Abstract
Keywords
Introduction
Zingiber monophyllum Gagnep., belongs to the Zingiber genus (Zingiberaceae family), is a native plant from Vietnam and Laos. 1 Z. monophyllum is commonly found in Vietnam in several provinces, such as Thanh Hoa, Thua Thien-Hue, and Ha Nam. 2 The antibacterial activity of essential oils from the rhizomes and leaves of Z. monophyllum has been reported previously. 2 Additionally, some compounds, including β-pinene, borneol, α-pinene, and myrtenal found in the rhizomes and leaves of Z. monophyllum were proven to exhibit potent cytotoxicity against cancer cells (eg, breast cancer and leukemia), as well as anti-tumor, anti-inflammatory, antioxidant, antibacterial, antifungal, antiviral, bronchodilatory, antiaggregative and antihemolytic, neuroprotective, and gastroprotective activities.3–5
In previous studies, the extracts from the rhizomes of Zingiber species also had antimicrobial properties. For example, several strains (eg, Bacillus cereus, Pseudomonas aeruginosa, etc) proved to be highly susceptible to the activity of the ethyl acetate extract of Z. officinalis rhizomes. 6 Similarly, the ethyl acetate extract from Z. neesanum rhizomes showed potential microbicidal effects. 7 Other extracts examined in previous reports also revealed interesting antibacterial activities. 8 However, there has been little information on the antimicrobial activity of the extracts from the rhizomes of Z. monophyllum, indicating the need for further study to diversify the source of promising antibacterial materials.
Previous studies have reported that antioxidant compounds have been widely used in medicine to treat various diseases and illnesses. They have the ability to interrupt, delay, or prevent oxidation. 9 As a result, antioxidants can protect the body by preventing aging, cell damage, and oxidative stress-related diseases. 10 Research on antioxidant compositions from plant-based extracts has received great interest for their potential as herbal drugs. Furthermore, antioxidants also exhibit various pharmacological effects including antibacterial, antiviral, anti-inflammatory, antiaging, and antitumor activities. 11 Because of these beneficial properties of antioxidants, the general aim of many studies has been carried out to find new natural sources of antioxidant properties. Up to now, the information about the phytochemical components and biological activities of the extract from Z. monophyllum rhizomes is still limited. In this work, we reported the volatile compositions of the EtOAc fractionated extract from Z. monophyllum rhizomes, in vitro antioxidant, antimicrobial activities combined with the density functional theory (DFT) approach, and molecular docking simulation.
Results and Discussion
Chemical Composition of the EtOAc Fractionated Extract
From the EtOAc fractionated extract of Z. monophyllum rhizomes, nine components were identified by gas chromatography-mass spectrometry (GC-MS) analysis (Supplemental Figure S1-S10). These compounds belong to three terpenoid groups, including guaiane (

Chemical structures of nine main compounds identified from the EtOAc fractionated extract of Z. monophyllum rhizomes by GC-MS analysis.
Major Compounds Detected from the EtOAc Fractionated Extract of Z. monophyllum Rhizomes.
