Abstract
Background
There is growing evidence that antibiotic resistance is increasing. As a result, the efficacy of these antibiotics is gradually declining. This poses a serious health risk in terms of treatment strategies. Therefore, the scientific community is continuously putting all efforts into looking for novel and green therapeutics to overcome the problem of antibiotic resistance.
Objectives
The aim of this study is to investigate the chemical composition of the methanolic extracts obtained from Phoenix dactylifera seeds, known agricultural waste, to identify new bioactive compounds and to examine the antibacterial and antifungal potentials.
Materials and Methods
Phytochemical profiling was performed by the gas chromatography-mass spectrometry (GC-MS) technique. For the antibacterial activity of the methanolic extract, 12 microorganisms were selected, including eight bacteria (Escherichia coli ATCC 35218, Pseudomonas aeruginosa ATCC 27853, Enterobacter cloacae, Enterococcus faecium, Staphylococcus hominis, Staphylococcus aureus, Staphylococcus epidermidis, and Klebsiella pneumoniae ATCC 27736) and four Candida species (C. albicans ATCC 10231, C. tropicalis ATCC 1362, C. utilis ATCC 9255, and C. guilliermondii ATCC 6260). The antimicrobial activity of the plant extract was supported by in silico molecular docking analysis.
Results
The obtained results from the GC-MS technique revealed the presence of 51 compounds, of which 6-octadecenoic acid (Z) was identified as the major component. The tested extract exhibited the highest zone of inhibition against S. aureus (growth inhibition zone 14.66 ± 0.57 mm). In addition, minimal inhibitory concentration (MIC) and minimal bactericidal concentration values obtained against S. aureus were 1.4 and 5.6 mg/mL, respectively. Regarding the antifungal assay, C. albicans was the most sensitive one to the extract among all the tested Candida strains (14.33 mm) (MIC: 1.4 mg/mL, minimal fungicidal concentration (MFC): 11.2 mg/mL). The binding free energies and root mean square deviation values obtained from the interaction of phytochemicals with two different target receptors (1JIJ and 2QZW for TyrRS from S. aureus and aspartic proteinase from C. albicans) were also determined. Ergost-5-en-3-ol, (3beta) established hydrogen, electrostatic, alkyl, and pi-alkyl interactions with five different amino acid residues (Asp40, Asp80, Pro53, His50, and Phe54) located at the TyrRS receptor of S. aureus. On the other hand, hexadecanoic acid and methyl ester established hydrogen, electrostatic, pi-sigma, and alkyl interactions with five different amino acid residues (Thr222, Asp86, Asp218, Tyr84, and Ile123) located at the aspartic proteinase receptor of C. albicans. These interactions contributed to the stability of the complex and, hence, to the biological activity of the P. dactylifera compounds.
Conclusion
Our results highlighted that methanolic extract from date seeds can be a natural source of chemical compounds with promising antibacterial and antifungal activities, as demonstrated using both in vitro and in silico approaches.
Introduction
Natural compounds derived from plant species are promising candidates to counteract the widespread prevalence of drug-resistant microorganism. Thus, scientists focused on the identification of new molecules from various natural sources to combat such kinds of bacterial, candida, and fugal strains. (Mallhi et al., 2014). The latest reported methods and strategies include the extraction of bioactive substances from marine microorganisms or lactic acid bacteria, the synthesis of new drugs, in particular the drug development processes in the field of proteomics, and robust site-specific drug delivery using polymers or nanotechnological methods (Naz et al., 2012;). Apart from the aforementioned techniques, researchers thoroughly work on plant extracts, which are widely recognized for their potential therapeutic benefits. Plants or microorganism-derived bioactive compounds exhibit higher applicability compared to synthetic drugs. The pharmacological effects of some of the investigated compounds are well known, such as anti-Parkinsonian, cytotoxic, hypotensive, bronchodilator, antimicrobial, anti-inflammatory, amoebicidal, hepatoprotective, antidiarrheal, antifertility, hypoglycemic, antioxidant, and spasmolytic (Ali et al., 2013; Janbaz et al., 2013).
Phoenix dactylifera L. is a tree of Mesopotamian origin. Almost all parts of this tree are used industrially for certain purposes. While wood and timber can be obtained from their trunks, they are also used as fuel. The fibers obtained from both the stem and the leaves are very strong and are used in the manufacture of household items, such as camel saddles, baskets, trays, furniture, bags, twine, rope, paper, chests, food covers, fans, cords, and mats. Dried leaves are also useful; these leaves can be used as a covering material on the roofs of buildings as well as to make shade. On the other hand, since dried leaves can be easily ignited, they are often used as fuel by the local people. The fruits of the tree are known as dates and are consumed both fresh and dried. Dates are generally consumed after they are fully ripe; thus, they have high sugar and low tannin content, but they can be consumed before they are ripe in North Africa, in which case more tannins are taken into the body. The oil obtained from the palm kernel is also used in soap making (Qamar et al., 2019).
