Abstract
Background
Epilepsy is a neurological disease, and its clinical treatment presents a significant medical challenge. Nevertheless, Chloriti Lapis (CL), a traditional Chinese Mineral Medicine, has been employed for centuries to address neurological disorders and has shown notable effectiveness.
Objectives
The objectives of this study are to further explore whether CL can modulate the abundance and diversity of the small intestinal flora in rats with epilepsy induced by pentylenetetrazol (PTZ) and to investigate the interrelationship among the flora.
Materials and Methods
Operational taxonomic units (OTUs), α diversity, β diversity, hierarchical clustering tree, and principal component analysis (PCA) were utilized to analyze the small intestinal flora of PTZ-kindled epileptic rats.
Results
The predominant bacterial groups in the small intestine mainly belonged to the phyla Firmicutes, Actinobacteria, Proteobacteria, and Bacteroidetes. The administration of CL was found to effectively increase the abundance and diversity of the small intestinal flora, modify its structural composition, reduce the relative abundance of PTZ-elevated microflora, and enhance the abundance of Streptococcus, Staphylococcus, and Gemella Berger.
Conclusion
CL exhibits promising potential, reliability, and unique value in managing epilepsy through the regulation of intestinal flora.
Keywords
Introduction
Epilepsy is a prevalent neurological condition characterized by its chronicity, recurrence, and significant burden on both individuals and society (Fisher et al., 2014). Recent research has estimated that there are approximately 65 million epilepsy patients worldwide, and this number continues to grow. Unfortunately, a substantial portion of these individuals lack access to adequate intervention and treatment, which in turn increases the risk of recurrent epilepsy episodes (Kanner & Bicchi, 2022). The etiology of epilepsy is complex, with factors such as head injuries, brain diseases, and central nervous system issues playing pivotal roles in its development (Hu et al., 2012). Epileptic seizures exert a profound impact on the physical, psychological, social, and economic aspects of the lives of those affected, and in severe cases, can even lead to fatalities.
Currently, there are approximately 20 types of antiepileptic medications available globally, including drugs such as phenobarbital, Phenytoin Sodium, Carbamazepine (CBZ), and sodium valproate. However, a significant proportion of epilepsy patients, roughly one-third, experience drug-resistant epilepsy, posing a considerable challenge in effectively managing their condition through medication (Löscher et al., 2020). Presently, the primary treatment methods for drug-resistant epilepsy include corpus callosotomy (Ball et al., 2018; Mallela et al., 2022), nerve stimulation therapy (Starnes et al., 2019), and electrical stimulation of the anterior nucleus of the thalamus (Fisher et al., 2010), among others. Regrettably, these approaches often lead to irreversible side effects and do not consistently achieve the desired treatment outcomes. Recent research has unveiled the dynamic and intricate microbiota residing in the human gastrointestinal tract, commonly referred to as the “intestinal flora.” This microbial community plays a pivotal role in the growth and metabolism of the human body while maintaining a complex and mutually beneficial relationship with it (Mohr et al., 2020). Notably, Perez et al. (2021) observed that a significant proportion, up to 60%, of patients with intestinal disorders exhibit symptoms associated with psychiatric disorders. Furthermore, Xu (2020) demonstrated that the onset of epilepsy is frequently accompanied by gastrointestinal discomfort, such as nausea and vomiting. Intriguingly, Bagheri et al. (2019) conducted an evaluation of the impact of a probiotic mixture on various aspects of epilepsy in rats and observed a substantial reduction in seizure severity with the administration of probiotics. Additionally, Xie et al. (2017) administered a ketogenic diet to children with epilepsy for 1 week and found that Cronobacter was the predominant bacterial group during the occurrence of epilepsy. These findings underscore the intricate and interconnected relationship between intestinal microorganisms and brain diseases.
During the 99th “Infection, Inflammation, and Chronic Diseases” conference held in Berlin, a presentation was delivered that provided compelling evidence in support of the concept known as the “microbe-brain-gut axis” (Ehlers et al., 2010). The theory of the “brain-gut axis” postulates a bidirectional relationship between the brain and the gastrointestinal tract, mediated through three primary pathways: neurological, endocrine, and immune. Within this axis, the intestinal flora plays a pivotal role, with the brain influencing the composition of the gut microbiota through the vagus nerve, the immune system, and the neuroendocrine system. Conversely, the gut microbiota produces neurotransmitters and modulators that impact the enteric nervous system, thus exerting regulatory effects on the central nervous system (Bienenstock & Collins, 2010). Notably, Holmes et al. (2020) conducted experiments that demonstrated how manipulating the gut microbiota in a drug-resistant epilepsy mouse model could alter the release of neurotransmitters in the brain, subsequently affecting the occurrence of seizures in drug-resistant epilepsy. This discovery holds promising potential as a potential breakthrough in the treatment of epilepsy, including cases that do not respond to conventional drug therapies.
Anti-convulsant action in modern pharmacology refers to the ability of a drug to inhibit or reduce seizures. These drugs often achieve anti-convulsant effects by regulating the release or mechanism of neurotransmitter substances (such as γ-aminobutyric acid) to inhibit the abnormal excitatory activity of brain neurons. In traditional Chinese medicine theory, some drugs or prescriptions have a calming effect that can regulate the function of the nervous system, calm the mind, and promote relaxation. This sedative effect can be achieved by affecting the central nervous system and regulating the flow of qi and blood. Both of them stabilize and inhibit abnormal excitation by regulating nervous system function. Both of them achieve the effect of stabilizing and inhibiting abnormal excitation by regulating the function of the nervous system and have certain commonalities.
