Abstract
Aim
Doxorubicin (DOX) has long been criticized for its cardiotoxicity, yet one cannot overlook its hepatotoxicity. Deoxyschizandrin (Deo) is a type of monomer used in traditional Chinese medicine that has a powerful anti-inflammatory action and is frequently used to treat a variety of inflammatory diseases. However, its therapeutic effect on DOX-induced hepatotoxic inflammatory reactions is still unknown.
Materials and Methods
In this study, DOX was used to induce hepatotoxicity in rats, and the effects of Deo on hepatotoxicity and hepatic inflammation were investigated.
Results
The results show that Deo protects liver function by inhibiting PETN, which regulates DOX-induced liver inflammation.
Conclusion
The anti-inflammatory effect of Deo in DOX-induced hepatotoxicity is attributed to the inhibition of phosphatase and tensin homolog deleted on chromosome ten (PTEN) and the increase in AKT phosphorylation.
Introduction
Doxorubicin (DOX) is a commonly used anthracycline anticancer chemotherapy drug that has a high activation effect on a variety of tumors. 1 However, there are numerous adverse reactions, such as toxic effects on the heart, liver, kidney, and so on, which lead to certain restrictions on its application.1–3 The liver is an important organ for DOX metabolism. 2 DOX can be embedded between DNA base pairs and tightly bound to DNA to prevent the RNA transcription process, inhibit RNA synthesis, and also prevent DNA replication in the liver.4, 5 Inflammation is an important pathophysiological basis of DOX-induced hepatotoxicity. 6 DOX has been shown in studies to activate NF-кB mediated inflammation in hepatocytes and the explosive release of inflammatory factors (TNF-α, IL-1, and IL-6), ultimately leading to the death of hepatocytes.2, 6, 7
Deoxyschizandrin (Deo) is one of the main lignans isolated from the fruit of Schisandra chinensis (Turcz.) Baill. 8 It has extensive pharmacological effects in anti-inflammation, anti-oxidation, and immune regulation.8, 9 Deo has been shown in studies to improve ulcerative colitis in mice, and its mechanism may be related to its inhibition of inflammatory factors such as NO, IL-6, and TNF-α. 10 Some studies have also shown that Deo can effectively inhibit inflammation induced by lipopolysaccharide. 11 However, the therapeutic effect of Deo on DOX-induced liver inflammation has not been reported.
Phosphatase and tensin homolog deleted on chromosome ten (PTEN) is a key molecule in the development of some inflammatory diseases and is widely expressed in the liver, kidney, heart, and other important target organs.12, 13 It regulates AKT expression, cell survival/apoptosis, and metabolism.13, 14 It has been confirmed that activation of the AKT pathway can reduce the expression of NLRP3 inflammatory bodies in the liver of rats with liver ischemia/reperfusion, and inhibition of the PTEN signal can activate the AKT-mediated anti-inflammatory effect, reduce the expression of NLRP3 inflammatory bodies in rats after liver transplantation, and slow down the injury of hepatocytes caused by ischemia/reperfusion.15, 16
However, it is rarely reported whether Deo can inhibit the PTEN signal and regulate the liver inflammatory response caused by DOX to protect liver function. This study is to explore the effect of Deo on the expression of PTEN and the inflammatory reaction in DOX-induced rat liver and its possible mechanism, so as to provide an experimental basis for Deo to improve the pathological process and mechanism of DOX-induced rat liver toxicity and to screen therapeutic targets.
Materials and Methods
Reagents
DOX (HY-15142A) and Deo (HY-N0693) were provided by MedChemExpress (MCE). Hematoxylin and Eosin (HE) Staining Kit (C0105M) and Cell Counting Kit-8 (CCK-8, C0039) were purchased from Shanghai Biyuntian Biotechnology Co., Ltd. (Shanghai, China). The alanine aminotransferase (ALT, C009-2-1), aspartate aminotransferase (AST, C010-2-1), C-reactive protein (CRP, H126-1-2), macrophage chemoattractant protein 1 (MCP-1, H115), and tumor necrosis factor-α (TNF-α, H052-1-2) assay kits were purchased from Nanjing Jiancheng Bioengineering Institute (Nanjing, China). The primary antibodies PTEN (22034-1-AP), against protein kinase B (AKT, 10176-2-AP), phospho-protein kinase B (p-AKT, 80455-1-RR), and GAPDH were purchased from Wuhan Proteintech Biotechnology Co., Ltd., Wuhan, China.