The nutritional value of dates is quite high. In addition, it contains numerous important bioactive compounds bearing pharmacological significance (Al-Snafi & Thuwaini, 2023). The dates are also used as an aphrodisiac, heart tonic, expectorant, sedative, and laxative supplement. In traditional medicine, date fruits are used in the treatment of diseases such as gastroenteritis, cough, hypertension, fatigue, asthma, diabetes, and chest pains (Atif et al., 2017). Furthermore, it is stated that it has important therapeutic effects in treating liver disorders (Al-Mamary et al., 2014). It is also said that the plant’s pollen and fruits increase a woman’s ability to conceive (Ali et al., 2020). According to Hassan (2011), fruits help with nerve and memory disorders, infection-related fever and inflammation, paralysis, and loss of consciousness.
In addition to its fruit, the seeds of this plant, known as agricultural waste and/or byproducts, are also rich in bioactive compounds. In fact, date seeds are a rich source of antioxidants (Juhaimi et al., 2012). Several studies have reported that the high antioxidant activity is due to the high content of phenolic and flavonoid compounds and the high content of vitamin C in the seeds (Juhaimi et al., 2012). Also, the seeds may contain 31–44 g of gallic acid-equivalent phenolic compounds per kilogram. Moreover, date seeds contain significant amounts of polyphenols compared to many other seeds, like flaxseed, tea, the date fruit itself, and grapes (Habib et al., 2014). Interestingly, roasted seeds contain higher amounts of polyphenols than raw ones (Paranthaman et al., 2012).
The aim of this study is to determine the chemical composition of methanolic extracts obtained from Ajwa palm date seeds, followed by the identification of new bioactive compounds. Further, to investigate their antibacterial and antifungal potentials. We also aim to explore the antibacterial and antifungal activities of the methanolic extract using in silico molecular docking analysis by targeting the active sites of the TyrRS from Staphylococcus aureus (PDB ID: 1JIJ) and the aspartic proteinase from Candida albicans (PDB ID: 2QZW).
Materials and Methods
Plant Material Sampling and Extract Preparation
Ajwa palm date seeds were collected in 2021 from local date markets in Al-Madinah Munawara, Kingdom of Saudi Arabia. The specimen was recorded and deposited at the herbarium (Department of Biology, College of Science, University of Hail, Hail, Kingdom of Saudi Arabia), under a specimen voucher (APDS01) (Alrashidi et al., 2023). Briefly, 40 g of seeds were macerated in 400 mL of methanol for 48 h at room temperature, and then the mixture was extracted three more times following the same method. Amorphous powder was produced by filtering the resulting extract via a Millirock Technology device (Kingston, NY, USA) and lyophilizing water.
Phytochemical Profiling
The bioactive molecules from P. dactylifera seed methanolic extract were identified using a Shimadzu Nexis GC-2030 gas chromatograph system outfitted with a QP2020 NX mass spectrometer. In the constant flow mode, helium was used as a carrier gas at a rate of 1 mL/min. The column’s initial temperature was 70°C. The temperature was held at this level for 2 min before being steadily raised by 10°C–280°C. At an increased rate of 5°C/min, the oven temperature was raised to 280°C and kept there for 9 min. Helium was flowing at a rate of 1 mL/min, and the injection port temperature was 250°C. In addition, 70 eV served as the ionization voltage. A 30-m-long RTS volatile column achieved separation. Compounds were detected using a quadrupole mass spectrometer as they were expelled from the column. The detector had a temperature of 300°C. Compound identification was done by analyzing the spectrum using MS data libraries like WILEY8.LIB and NIST08 (Bouali et al., 2023; Snoussi et al., 2022a).
Antimicrobial Activity
The obtained methanolic extract was tested for its ability to act as an antimicrobial agent using the same protocol described by Haddaji et al. (2021). For the experiments, 12 microorganisms were selected, including eight bacteria (Escherichia coli ATCC 35218, Pseudomonas aeruginosa ATCC 27853, Enterobacter cloacae, Enterococcus faecium, Staphylococcus hominis, S. aureus, Staphylococcus epidermidis, and Klebsiella pneumoniae ATCC 27736) and four Candida species (C. albicans ATCC 10231, C. tropicalis ATCC 1362, C. utilis ATCC 9255, and C. guilliermondii ATCC 6260). For antibacterial activity, the CLSI document M02-A11 (CLSI, 2012) was used. For antifungal activity, CLSI document M44-A was followed (CLSI, 2004). The diameter of the growth inhibition zone measured on agar media was determined using the disk diffusion assay (Noumi et al., 2022).
The diameter of the growth inhibition zone was measured on agar media and determined using the disk diffusion assay. The minimal inhibitory concentrations (MICs) and the minimal bactericidal/fungicidal concentrations (MBC/MFC) were determined using the microdilution technique in the presence of extract concentrations ranging from 0.02 to 11.2 mg/mL. The calculated (MBC/MIC) and (MFC/MIC) ratio values were analyzed using the same scheme proposed by Gatsing et al. (2009).
