Abstract
Background
The hepatotoxicity of doxorubicin (DOX) is one of its main toxic side effects that cannot be ignored. As an important initiating factor for hepatocyte apoptosis and necrosis, oxidative stress is crucial in DOX-induced hepatotoxicity. Deoxyschizandin (Deo) is a highly effective antioxidant flavonoid that has potential therapeutic potential in liver diseases.
Objectives
It is of great clinical significance to demonstrate that Deo can effectively treat DOX-related hepatotoxicity.
Materials and Methods
The hepatotoxicity animal model was established by DOX, and Deo was used for intervention to observe the therapeutic effect. At the cellular level, phosphatase and tensin homolog deleted on chromosome ten (PTEN) is used as a therapeutic target for Deo to explore its impact on oxidative stress in hepatocytes.
Results
Liver function damage and oxidative stress successfully appear in animal bodies. Specific manifestations include biomarkers of liver function damage, abnormal biomarkers of oxidative stress, and excessive production of reactive oxygen species (ROS) in hepatocytes. PTEN signaling plays an important role in Deo’s inhibition of oxidative stress.
Conclusion
Our studies indicate that Deo improved DOX-induced liver oxidative stress, whose mechanisms may be related to PTEN signaling.
Introduction
As an antitumor drug discovered in the last century, doxorubicin (DOX) is indispensable in the treatment of various tumors (Khasraw et al., 2012). However, the toxic side effects that occur during use (mainly concentrated in the liver, heart, bone marrow, etc.) seriously restrict its anti-tumor application (Khasraw et al., 2012; Prasanna et al., 2020; Wallace et al., 2020). When DOX is metabolized in the liver, it can interfere with RNA transcription and synthesis and DNA replication in hepatocytes, hindering basic life activities (Kimura et al., 2010; Liu et al., 2017). When hepatocytes are damaged, the liver’s oxidative stress state is abnormally active, ultimately leading to various types of hepatocyte death (Brown et al., 2021; Prasanna et al., 2020; Wan et al., 2021).
Deoxyschizandin (Deo) is one of the most important flavonoids in the fruit of the herb Schisandra chinensis. It is a common material in traditional Chinese dietary therapy, with various effects such as anti-inflammatory, antibacterial, and immune enhancement (Fu et al., 2022; Yang et al., 2022). Pharmacological experiments have confirmed that Deo inhibits the generation of osteoclasts by inhibiting reactive oxygen species (ROS) and activating Nrf2 signals (Ni et al., 2020). It has also been shown that Deo can produce ROS in C2C12 cells and prevent DNA damage and apoptosis induced by oxidative stress (Choi, 2018). However, among the numerous studies on Deo, no reports have been found on DOX-induced hepatotoxicity.
Phosphatase and tensin homolog deleted on chromosome ten (PTEN) is widely expressed in various cells and is a key target for various diseases (Worby and Dixon, 2014; Yehia et al., 2019). It activates ROS production by targeting the upregulation of nuclear factor kappa B (NF-кB) expression, thereby regulating the survival, apoptosis, and metabolism of cells (Fu et al., 2020; Lai et al., 2009; Schabbauer et al., 2010). Abnormal NF-κB can stimulate the liver to release ROS (Bang et al., 2021; Xu et al., 2018).
Based on the research gap of Deo in DOX-induced hepatotoxicity, this study aims to explore its potential therapeutic mechanism by administering Deo as a therapeutic drug to DOX-induced hepatotoxicity rats.
Materials and Methods
Animal
The specific grouping scheme is as follows (random number method): (1) control group: solvent control of drugs (physiological saline); (2) Deo group: 20 mg/kg Deo (HY-N0693, MedChemExpress), gavage daily; (3) DOX group: 3 mg/kg DOX (HY-15142A, MedChemExpress), intravenous injection once a week; and (4) Deo+DOX group: DOX intravenous injection, Deo gavage (dose and frequency as before). Measure the body weight of each group of rats in a weekly routine and calculate feed consumption. The corresponding animal model establishment plan has been used and successfully achieved in our previous studies (Wang and Zhou, 2023).
