Abstract
Background
Ovarian cancer is a devastating disease that affects the female reproductive system, specifically the ovaries, which are responsible for producing eggs and the female reproductive hormones. Despite improvements in therapy, the prognosis for many ovarian cancer patients remains serious, with a high case-to-fatality ratio compared to other gynecological cancers.
Objectives
Therefore, the major aim of this research study is to assess the anti-cancer activity of scopoletin against ovarian cancer cells.
Materials and Methods
The efficacy of scopoletin at various concentrations (5–100 µM) on ovarian cancer A2780 cell viability was assessed utilizing the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) test. The accumulation of endogenous reactive oxygen species (ROS) and apoptosis was assessed using fluorescent staining assays. The concentrations of oxidative stress-related biomarkers, apoptotic proteins (Bax and Bcl-2), and caspase enzymes were studied in the control and treated cell lysates.
Results
The findings of the MTT test indicated that scopoletin treatment substantially reduced the vitality of A2780 cells. The half maximal inhibitory concentration (IC50) value of scopoletin for A2780 cells was determined to be 50 µM, which is designated for subsequent fluorescence staining and biochemical assays. The scopoletin treatment induced endogenous ROS accumulation and facilitated apoptosis in A2780 cells, as suggested by the findings of the fluorescent-staining assays. In the scopoletin-treated A2780 cells, thiobarbituric acid reactive substances (TBARS) levels were elevated, whereas antioxidant levels were diminished. Scopoletin treatment also enhanced pro-apoptotic protein concentrations while concurrently reducing the anti-apoptotic Bcl-2 concentration in the A2780 cells.
Conclusion
This study demonstrates that scopoletin is effective in enhancing apoptosis in A2780 cells. It can suppress cell proliferation and facilitate apoptosis in A2780 cells. These results highlight that scopoletin can be a useful anti-cancer candidate to treat ovarian cancer. However, further studies are warranted in the future to advocate for scopoletin as a useful anti-cancer agent to treat ovarian cancer.
Introduction
Ovarian cancer is a devastating disease that affects the female reproductive system, specifically the ovaries, which are responsible for generating eggs and the female reproductive hormones. This type of cancer is particularly challenging because of its non-specific symptoms and lack of early detection methods, often leading to late-stage diagnoses (Lliberos et al., 2024). Ovarian cancer is a complex and heterogeneous condition, with various subtypes that differ in their genetic and molecular profiles, as well as their clinical presentation and response to treatment. The pathogenesis of ovarian cancer is not yet fully reported, but it is highlighted to involve various environmental, hormonal, and genetic causes. Some of the known causes of ovarian cancer include age, genetic mutations, and certain reproductive factors, such as nulliparity and infertility (Tavares et al., 2024). The elevated death rate linked to ovarian cancer is attributable to its frequent diagnosis at an advanced stage, when the malignancy has disseminated to other regions of the body (Webb & Jordan, 2024). The impact of ovarian cancer extends beyond the personal toll on patients. It also imposes a substantial economic burden on healthcare systems and society as a whole. Despite improvements in therapy options, the 5-year survival rate for ovarian cancer remains low, at only 47%. Contrastingly, the 5-year survival rate for breast cancer is 85%. The disparity in survival rates highlights the urgent need for improved early detection methods and more effective treatment strategies (Zhuang et al., 2025).
Ovarian cancer exhibits remarkable heterogeneity, particularly within the high-grade serous subtype, which has been the focus of extensive research efforts. It has been suggested that the underlying pathophysiology of ovarian cancer is multifactorial, with a complex interplay between genetic predisposition, hormonal imbalances, and environmental factors. The role of oxidative stress and inflammation in the development of ovarian epithelial cancer has been highlighted, with incessant ovulation and the associated cyclical exposure of the ovarian surface epithelium to harmful agents playing a potential etiologic role (Pu et al., 2024). Hormonal dysregulation, particularly of the hypothalamic–pituitary–ovarian axis, has also been associated with the onset of ovarian cancer, with alterations in the production and signaling of steroid hormones potentially contributing to the onset of the disease (Madakkatel et al., 2025). The comprehensive understanding of the molecular and physiological mechanisms involved in ovarian carcinogenesis will be essential in advancing more effective diagnostic, prognostic, and salutary methods to improve patient outcomes and reduce the burden of this disease (Bryant et al., 2022).
