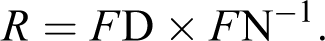Abstract
In a previous study, we have found that organophosphate (OP) pesticides such as chlorpyrifos (CPF), methyl parathion (MPT), and malathion (MLT) significantly induced genotoxicity in peripheral blood lymphocytes of rats. To explore the mechanism of OP-induced genotoxicity, we measured the formation of DNA interstrand cross-links (DICs) and apoptosis in peripheral blood lymphocytes of rats. Peripheral blood lymphocytes of rats were treated with CPF, MPT, and MLT individually and in combination at concentrations of 0.1 and 0.25 LC50 for 2, 4, 8, and 12 h at 37°C. Lipid peroxidation (LPO) was measured as a biomarker of oxidative stress. Apoptosis induced by CPF, MPT, and MLT individually and in combination was determined by measuring the intracellular level of active caspase-3 and caspase-9 by spectrofluorimetry. We found significant dose- and time-dependent increases in LPO, DICs formation and increase of intracellular active caspase-3 and caspase-9 in exposed peripheral blood lymphocytes of rats. These findings suggest that the studied pesticides have potential to induce oxidative stress, cause DNA adduct formation, and cause failure of adduct repair, which leads to apoptosis that is partially mediated by activation of intracellular caspase-3 and caspase-9.
Introduction
Increased crop production demands increased use of pesticides. 1 Organophosphate (OP) compounds are extensively used as pesticides. They exert their acute toxic effects to pests by phosphorylation of serine residues in the active center of acetylcholinesterase, which leads to accumulation of acetylcholine. 2 Humans are exposed to the ubiquitous pesticides in the houses, workplace, hospitals, schools, and during manufacturing. 3 The extensive use of pesticides exposes the community to both long-term and acute occupational health problems such as mutation, 4 cancer, and birth defects. 5,6
In a previous study, we have found that chlorpyrifos (CPF), methyl parathion (MPT), and malathion (MLT) OP pesticides caused significant increase in DNA–protein cross-links formation and DNA damage in peripheral blood lymphocytes of rats. 7,8 DNA adducts are responsible for inhibition of replication and transcription and thus directly or indirectly involved in generation of several diseases. DNA adduct formation is used as a biomarker for oxidative stress and genotoxicity studies. 9 Pesticides can produce covalent adducts with DNA bases on both strands of DNA, ultimately leading to the formation of interstrand cross-links (ICLs). 10 DNA interstrand cross-links (DICs) covalently connect the Watson and Crick strands of DNA and thereby block DNA replication, transcription, and any other processes requiring strand separation. As a result, ICLs are extremely damaging, particularly to dividing cells. If left intact, ICLs can lead to cell death. It has been estimated that as few as 20 ICLs in the bacterial or mammalian genome can be lethal to cells that lack the ability to remove the cross-links. 11
It has been reported that OP pesticides induce chromosomal aberration, sister chromatid exchange, and micronucleus frequency in humans (occupationally or accidentally exposed to OP pesticide) peripheral blood lymphocytes and rat bone marrow cells. 12,13 It is also reported that OP pesticides induce genetic damage in erythrocytes of amphibian tadpoles. 14,15 There are only a few reports on acute exposure. Exposure to the OP pesticide demonstrated to cause apoptosis both in vitro and in vivo. 16 –18 It is reported that low dose exposure to CPF, paraoxon (metabolite of parathion), and malaoxon (metabolite of MLT) leads dopaminergic neurons to undergo apoptosis, or programmed cell death. 19,20 MLT induces oxidative stress, cytotoxicity, and genotoxicity in human liver carcinoma (HepG2) cells. 21 It has been reported that the metabolism of MLT produces reactive oxygen species (ROS) that lead to the onset of oxidative stress. 22 Recently, it is demonstrated that another OP pesticides diazinon have genotoxic and cytotoxic potential on human lymphocytes. 23 Chronic exposure to OP pesticides induced neurodegenerative diseases such as Alzheimer’s disease and Parkinson’s disease. 24,25 OP pesticides are capable to induce apoptosis (programmed cell death) by multifunctional pathways. Caspases are a family of genes important for maintaining homeostasis through regulating cell death and inflammation. Although caspase-mediated processing can result in substrate inactivation, it may also generate active signaling molecules that participate in ordered processes such as apoptosis and inflammation. Proapoptotic signaling activates initiator caspases, such as caspase-2, -8, and -9. Caspase-3 is one of the key executioners of apoptosis, being responsible either partially or totally for the proteolytic cleavage of many structural and regulatory proteins. 26 However, the molecular pathways leading to OP pesticides–induced apoptosis have not been established.