Traditional Chinese medicine encompasses botanicals, animal-derived medicines, and mineral-based treatments. The use of minerals as therapeutic agents for preventing and treating diseases has a rich history in China, dating back several millennia. This practice represents a culmination of knowledge and experiences passed down through generations and holds deep traditional significance. Mineral-based medicines have been integral to the treatment of neurological disorders since ancient times, with roots tracing back to around 1600 BC. They were predominantly valued for their sedative and calming effects. Among these mineral medicines is Chloriti Lapis (CL), depicted in Figure 1, which falls under the category of magnesium-based mineral Chinese medicine. CL is sourced from metamorphic biotite schist or chlorite mica carbonate schist (Editorial Board of the State Administration of Traditional Chinese Medicine, 1998, pp. 293−295). Its historical documentation can be traced back to the Chinese book “Jia You Ben Cao,” originating during the Song Dynasty (1057–1059 AD). The 2020 edition of the Chinese Pharmacopoeia (Part I) delineates the therapeutic properties of CL, including its abilities to resolve phlegm, direct qi downward, pacify the liver, and alleviate fright. CL finds common clinical use in treating epilepsy and contains a spectrum of metal elements, such as Si, Fe, Mg, Al, Ca, K, and Na, along with trace amounts of Li, Ba, Ni, Zn, Ti, Sr, Se, Mo, Mn, Co, V, and Sn, among others (Liu et al., 2010). Furthermore, research has unveiled a complex interplay between mineral elements and the intestinal flora, which significantly influences the restoration of intestinal microecology (Chen et al., 2006).
Chloriti Lapis Sample.

In the previous phase of our research, our team delved into the impact of the mineral CL on a PTZ-kindling rat model of epilepsy. We meticulously examined shifts in metal element levels within brain tissue and plasma, as well as variations in amino acid neurotransmitter levels within brain tissue. Additionally, our investigation encompassed the assessment of differentially expressed proteins within the hippocampus (Bao et al., 2018; Liu et al., 2016; Liu et al., 2021a; Wu et al., 2016). We conducted a metabonomic analysis of brain tissue and scrutinized the composition of the intestinal microflora in the rat colon (Liu et al., 2021b; Yuan et al., 2021). Within our research paper, we probed the influence of CL on the small intestinal flora of PTZ-kindling epileptic rats, approaching the subject from the vantage point of intestinal flora. This exploration allowed us to uncover potential mechanisms through which CL may intervene in epileptic disorders.
Materials and Methods
Materials
The CL used in this study was sourced from the Bozhou mineral franchise (batch number 20140412). The analysis conclusively identified the rock as belonging to the metamorphic rock type known as Biotite Schist.
CBZ tablets were procured from Beijing Novartis Pharmaceutical Co., Ltd. (Lot No. X1395), while Pentylenetetrazol (PTZ) was obtained from Sigma Corporation, USA (Lot No. 1002905179). The E.Z.N.A.@ soil DNA Reagent Kit was purchased from Omega Bio-tek, Norcross, GA, USA, and the AxyPrep DNA Gel Extraction Kit was sourced from Axygen Biosciences, Union City, CA, USA. Additionally, 1,2-ethanediol homopolymer (PEG 400) of chemical purity was acquired from Shanghai Lingfeng Chemical Reagent Co. (batch number: 20170515).
Animals
A total of 48 male Sprague–Dawley (SD) rats in the specific pathogen-free (SPF) stage, weighing between 180 and 220 g, were procured from Zhejiang Academy of Medical Sciences (Certificate No. SCXK (Zhe) 2019-0002). These animals were acclimated in a controlled environment for 1 week, maintaining a temperature of 22 ± 2°C, a 12-h day/night cycle, and a humidity level of 50%–60%. During this period, the rats had unrestricted access to food and water.
The animals were housed in a barrier environment with alternating 12-h light and dark cycles, where room temperature was maintained between 20 and 24°C, and relative humidity remained around 50%.
PTZ-kindling Epileptic Rats Model
The epileptic rat model was established using the PTZ-kindling method (Liu et al., 2021a; Wang et al., 1993). PTZ (Sigma-Aldrich, USA) was dissolved in saline to create a solution with a concentration of 0.01 g/mL. Intraperitoneal injections of 35 mg/kg of PTZ were administered daily at a consistent time. After 4 weeks of continuous administration, the drug was discontinued for 1 week before testing with the same dose of PTZ. Seizures were classified according to the modified Racine scale as follows (Figure 2) (Racine, 1972): stage I: Facial clonus; stage II: Facial clonus and rhythmic nodding; stage III: Facial clonus, rhythmic nodding, and forelimb clonus; stage IV: Facial clonus, rhythmic nodding, forelimb clonus, and hind limb standing; and stage V: Facial clonus, rhythmic nodding, forelimb clonus, hind limb standing, and falling.
Molding Process of PTZ-kindling Epileptic Rats.
The kindling standard was defined as the occurrence of stage II or higher convulsion reactions in rats for five consecutive times. In cases where rats remained in stage V seizures for an extended period without showing signs of cessation, a 1 mL dose of 7% chloral hydrate was administered to alleviate the seizures. Throughout the experimental period, close attention was paid to any changes in the rats’ dietary habits, coat color, stress levels, as well as the presence of abnormal physiological behaviors such as mania or sluggishness in order to assess potential alterations in their mental state.
Preparation of Drug Administration
The CBZ tablets were dissolved in saline to yield a solution with a concentration of 0.02 g/mL. After removing quartz and other impurities from the CL, it was finely ground using an agate mortar. The resulting powder was then sieved through the Chinese Pharmacopoeia No. 6 sieve (100 mesh, 150 ± 6.6 µm). CL was further processed into a 0.5 g/mL suspension using PEG 400, with thorough ultrasonic mixing before each administration.