In silico Analyses
The antimicrobial activity of the plant extract was supported by in silico molecular docking and analysis of the interactions with some targeted receptors. The active sites of the TyrRS from S. aureus (PDB ID: 1JIJ) and the aspartic proteinase from C. albicans (PDB ID: 2QZW) have been targeted to assess the antibacterial and antifungal effects, respectively. The 3D structure of the targeted proteins has been retrieved from the RCSB data bank. ChemDraw was used to draw the chemical structures of the identified compounds whenever they did not exist on the PubChem website. The docking parameters were set: deletion of water molecules, addition of polar hydrogens and Kollman charges and optimization for complex units: ligands and proteins. The binding sites have been defined as follows: center_x = –12.39315, center_y = 14.00218 and center_z = 86.50831 for 1JIJ and center_x = –15.01393, center_y = –21.10501, and center_z = –16.53591 for 2QZW. The analyses have been carried out using Autodock vina software packages following DS visualization. The binding affinity and the hydrogen bonds calculations were assessed as previously reported (Akacha et al., 2022; Badraoui et al., 2022). The major reason behind the selection of 1JIJ and 2QZW is their high implication in the pathogenesis of infectious diseases, particularly S. aureus and C. albicans (Hamrita et al., 2022; Othman et al., 2021). Furthermore, 1JIJ and 2QZW are commonly targeted in several combined in silico and in vitro antimicrobial studies (Alreshidi et al., 2023; Hamrita et al., 2022; Noumi et al., 2022).
Statistical Analysis
The mean and SD are used to express the data. Utilizing the GraphPad software package 8.3.1 (SPSS Inc., Chicago, IL, USA), the statistical analyses were performed. Both the Tukey’s post hoc test and the one-way analysis of variance (ANOVA) were carried out. With p < 0.05, the nominal statistical significance was considered.
Results and Discussion
Phytochemical Composition
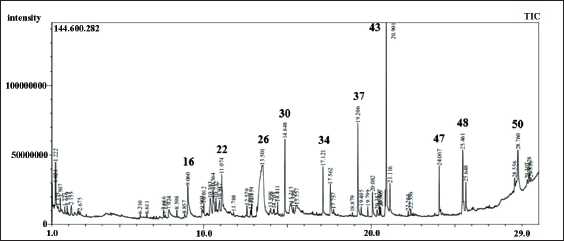
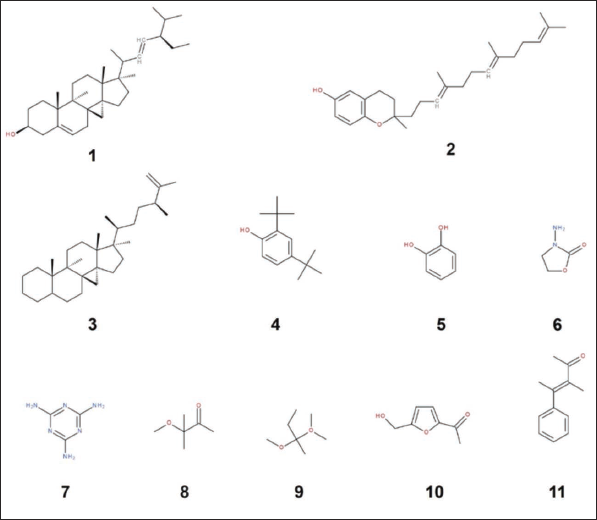
The quantitative chromatographic analysis of the methanolic extract of P. dactylifera seeds is presented in Table 1. The chromatogram of seed methanol extracts and the chemical structures of the most active identified compounds are also given in Figures 1 and 2, respectively. The data in Table 1 indicates that 51 compounds were identified, representing 75.07% of the extract. The remaining 23.36% of the extract consisted of two unidentified compounds (21.32% and 2.04%, respectively). The data also revealed that a significant part of the extract consists of monounsaturated fatty acids. The most important monounsaturated fatty acid found was 6-octadecenoic acid (Z) with 12.92%. Apparently, the extract also contained acids and phytochemicals from classes of acyclic alkanes, alcohols, aldehydes, alkanes, alkyl benzenes, azoles, aromatic benzene derivatives, carbohydrates, carboxylic acids, acyclic acetates, cresols, cyclic hydrocarbons, aromatic benzene derivatives, fatty acid derivatives, glycerides, heterocyclic compounds, ketones, lipids, phenols, phytosterols, terpenes, steroids, sugar acids, and vitamins were also detected.
Phytochemical Composition of the Methanolic Extract of P. dactylifera Using the GC-MS Technique.