Pesticide exposure can be evaluated with respect to a single compound or to complex mixtures. The mixtures of pesticides may influence and even amplify the toxicity of individual components through synergies, potentiating, antagonism, inhibition or additive effects. 27 In continuation of our previous study, the present study shed some light on the molecular mechanisms responsible for apoptosis initiated by CPF, MPT, and MLT, individually and in combination, in peripheral blood lymphocytes of rats. To identify the apoptotic pathways, peripheral blood lymphocytes of rats were exposed to 0.1 and 0.25 LC50 concentrations of CPF, MPT, and MLT individually and in combination for 2, 4, 8, and 12 h. After exposure, lipid peroxidation (LPO), DICs, and activity of caspases were measured. LPO was measured as a biomarker of oxidative stress.
Material and methods
Chemicals
Ethylenediaminetetraacetic acid (EDTA), Tris hydrochloride, trypan blue, dimethyl sulfoxide (DMSO), formaldehyde (HCHO), thiobarbituric acid (TBA), trichloroaceticacid (TCA), dipotassium hydrogen phosphate (K2HPO4), potassium dihydrogen phosphate, ethidium bromide (EB), digitonin, and 1,1,3,3-tetramethoxypropane were purchased from Merck, Germany. Culture medium (RPMI 1640) and lymphocytes separation medium (HiSep) were purchased from Himedia Laboratories Pvt. Ltd (Mumbai, India). Ac-DEVD-AMC (caspase-3 substrate) and Ac-LEHD-AMC (caspase-9 substrate) were procured from Enzo Life Sciences (ELS) AG (Switzerland). Calf thymus DNA was purchased from Sigma-Aldrich. CPF, MPT, and MLT were kind gift from Devidayal (Sales) Limited (Mumbai, India).
Experimental animals
Adult male albino rats of Wistar strain (Rattus norvegicus), weighing 150 ± 20 g, were used in all experiments. Rats were obtained from animal facility division of All India Institute of Medical Sciences (New Delhi, India) and were maintained in a light (light–dark cycle of 12 h each) and temperature (25 ± 2°C) controlled animal room of our department. The animals were fed on a standard pellet diet and tap water ad libitum. Rats were acclimatized for 1 week prior to the start of the experiment. The care and maintenance of animals were as per the approved guidelines of the ‘Committee for the Purpose of Control and Supervision of Experiments on Animals’ (India).
Lymphocytes isolation
Peripheral blood was collected from healthy male Wistar rats (150 ± 20 g) from eye orbital in vacutainer test tubes containing EDTA (Becton-Dickinson, Cedex, France). Blood samples were layered on the top of a Ficoll solution (1.077 g/ml), and the supernatant containing the leukocytes was removed after sedimentation of erythrocytes at 1000 × g for 10 min at room temperature. Lymphocytes sediment at the interface of the Ficoll layers and were collected and washed twice with phosphate buffer solution (pH 7.4) at 20°C. The cell viability was checked by trypan blue exclusion test 28 and should be at least 95% for healthy log-phase cultures. The final concentration of the lymphocytes was adjusted to 1–3 × 105 cells per milliliter by adding RPMI 1640 to the single cell suspension:
Pesticide treatment
CPF (4 h LC50 0.2 mg/l), 29 MPT (4 h LC50 0.135 mg/l), 30 and MLT (4 h LC50 >5.2 mg/l) 31 individually and in the mixture were taken from DMSO stock solutions (LC50) and added to the lymphocyte suspension to give final concentrations of 0.1 and 0.25 LC50 individually and in the mixture (equivalent to 1/30 and 1/12 LC50 of individual pesticides). The control cells were treated with DMSO alone. The lymphocytes were incubated with (0.1 and 0.25 LC50 concentrations of CPF, MPT, and MLT, individually and in the mixture) pesticides for 2, 4, 8, and 12 h at 37°C in a CO2 incubator. At the end of each incubation time, cell viability was determined again by trypan blue dye exclusion.