Grouping, Administration, and Sampling
One group served as the blank (B) and received saline treatment. Rats that successfully developed the model were then randomly assigned to four groups: the model group (M), the CBZ positive control group (C), the high-dose CL group (H), and the low-dose CL group (L) (Table 1). The B and M groups were administered 5 mL/kg of saline via gavage. The K group received a dose of 5 mL/kg. The CL dosage was 20 times the clinical dosage (equivalent to four times the clinical equivalent dose) in the H group, while the L group received a dosage five times the clinical equivalent dose, at 4 and 1 mL/kg suspension, respectively. In the clinical application process, CL in the treatment of epilepsy generally needs three to four courses of medication, and each course of time is 1 week. Thus, during the experiment, the epileptic rats were given the drug once a day at a fixed time for 4 weeks. Rat body weights were measured twice a week, and dosages were adjusted accordingly. On the day following the final administration, rats were anesthetized using isoflurane inhalation and subsequently sacrificed. The contents of the jejunum and ileum were collected and stored at –80℃ for subsequent testing.
Grouping of Experimental Animals.
DNA Extraction and High-throughput Sequencing
Microbial DNA extraction was carried out using the E.Z.N.A.® soil DNA Kit (Omega Bio-tek, Norcross, GA, USA) following the provided instructions. The concentration and purity of the isolated DNA were assessed using a NanoDrop 2000 UV–vis spectrophotometer (Thermo Scientific, Wilmington, USA), while the DNA’s quality was confirmed through 1% agarose gel electrophoresis. The V3–V4 hypervariable regions of the bacteria’s 16S rRNA gene were then amplified using the GeneAmp 9700 thermocycler PCR system (ABI, USA) with primers 338F (5′-ACTCCTACGGGAGGCAGCAG-3′) and 806R (5′-GGACTACHVGGGTWTCTAAT-3′). The resulting PCR products were extracted from a 2% agarose gel and further purified using the AxyPrep DNA Gel Extraction Kit (Axygen Biosciences, Union City, CA, USA). The quantification was carried out using QuantiFluorۛ-ST (Promega, USA) in accordance with the manufacturer’s protocol. Subsequently, sequencing was performed on an Illumina Miseq platform, following the standard protocols by Majorbio Bio-Pharm Technology Co. Ltd. (Shanghai, China).
Bioinformatics Analysis
Operational taxonomic units (OTUs) were clustered at a 97% similarity cutoff utilizing UPARSE (version 7.1,
Various analytical techniques were employed to examine the microbiota composition. This included generating rarefaction curves and conducting α diversity analyses (Chao1, Ace, Shannon, and Simpson) through OTU clustering. Furthermore, β diversity, hierarchical clustering trees, principal component analysis (PCA), and principal coordinate analysis (PCoA) were employed to explore the microbiota structure. The microbiota composition was scrutinized and compared at both phylum and genus levels.
To pinpoint taxa exhibiting significant differences between groups, linear discriminant analysis effect size (LEfSe) measurements were employed. Significance was determined at a level of p < 0.05, with an LDA score threshold of 5.
Statistical Analysis
The data were expressed as the mean ± standard error of the mean (SEM). To assess differences between two groups, the least significant difference (LSD) post hoc test was applied, while for comparisons involving multiple groups, the one-way ANOVA was used. A significance level of p < 0.05 was considered statistically significant.
Results
General Situation and Model Establishment of Rats
The growth rate of rats’ body weight in each group was consistent, with no significant differences observed. Approximately 1 week into the experiment, the rats’ fur color gradually transitioned to a yellowish hue, followed by the onset of stage Ⅱ convulsions in the second week. By the fourth week after modeling, all rats exhibited convulsions of stage Ⅱ or higher intensity. Throughout the modeling test, a majority of rats experienced stage Ⅱ or higher convulsions on five occasions, meeting the criteria for the PTZ-kindling epilepsy rat model. In the development of the PTZ-kindling epilepsy rat model, a total of 40 rats were utilized; out of which, 32 rats were successfully prepared, while 8 rats unfortunately did not survive the procedure. This yielded a modeling success rate of 80.0%.
OTU and Rarefaction Curve Analysis
The sequences deemed effective were found in both the jejunum and ileum. Upon conducting a rank-abundance curve analysis of OTU numbers, it was evident that the curves for each group in both the jejunum and ileum demonstrated a gradual, steady decline. This implies that the gut microbiota within the rat contents exhibited a diverse array of species, with a well-balanced distribution in terms of species abundance (Figure 3 A, B).
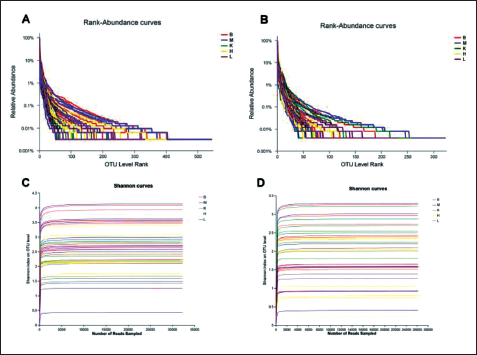
To evaluate the relationship between the number of randomly sampled sequences and the corresponding number of OTUs they represented, rarefaction curves were generated. The results demonstrated that as the number of species exceeded 2000, the Shannon curves began to plateau, signifying that the sequencing data volume was ample, an appropriate number of samples had been included, and the sequencing depth was reliable (Figure 3 C, D).