Animal
A total of 20 male Sprague Dawley rats weighing 180–220 g were obtained from the Medical Animal Center of Nanjing Medical University. All experimental procedures in this study follow the guidelines of the Research Ethics Committee of Nanjing Medical University. The rats were randomly divided into four groups: (1) Control group: 0.9% normal saline by gavage and tail vein injection; (2) Deo group: 0.9% normal saline by tail vein injection and 20 mg/kg Deo by gavage; (3) DOX group: 0.9% normal saline by gavage and 3 mg/kg DOX by tail vein injection; and (4) Deo+DOX group: 3 mg/kg DOX by tail vein injection and 20 mg/kg Deo by gavage. All the animals were in a standard laboratory (12-h light/dark cycle) and had free access to drinking water. Weight and food intake were measured weekly.
Liver and Serum Sample Collection
The rats were anesthetized by intraperitoneal injection of pentobarbital and then killed by cervical dislocation. The rat blood and serum were obtained immediately from the aorta (3000 rpm, 20 min at room temperature). Then the liver was excised, partially immersed in 10% formalin, fixed overnight, and partially frozen in a –80°C refrigerator for a follow-up study.
Histopathological Examinations
The liver tissue fixed in formalin was dehydrated in an ethanol solution with increasing concentrations (70%, 80%, 96%, and 100%). The tissue sample was then embedded in paraffin and cut into 5-µM thick sections. The sections were placed on normal slides and stained with HE for 2 h for histological examination. The specimens were examined under an optical microscope (Olympus BX-50; Olympus, Tokyo, Japan).
Serum Biochemical Markers
The contents of ALT, AST, CRP, MCP-1, and TNF-α in the serum of each group were measured in 96-well plates according to the operating instructions of several kits, and the absorbance values of each hole were read at 450 nm in the microplate reader.
Cell
Primary Hepatocyte Extraction
One male SD rat (6 w) was purchased from the medical laboratory of Nanjing Medical University. The rats were anesthetized by intraperitoneal injection of pentobarbital and sterilized with alcohol, and the abdominal cavity of the mice was opened under sterile conditions. Perfusion (5 ml/min) starts from the portal vein of the liver until there is no blood in the perfusate. Replace the perfusate with collagenase IV to continue perfusion digestion until the liver is soft. Then use a 100-mesh sieve to filter and clean the liver tissue, centrifuge, resuspension, count, and lay plates. Change the solution after 6 h and conduct a grouping experiment after 48 h. For the extraction scheme of primary hepatocytes, refer to the relevant literature. 17
Lentivirus Processing
In this study, the PTEN gene was silenced by lentivirus (lv). PTEN-specific lv and non-targeted control lv were purchased from Qingke Biotechnology Co., Ltd., Beijing, China. According to the manufacturer’s instructions, the lv vector was transferred into primary hepatocytes under the 15 µg/mL polybrene with a complex number of infection (MOI) of 10. The DMEM was changed 24 h after the infection. After 72 h, primary hepatocytes were screened with 2.0 µg/mL purine and cultured in an incubator (95% air, 5% carbon dioxide, 37°C).
Cell Grouping
Hepatocytes were divided into 6 groups: (1) Control group; (2) DOX group: 3 µM DOX; (3) Deo+DOX: 10 µM Deo+3 µM DOX; (4) lv-Control; (5) lv-PTEN; and (6) lv-PTEN+Deo.
Hepatocyte Viability
Hepatocytes were inoculated into 96-well plates and treated with drugs according to the above groups. Then add CCK-8 reagent to incubate for 1 h and measure the absorbance at 450 nm on the microplate reader. The hepatocyte viability of the Control group was set at 100%.
Western Blot
The liver tissue or liver cells were immersed in the RIPA lysis buffer (Shanghai Biyuntian Biotechnology Co., Ltd., Shanghai, China) containing 1% protease inhibitor and penicillin, ground into a chyle shape in a grinder, and cracked at 4°C for 2 h. Then it was centrifuged in a cryogenic centrifuge for 15 min (12000 rpm, 4°C) to retain the supernatant. Detect the protein concentration according to the operation instructions of the BCA protein concentration detection kit. Mix with the protein loading buffer (Shanghai Biyuntian Biotechnology Co., Ltd., Shanghai, China) and take a water bath at 100°C for 30 min. Then the protein lysate was used for 12% SDS-PAGE, membrane transfer, and protein blocking. Subsequently, it was incubated in the primary antibodies p-AKT (1:1000), t-AKT (1:1000), PTEN (1:1000), and GAPDH (1:5000) at 4°C for 14 h and then soaked in the secondary antibody (1:10000) for 1.5 h. Finally, BeyoECL Plus and Image Lab software were used to analyze the results of protein imprinting. GAPDH is used as an internal reference.