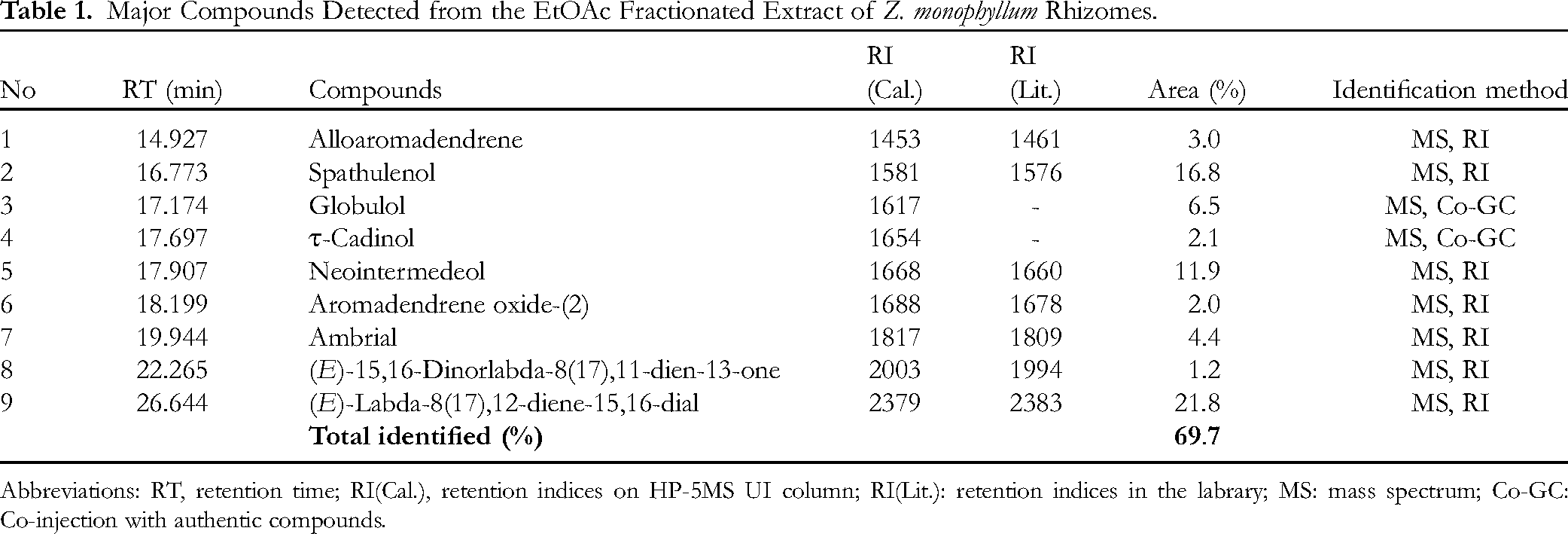
Abbreviations: RT, retention time; RI(Cal.), retention indices on HP-5MS UI column; RI(Lit.): retention indices in the labrary; MS: mass spectrum; Co-GC: Co-injection with authentic compounds.
Antioxidant Activity
The EtOAc fractionated extract of Z. monophyllum rhizomes was evaluated for its antioxidant activities via 2,2-diphenyl-1-picrylhydrazyl (DPPH) radical scavenging and ferric reducing antioxidant power (FRAP) assays (Supplemental, Figures S11 and S12). As a result, the EtOAc fractionated extract showed moderate antioxidant effects, with an IC50 value of 1.862 ± 0.730 mg/mL in the DPPH assay using ascorbic acid as the reference drug (IC50 value of 0.015 ± 0.000 mg/mL) and an EC50 value of 1.519 mg/mL in the FRAP assay using ascorbic acid as the standard compound (IC50 value of 0.070 ± 0.000 mg/mL). Comparing to other ginger extracts, the EtOAc fractionated extract of Z. monophyllum rhizomes showed relatively smaller activities. Specifically, the high-pressurized CO2 extract of ginger roots from Vietnam showed an IC50 of 0.64 µg/mL against DPPH free radicals. 14 In another study, the n-hexane and methanol extracts of ginger displayed the highest scavenging percentage of 82% to 88% against DPPH free radicals. 15 The smaller activities of the EtOAc fractionated extract of Z. monophyllum rhizomes than other ginger extracts could be explained based on the difference in extracted solvent and species. So far, there has been limited information on the antioxidant activity of Z. monophyllum rhizomes in the literature.
Antimicrobial Activity
The antimicrobial activity of the EtOAc fractionated extract of Z. monophyllum rhizomes was evaluated against pathogenic microorganisms, and the resulting IC50 values are presented in Table 2. This extract exhibited notable inhibitory activity against all examined microbial strains, except for P. aeruginosa. Notably, the EtOAc fractionated extract of Z. monophyllum rhizomes demonstrated significantly higher antibacterial and anti-yeast activities when compared to the 2 drugs, streptomycin and cycloheximide, with IC50 values ranging from 4.89 to 5.34 µg/mL. Specifically, within the context of the antimicrobial assay, the EtOAc fractionated extract exhibited the most potent inhibition against Staphylococcus aureus (IC50 = 4.89 µg/mL), followed by B. cereus (IC50 = 4.97 µg/mL) and C. albicans (IC50 = 4.98 µg/mL). Regarding Gram-negative bacteria, E. coli and Salmonella enterica displayed similar sensitivity to the fractionated extract, with IC50 values of 5.23 and 5.18 µg/mL, respectively. However, no antibacterial activity against P. aeruginosa was observed. Kader et al 16 found that the crude ethanol extract of Z. zerumbet rhizomes and its fractions exhibited mild to moderate antimicrobial activity against 2 Gram-positive and 4 Gram-negative bacteria with MIC values of 128 to 256 µg/mL. In another study, various extracts of Z. officinale var. officinale and Z. officinale var. rubrum showed weak antimicrobial activity. 17 These findings may demonstrate P. aeruginosa's relatively insensitive to the extract among the tested organisms, suggesting that Gram-positive bacteria tend to be more susceptible to the EtOAc fractionated extract than their Gram-negative counterparts.