α-Diversity Analysis
Figure 4 displays the richness index (Chao 1 and Ace) and diversity index (Shannon and Simpson) of bacterial communities in the jejunum of rats. The findings revealed that there was no notable change in the Chao 1 and Ace richness index between the M and B groups. However, both the K and CL groups, particularly the L group, exerted a more significant impact on community richness compared with the B group.
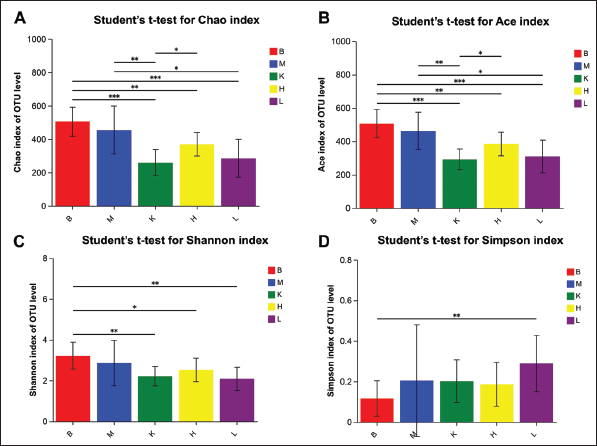
In terms of the Shannon and Simpson diversity index, no significant difference was observed between the M and B groups. Nonetheless, both the K and CL groups, especially the L group, exhibited noteworthy distinctions. Furthermore, the H group affected the Shannon index but did not have an impact on the Simpson index. These outcomes indicate that there was no substantial effect on the richness and diversity of the jejunal gut microbiota in PTZ-kindling epileptic rats. However, the mode of administration did influence both the richness and diversity of the intestinal microbiota. Figure 5 presents the richness index (Chao 1 and Ace) and diversity index (Shannon and Simpson) of bacterial communities in the ileum of rats. The findings revealed no significant differences in the Chao 1 and Ace richness index within each group, indicating that the richness of gut microbiota remained unaffected by the various treatments. Similarly, the Shannon and Simpson community diversity index did not show any significant distinctions between the M and B groups.
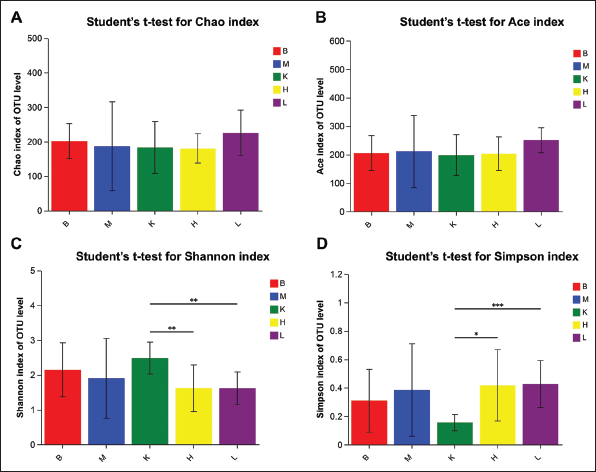
However, it was noteworthy that the K group exhibited significant differences when compared with the CL-treated group, whereas CL treatment did not exert a significant impact on community diversity. These results indicate that the PTZ-kindling method did not have a significant effect on the richness and diversity of gut microbiota in ileum contents. Conversely, CBZ primarily influenced the diversity of intestinal flora in the small intestine.
β-Diversity Analysis
Hierarchical clustering analysis, PCA, and PCoA of the intestinal microbiota of rats were conducted at the OTU levels (Figure 6). The hierarchical clustering results demonstrated that there was no distinct classification observed in either the jejunum or ileum. Notably, in the ileum, groups administered CL exhibited similar microbial community structures. This suggests that the various treatments had minimal impact on the overall structure of the microbial community. However, it was observed that both the high-dose and low-dose CL had comparable intervention effects on the intestinal flora structure of the ileum.
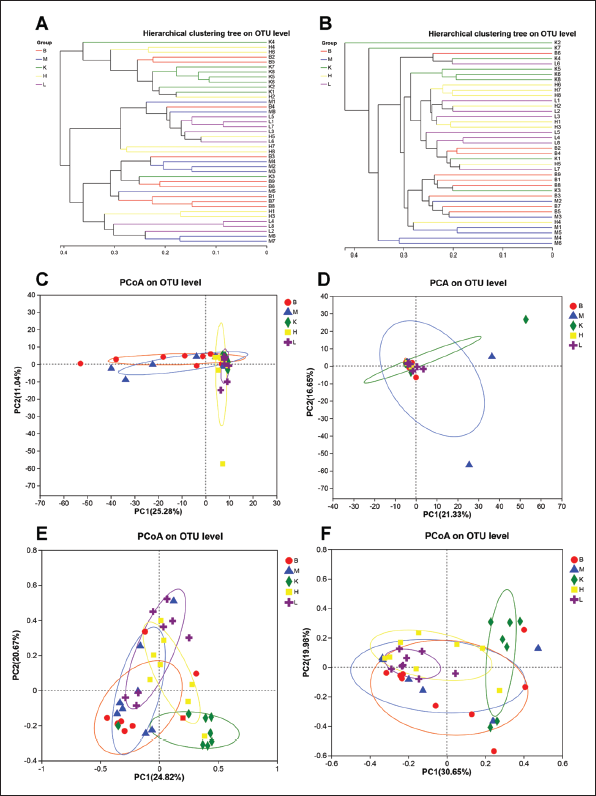
PCA and PCoA analyses demonstrated that the microbiota structure outlines of each group did not exhibit obvious separation. Most samples within each group clustered together in a relatively centralized manner. This suggests that both the modeling process and drug administration had minimal impact on the overall structure of the intestinal microbiota in rats. However, upon closer examination of the PCoA diagram, it became evident that the microbiota structure in both the jejunum and ileum of rats displayed a certain degree of separation following the administration of CBZ.