PCR
Using the TRIzol reagent (Invitrogen, Shanghai, China), we isolated RNA from liver tissue according to the instructions. The gene expression level was quantified by qRT-PCR. Adopt 2-∇∇CT calculation is used to calculate the relative number of tested genes. GAPDH was used as an internal control. The primer sequences of these genes are as follows: GAPDH: forward 5-CGCTAACATCAAATGGGGTG-3, reverse 5-TTG CTGACAATCTTGAGGGAG-3; NFκB: forward 5-AGAGGATTTCGATTCCGCTA-3, reverse 5-CGTGAAGTATTCCCAGGTTTG-3; IL-1: forward 5-GACCTGTTCTTTG AGGCTGAC-3, reverse 5-TTCAT CTCGAAGCCTGCAGTG-3; IL-6: forward 5-AACC ACGGCCTTCCCTACTTC-3, reverse 5-GATGAATTGGATGGTCTTGGTC-3; TNF-α: forward 5-GCCT CTTCTCATTCCTGCTT-3, reverse 5-TGGG AACTTCT CATCCCTTTG-3; PTEN: forward 5-CAATGACAGCCATCATCAAAGAG-3, reverse 5-GCTCAGACTTTTGTAATTTGTG-3; and MCP-1: forward 5-AGCCAACTCTCACTGAAGCC-3, reverse 5-AACTGTGAACAACAGGCCCA-3.
Results
Effects of Deo and DOX on Body Weight and Feed Consumption
During the 6-week model establishment period, compared with the Control group, the weight of rats in the Deo group did not change significantly (p > 0.05), while the weight of rats in the DOX group decreased significantly from the 4th week to the 6th week (Figure 1A); however, compared with DOX group, the weight of rats in the Deo+DOX group increased significantly from the 5th week (Figure 1A).
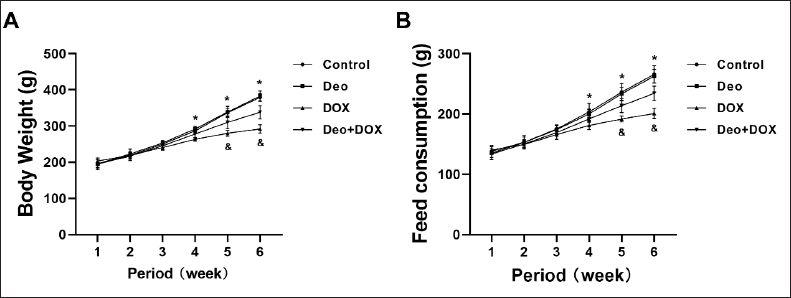
At the same time, compared with the Control group, the feed consumption of rats in the Deo group showed no significant difference (Figure 1B). The feed consumption of rats in the DOX group decreased significantly from the 4th week to the 6th week (Figure 1B); however, compared with the DOX group, the feed consumption of rats in the Deo+DOX group increased significantly from the 5th week (Figure 1B).
Effects of Deo and DOX on Liver Morphology and Function
As shown in Figure 2A, the liver tissue structure of rats in the Control group and Deo group is normal, while the liver of rats in the DOX group has obvious histomorphological abnormalities, such as unclear cell boundaries, fusion and arrangement disorder, increased cell gap, and abnormal nucleus, while the above abnormalities in the liver of rats in the Deo+DOX group are significantly alleviated.