Antimicrobial Activity of the EtOAc Fractionated Extract of Z. monophyllum Rhizomes.

Note: Mean ± SD, n = 3
Previous studies have reported a range of biological effects associated with the EtOAc fraction of Stachys schtschegleevii leaves and stems, including antibacterial, antifungal, and antioxidant activities, among others. Therefore, several EtOAc fractions of various plant sources have been identified as potential reservoirs of antimicrobial agents. For example, the EtOAc fraction obtained from Ruta officinalis flowers was demonstrated to exhibit in vitro antibacterial, antioxidant, anti-inflammatory, and analgesic activities, likely attributed to their rich polyphenolic content. 18 In the present study, the EtOAc fractionated extract was found to contain a substantial quantity of bioactive compounds, including spathulenol (16.8%), which has previously been reported to possess antimicrobial effects against Mycobacterium tuberculosis. Additionally, neointermedeol (11.9%), a compound also found in Artemisia argyi essential oil, exhibited antibacterial effects against Listeria monocytogenes, E. coli, Proteus vulgaris, Salmonella enteritidis, and Aspergillus niger. 19 Consequently, these results are consistent with the robust antibacterial activities observed in the EtOAc fractionated extract derived from Z. monophyllum rhizomes in this study. Given these findings, further comprehensive investigations of Z. monophyllum are warranted to illuminate and substantiate its potential applications in the pharmaceutical field.
DFT-based Optimized Structures and Quantum Chemical Properties
The DFT analysis was performed on the identified compounds in the EtOAc fractionated extract of Z. monophyllum rhizomes. These chemical constituents are responsible for the antioxidant potential of Z. monophyllum's rhizomes. Through DFT, various characteristics were determined, including the energies of frontier orbitals (highest occupied molecular orbital [HOMO] and lowest unoccupied molecular orbital [LUMO]), as well as several reactivity parameters such as dipole moment, electron affinity, ionization energy, hardness, softness, electronegativity, chemical potential, electrophilicity, electron-accepting power, and electron-donor power, which are significant tools to describe the reactivity, stability, and binding capacity of molecules. 20 The M052X/def2-TZVPP method was employed for these calculations (Supplemental, Table S1 and Figure S13).
Pi-electron delocalization is a crucial factor in the stabilization of molecules and the reactivity of donor-acceptor sites, as per theoretical understanding. The electron transfer characteristics of the HOMO indicate the tendency for intermolecular electron donation, whereas the LUMO reflects electron-accepting capability. A greater EHOMO (corresponding to a lower ionization potential, Io) and a lower ELUMO (corresponding to a higher electron affinity, A) contribute to enhanced electron-donating ability and increased sensitivity to receiving electrons, respectively. Meanwhile, lower gap energy, indicating easier electron transfer, signifies improved antioxidant reactivity. Figure 2 illustrates that the HOMO and LUMO profiles of several molecules, such as

Egap = ELUMO−EHOMO and dipole moment of the structures 1–9. Abbreviations: LUMO, lowest unoccupied molecular orbital; HOMO, highest occupied molecular orbital.