Species Composition Analysis
At the phylum level, discernible differences were noted in the composition of intestinal flora among groups, both in the jejunum and ileum (Figure 7). The dominant bacterial taxa in these segments primarily belonged to four phyla: Firmicutes, Actinobacteria, Proteobacteria, and Bacteroidetes. Firmicutes constituted the largest proportion, while proteobacteria and actinobacteria exhibited comparable levels of abundance. On the contrary, Bacteroidetes exhibited relatively lower levels of abundance.
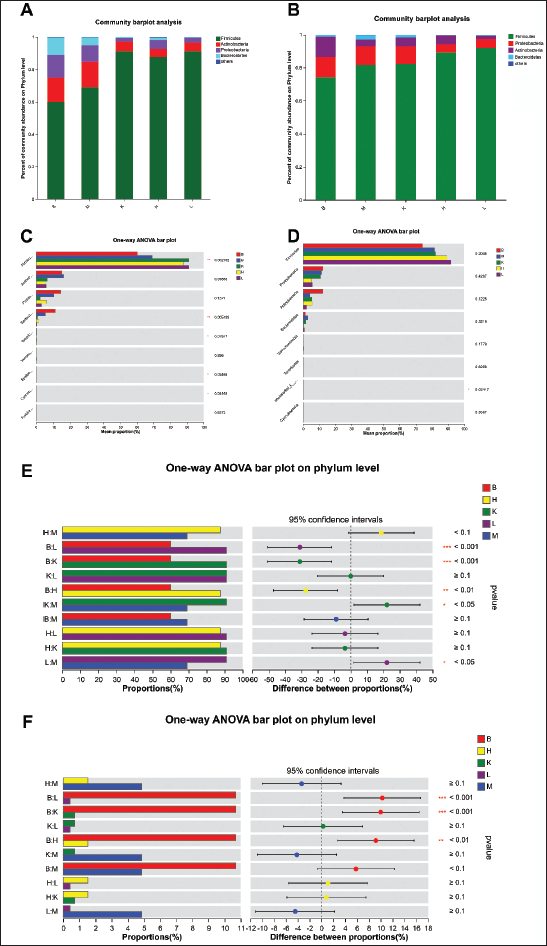
In the jejunum, the model group displayed an increase in the relative abundance of Firmicutes and a decrease in Bacteroidetes. Additionally, it was observed that the administration of CBZ, both at high and low doses, exacerbated this shift (p < 0.05). Furthermore, the abundance of proteobacteria and actinobacteria also decreased.
In the ileum samples, in comparison with the B group, the other groups demonstrated an increase in the relative abundance of firmicutes and a decrease in proteobacteria and actinobacteria. However, in contrast to the alterations observed in the jejunum contents, the abundance of Bacteroidetes in the ileum increased after modeling and decreased following administration.
At the genus level, there were 25 and 24 dominant bacteria identified in the jejunum and ileum of rats in each group (Figure 8 A, B). In the jejunum, 13 species of microbiota exhibited significant differences, including Romboutsia, Streptococcus, Corynebacterium_1, Aerococcus, Turicibacter, Staphylococcus, Psychrobacter, Gemella, Jeotgalicoccus, Alistipes, Bacteroides, Clostridium_sensu_stricto_1, and Faecalibacterium (Figure 8 C). In the ileum, there were seven species that showed significant differences, which are Romboutsia, Aerococcus, Corynebacterium_1, Rodentibacter, Staphylococcus, Rothia, and Jeotgalicoccus (Figure 8 D).
Composition of Gut Microbiota on Genus Level and Comparison of Differences Between Groups. (A) Average Flora of Each Group in Jejunum; (B) Multispecies Comparison of Each Group in Jejunum; (C) Average Flora of Each Group in Ileum; and (D) Multispecies Comparison of Each Group in Ileum.
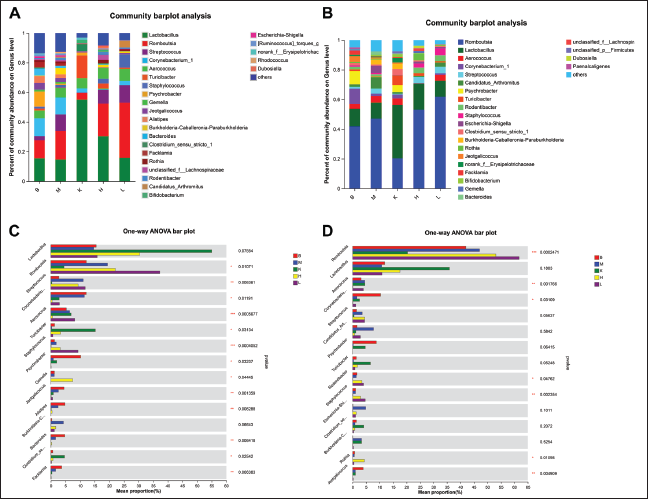
As depicted in Figure 9 A, the jejunum of the M group exhibited a significant increase in the relative abundance of Streptococcus and Bifidobacterium-Caballeronia-Paraburkholderia, compared with the B group. Conversely, the relative abundance of Psychrobacter showed a significant decrease. Following treatment with CBZ and CL, the relative abundances of Streptococcus and Bifidobacterium-Caballeronia-Paraburkholderia decreased to some extent, while the abundances of Psychrobacter increased, notably in the K group.
Comparison of Species Among Gut Microbiota of Each Group on Genus Level. (A) Post hoc Comparison of B and M in Jejunum and (B) post hoc Comparison of B and M in Ileum.