Estimation of LPO in lymphocytes
LPO was measured by estimation of thiobarbituric acid–reactive substances (TBARSs). 32 Briefly, after the incubation period, cells were collected in test tubes and centrifuged. The cell pellets were resuspended in 0.5 ml of Tris-hydrochloric acid (Tris-HCl), pH 7.4, and lysed by keeping it in a 60°C water bath for 30 min. Protein was precipitated with TCA (10% w/v). The precipitate was then dissolved in 1 ml of 0.05M H2SO4. A volume of the mixture (600 µl) was added to 150 µl of TBA (1% w/v) and then heated in boiling water for 15 min. TBARSs were measured with an ultraviolet–visible spectrophotometer set at 532 nm. The value of LPO was calculated using a standard 1,1,3,3-tetramethoxypropane with millimolar extinction coefficient of 25,000/mM/cm, and the results were expressed as percentage of increase in LPO as compared to the control.
Estimation of DICs
The estimation of DICs was performed according to Cui et al. 33 with some modifications. An aliquot of 1 ml of lymphocyte suspension was incubated with 0.1 and 0.25 LC50 concentrations of each pesticides, individually and in combination at 37°C for 2, 4, 8, and 12 h in the absence and presence of 100 µM SKF-525A. After pesticide treatment, 2 ml of free DNA from rat lymphocytes was mixed with 105 µl of 200 µg/ml EB in the buffer (20 mM K2HPO4, 2 mM EDTA, pH 7.4), while double-stranded calf thymus DNA, dissolved in TE buffer (10 mM Tris, 1 mM EDTA, pH 7.4), was mixed with 1.45 ml of 14.5 µg/ml EB. Fluorescence of DNA was determined in the spectrofluorometer. The λ ex (excitation) was 530 nm, and the maximum λ em (emission) was 580 nm. The slits of λ ex and λ em were 5 nm. After DNA was denatured by heating, the fluorescence of DNA was determined again as above. The ratio (R) of DNA fluorescence intensity after denaturing (FD) to that before denaturing (FN) was calculated according to the following equation:
Estimation of caspase-3 and caspase-9
Caspase-3 and caspase-9 activities were determined according to Kaul et al. 34 with some modifications. After treatment, the cells were lysed with 0.8 ml of lysis buffer (50 mM Tris-HCl, 1 mM EDTA, 10 mM EDTA, 10 µM digitonin, pH 7.4) for 20 min at 37°C. Lysates were then centrifuged at 10,000 r/min for 15 min, and supernatants were incubated with 50 µM Ac-DEVD-AMC (caspase-3 substrate) or Ac-LEHD-AMC (caspase-9 substrate) at 37°C for 60 min. Caspase activity was then determined by measuring the levels of released AMC using a spectrofluorometer with excitation at 380 nm and emission at 460 nm. The activity was calculated as fluorescence units per milligram of protein and expressed as percentage of the control.
Statistical analysis
Results were expressed as mean ± SE of six set of observations. Statistical analyses were performed using SPSS statistical software, version 11.5 (SPSS Inc, Chicago, Illinois, USA). All the statistical analyses were performed using one-way analysis of variance with post hoc Bonferroni’s multiple comparison test applied across the treatment groups. Significance was based on a p value of <0.05.
Results
% viability estimation
The results of the present study showed that lymphocytes exposed to 0.1 and 0.25 LC50 equivalents of CPF, MPT, MLT, and their mixture caused time-dependent decreases in viability of rat peripheral blood lymphocytes (Table 1). The data showed significant decreases of 8–14% and 14–26% in cell viability after a 2-h exposure of rat lymphocytes with 0.1 and 0.25 LC50 concentrations of CPF, MPT, MLT, and their mixture, respectively, when compared with the control. Incubation of rat lymphocytes with the same concentration of CPF, MPT, MLT, and their mixture for 4 h caused decreases of 10–14% and 13–28% in cell viability, respectively, as compared with the control. Similarly, incubation of rat lymphocytes with 0.1 LC50 and 0.25 LC50 concentrations of CPF, MPT, MLT, and their mixture for 8 and 12 h causes significant decreases of 11–32% in cell viability, as compared to the control. Incubation of rat lymphocytes with HCHO (30 µg/ml) for 2, 4, 8, and 12 h causes significant decreases of 22–28% in cell viability as compared to the control.