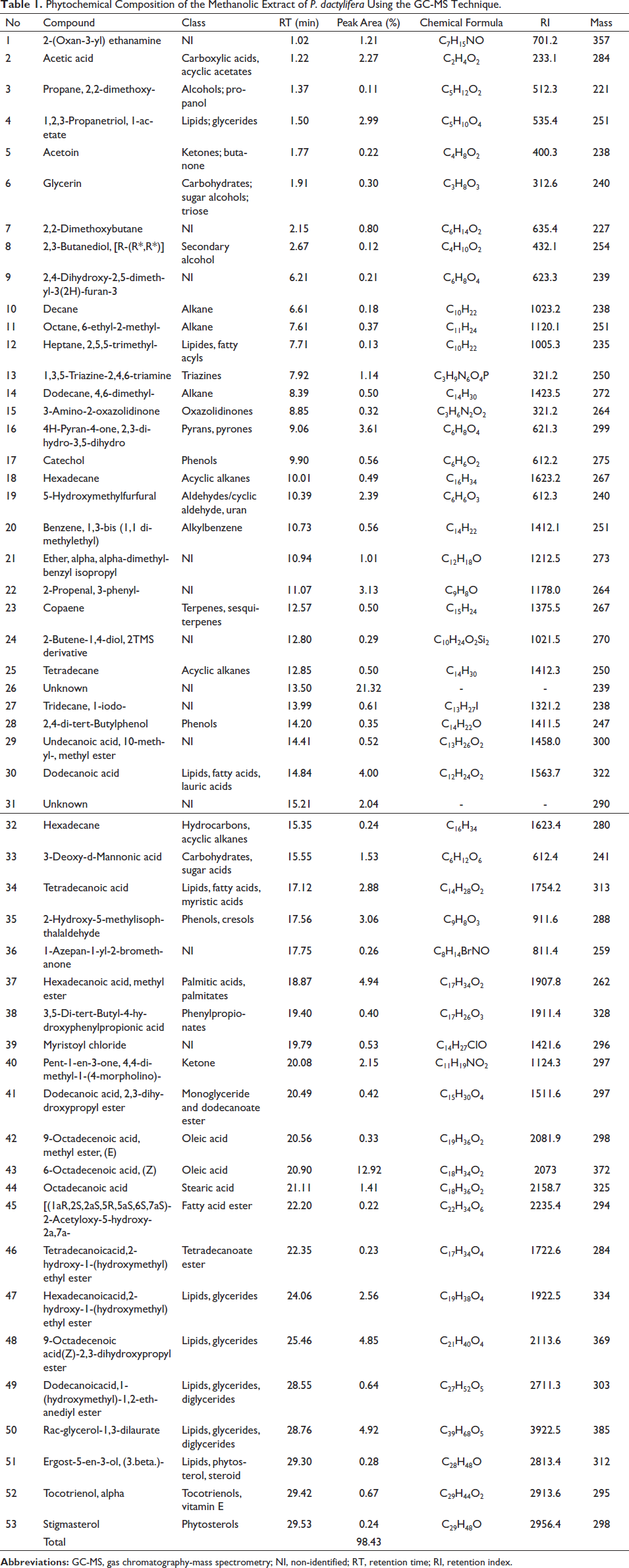
Nehdi et al. (2018) showed that the date seeds are predominantly composed of fatty acids such as oleic (48.67%), lauric (17.26%), stearic (10.74%), palmitic (9.88%), and linoleic acids (8.13%). Likewise, the seeds of P. dactylifera appear to contain plentiful of fatty acids (oleic, lauric, and myristic), triacylglycerols (1-myristoyl 2-oleoyl, 3-linoleoyl glycerol, 1-linolenoyl 2-oleoyl 3-linoleoyl glycerol, 1-2-linolenoyl 3-linoleoyl glycerol, and 1-linolenoyl 2-myristoyl 3-linoleoyl glycerol), bioactive phenols, catechin, vanillin, and tocopherols (Harkat et al., 2022). In parallel, Alkhoori et al. (2022) established that seeds are primarily a rich source of carbohydrates, fatty acids, dietary fibers, proteins, and bioactive phenolic compounds. Numerous similar reports in the literature confirm our current findings regarding the chemical composition of P. dactylifera seeds.
Antimicrobial Activity
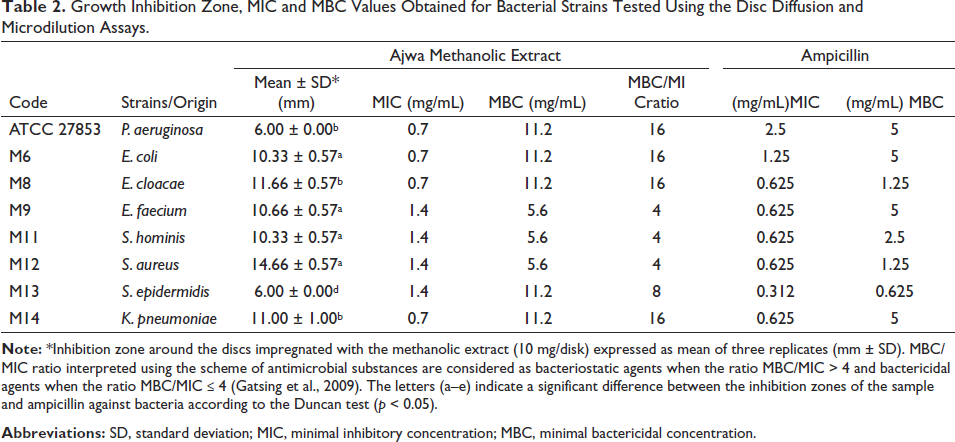
Growth inhibition of bacterial and fungal strains depicted by MIC, MBC, and MFC values is presented in Tables 2 and 3. Ampicillin and amphotericin B were used as positive controls in the antibacterial and antifungal assays, respectively. The data in Table 2 suggests that P. dactylifera methanolic extract exhibited the highest zone of inhibition against S. aureus (14.66 ± 0.57 mm). MIC and MBC values obtained against S. aureus in the presence of the extract were 1.4 and 5.6 mg/mL, respectively. In addition, the MBC/MIC ratio of the methanolic extract was determined to be 4. While the extract exhibited inhibition zones varying between 10.33 and 11.00 mm against E. coli, E. cloacae, E. faecium, S. hominis, and K. pneumonia. The most resistant strains among all the tested microorganisms were K. pneumoniae and S. epidermidis (6.00 mm). The results of the MIC and MBC tests were validated by disk diffusion analysis.