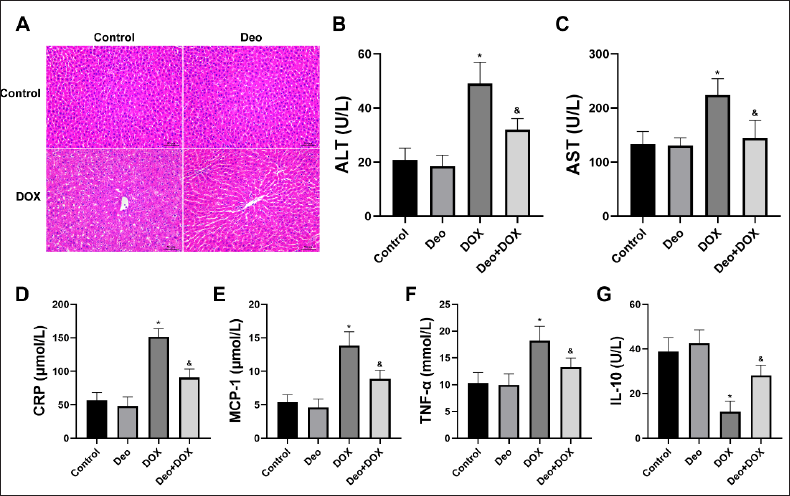
At the same time, compared with the Control group, the serum biochemical markers ALT (Figure 2B) and AST (Figure 2C) of liver function in the DOX group were significantly increased. However, compared with the DOX group, the serum biochemical markers ALT and AST of rats in the Deo+DOX group decreased (Figure 2B and 2C).
Effects of Deo and DOX on Serum Inflammatory Factor
Compared with the Control group, the serum inflammatory factors such as CRP (Figure 2D), MCP-1 (Figure 2E), and TNF-α (Figure 2F) were significantly increased, and IL-10 (Figure 2G) was significantly decreased in DOX group. However, compared with the DOX group, all the serum inflammatory factors in the Deo+DOX group decreased (Figure 2D–G).
Effects of Deo and DOX on Liver PTEN/AKT Protein
As shown in Figure 3A, compared with rats in the Control group, the expression of p-AKT/t-AKT proteins decreased, and PTEN protein increased in the liver of rats in the DOX group. However, compared with the DOX group, the above protein expression in the liver of rats in the Deo+DOX group reversed. Semi-quantitative analysis further confirmed the above results (Figure 3B and 3C).
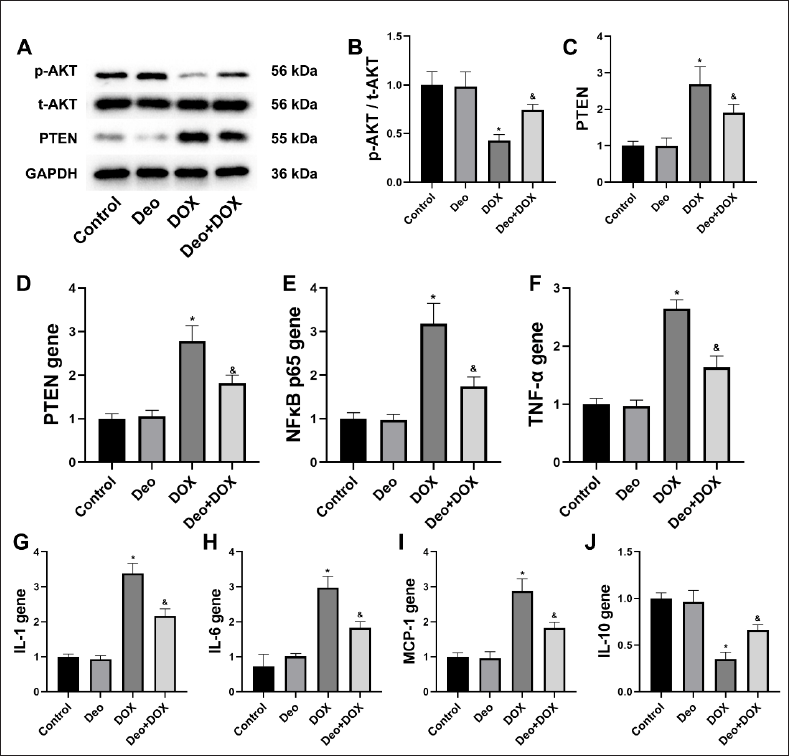
Effects of Deo and DOX on Liver Inflammatory Gene
Compared with the Control group, the liver inflammatory genes such as PTEN (Figure 3D), NFкB p65 (Figure 3E), TNF-α (Figure 3F), IL-1 (Figure 3G), IL-6 (Figure 3H), and MCP-1 (Figure 3I) were significantly increased, and IL-10 (Figure 3J) was significantly decreased in the DOX group. However, compared with the DOX group, all the liver inflammatory genes in the Deo+DOX group reversed (Figure 3D–J).