% viability in rat lymphocytes exposed to 0.1 and 0.25 LC50 doses of CPF, MPT, and MLT individually and in mixture (PM) for 2, 4, 8, and 12 h.a

CPF: chlorpyrifos; MPT: methyl parathion; MLT: malathion; DMSO: dimethyl sulfoxide; HCHO: formaldehyde.
aResults are expressed as mean ± SE of six set of observations. Rat lymphocytes were given 0.1 and 0.25 LC50 equivalent of individual pesticide and their mixture suspended in DMSO for 2, 4, 8, and 12 h, and after incubation, % viability is calculated.
b p < 0.05 when compared with the respective control.
c p > 0.05 when compared with the respective control.
Estimation of LPO
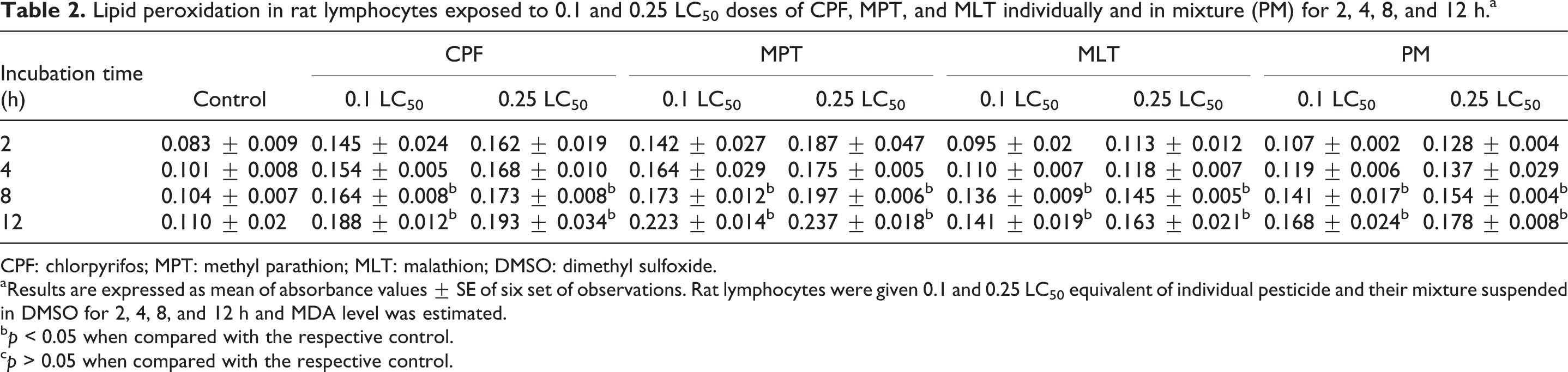
The results of the present study showed that lymphocytes exposed to 0.1 and 0.25 LC50 equivalents of CPF, MPT, MLT, and their mixture caused time- and dose-dependent increase in LPO in rat peripheral blood lymphocytes (Table 2). Incubation of rat lymphocytes with 0.1 LC50 and 0.25LC50 concentrations of CPF, MPT, MLT, and their mixture for 8 and 12 h caused significant increases of 28–115% in LPO, as compared to the control (Table 2).
Lipid peroxidation in rat lymphocytes exposed to 0.1 and 0.25 LC50 doses of CPF, MPT, and MLT individually and in mixture (PM) for 2, 4, 8, and 12 h.a
CPF: chlorpyrifos; MPT: methyl parathion; MLT: malathion; DMSO: dimethyl sulfoxide.
aResults are expressed as mean of absorbance values ± SE of six set of observations. Rat lymphocytes were given 0.1 and 0.25 LC50 equivalent of individual pesticide and their mixture suspended in DMSO for 2, 4, 8, and 12 h and MDA level was estimated.
b p < 0.05 when compared with the respective control.
c p > 0.05 when compared with the respective control.