Growth Inhibition Zone, MIC and MBC Values Obtained for Bacterial Strains Tested Using the Disc Diffusion and Microdilution Assays.
Growth Inhibition Zone, MIC and MFC Values Obtained for Fungal Strains Tested Using the Disc Diffusion and Microdilution Assays.
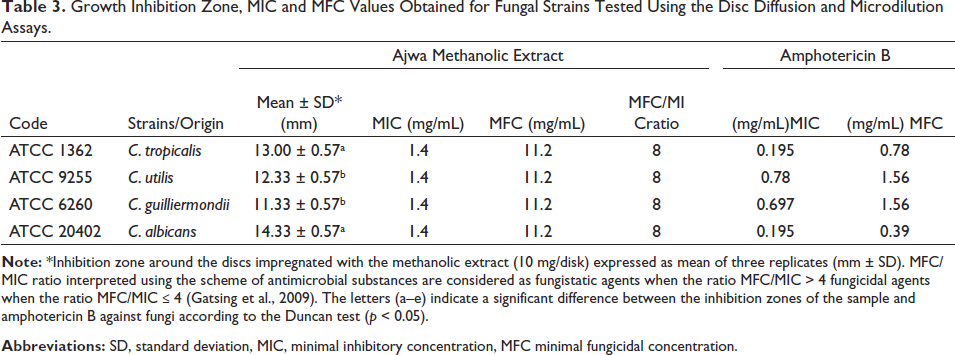
Table 3 denotes the antifungal data; it is implied that the extract exhibited the same MIC and MBC values against all the tested fungal strains (1.4 and 11.2 mg/mL, respectively). However, the results obtained from the disk diffusion test varied between 11.33 and 14.33 mm (albeit statistically not significantly different from each other). Considering the disk diffusion data, C. albicans was the most sensitive one to the extract among all the tested fungi (14.33 mm). It was followed by C. tropicalis, C. utilis, and C. guilliermondii, respectively.
In fact, the data suggested that the methanolic extract of the seeds of P. dactylifera demonstrated the least antimicrobial activity. Our current findings are supported and in line with the previous findings. For instance, (Al-Daihan & Bhat, 2012) compared the antimicrobial activity of dried leaf, fruit, seed, and tree bark of the plant and reported that the highest activity was exhibited by the fruit extract. Additionally, when the plant parts were ranked according to their antimicrobial activities, seemingly the seed extract exhibited the minimal activity. The likely explanation is that seeds contain lower amount of bioactive phenolic compounds compared to other plant parts. In another similar study, the antimicrobial effect of the extracts of P. dactylifera and Annona squamosa was compared; the P. dactylifera extract showed weak antimicrobial activity alone, but the activity level increased, when used together with A. squamosa extract (Aamir et al., 2013). Moreover, comparison of the antimicrobial activities of extracts of P. dactylifera seeds and pollen was carried out, the results implied that the pollen extract showed stronger activity than the seed extract against E. coli, S. aureus, S. aureus MRSA, and Enterococcus faecalis (Bentrad et al., 2017). Certainly, the course of the antimicrobial activity of the extracts obtained from P. dactylifera seeds are long established consistently (Abuelgassim et al., 2020; Radfar et al., 2019; Shakiba et al., 2011). Contrary to our findings, the use of the organic solvent acetone versus chloroform during the extraction from the seeds of P. dactylifera confirmed that the acetone extract contained a higher amount of aliphatic molecules, and accordingly, its antimicrobial and/or cytotoxic activity was higher (Ravi, 2017). Overall, the majority of the related studies carried out over a decade or so employing similar settings which supports our current findings.
In silico Analyses
Through molecular docking analysis, researchers can simulate interactions between molecules, predict the binding modes between molecules, and determine the affinity of these molecules for each other (Badraoui et al., 2022, Ben Saad et al., 2023). In this way, there is no need for real-time experiments for molecules subjected to docking analysis, research costs are significantly reduced, and the prediction capacity can be significantly increased by making some changes to the parameters (Fan et al., 2019; Jedli et al., 2022).