Effects of Deo and DOX on Hepatocyte PTEN/AKT Protein
As shown in Figure 4A, compared with the Control group and lv-Control group, the expression of p-AKT/t-AKT proteins decreased and PTEN protein increased in the hepatocytes of the DOX group and lv-PTEN group. However, compared with the DOX group, the above protein expression in the hepatocytes of the Deo+DOX group reversed, and compared with the lv-PTEN group, the above protein expression in the hepatocytes of the lv-PTEN+Deo group reversed. Semi-quantitative analysis further confirmed the above results (Figure 4B and 4C).
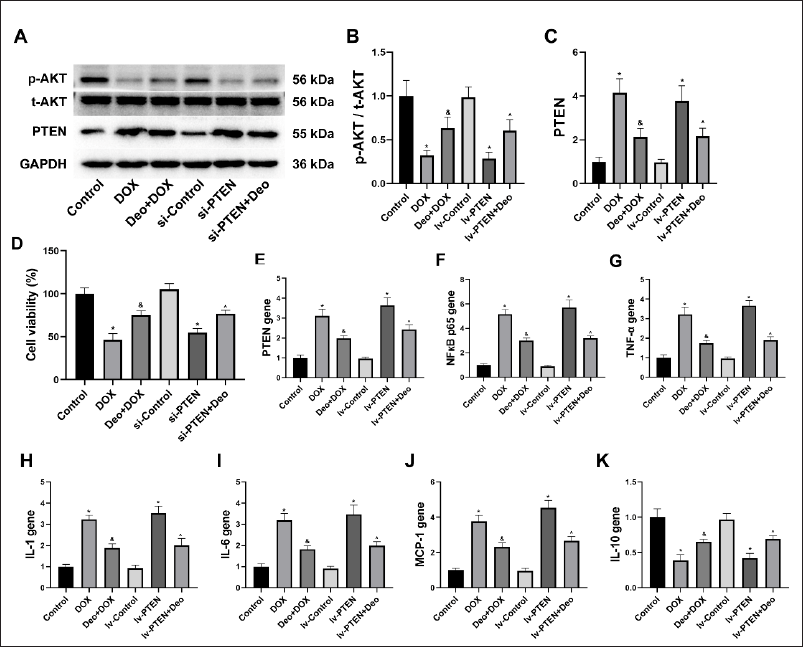
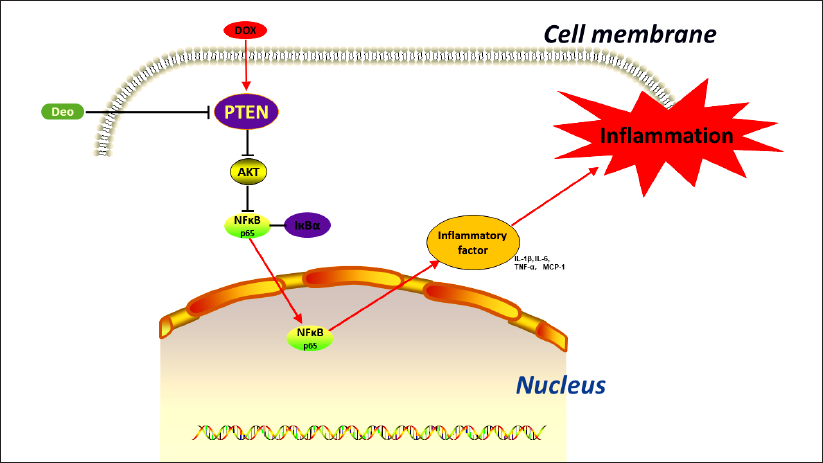
Deo Regulates the PTEN/AKT/NFкB Signaling Pathway in the Liver. Deo Inhibits PTEN, Regulates AKT/NFкB-mediated Inflammatory Signaling Pathways, and Prevents DOX-induced Liver Inflammation.
Effects of Deo and DOX on Hepatocyte Viability
As shown in Figure 4D, compared with the Control group, the hepatocyte viability in the DOX group and the lv-PTEN group decreased. However, compared with the DOX group, the hepatocyte viability in the Deo+DOX group increased, and compared with the lv-PTEN group, the hepatocyte viability in the lv-PTEN+Deo group increased.