DICs estimation
DICs were determined by calculating the ratio (R) of DNA fluorescence intensity after denaturing to that before denaturing. Calf thymus DNA and rat lymphocytes with or without SKF-525A, a cytochrome P450 inhibitor, were tested to determine whether the insecticides themselves or their active metabolites formed the DICs. Table 3 shows that HCHO significantly increased the R of calf thymus DNA and rat lymphocyte DNA in the absence or presence of SKF-525A. Incubation of rat lymphocytes with 0.1 LC50 concentration of CPF, MPT, MLT, and their mixture for 2, 4, 8, and 12 h caused 3–99% increase in the R of calf thymus DNA, when compared with the control in the presence or absence of SKF-525A. The incubation of rat lymphocytes with a 0.25 LC50 concentration of CPF, MPT, MLT, and their mixture for 2, 4, 8, and 12 h caused 8–143% increase in DICs formation as compared to the control (Table 3).
DNA interstrand cross-link formation in rat lymphocytes exposed to 0.1 and 0.25 LC50 doses of CPF, MPT, and MLT individually and in mixture (PM) for 2, 4, 8, and 12 h.

CPF: chlorpyrifos; MPT: methyl parathion; MLT: malathion; DMSO: dimethyl sulfoxide; HCHO: formaldehyde.
aResults are expressed as mean ± SE of ratios (R) of fluorescence intensity of calf thymus DNA and rat lymphocytes DNA after denaturing to that before. Rat lymphocytes were treated with 0.1 and 0.25 LC50 equivalent concentrations of individual pesticide and their mixture suspended in DMSO for 2, 4, 8, and 12 h.
b p < 0.05 when compared with the respective control.
c p > 0.05 when compared with the respective control.
Estimation of caspase-3 and caspase-9 activity
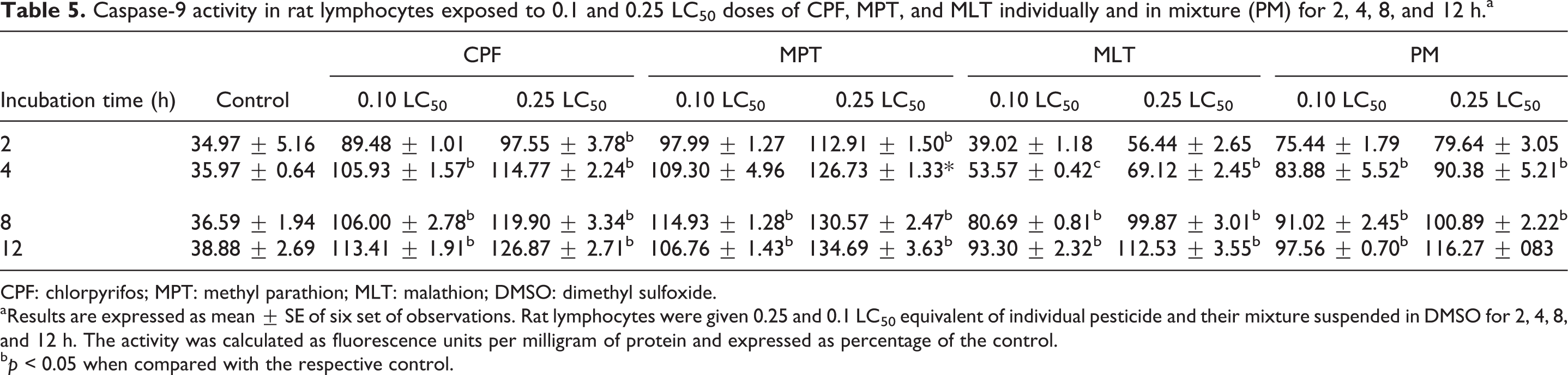
The results of the present study showed that in vitro exposure of rat lymphocytes to 0.1 and 0.25LC50 concentrations of OP pesticides, CPF, MPT and MLT, individually and in combination for 2, 4, 8, and 12 h caused a significantly marked increase in caspase-3 and caspase-9 activity, as shown by the increase in the florescence. Two-hour exposure of rat lymphocytes with 0.1 and 0.25LC50 concentrations of CPF, MPT, MLT, and their mixture caused significant increases of 12–223% in caspase 3 activity and 5–26% in caspase 9 activity, respectively, as compared to the control. Similarly, 4-h exposure of rat lymphocytes with 0.1 and 0.25LC50 doses of CPF, MPT, and MLT, individually and in combination, caused significant increases of 49–252% in caspase-3 activity and 33–70% in caspase-9 activity, as compared to the control. Similarly, 8- and 12-h exposure of rat lymphocytes with 0.1 and 0.25LC50 concentrations of CPF, MPT, and MLT, individually and in combination, caused significant increases of 47–270% in caspase-3 activity and 121–257% in caspase-9 activity as compared to the control (Tables 4 and 5).