In the current study, molecular docking was performed to further validate our in vitro results. For this purpose, first of all, the binding energies and root mean square deviation values obtained from the interaction of all phytochemicals given in Table 1 with two different target receptors (1JIJ and 2QZW for TyrRS from S. aureus and aspartic proteinase from C. albicans) were determined (Table 4). Nine poses have been evaluated for each complex. The best poses have been reported in this study, together with their best interaction affinities. According to the data in the table, the phytochemicals with the highest binding affinity to target receptors were those 37, 50, 51, and 52. The binding energies of these compounds appear to range from −7.2 to −9.9 kcal/mol, which satisfactorily supports their biological activity, as previously reported (Akacha et al., 2022; Jedli et al., 2022).
Binding Energy and RMSD of the Identified Compounds and the Two Targeted Receptors: 1JIJ and 2QZW for TyrRS from S. aureus and Aspartic Proteinase from C. albicans, Respectively.
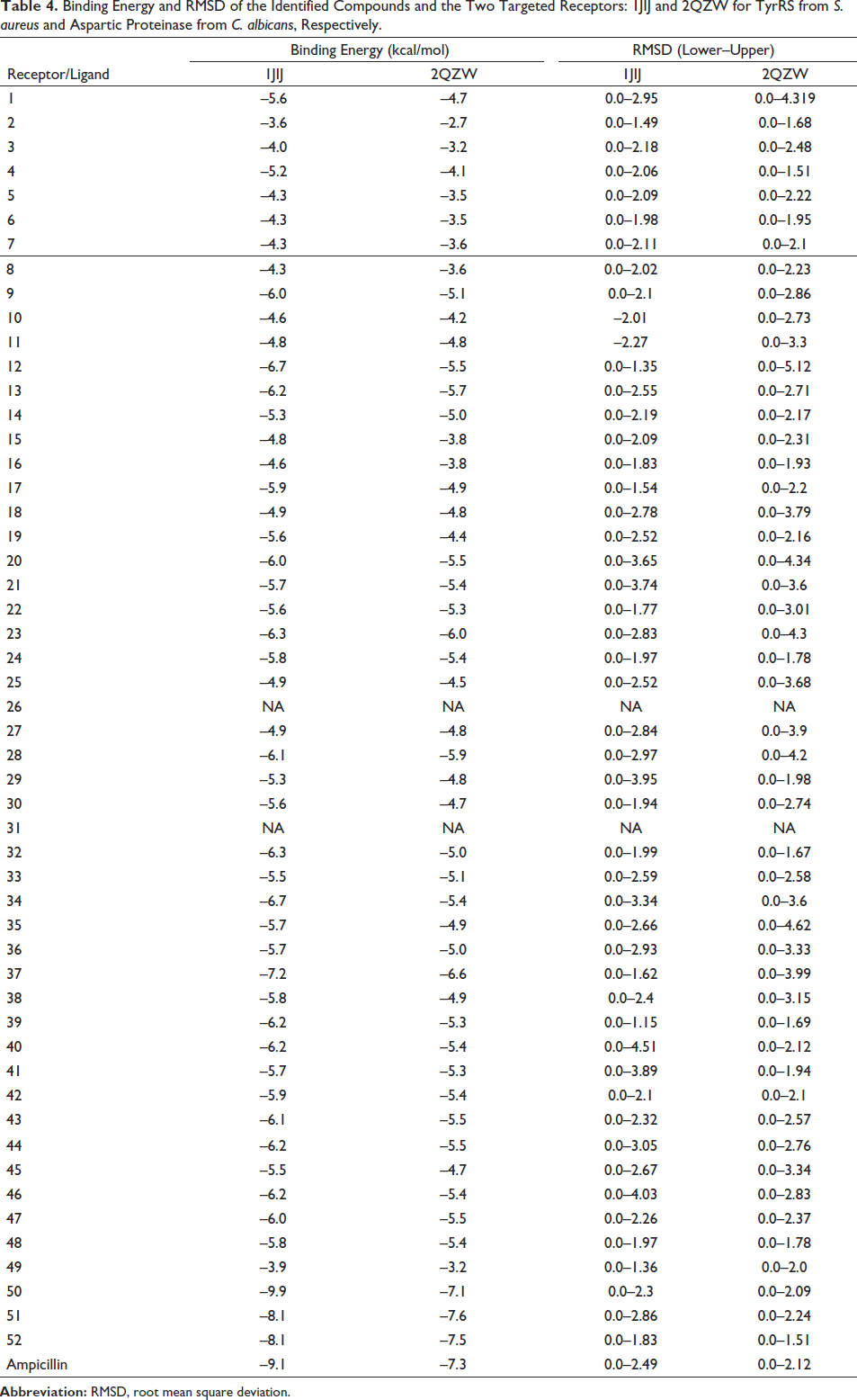
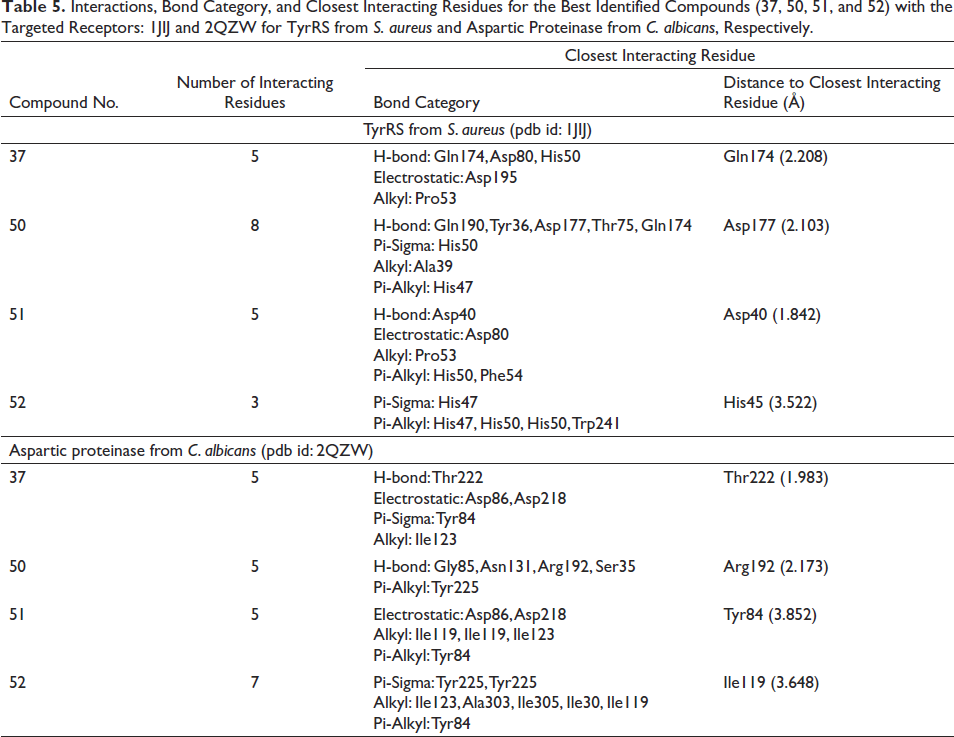
Based on the results given in Table 4, it was determined that the compounds with the highest binding energy (Compounds Nos. 37, 50, 51, and 52) interacted with each other, which was further explored to reveal the interacting amino acids in the targeted receptors and how far they are from the amino acid with which they interact most closely, the embedding distances (Table 5, Figures 3 and 4). According to the data in Table 5, compound 51 [ergost-5-en-3-ol, (3beta)-] established hydrogen, electrostatic, alkyl and pi-alkyl interactions with five different