL group showed higher IBD activity during anti-TNF treatment compared with the non-ATIL cases in our series. This may be due to the association between anti-drug antibodies (ADAs) and ATIL development. ADAs are known to contribute to poor outcomes in IBD through an accelerated drug clearance.
24
Ishikawa
In our present analyses, UC and a long disease duration were found to be associated with ATIL occurrence. In previous reports, female gender, former smoker, old age, and no concomitant treatment were suggested risk factors for ATIL.16,19,21,23 The coexistence of SLE and IBD is rare. UC is reported in 0.4% of SLE patients, and the concurrence of SLE and CD is even rarer.
28
The contribution of UC compared with CD as a risk factor for ATIL is poorly understood, but the different mechanisms between UC and CD may provide insights in this regard. A prior report has indicated that a cytokine shift from Th1 to Th2 mechanisms caused by anti-TNF agents may contribute to the pathogenesis of ATIL.
29
CD is associated mainly with Th1 mechanisms, whilst UC produces Th2 immune responses.
30
On the other hand, Beigel
Our current study had some limitations of note, including those inherent to its retrospective design. There may have been a selection bias resulting from our analysis only of patients with suspected rheumatic diseases after anti-TNF exposure. Also, the incidence of ATIL may have been underestimated because the symptoms of ATIL may be assessed as extra-intestinal symptoms of IBD, and there may have been ATIL patients who have not visited a rheumatology clinic. Because the IBD activity score also includes symptoms similar to ATIL, the symptoms of ATIL may have contributed to the high IBD activity score in the ATIL group. An information bias may have also been present in the medical records that could have affected the results. Moreover, ANAs and anti-dsDNA antibodies were not measured prior to anti-TNF treatment and therapeutic drug monitoring and the results of ADA levels were not available. Nonetheless, our study findings have robustly demonstrated the characteristics and disease course aspects of ATIL in IBD patients from a single-center cohort. Based on our present observations, the likelihood of ATIL onset can be evaluated when there are suspected symptoms of lupus in patients undergoing anti-TNF treatment. It is notable that we used the diagnostic criteria for ATIL that are included the ACR criteria for SLE diagnosis. Our present data may help to predict the prognosis and develop future treatment strategies for ATIL in the IBD population.
In conclusion, ATIL has a low prevalence in anti-TNF treated IBD patients and UC and a long disease duration were associated with its development. The possibility of an ATIL onset should be considered if new lupus-related symptoms develop during anti-TNF treatments.
Footnotes
Author contribution
Study concept and design: SJC, SHP and YGK
Acquisition of data: SJC, SMA and SH
Analysis and interpretation of data: JSO, CKL, BY, BDY, SKY, SHP and YGK
Drafting of the manuscript: SJC, SHP and YGK
Critical revision of the manuscript for important intellectual content: all authors
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grant to Y-G Kim from the Asan Institute for Life Sciences (grant number: 2020IL0037).