Sample Collection
The experimental animals were executed under anesthesia (50 mg/kg pentobarbital, intraperitoneal). Perform chest hair removal on rats, open the chest cavity to expose the left ventricle, and obtain rat blood. Centrifuge under certain conditions (3000 rpm, room temperature, 20 min) to obtain serum. Subsequently, the liver was perfused with a phosphate-buffered solution (PBS) solution to remove residual blood, and the liver was obtained and stored at −80°C.
Histopathological Examinations
Immerse fresh liver tissue in a formaldehyde#Forms solution for more than 48 h, then dehydrate the liver tissue in ethanol solution of 70%, 80%, 96%, and 100% concentration gradients in turn. Finally embed the dehydrated liver tissue in paraffin, cut it into tissue sections of 5 µM thickness through a slicer, and lay it on the slide. Dye the sections with the hematoxylin and eosin (H&E) staining kit (C0105M, Shanghai Biyuntian Biotechnology Co., Ltd.). The instructions of the ROS staining kit were used to stain the ROS produced by hepatocytes in each group.
Serum Liver and Hepatocyte Biochemical Markers
Detect the content in rat serum or hepatocytes according to the experimental instructions of commercial kits for alanine transaminase (ALT), aspartate transaminase (AST), superoxide dismutase (SOD), malondialdehyde (MDA), glutathione (GSH), glutathione-peroxidase (GSH-px), catalase (CAT), and Total antioxidant capacity (T-AOC) (C009-2-1, C010-2-1, A001-3-2, A003-1-2, A006-2-1, A005-1-1, A007-1-1, and A015-1-2) and obtain the CD value at 450 nm in a spectrophotometer.
Cell
Primary Hepatocytes
Under the anesthesia of 50 mg/kg pentobarbital, the abdominal cavity of the mouse was opened aseptically. Primary liver cells were obtained through steps such as perfusion, digestion, filtration, centrifugation, and culture (Wang and Zhou, 2023). Subsequently, group experiments were conducted.
PTEN Lentivirus (lv)
Qingke Biotechnology Co., Ltd. (China) provided specific lentiviruses targeting PTEN (shRNA sequence: GCTAACTTACAACCTT) and non-targeted lentiviruses (shRNA sequence: CAACAGAGAGACCAA) for this study and completed packaging, transfection, and cell screening. Simply put, plasmid and 1µg/µL PEI were mixed in Optimem centrifuged at 4°C and 3,000 rpm for 10 min before transfection. Mix the supernatant with PEG8000 and centrifuge at 4°C, 4,000 rpm, and 20 min to obtain the virus. The lentivirus vector was transferred to primary liver cells with a complex infection of 10 in 15 µg/mL polybren. After 72 h, 2 µg/mL purine was used for screening primary liver cells.
Cell Grouping
Primary hepatocytes were cultured in a complete medium of Dulbecco’s modified eagle medium (DMEM) +10% PBS. Group as follows: (1) control group; (2) DOX group; (3) Deo+DOX group; (4) lv-control group; (5) lv-PTEN group; and (6) lv-PTEN+Deo group. All drugs used in cell research were dissolved in dimethyl sulfoxide (DMSO), with a DMSO content of 1/10,000 in each cell culture medium to ensure the exclusion of the impact of DMSO on the study. The corresponding cell model establishment scheme has been used and successfully achieved in our previous research (Wang and Zhou, 2023).
Cell counting Kit-8 (CCK-8)
Cells grouped according to the above will be treated with drugs and incubated in the dark at 37°C for 1 h according to the operating instructions of the CCK-8 commercial reagent kit (C0039, Shanghai Biyuntian Biotechnology Co., Ltd.). Finally, the CD value will be measured at 450 nm on a spectrophotometer.