Despite advances in surgery and the introduction of targeted therapies, the failure of existing treatments is underscored by ovarian cancer’s position as the major cause of gynecological-related cancer mortality. The current standard of care, comprising cytoreductive surgery and platinum-based chemotherapy, has proven inadequate in maintaining long-term disease control and preventing recurrence in most patients. This suggests the need for novel therapies that can effectively target the molecular underpinnings of ovarian cancer and improve patient outcomes (Kurnit et al., 2021). It has already been highlighted the efficacy of plant-derived bioactive compounds in the management of cancers. These natural products have demonstrated promising anti-tumor effects, targeting various pathways involved in tumor cell proliferation and metastasis. Additionally, plant bioactive compounds have shown the ability to overcome common challenges associated with conventional chemotherapy, such as drug resistance and adverse effects (Auti et al., 2024). The application of plant bioactive compounds in the management of cancer is further supported by their ability to modulate various aspects of the tumor microenvironment (Kangra et al., 2025). Scopoletin is a well-known bioactive, phenolic-coumarin compound derived from several plants, exhibiting multiple pharmacological properties. The numerous previous studies have already highlighted several pharmacological properties of scopoletin, including anti-arthritis (Wan Osman et al., 2017), neuroprotective (Gay et al., 2020), anti-hypertensive (Armenia et al., 2019), anti-hyperglycemia (Jang et al., 2018), anti-steatosis (Choi et al., 2017), hepatoprotective (Lee et al., 2014), and anti-anxiety (Luo et al., 2020) properties. Furthermore, it has been well reported that scopoletin enhances apoptosis in various tumor cells, including cervical cancer (Tian et al., 2019), breast cancer (Liu et al., 2019), and leukemia (Ahmadi et al., 2019). However, there are no such reports on the anti-cancer properties of scopoletin against ovarian cancer. Consequently, the major aim of this research is to assess the anti-cancer effects of scopoletin against ovarian cancer cells.
Materials and Methods
Chemicals
Scopoletin, antibiotics, Dulbecco’s Modified Eagle Medium (DMEM), and additional chemicals were obtained from Sigma–Aldrich, USA. The diagnostic kits for biochemical evaluations were procured from Elabscience and Abcam, USA, respectively.
Cell Culture Collection and Maintenance
The ovarian cancer A2780 cells were obtained from ATCC, USA, and cultured in DMEM media with antibiotics (1%) and heat-inactivated fetal bovine serum (FBS) (10%) at 37°C with 5% CO2. Cells were trypsinized and utilized for further tests upon achieving 80% confluence.
3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide (MTT) Cytotoxicity Assay
The effects of scopoletin at several doses on the growth of ovarian cancer A2780 cells were examined using the MTT test (Mosmann, 1983). The 96-well plate was employed to grow the cells for 24 h. A2780 cells were subsequently exposed to different doses (5, 10, 25, 50, 75, and 100 µM) of scopoletin and incubated for 24 h. Subsequent to incubation, MTT (20 µL) and DMEM (100 µL) were added to the wells and incubated for 4 h. After that, dimethyl sulfoxide (DMSO) (100 µL) was added to the wells to dissolve the formazan deposits. The absorbance was ultimately taken using the microplate reader at 570 nm.
2′,7′-Dichlorodihydrofluorescein Diacetate (DCFH-DA) Staining
The DCFH-DA staining (Hempel et al., 1999) was employed to quantify the accumulation of reactive oxygen species (ROS) in control and treated A2780 cells. The cells were cultured on a 24-well plate for 24 h before the addition of the half maximal inhibitory concentration (IC50) of scopoletin and/or 1 µg/mL of the standard drug cisplatin (CIS) for 24 h. Subsequently, 10 µg of DCFH-DA stain was introduced to each well for a duration of 1 h. A fluorescent microscope was employed to measure fluorescence intensity to assess the intracellular ROS generation.