Upon analyzing the detailed parameters, all structures (
Molecular Docking
Docking studies have been conducted in an endeavor to predict the antimicrobial mechanisms of action of test molecules. Enterococcus faecalis DNA gyrase B (PDB ID: 4GEE), E. coli DNA gyrase B (PDB ID: 4DUH), S. aureus biotin protein ligase (PDB ID: 3V7R), E. faecalis Alanine racemase (PDB ID: 3E6E), and C. albicans N-myristoyltransferase (PDB ID: 1IYK) have been selected as potential target proteins for antimicrobial activity. Based on available information in the RCSB Protein Data Bank, proteins 4GEE and 4DUH are essential enzymes that regulate the structural state of DNA binding during replication, while the binding strategy of the biotin ligase enzyme of S aureus as depicted by protein 3V7R suggests a potential avenue for selective inhibition.22–24 As a constituent of the Alanine racemase family, 3E6E serves as a prevalent bacterial enzyme facilitating the interconversion between ʟ- and ᴅ-alanine, critical precursors for cell wall peptidoglycan synthesis. 25 Myristoyl-CoA:protein N-myristoyltransferase (PDB ID: 1IYK) emerges as a promising target for antifungal drugs, recognized as a monomeric enzyme catalyzing the transfer of myristate from myristoyl-CoA to the N-terminal glycine residue of various viral and eukaryotic proteins. 26
Verification prior to screening is executed to ensure the reliability and precision of the AutoDock Vina v1.2.3 program. Consequently, validation is established through the redocking of the inhibitory compound in the complex with the crystal structure of E coli DNA gyrase B, yielding a reasonable low root-mean-square deviation (RMSD) value (< 2 Å) of 0.461546 Å. 27 The co-crystallized structures of 4-[[4′-methyl-2′-(propanoylamino)-4,5′-bi-1,3-thiazol-2-yl]amino]benzoic are depicted in Figure S14 (Supplemental information). In this study, molecular docking analysis has been conducted to assess the interactions and binding affinities of the main compounds of the EtOAc fractionated extract from Z. monophyllum rhizomes against target proteins. Table 3 demonstrates binding energies, and Figures S15 to S19 (Supplemental information) depict the amino acid residues within the receptor with which the ligands interact.
The Binding Affinity of Main Compounds in the EtOAc Fractionated Extract with Enterococcus faecalis DNA gyrase B, Escherichia coli DNA gyrase B, Staphylococcus aureus Biotin Protein Ligase, E. faecalis Alanine Racemase, and C. albicans N-myristoyltransferase.

As evident from Table 3, binding affinities range from −5.772 to −7.617 kcal/mol for the 4GEE protein, −4.511 to −6.85 kcal/mol for protein 4DUH, −5.04 to −7.293 kcal/mol for protein 3V7R, −6.024 to −8.194 kcal/mol for protein 3E6E, and −6.03 to −7.67 kcal/mol for protein 1IYK. Intriguingly, compound
Conclusions
The current research marks the first concentrated effort to establish the volatile compositions of the EtOAc fractionated extract of Z. monophyllum through the GC-MS analysis. The research also demonstrated the antioxidant and antibacterial potential of the EtOAc fractionated extract of Z. monophyllum rhizomes. Nine compounds along with their relative content were identified, including alloaromadendrene, spathulenol, globulol, τ-cadinol, neointermedeol, aromadendrene oxide-(2), ambrial, (E)-15,16-dinorlabda-8(17),11-dien-13-one, and (E)-lambda-8 (17),12-diene-15,16-dial. The antioxidant screening revealed that the extract showed moderate antioxidant activity and this was supported by the DFT study. Furthermore, the extract exhibited remarkable antimicrobial activity except for P. aeruginosa. Molecular docking studies were also performed to display binding affinities of identified compounds with selected antibacterial target proteins. The isolation of these bioactive compounds and more in-depth studies to analyze their biological activities and conduct clinical trials for exploring and developing novel drug formulations will be carried out and reported in the future.
Materials and Methods
Chemicals and Reagents
Ascorbic acid (99%), Folin–Ciocalteu, 2,2-di(4-tert-octylphenyl)-1-picrylhydrazyl (DPPH), 24,6-Tris(2-pyridyl)-s-triazine, dimethyl sulfoxide (DMSO), n-hexane, ethyl acetate, methanol, streptomycin and cycloheximide were purchased from Sigma-Aldrich. All microorganisms used in this work were purchased from the National Institute for Food Control (Hanoi, Vietnam). All other chemicals were of the highest grade available.