Western Blot
Tissues or cells were split in radioimmunoprecipitation lysis buffer (containing a 1% protease inhibitor) provided by Biyuntian Biotechnology Co., Ltd. for more than 30 min to obtain the supernatant (12,000 rpm at 4°C for 15 min). Use the bicinchoninic acid protein concentration detection kit provided by Biyuntian Biotechnology Co., Ltd., and perform concentration testing on the protein solution according to the operating instructions. Biyuntian Biotechnology Co., Ltd. provided a protein-loaded buffer solution for this experiment. Mix the protein stock solution and protein-loaded buffer solution in a 4:1 ratio and heat them in a 95°C environment for 15 min. The protein sample was used for electrophoresis, electroporation, blocking, primary antibody PTEN (Wuhan Proteintech Biotechnology Co., Ltd., 1:1000, 22034-1-AP) and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) (Wuhan Proteintech Biotechnology Co., Ltd., 1:1000, 10494-1-AP) overnight, and incubated in secondary antibody for 1.5 h the next day. Finally, semiquantitative analysis was performed on the results of protein imprinting using BeyoECL Plus and Image Lab software.
Quantitative Real-Time Polymerase Chain Reaction (qRT-PCR)
Sangon Biotech provided primer sequences for this study, as follows: PTEN: forward 5-GCCAAATTTAACTGCAGAGTT GC-3, reverse 5-TACATAGCGCCTCTGACTGG-3; GAPDH: forward 5-CGCTAACATCAAATGGGGTG-3, reverse 5-TTGCTGACAATCTTGAGGGAG-3. Invitrogen (Shanghai, China) provided TRIzol reagents for this study and isolated RNA from tissues or cells according to the user manual. Takara Bio, Inc. provided the PrimeScript reverse transcriptase kit for this study and synthesized RNA cDNA according to the instruction manual. Similarly, Takara Bio, Inc. provided qRT-PCR using the SYBR PremixEX Taq kit for this study. Initial denaturation for 10 min (95°C), followed by cyclic denaturation for 15 s (45°C). Last 30 s (64°C) is the thermal cycling conditions of qRT-PCR. The internal control is GAPDH. Refer to the following information for the PCR operation scheme (Livak and Schmittgen, 2001).
Statistical Analysis
The normal distribution and homogeneity of variance of the data were tested using one-way or two-way ANOVA, followed by significant differences between groups (p ≤ 0.05 considered statistically significant) analyzed by Tukey’s multiple comparison post hoc test.
Results
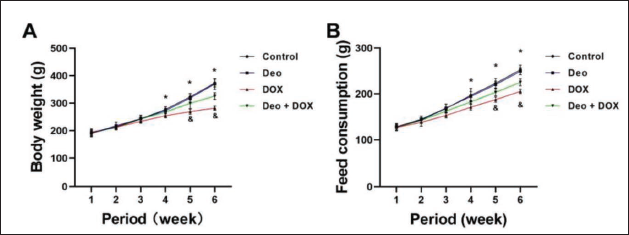
Deo Improves the Effects of DOX on Body Weight and Food Intake in Rats
The DOX group of rats showed a decrease in weight and food intake from week 4 onwards (vs. Control, Figure 1A, B). The weight and food intake of the Deo+DOX group rats significantly increased from the fifth week onwards (vs. DOX, Figure 1A, B).
Deo Effectively Alleviated the Abnormal Liver Morphology and Function Caused by DOX in Rats
The control liver tissue structure was clear without abnormalities, while DOX caused unclear boundaries, fusion, disordered arrangement, an abnormal nucleus, and increased intercellular space in the rat liver cells (Figure 2A). However, the above changes were significantly alleviated after Deo treatment (Figure 2A). In Figure 2B and C, DOX caused a significant increase in serum biochemical markers ALT and AST in rats (compared to the control group), but decreased ALT and AST after Deo treatment (compared to the DOX group).
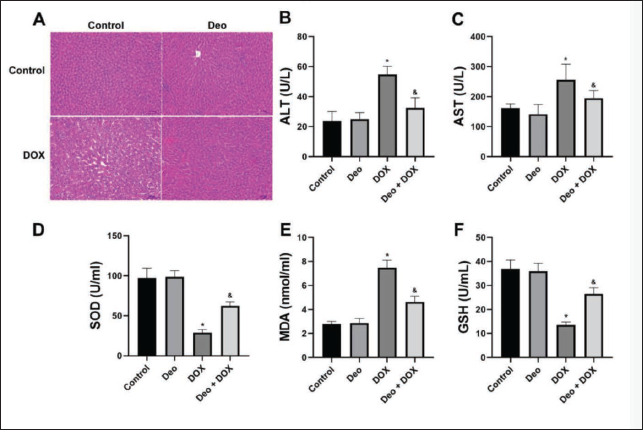
Deo Improved the Abnormal Serum Oxidative Stress State Caused by DOX in Rats
As shown in Figure 2D–F, DOX caused a decrease in serum SOD and GSH, while MDA significantly increased in rats (vs. control). Deo alleviated all abnormal serum oxidative stress biochemical markers (vs. DOX).