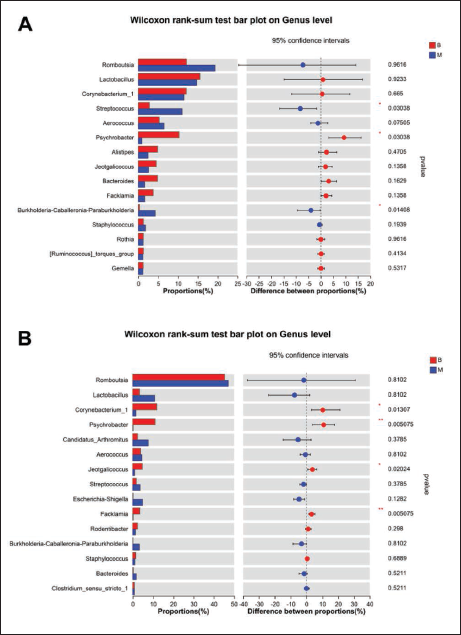
It is worth noting that post-modeling, the relative abundance of Jeotgalicoccus, Alistipes, Bacteroides, and Faecalibacterium decreased. However, their abundance did not recover even after the administration of CBZ and CL suspension. Additionally, the relative abundance of Lactobacillus and Turicibacter saw a significant increase in the K group.
In the ileum (Figure 9 B), specific bacterial species including Corynebacterium_1, Psychrobacter, Jeotgalicoccus, and Facklamia were found to have significantly lower relative abundances in epileptic rats compared with rats in the B group. However, after the administration of CBZ, there was a noticeable increase in the relative abundance of these bacterial species. Conversely, the administration of high and low doses of CL did not exhibit any notable improvement in the abundance trend.
LEfSe Analysis
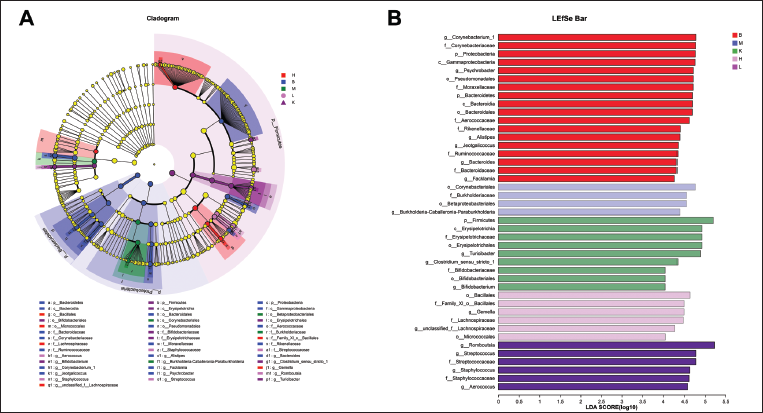
LEfSe analysis was applied to scrutinize changes in microbial composition at both the phylum and genus levels within the jejunum and ileum contents of rats across different groups. This was done to pinpoint specific bacteria that might be associated with epilepsy.
In the jejunum contents, the LEfSe analysis showcased significant disparities in gut microbiota composition among the groups (Figure 10). While Proteobacteria were primarily enriched in the B group, Bifidobacterium-Caballeronia-Paraburkholderia, a constituent of Proteobacteria, was notably enriched in the M group. Its relative abundance saw a decrease following drug administration. Turicibacter, Clostridium_sensu_stricto_1, and Bifidobacterium were prominently enriched in the K group. In the H group, the highest relative abundance was observed in Gemella Berger, whereas in the L group, Romboutsia, Streptococcus, Staphylococcus, and Aerococcus were notably enriched.
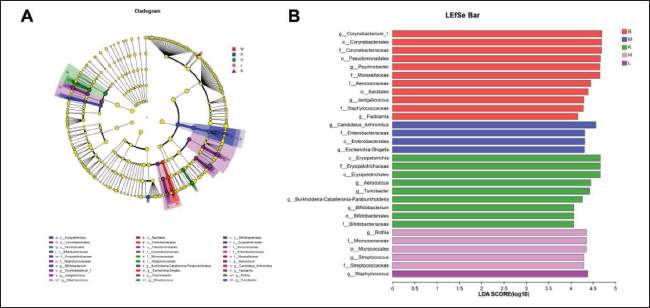
In the ileum, the contents of modeled rats exhibited a notable enrichment of Bifidobacterium-Caballeronia-Paraburkholderia. Moreover, Turicibacter and Bifidobacterium were significantly enriched in the K group, while the H group displayed the highest relative abundance of Staphylococcus. Conversely, the L group demonstrated a significant enrichment of Streptococcus and Gemella. The enrichment pattern of microbiota mirrored that observed in the jejunum (Figure 11).
Discussion
In this study, it was surprising to observe that the abundance and diversity of intestinal flora in the small intestine (jejunum and ileum) of the PTZ-kindling rat model of chronic epilepsy did not exhibit significant alterations when compared with normal rats. This finding contrasts with some previous studies. For instance, Peng et al. (2018) analyzed the gut flora of epileptics and normal subjects and found no substantial differences between patients with general epilepsy and normal subjects. However, refractory epilepsy showed higher α diversity compared with the other two groups. Lindefeldt et al. (2019) also discovered that the diversity of intestinal flora in specific epilepsy patients differed from that in both general epilepsy patients and ordinary individuals.