Caspase-3 activity in rat lymphocytes exposed to 0.1 and 0.25 LC50 doses of CPF, MPT, and MLT individually and in mixture (PM) for 2, 4, 8, and 12 h.a

CPF: chlorpyrifos; MPT: methyl parathion; MLT: malathion; DMSO: dimethyl sulfoxide.
aResults are expressed as mean ± SE of six set of observations. Rat lymphocytes were given 0.25 and 0.1 LC50 equivalent of individual pesticide and their mixture suspended in DMSO for 2, 4, 8, and 12 h. The activity was calculated as fluorescence units per milligram of protein and expressed as percentage of the control.
b p < 0.05 when compared with the respective control.
Caspase-9 activity in rat lymphocytes exposed to 0.1 and 0.25 LC50 doses of CPF, MPT, and MLT individually and in mixture (PM) for 2, 4, 8, and 12 h.a
CPF: chlorpyrifos; MPT: methyl parathion; MLT: malathion; DMSO: dimethyl sulfoxide.
aResults are expressed as mean ± SE of six set of observations. Rat lymphocytes were given 0.25 and 0.1 LC50 equivalent of individual pesticide and their mixture suspended in DMSO for 2, 4, 8, and 12 h. The activity was calculated as fluorescence units per milligram of protein and expressed as percentage of the control.
b p < 0.05 when compared with the respective control.
Discussion
CPF, MPT, and MLT are the most commonly used OP pesticides in agriculture and homes. In our previous studies, we have found that in vitro exposure of peripheral blood lymphocytes of rats to OP pesticides CPF, MPT, and MLT, individually and in combination, caused significant DNA damage and DNA–protein cross-links formation. 7,8 In the present study, we tried to explore the previously studied molecular mechanism of OP pesticides that induced oxidative stress. We have also studied the role of DICs and DNA damage in apoptosis in pesticide-exposed peripheral blood lymphocytes of rats.
Previous studies showed that some insecticides such as CPF and cypermethrin have potential to form DNA adducts. 10 A connection between DNA damage and apoptosis is supported by a large body of literature. In vitro exposure of Drosophila S2 cells to Fipronil induces apoptosis through caspase-dependent mitochondrial pathways. 26 In recent literature, it is reported that CPF confers its genotoxic effects by inducing DNA damage and cell apoptosis. 35 However, the rules that govern this connection, which must be made judiciously, have remained unsolved. DNA adducts derive from covalent binding of electrophilic molecules to DNA. 36 If DNA adducts are not repaired or are mistakenly repaired before DNA replication, they may lead to gene mutations and initiate carcinogenesis. 37 DNA adducts include DNA monoadducts, DNA intrastrand cross-links, DICs, and DNA–protein cross-links. DICs derive from DNA monoadducts forming an additional covalent bond with the complementary strand of DNA. DICs prevent the separation of two DNA strands and therefore essential cellular processes including DNA replication and transcription. 38
The appearance of DNA adducts in blood or tissues is the result of the interaction of carcinogenic factors, such as tobacco constituents, environmental exposure to different chemicals, and the body reaction that is determined by individual characteristics of metabolic and repair systems. So, DNA adducts may be considered as a cumulative mirror of heterogeneous response of different individuals to carcinogenic chemicals, which finally could determine the risk for cancer. The present study showed potential of the studied pesticides to form DNA adducts and thus demonstrated their usefulness as cancer-associated markers. It is known that all DNA adducts are immunogenic, and their accumulation over some threshold concentration leads to the appearance of long-living autoantibodies. 39
In the present study, we have found a significant dose-and time-dependent decrease in the viability of lymphocytes on exposure to the studied pesticides. The decrease in viability may also be due to increased production of ROS. 7,8 Involvement of ROS has already been reported in cytotoxicity of many OP pesticides in cultured cells. For example, enantiomer-specific profenofos induces cytotoxicity and DNA damage in rat adrenal pheochromocytoma (PC12) cells mediated by oxidative stress. 40 We have also found that CPF, MPT, and MLT, individually and in combination, induce LPO as shown by an increase in the MDA levels. MDA is a marker of LPO resulting from the interaction of ROS and the cellular membrane. 41
In the present study, only MPT caused a significant increase in R of calf thymus and rat lymphocyte DNA in the absence or presence of SKF-525A, showing DICs formation capacity in time-dependent manner. The pesticide mixture did not show any dose- and time-dependent increase in DICs formation. These DICs may be formed by the active metabolites of CPF, MPT, and MLT instead of CPF, MPT, and MLT itself. The oxidation through cytochrome P450 may be involved in the activation of these OP pesticides. Incubation of primary mouse hepatocytes with different concentrations of CPF in the presence and absence of SKF-525A for 4 h could not induce any DNA adducts. 10 In the present study, we have also found that incubation of rat lymphocytes with 0.1 and 0.25 LC50 concentrations of CPF for 2 and 4 h did not induce any significant increase in DICs formation.