Plant Material
Rhizomes of Z. monophyllum were collected from Kon Ka Kinh National Park, Gia Lai Province, Vietnam in December 2022. Identification of the plant material was performed by Assoc. Prof. Dr Nguyen Hoang Tuan (Faculty of Pharmacognosy and Traditional Medicine, Hanoi University of Pharmacy, Vietnam). A voucher specimen (No. G1LE) was deposited in the Herbarium of the Department of Chemistry, Vinh University, Vinh City, Nghean, Vietnam.
Preparation of Ethyl Acetate Fractionated Extract
Dried rhizomes of Z. monophyllum (4.5 kg) were crushed into coarse powder, and extracted with methanol (5 times, 6.0 L each) at room temperature. The combined methanol extracts were concentrated under reduced pressure to give a residue that was suspended in water and partitioned successively with hexane and ethyl acetate (EtOAc). The crude extracts were stored in a refrigerator (4 °C) to future analyses.
GC-MS Analysis
The GC-MS analysis was conducted with an Agilent 7890B GC coupled with an Agilent 7890B GC system, equipped with HP-5MS UI column (30 m × 0.25 mm i.d. × 0.25 μm film thickness). Helium was the carrier gas, at a flow rate of 1.5 mL/min. A volume of 1.0 μL of diluted samples (1000 ppm) was injected with a split ratio was 10:1. The oven temperature rose from 80 °C (1 min kept) up to 300 °C at the rate of 20 °C/min, then kept constant at 300 °C for 15 min. Mass spectra were recorded at 70 eV. The mass range was from 50 to 550 m/z (2.0 scan/s). The chemical components of the EtOAc fractionated extract were identified by comparison of their MS fragmentation patterns, retention indices with those in the literature (NIST17) and by co-injection with authentic compounds. The percentage of each component was calculated by comparing the average area of its peak to the total area of all the peaks.28,29
In Vitro Antioxidant Activity
The antioxidant activity of the EtOAc fractionated extract was determined using the DPPH method,
30
with ascorbic acid used as a positive control. The samples (0.1 mL) or negative control (ionized water) were mixed with the 3 mM DPPH solution (0.1 mL) and incubated for 30 min. The absorbance of the mixture was measured at 517 nm. The DPPH scavenging activity (%) was calculated using the following equation: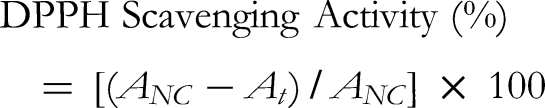
The ferric-reducing power was determined using the FRAP method in previous studies.31,32 The working FRAP (240 mL) solution was mixed with the fractionated extract (10 mL) and incubated for 15 min. The absorbance was then spectroscopically measured at 593 nm. The reducing power was expressed as an absorbance. The concentration of fractionated extract with an absorbance of 0.5 is known as EC50.
In Vitro Antimicrobial Activity
E. coli ATCC 25922, P. aeruginosa ATCC 27853, S. enterica ATCC 13076, E. faecalis ATCC 299212, S. aureus ATCC 25923, B. cereus ATCC 14579, and C. albicans ATCC 10231 were purchased from the National Institute for Food Control (Hanoi, Vietnam). To assess the antibacterial and antifungal properties of the EtOAc fractionated extract of Z. monophyllum rhizomes, we followed the methodology outlined by Hadacek and Greger. 33
Stock solutions of the EtOAc fractionated extract were prepared in 1% DMSO. In brief, the bacterial strains and yeast were cultured to achieve an approximate concentration of 2 × 105 CFU/mL. Subsequently, 50 µL of the bacterial or yeast culture was introduced into Luria-Bertani medium supplemented with various concentrations of the EtOAc fractionated extract, along with DMSO as a control. The mixtures were then incubated at 37 °C for a duration of 24 h. After the 24 h incubation period, we measured the optical density of the culture wells using a spectrophotometer (BioTeK Instruments, Inc., Highland Park) equipped with Rawdata software. The antimicrobial properties of the EtOAc fractionated extract were quantified in terms of Half-Maximal Inhibitory Concentration (IC50), which represents the concentration of the EtOAc fractionated extract required to reduce cell growth by half after 24 h of incubation. As a positive control, bacterial and yeast cells were subjected to exposure to streptomycin and cycloheximide, respectively. All experimental procedures were performed in triplicate.