Effects of Deo and DOX on Hepatocyte Inflammatory Gene
Compared with the Control group and lv-Control group, the hepatocyte inflammatory genes such as PTEN (Figure 4E), NFкB p65 (Figure 4F), TNF-α (Figure 4G), IL-1 (Figure 4H), IL-6 (Figure 4I), and MCP-1 (Figure 4J) were significantly increased, and IL-10 (Figure 4K) was significantly decreased in the DOX group and lv-PTEN group. However, compared with the DOX group, all the liver inflammatory genes in the Deo+DOX group reversed (Figure 3D–K), and compared with the lv-PTEN group, all the liver inflammatory genes in the lv-PTEN+Deo group reversed (Figure 3D–K).
Discussion
DOX is an indispensable part of many malignant tumor treatment schemes, but its use is limited due to serious toxic side effects. 18 Hepatotoxicity is one of the more serious side effects of DOX, which cannot be ignored. 19 The main clinical manifestation of it is the elevation of ALT and AST, 20 which has been verified in our animal models. Further studies showed that the inflammatory reaction was stimulated in the liver of DOX rats, which was related to the upregulation of PTEN in the liver. However, the role of PTEN in the pathogenesis of DOX-induced hepatotoxicity remains unclear. In this study, we confirmed that PTEN was upregulated and AKT was downregulated in DOX-treated rat liver and hepatocytes. We also found that the expression of inflammatory factors or genes was significantly upregulated in both serum and liver, such as NFкB p65, TNF-α, IL-1, IL-6, and MCP-1. The use of traditional Chinese medicine monomer Deo inhibited the liver inflammation, the upregulation of PTEN, and the inhibition of AKT in DOX rats, as well as the inflammatory factors and gene abnormalities in rat serum, liver, and hepatocytes. Therefore, regulating inflammation may be an effective channel for Deo to intervene in DOX-related hepatotoxicity, and PTEN may be a key target for Deo to exert a therapeutic effect by inhibiting an inflammatory reaction.
PTEN is a newly discovered tumor suppressor gene, located at 10q23.3, and its transcript is 515 kb of mRNA, which belongs to the protein tyrosine phosphatases gene family. 21 PTEN exists in almost all tissues in the body and plays an important role in cell growth, apoptosis, adhesion, migration, and infiltration.12, 22 PTEN is the first tumor suppressor gene with double-specific phosphatase activity found so far, which can participate in cell regulation through dephosphorylation.12, 23, 24 For example, the lipid phosphatase activity of PTEN can phosphorylate phosphatidylinositol triphosphate (PIP3) at D3 to produce phosphatidylinositol diphosphate (PIP2), thus activating the phosphorylation of AKT and affecting the expression of NFκB, the core target of inflammation, and ultimately regulating the normal function of cells.25, 26 Mayo et al. proposed that the up-regulation of PTEN is a necessary and sufficient condition for maximum activation of NF-κB. 27 In the study of RAW264.7 cells, it was found that PTEN, as a lipid phosphatase, antagonized AKT signal transduction through dephosphorylation, thereby regulating the activation of NF-κB. 28
It is well known that Deo has a strong anti-inflammatory effect in various inflammatory diseases, such as diabetes, kidney disease, and cardiovascular disease, and is significantly related to the treatment and prognosis of these diseases.29–31 Relevant studies also indicate that Deo has an effect in the treatment of inflammation. For example, Deo can treat ulcerative colitis through the TLR4/NF-κB signal pathway. 10 Another study indicated that Deo inhibited inflammation and oxidative stress induced by lipopolysaccharide in RAW 264.7 macrophages by inhibiting NF-κB, MAPKs, and PI3K/AKT pathways and activating Nrf2/HO-1 signals. 11 In this study, Deo could inhibit the inflammatory reaction in the liver of DOX rats. Further studies found that this effect was achieved by inhibiting PTEN, regulating the AKT/NF-κB signaling pathway, and then inhibiting liver inflammation.
As far as we know, no research has shown that Deo can effectively treat DOX related hepatotoxicity, and no research has clearly pointed out the role and specific mechanism of PTEN in treating DOX-related hepatotoxicity. More importantly, there is no report on Deo regulating liver inflammation by inhibiting PTEN. Our findings show that Deo, as a natural molecule, is also a natural inhibitor of PTEN and that it can reverse the inflammatory response in DOX-induced liver injury by inhibiting PTEN, highlighting its important potential mechanism.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All experimental procedures in this study follow the guidelines of the Research Ethics Committee of Nanjing Medical University.
Funding
The author received no financial support for the research, authorship and/or publication of this article.