Deo Improved the Abnormal Liver Oxidative Stress State Caused by DOX in Rats
As shown in Figure 3A–F, DOX caused a significant decrease in the liver oxidative stress biomarkers SOD, GSH, GSH-px, CAT, and T-AOC, while MDA significantly increased in rats (vs. Control). Deo alleviated all abnormal liver oxidative stress biochemical markers (vs. DOX).
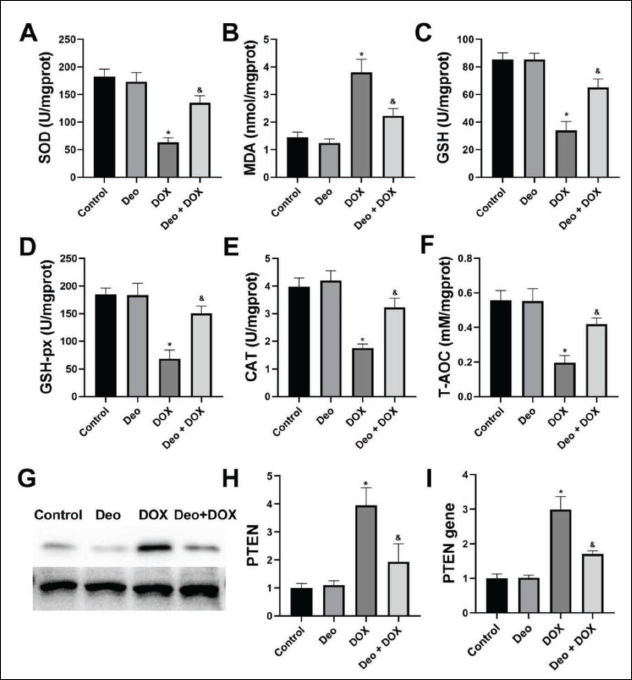
Deo Inhibits the Activation of PTEN Proteins and Genes in the Liver by DOX
The PTEN protein (Figure 3G) and gene (Figure 3I) in the DOX group increased (vs. Control). The PTEN protein and gene expression in the Deo+DOX group decreased (vs. DOX). Figure 3H is an analysis of the grayscale values of the image.
Deo Alleviates the Eexcessive Production of ROS in Hepatocytes
The ROS production of DOX and lv-PTEN hepatocytes increased significantly (vs. Control, Figure 4A). However, the ROS production of Deo+DOX hepatocytes was significantly reduced (vs. DOX). The ROS production of lv-PTEN+Deo hepatocytes was significantly reduced (vs. lv-PTEN).
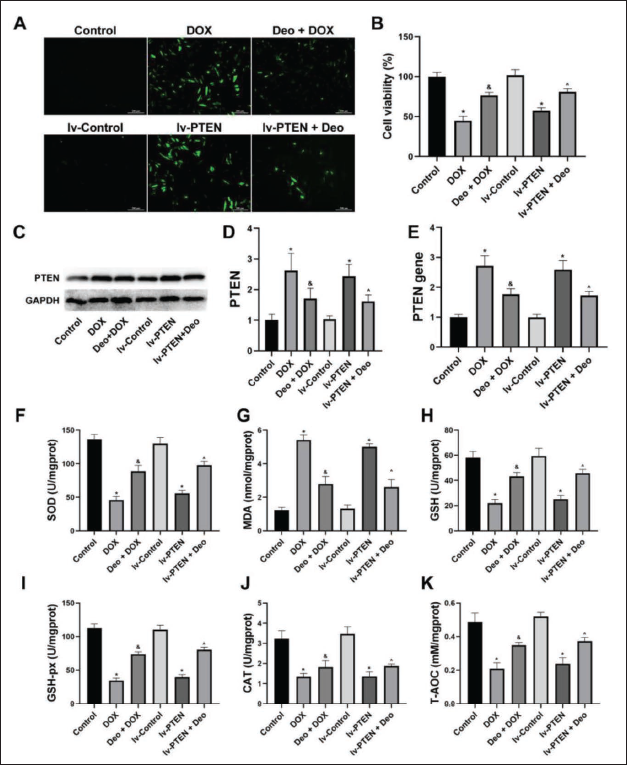
DOX and lv-PTEN significantly reduced hepatocyte viability (compared to the control group), while Deo intervention significantly increased hepatocyte viability (compared to the DOX and lv-PTEN groups) (Figure 4B).