Dual Staining
The dual-staining experiment (Gherghi et al., 2003) was done to study the impact of scopoletin on apoptotic induction in A2780 cells. The 24-well plate was employed to culture A2780 cells for 24 h and was exposed to the IC50 of scopoletin and 1 µg/mL of the CIS for an additional 24 h. Subsequent to incubation, a 1:1 combination of acridine orange/ethidium bromide (AO/EB) fluorescent dye (100 µg/mL) was added to the wells for 5 min. Ultimately, to identify apoptotic occurrences, the resultant fluorescence was assessed using a fluorescence microscope.
Analysis of Oxidative Stress Markers
The experimental A2780 cells were collected, lysed, and the resultant suspension was centrifuged. The cell lysate was subsequently used for biochemical evaluations. The levels of superoxide dismutase (SOD), glutathione (GSH), catalase (CAT), and thiobarbituric acid reactive substances (TBARS) in the cell lysates were studied using the appropriate kits. The tests were performed in triplicate using the manufacturer’s guidelines (Elabscience, USA).
Analysis of Apoptotic Protein Levels
The concentrations of apoptotic proteins Bcl-2, Bax, caspase-3, and caspase-9 in the cell lysates were evaluated using commercial diagnostic kits, following the manufacturer’s recommended guidelines (Abcam, USA).
Statistical Analysis
The data were given as a Mean ± Standard Deviation (SD) of triplicates following analysis with GraphPad Prism software (version 10.1.2). One-way analysis of variance (ANOVA) and Duncan’s Multiple Range Test (DMRT) were performed to assess the variations in the treatment group values, utilizing a significant threshold of p < .05. The IC50 value of scopoletin was determined using GraphPad software.
Results
Scopoletin Treatment Decreases the Cell Growth of A2780 Cells
The effect of scopoletin on the growth of A2780 cells was examined using the MTT test. The administration of scopoletin at diverse dosages (5–100 µM) significantly inhibited the growth of A2780 cells. These findings illustrate the cytotoxic effects of scopoletin on the growth of A2780 cells (Figure 1). The IC50 concentration of scopoletin for A2780 cells was established at 50 µM, which was selected for subsequent fluorescence staining and biochemical tests.
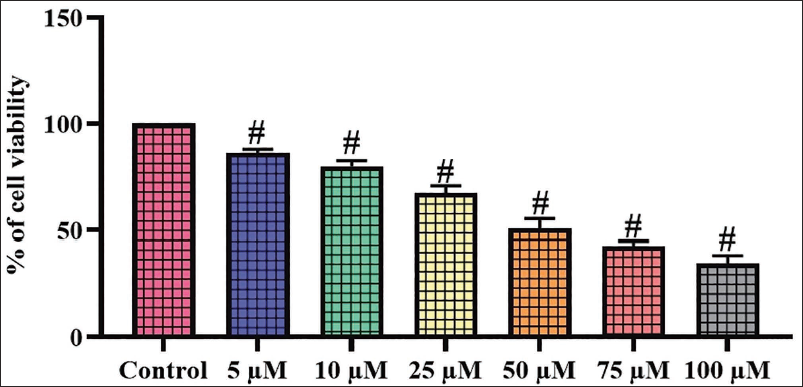
Scopoletin Treatment Increases Intracellular ROS in A2780 Cells
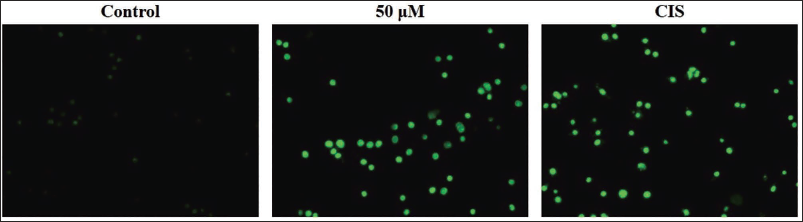
The effect of scopoletin treatment on the endogenous ROS in A2780 cells was assessed, with the findings illustrated in Figure 2. The A2780 cells treated with 50 µM of scopoletin exhibited a pronounced increase in green fluorescence than untreated control cells, indicating enhanced intracellular ROS production. A similar pattern was also observed in the CIS-exposed A2780 cells (positive control), signifying increased ROS production.