Additionally, the administration of CBZ, a commonly prescribed drug for epilepsy, resulted in notable changes in the abundance and diversity of intestinal flora in the model rats compared with both the model and normal groups. However, the effect of different doses of CBZ did not show significant distinctions. Gómez-Eguílaz et al. (2018) employed a mixture of microorganisms for treating epilepsy patients, and He et al. (2017) induced epilepsy by transplanting microbiota from feces. Remarkably, in the absence of antiepileptic drugs, there was no recurrence of epilepsy for up to 20 months after treatment.
Based on these experimental findings, it is reasonable to suggest that the alterations in intestinal flora following drug administration may be attributed to the impact of CBZ on the diversity and abundance of probiotic, harmful, and neutrophilic bacteria. This shift in bacterial distribution and dominance may contribute to therapeutic efficacy in epilepsy through changes in metabolite ratios.
Hierarchical clustering analysis, as well as principal component analysis (PCA) and principal coordinate analysis (PCoA), were employed to evaluate the influence of different treatments on the flora structure in the small intestine. The outcomes did not indicate a significant segregation of the flora profiles, signifying that the treatments had a modest impact on the flora structure in the small intestine. Notably, both the high and low-dose groups of CL demonstrated a comparable intervention effect on the flora structure in the ileum. However, the CBZ group exhibited a certain degree of segregation in the profiles.
In clinical experiments, Zakrzewska et al. (2017) observed that CBZ often leads to diarrhea and subsequent dysbiosis of the gut microbiota. This dysbiosis could plausibly account for the segregation observed in the CBZ group in the PCoA analysis. The reduction in the high abundance of Aspergillus clades in the small intestine of rats in the model group following the administration of CBZ and different doses of CL is noteworthy. This implies that a healthy flora composition is characterized by a low abundance of Aspergillus clades, and an abnormally high expression of these clades may trigger an inflammatory response (Leite et al., 2020).
The abundance of Streptococcus and Bifidobacterium in the jejunum of epileptic rats was significantly higher compared with healthy rats, and their abundance decreased somewhat after drug administration. Streptococcus is a group of bacteria known for disrupting the balance of intestinal microorganisms and affecting the permeability of the intestinal barrier. It has been observed that abnormal expression of Streptococcus is associated with patients suffering from meningitis, which can promote neuroinflammation and nerve damage (Hattori et al., 2020). On the contrary, Bifidobacterium is a common group of pathogenic bacteria whose abnormal presence in the small intestine can disrupt the microbial community balance (Dong et al., 2010).
The administration of the drug carbamazepine (CBZ) had a more pronounced effect on the small intestinal flora, increasing the abundance of Turicibacter and Bifidobacterium. Turicibacter is involved in fermentation metabolism, with lactic acid as its primary metabolite. Lactic acid can aid in the inactivation of pathogenic bacteria, thus protecting the intestinal environment (Collins et al., 2014). Bifidobacterium is a well-known class of probiotics capable of inhibiting pathogenic bacteria such as Staphylococcus aureus. Additionally, it can break down abundant conjugated bile acids in the small intestine into free bile acids, which possess stronger antibacterial properties. Bifidobacterium also produces various vitamins essential for the human body, including VB6, which plays a role in epilepsy treatment as a coenzyme for the decarboxylation of glutamate decarboxylase into γ-aminobutyric acid in the brain. Bifidobacterium is also involved in the detoxification of nitrogenous substances, thus safeguarding neurological functions. Moreover, Bifidobacterium can stimulate intestinal immune cells and enhance the body’s immune function, protecting the human body (Mager et al., 2020).
Although different doses of CL administration did not have a significant impact on intestinal flora diversity and abundance, it did intervene to some extent with Streptococcus, Staphylococcus, and Gemella Berger. Streptococci, as conditionally pathogenic bacteria, are typically part of the normal flora in a healthy organism. In vitro studies have shown that Streptococci are involved in the production of 5-HT, which, as an inhibitory neurotransmitter, is closely linked to epilepsy development. The increase in the relative abundance of streptococcal species in the CL group may contribute to the synthesis of 5-HT and help suppress seizures (Singhal et al., 2019). This further supports the rationale for the changes in intestinal flora diversity and abundance observed in the model rats after drug administration as mentioned earlier.
Both Staphylococcus and Gemella Berger are associated with intestinal infections, and their higher expression in the CL group may be related to the mineral drug attributes and the method of administration. Additionally, it was noted that the CL sample (in powder form) may cause some degree of damage to the small intestinal mucosa of rats (Shinagawa et al., 2014). This underscores the importance of selecting an appropriate drug delivery method that balances efficacy and safety to enhance drug utilization and user convenience.
A healthy intestinal flora plays a pivotal role in human growth and development. It accomplishes this by synthesizing essential vitamins and amino acids, participating in the metabolism of sugars and proteins, and aiding in the absorption of crucial minerals such as iron (Fe), magnesium (Mg), and zinc (Zn) (Derrien et al., 2017; Mayneris-Perxachs et al., 2021). Numerous studies have explored the effects of metal ions on the intestinal microbiota, shedding light on the mechanisms through which various metal ions promote the growth of beneficial bacteria and maintain a harmonious microbial balance in both animal models and humans (Li et al., 2021; Huynh et al., 2022). The equilibrium of the intestinal microecology is paramount for sustaining overall health, as any disruption in the flora can lead to a range of diseases.