Deo Inhibits the Activation of Hepatocyte PTEN Proteins and Genes by DOX
The expression of the PTEN protein (Figure 4C) and gene (Figure 4E) in DOX and lv-PTEN hepatocytes increased (vs. Control). However, these protein in Deo+DOX hepatocytes decreased (vs. DOX), and in lv-PTEN+Deo hepatocytes decreased (vs. lv-PTEN). Figure 4D is an analysis of the grayscale values of the image.
Deo Alleviated Abnormal Biochemical Markers of Oxidative Stress in Hepatocytes Caused by DOX
The DOX and lv-PTEN hepatocyte oxidative stress biochemical markers MDA (Figure 4G) were significantly increased and the SOD (Figure 4F), GSH (Figure 4H), GSH-px (Figure 4I), CAT (Figure 4J), and T-AOC (Figure 4K) were significantly decreased (vs. Control). However, all of these reversed after Deo treatment.
Discussion
Under physiological conditions, the level of ROS in hepatocytes is in dynamic equilibrium and plays a basic role in regulating cell activities (Cichoż-Lach and Michalak, 2014; Rezzani and Franco, 2021). Under pathological conditions, such as DOX-related hepatotoxicity, the content of endogenous antioxidant enzymes decreases, leading to high levels of ROS, leading to cell function inactivation, and gene expression changes (Cichoż-Lach and Michalak, 2014; Rezzani and Franco, 2021; Samarghandian et al., 2017). Therefore, toxic free radicals can be used as an early indicator of hepatotoxicity in clinical practice. Similarly, in this study, DOX caused liver function damage in rats. Further studies showed that the level of ROS in the injured liver was significantly increased; specifically, the content of antioxidant enzymes such as SOD, GSH, and CAT decreased, and the lipid oxidation end product MDA increased. Therefore, we speculate that reducing the level of oxidation in the liver may be the key to treating DOX-related hepatotoxicity.
A large amount of ROS appears during liver injury (Cichoż-Lach and Michalak, 2014; Zhang et al., 2018). Typical changes in ROS increase can be detected after hepatocyte injury, such as the abnormality of the antioxidant enzymes SOD, GSH, and CAT (Wang et al., 2022; Zhang et al., 2018). Studies have shown that the expression of PTEN increases when the body is injured, which reduces the phosphorylation of endothelial nitric oxide synthase (NOS) and then increases the expression of inducible NOS and gp91phox, which ultimately leads to the increase of nitric oxide, the superoxide anion and its cytotoxic reaction product peroxynitrite (ONOO−) and MDA production, the decrease of antioxidant enzymes, and ultimately damage target organs (Parajuli et al., 2012; Yu et al., 2018; Zhang et al., 2015). The ability to reduce or neutralize ROS in and out of cells, as well as inhibit its production, is critical for reducing oxidative damage to tissues and organs.