Effect of Scopoletin on the Intracellular Reactive Oxygen Species (ROS) Accumulation in the A2780 Cells. The A2780 Cells Treated with 50 µM of Scopoletin and/or 1 µg of Cisplatin (CIS) Exhibited Marked Increase in Green Fluorescence in Comparison to the Untreated Control Cells, Indicating Enhanced Intracellular ROS Generation.
Scopoletin Treatment Enhances Apoptosis in A2780 Cells
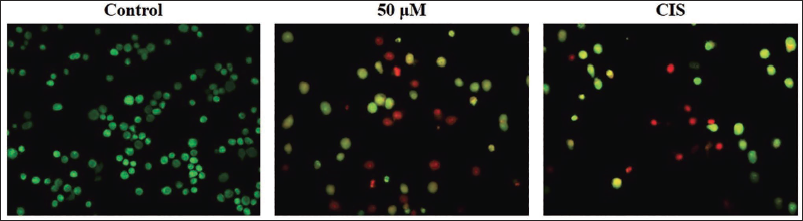
The effect of scopoletin on apoptosis in the A2780 cells is assessed using a dual-staining technique. The untreated control cells exhibited green-fluoresced viable cells; however, the A2780 cells treated with 50 µM of scopoletin showed increased cellular changes and apoptotic occurrences, as confirmed by intense red fluorescence in comparison to the untreated control (Figure 3). The CIS treatment also exhibited enhanced red fluorescence, signifying the presence of apoptotic events, and hence corroborating scopoletin’s potential to induce apoptosis in A2780 cells.
Effect of Scopoletin on the Apoptosis in the A2780 Cells. The A2780 Cells Treated with 50 µM of Scopoletin and/or 1 µg of Cisplatin (CIS) Showed Increased Ethidium Bromide (EB)-stained Red Fluorescence in Comparison to the Untreated Control, Signifying the Presence of Apoptotic Cell Deaths.
Scopoletin Treatment Induces Oxidative Stress in the A2780 Cells
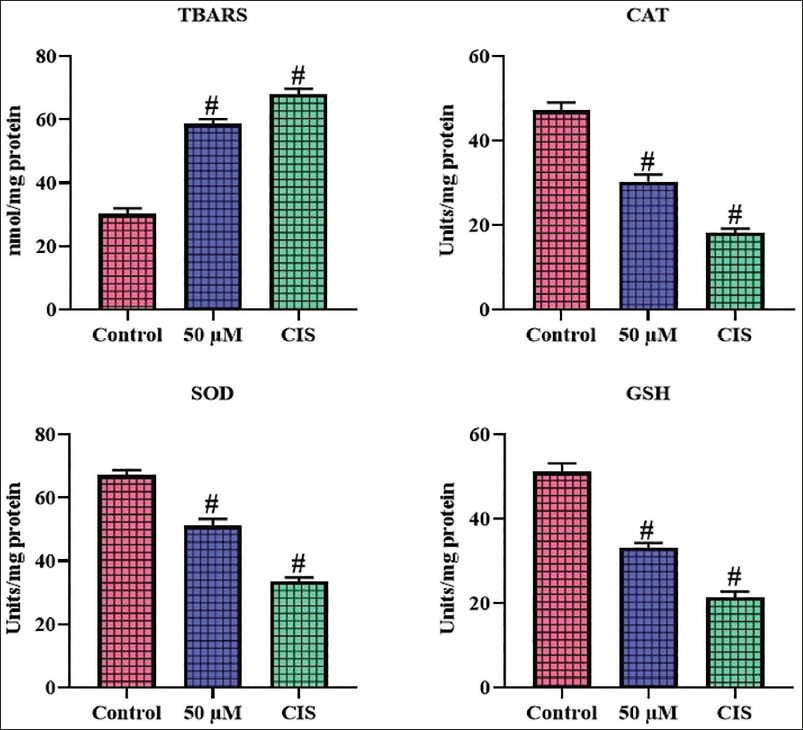
The levels of antioxidants and TBARS in the experimental A2780 cells were assessed, with the results presented in Figure 4. The concentrations of TBARS were considerably elevated in A2780 cells treated with 50 µM of scopoletin in comparison with untreated control cells. Moreover, the scopoletin treatment also significantly decreased the CAT, SOD, and GSH concentrations in the A2780 cells. These results indicate that scopoletin treatment effectively diminishes antioxidants while enhancing oxidative stress in A2780 cells. The CIS treatment also dramatically elevated oxidative stress in A2780 cells, hence corroborating the efficacy of scopoletin (Figure 4).