Statistical Analysis
All the experiments were conducted in triplicate. The results were represented as mean ± standard deviation. The statistical comparison was performed using F-test 2-sample (Microsoft Excel, Microsoft, 2018). P values less than .5 were considered significant.
Quantum Chemical Calculation
Using theoretical chemistry simulations, the bioactivity of compounds was connected with their molecular electronic structures in order to reveal probable interaction pathways with active sites in biological molecules.
34
The chosen compounds were subjected to geometric optimization using the Gaussian 09 software in the gas phase.
35
The frozen-core approximation for nonvalence-shell electrons with a greater basis set def2-TZVPP obtained single-point energies at M052X/6-311++G(d,p)-level optimized geometries. Frontier orbital analysis was carried out using NBO 5.1 at the level of theory M052X/def2-TZVPP.
36
Following that, the conformational analysis of the HOMO, LUMO, and molecular electrostatic potential surface were used to assess the local reactivity. The following relationships were used to determine molecular characteristics, including the HOMO energy (EHOMO), LUMO energy (ELUMO), band gap energy (Egap), electron chemical potential (μ), softness (σ), hardness (η), electrophilicity (ω), and electronegativity (χ).37,38






Molecular Docking Simulation
To identify the manner of contact and binding energy via ligand-enzyme interaction that supports the biological activity (antimicrobial activity) of the main compounds in the EtOAc fractionated extract, a molecular docking study was employed. The PDB format files of E. faecalis DNA gyrase B, E. coli DNA gyrase B, S. aureus biotin protein ligase, E. faecalis Alanine racemase, and C. albicans N-myristoyltransferase target proteins were downloaded from the RCSB protein data bank (https://www.rcsb.org/), with PDB entries including 4GEE, 4DUH, 3V7R, 3E6E, and 1IYK, respectively.20–24 The target proteins were then thoroughly prepared for established docking by adding missing hydrogen, Kollman charge, and energy minimization using the software AutoDockTools and Swiss-PdbViewer. 39 The generic docking technique was used in accordance with the default parameters, and the AutoDock Vina 1.2.3 program carried out the docking operation for the chosen proteins and compounds.40–42 The docked pose with the highest binding affinity and most pronounced amino acid residue interactions for each docked ligand was chosen and visualized by the Discovery Studio Visualizer software.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241253443 - Supplemental material for Chemical Constituents, Antioxidant, and Antimicrobial Activities of Ethyl Acetate Fractionated Extract from Rhizomes of Zingiber monophyllum Gagnep.: In vitro and in silico Screenings
Supplemental material, sj-docx-1-npx-10.1177_1934578X241253443 for Chemical Constituents, Antioxidant, and Antimicrobial Activities of Ethyl Acetate Fractionated Extract from Rhizomes of Zingiber monophyllum Gagnep.: In vitro and in silico Screenings by Le D. Giang, Hieu Tran-Trung, Phan T. Thuy, Nguyen Thi Giang An, Hieu Nguyen-Ngoc, Trang H.D. Nguyen, Dang K. Nguyen, Anh V. Nguyen, Tran V. Chen, Nguyen X. Ha and Dau X. Duc in Natural Product Communications
Supplemental Material
sj-docx-2-npx-10.1177_1934578X241253443 - Supplemental material for Chemical Constituents, Antioxidant, and Antimicrobial Activities of Ethyl Acetate Fractionated Extract from Rhizomes of Zingiber monophyllum Gagnep.: In vitro and in silico Screenings
Supplemental material, sj-docx-2-npx-10.1177_1934578X241253443 for Chemical Constituents, Antioxidant, and Antimicrobial Activities of Ethyl Acetate Fractionated Extract from Rhizomes of Zingiber monophyllum Gagnep.: In vitro and in silico Screenings by Le D. Giang, Hieu Tran-Trung, Phan T. Thuy, Nguyen Thi Giang An, Hieu Nguyen-Ngoc, Trang H.D. Nguyen, Dang K. Nguyen, Anh V. Nguyen, Tran V. Chen, Nguyen X. Ha and Dau X. Duc in Natural Product Communications
Footnotes
Acknowledgments
The authors are grateful to Assoc. Prof. Dr Nguyen Hoang Tuan (Faculty of Pharmacognosy and Traditional Medicine, Hanoi University of Pharmacy) for assisting in the identification of the plant material.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