amino acid residues (Asp40, Asp80, Pro53, His50, and Phe54) located at the TyrRS receptor of S. aureus. It was determined that there is a distance of 1.842 Å between the phytochemical in question and Asp40, with which it interacts most closely. On the other hand, compound 37 (hexadecanoic acid, methyl ester) established hydrogen, electrostatic, pi-sigma, and alkyl interactions with five different amino acid residues (Thr222, Asp86, Asp218, Tyr84, and Ile123) located at the aspartic proteinase receptor of C. albicans. It was determined that there is a distance of 1.983 Å between the phytochemical in question and Thr222, with which it interacts most closely. Involvement of such amino acids was reported as a key role in the antimicrobial activities, specifically while interacting with electrostatic, attractive, or conventional H-bonds (Alreshidi et al., 2023; Hamrita et al., 2022). In fact, these bonds are commonly associated with pharmacotherapeutic mechanisms and efficacies (Badraoui et al., 2021; Hamadou et al., 2022).
Interactions, Bond Category, and Closest Interacting Residues for the Best Identified Compounds (37, 50, 51, and 52) with the Targeted Receptors: 1JIJ and 2QZW for TyrRS from S. aureus and Aspartic Proteinase from C. albicans, Respectively.
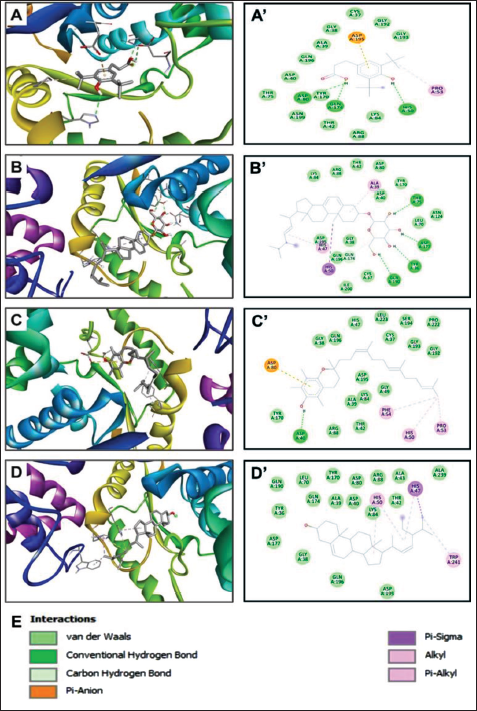
Three-dimensional Views of the Receptor–Ligand Complexes (A–D) and their Corresponding Two-dimensional Diagrams of Interactions (A’–D’) for the Plant Identified Compounds 37 (A and A’), 50 (B and B’), 51 (C and C’), and 52 (D and D’), which have the Best Binding Scores (−7.2, −9.9, −8.1, and −8.1 kcal/mol, respectively). The Compounds are Docked to the Pocket Region of TyrRS from S. aureus (1JIJ).