Another significant discovery in this study is that Deo can effectively alleviate the liver injury caused by DOX and shows excellent changes in a series of liver function indicators. Further studies showed that the beneficial liver protection effect of Deo could inhibit the expression of PTEN in hepatocytes and inhibit the production of ROS. In this study, the contents of SOD, GSH, GSH-PX, CAT, and T-AOC in the liver tissue of rats treated with Deo increased while MDA decreased. SOD is a mitochondrial oxidative stress antioxidant enzyme that inhibits lipid oxidation by scavenging ROS produced by mitochondrial metabolism and plays a role in cell membrane stabilization (Hassan et al., 2014). As an important antioxidant in the body, GSH can eliminate free radicals in the body, protect sulfhydryl groups in many proteins, enzymes, and other molecules from being oxidized by harmful substances, and enable its physiological functions to function normally (Harfield et al., 2012; Yin et al., 2015). GSH-px can specifically catalyze the hydrogen peroxide reduction reaction, causing hydrogen peroxide to lose its oxidation and protecting the integrity of cell membrane structure and function (Ren et al., 2014). CAT has the highest concentration in the liver, which can promote the decomposition of H2O2 into molecular oxygen and water so that it will not further produce highly toxic hydrogen and oxygen-free radicals, thus protecting the function of the antioxidant enzyme system (Hu et al., 2014). It is also of great significance for human growth and metabolism. T-AOC reflects the total antioxidant capacity of the body’s defense system, mainly including SOD, GSH, vitamin C, and vitamin E (Yang et al., 2019). MDA is one of the products of lipid peroxidation; its concentration can reflect the degree of cell damage and it is one of the biomarkers that respond to oxidative stress (Tangvarasittichai, 2015). Current research shows that the oxidative damage effect of the liver can be improved by the treatment of Deo, and the role of Deo in restraining oxidative stress is proposed. At present, many plants and their components with antioxidant activity have been studied to inhibit many diseases related to oxidative stress. In fact, PTEN may also be just a way for Deo to inhibit oxidative stress. Other targets or anti-inflammatory pathways may have an impact on ROS. In short, the treatment of natural drugs for diseases is extremely complex, but it is undeniable that they have excellent treatment effects.
Limitations
Although PTEN seems to induce ROS production in the liver in our study, inhibiting PTEN expression can effectively treat related diseases. However, PTEN seems to have played an excellent role in a few studies, such as tumor and vascular smooth muscle (Mayo et al., 2002; Yu et al., 2022). We believe that this may have different effects in different organs, tissues, or cells, but PTEN downregulates AKT and then plays a physiological role, which is the same conclusion. Another possible reason is that the proliferation of vascular smooth muscle cells or the appearance of tumor cells is a harmful phenomenon to organisms, and the intervention of PTEN by different treatment schemes (whether up or down) is to inhibit the proliferation of smooth muscle cells, promote tumor cell death and protect the function of normal cells (Lai et al., 2009; Mayo et al., 2002; Yu et al., 2022). Deeper research needs to be carried out, which also points out the direction for researchers. In addition, the extracted primary hepatocytes were not identified in this study. Although the corresponding extraction scheme was widely used more than 10 years ago and may have had little impact on the experimental results, it can also improve the rigor of the research if cell identification can be carried out.
Conclusion
Our research shows that Deo inhibits ROS production by inhibiting the PTEN signal and finally prevents DOX-induced hepatotoxicity. These findings will help us to further understand the role of PTEN in DOX-induced hepatotoxicity, find that Deo may be a potential PTEN inhibitor, and identify new targets and new drugs for the treatment and prevention of DOX-induced hepatopathy.
Abbreviations
DOX: Doxorubicin; Deo: Deoxyschizandrin; ROS: Reactive oxygen species; PTEN: Phosphatase and tensin homolog deleted on chromosome ten; HE: Histopathological examinations; ALT: Alanine aminotransferase; AST: Aspartate aminotransferase; SOD: Superoxide dismutase; MDA: Malondialdehyde; GSH: Glutathione; GSH-px: Glutathione peroxidase; CAT: Catalase; T-AOC: Total antioxidant capacity; CCK-8: Cell Counting Kit-8.
Authors’ Contributions
NZ and QZ: conception and design, key revision, approval publish, and responsible for this work. NZ: data acquisition, analysis, interpretation, and drafting. QZ: review and revision.
Availability of Data and Material
The data can be obtained from the corresponding author upon reasonable request.
Footnotes
Declaration of Conflict of Interests
The authors declare no conflicts of interest.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Statement of Informed Consent and Ethical Approval
All methods used in this study were conducted in accordance with relevant guidelines and regulations and approved by the Research Ethics Committee of Changzhou Second People’s Hospital (Approval No. CZ20220028).