CL has long been used in the treatment of neurological diseases such as epilepsy, and now it has been proved that the metal element in it can affect the intestinal flora. In addition, the studies of Jomova (Jomova et al., 2022; Jomova & Valko, 2011) showed the influence mechanism of metal ions on neurological diseases, indicating that CL affects epilepsy by influencing intestinal flora, maintaining the level of metal ions related to neurological diseases in the body, and activating and inhibiting related enzymes. Current studies have shown that drugs regulate the central nervous system through various mechanisms that can involve the balance of excitatory and inhibitory neurotransmitters, as well as the control of neural excitability. Among them, the related drugs that affect the intestinal flora make the intestinal flora regulate the release of neurotransmitters through the interaction between the microbial–brain–gut axis and the central nervous system, and the intestinal flora interfere with the synthesis and release of neurotransmitters through the release of metabolites and signal molecules, thus affecting the activity of neurons. The mechanism of the influence of intestinal microbial metabolites and signaling molecules on neurons includes the following processes (Chen et al., 2021; Sampson & Mazmanian, 2015; Sharon et al., 2016; Strandwitz, 2018; Yarandi et al., 2016): (1) Synthesis and release of neurotransmitters, the gut microbiota can produce neurotransmitters or their precursors, such as glutamate, aminobutyric acid, serotonin, dopamine, and acetylcholine, and neurotransmitters synthesized by the gut microbiota can enter the bloodstream and be transported to other parts of the body, including the brain; (2) Regulation of neurotransmitter synthesis, the gut microbiota can regulate neurotransmitter synthesis by regulating the availability of its precursors. These precursors can cross the blood–brain barrier and participate in the synthesis cycle of neurotransmitters in the brain; (3) Local signaling and communication, the gut microbiota can signal through its metabolites to promote the synthesis and release of neurotransmitters by intestinal endocrine cells. This local signal can affect the enteric nervous system and transmit rapid signals to the brain via the vagus nerve; and (4) Gut–brain communication via the vagus nerve: the vagus nerve plays a vital role in two-way communication between the gut and the brain. It not only transmits local sensory impulses mediated by enteric nerve podocytes but also acts as a physical bridge to the microbial–gut–brain axis. Bacteria-derived molecules or components can be transported to the brain via the vagus nerve, potentially affecting brain function and behavior. These mechanisms highlight the complex relationship between gut microbiota and neuronal function. Of course, what role do different bacterial communities play in the mechanism; The optimal proportion of bacteria; Whether there are other potential applications for the gut microbiota to influence neurotransmitters requires further investigation.
The experimental methods employed in this study can objectively demonstrate that CL can regulate the intestinal flora of PTZ-kindling epileptic rats, as well as the associated changes in the intestinal flora. However, these methods do not provide a comprehensive explanation of the correlation between the altered intestinal flora and neurotransmitters such as γ-aminobutyric acid and glutamate, which are closely related to epilepsy in brain tissues. This aspect requires further in-depth exploration and investigation.
Mineral Chinese medicine, characterized by its established efficacy, extensive historical usage, and robust contemporary research supporting its safety and effectiveness, is poised to emerge as a prominent approach to disease treatment. It offers a novel perspective on therapeutic strategies, promising new avenues for addressing various medical conditions.
Conclusion
This paper investigated the impact of CL on the intestinal flora of epileptic rats and drew several key conclusions:
Analysis of α diversity and β diversity in the contents of the small intestine (jejunum and ileum) revealed notable changes in microbial diversity and abundance.
Through species composition analysis and LEfSe analysis, the study examined the composition of the intestinal flora in the small intestine and identified potential factors that influence epilepsy treatment.
The anti-epileptic effect of CL was found to be closely associated with its ability to positively regulate the diversity and abundance of the intestinal flora in the small intestine.
Abbreviations
CL: Chloriti Lapis; PTZ: Pentylenetetrazol; CBZ: Carbamazepine; PEG 400: 1,2-ethanediol homopolymer; SD: Sprague–Dawley; SPF: Specific Pathogen-Free; B group: Blank group; M group: Model group; C group: CBZ positive control group; H group: High-dose CL group; L group: Low-dose CL group; OTU: Operational taxonomic units; PCA: Principal component analysis; OTUs: Operational taxonomic units; PCoA: Principal coordinates analysis; LEfSe: Linear discriminant analysis Effect Size.
Footnotes
Acknowledgments
We thank the organizations (4th National Survey of Chinese Materia Medica Resources-Mineral Chinese Medicine Resources, Strategic Research Project on the Modernization of Mineral Chinese Medicines of the Science and Technology Committee of the Ministry of Education of the People’s Republic of China) for their funding and support for our study.
Authors’ Contributions
YP, LFT, and QXL conceived and designed the experiments. YP, MYL, ZQ, and ZXY performed the experiments. QXL, YBF, ZL, YW, and PYQ analyzed and interpreted the data. DSL, FF, ZJ, and LSJ contributed reagents, materials, analysis tools, or data. QXL, YP, and LSJ wrote the paper. All authors have approved the manuscript.
Availability of Data and Materials
All the relevant data used in the study have been provided in the form of figures in the published article.
Declaration of Conflicting Interests
The authors indicated no conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the 4th National Survey of Chinese Materia Medica Resources-Mineral Chinese Medicine Resources Survey Project (GZY-KJS-2018-004) and Strategic Research Project on the Modernization of Mineral Chinese Medicines of the Science and Technology Committee of the Ministry of Education of the People’s Republic of China (2021), Bethune Charitable Foundation (No. BCF-XD-ZL-20220118-002).
Statement of Informed Consent and Ethical Approval
All animal experiments were approved by the Laboratory Animal Ethics Committee of Nanjing University of Chinese Medicine (No. 20190A029). The rock sample underwent thorough analysis and classification conducted by Professor Dekang Wu from the Teaching and Research Department of Chinese Medicine Identification at Nanjing University of Chinese Medicine, in collaboration with Professor Qingyou Kong from the School of Earth Science and Engineering at Nanjing University. The sample is presently housed within the Teaching and Research Department of Chinese Medicine Identification at Nanjing University of Chinese Medicine.