Three-dimensional Views of the Receptor–Ligand Complexes (A–D) and their Corresponding Two-dimensional Diagrams of Interactions (A’–D’) for the Plant Identified Compounds 37 (A and A’), 50 (B and B’), 51 (C and C’), and 52 (D and D’), which have the Best Binding Scores (−6.6, −7.1, −7.6, and −7.5 kcal/mol, respectively). The Compounds are Docked to the Pocket Region of Aspartic Proteinase from C. albicans (2QZW).
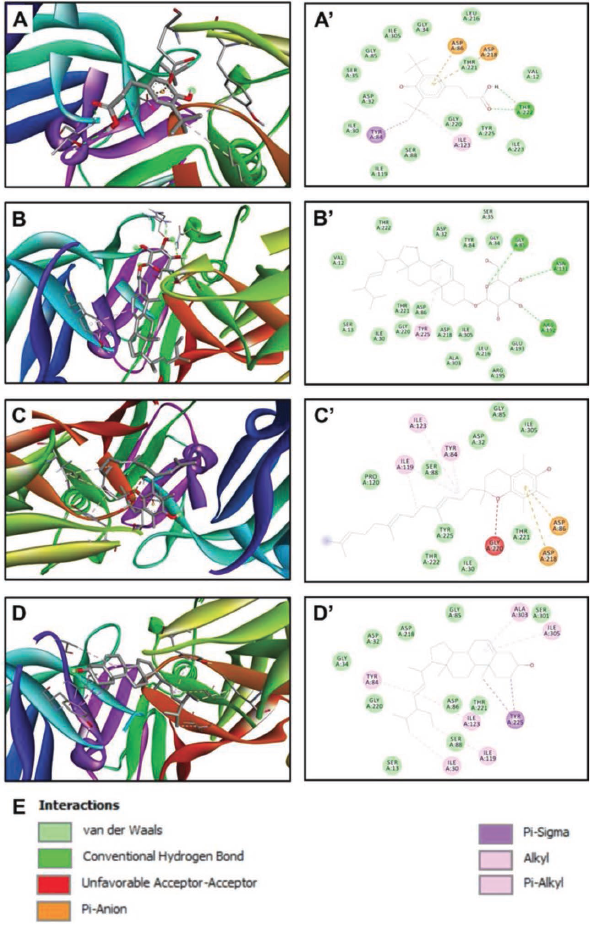
In the literature, there are some studies in which the interaction of some plant species with the target receptors analyzed in the current study was investigated through in silico analyses (Alreshidi et al., 2023; Gholam & Firdausy, 2022; Hamadou et al., 2022; Hamrita et al., 2022; Meenambiga et al., 2018; Noumi et al., 2022; Snoussi et al., 2022b). However, no study was found on the interaction of the plant sample examined in the current study with these targeted receptors. Therefore, the data presented in this part of the study is considered to be an original and important contribution to the literature.
Conclusion
Altogether, the methanolic extract from date palm seeds exhibited moderate antimicrobial activity. Considering the binding energies and the affinities of the compounds to the target residues, disk diffusion, MIC/MBC tests, and the results of in silico analysis support each other. We performed GC-MS analysis to investigate the chemical composition of the methanolic extract of date palm seeds, a well-known agricultural waste. This method was chosen based on the available technical and logistical possibilities. Moreover, the nature of this method could possibly alter the composition of the phenolic compounds in the extract since only volatile components could be detected. Presumably, not only non-polar compounds (e.g., fatty acids) contribute significantly to the biological activity; the polar phytochemicals (e.g., phenolic compounds) also exhibit robust biological activity potentials. Following up, we suggest to perform HPLC or LC-MS quantitative analysis to detect phenolic compounds that possess potential antimicrobial activity in the extract.
Footnotes
Abbreviations
MIC: Minimal inhibitory concentration; MBC: Minimal bactericidal concentration; MFC: Minimal fungicidal concentration; RMSD: Root mean square deviation; ANOVA: Analysis of variance; GC-MS: Gas chromatography-mass spectrometry; RT: Retention time; RI: Retention index.
Declaration of Conflict of Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding Sources
This research has been funded by Scientific Research Deanship at University of Ha’il - Saudi Arabia through project number RG-21 123.