To assess the role of CPF, MPT, and MLT, individually and in combination on apoptosis, we first examined the activity of caspase-3 and caspase-9. These caspases have recognized to play an important role in the execution phase of neuronal and nonneuronal cell apoptosis. 42 Caspases are a family of endoproteases that provide critical links in cell regulatory networks controlling inflammation and cell death. Activation of apoptotic caspases results in inactivation or activation of substrates and the generation of a cascade of signaling events permitting the controlled demolition of cellular components. Dysregulation of caspases underlies human diseases including cancer and inflammatory disorders, 43,44 and major efforts to design better therapies for these diseases seek to understand how these enzymes work and how they can be controlled.
In the present study, we have found that in vitro exposure of peripheral blood lymphocytes of rats with CPF, MPT, and MLT, individually and in combination, caused a modest dose- and time-dependent increase in caspase-3 and caspase-9 activity. Taken together, the presented data in this study establish a pathway of apoptosis initiated by CPF, MPT, and MLT exposure. It has been reported that OP pesticides induce apoptosis through caspase-3 activation. 45 A previous study suggests that caspase-3 plays an important role in mediating paraoxon-induced apoptosis in EL4 cells. It also supports the role of oxidative stress in the generation of apoptosis. 46 In the present study, there is a significant increase of caspase-3 and caspase-9 activity. Activated caspase-3 and caspase-9 is responsible for the breakdown of several cellular components related to DNA repair and regulation during apoptosis. 47 It is reported that in vitro exposure of dichlorvos on primary rat microglia and human colon cancer T116 cells shows the involvement of oxidative stress and apoptosis in DNA damage. 48 Recently, it has been reported that the pesticide metabolites paraoxon and malaoxon induce cellular death by a caspase-dependent pathway in cultured human pulmonary cells. 19 CPF, an OP pesticide, is estrogenic and alters embryonic hatching, cell proliferation, and apoptosis in zebra fish. 20 Exposure of Chinese hamster ovary (CHO-K1) cells to carbamate pesticides shows its potential to induce genotoxic, cytotoxic, and apoptotic effects. 49
Conclusion
The present in vitro study demonstrated that CPF, MPT, and MLT exposure decreases cell viability and increases LPO resulting from ROS formation. Studied pesticides have potential to induce DIC formation and apoptosis possibly by increasing caspase activity in dose- and time-dependent manner. These findings suggest that oxidative stress plays an important role in OP pesticides–induced genotoxicity in peripheral blood lymphocytes of rats. Overall, the present research demonstrated that OP pesticides exposure is associated with cytotoxic and genotoxic effects on peripheral blood lymphocytes of rats. Further investigations are needed to identify the intracellular processes and mechanisms of genotoxicity resulting from CPF, MPT, and MLT exposure.
Footnotes
Acknowledgment
Financial support from Indian council of Medical Research, New Delhi, India, in the form of individual research project to Dr Anupama Ojha, under the guidance of Professor YK Gupta, is thankfully acknowledged.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by Indian council of Medical Research, New Delhi, India.