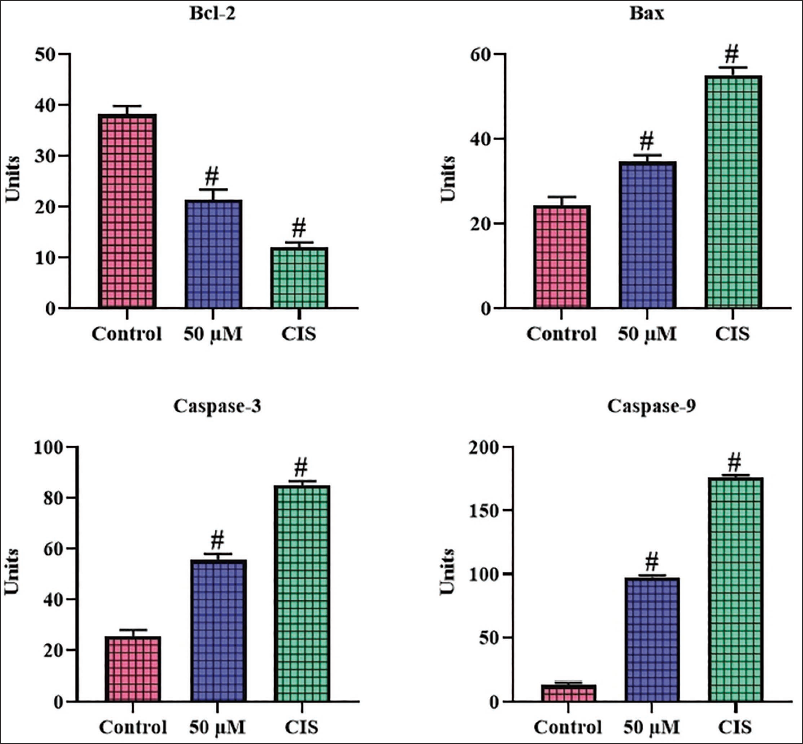
Scopoletin Treatment Regulates Apoptotic Protein Levels in A2780 Cells
The impact of scopoletin treatment on the concentrations of apoptotic proteins in A2780 cells was investigated, with results presented in Figure 5. The A2780 cells exposed to 50 µM of scopoletin exhibited increased concentrations of Bax, caspase-3, and caspase-9 in comparison to the untreated control cells. Furthermore, scopoletin treatment also reduced the Bcl-2 levels in A2780 cells. The standard drug CIS treatment also enhanced the concentrations of Bax, caspase-3, and caspase-9, while reducing Bcl-2 levels, as demonstrated in scopoletin-treated cells (Figure 5).
Discussion
Ovarian cancer is a severe disease that impacts women globally, imposing considerable physical, emotional, and financial burdens on healthcare systems. Despite improvements in therapies, the prognosis for many ovarian cancer patients remains serious, with a high case-to-fatality ratio compared to other gynecological cancers. One of the primary challenges in addressing ovarian cancer is the absence of a potential early detection method. The disease often presents with vague, non-specific symptoms, leading to delayed diagnosis and the majority of cases being identified at advanced stages when the cancer has already metastasized. The lack of potential screening methods for early diagnosis is a major contributor to the poor survival rates observed in ovarian cancer (Liberto et al., 2022). Despite the dismal prognosis, recent advancements in treatment strategies have offered some hope. Surgical debulking, systemic adjuvant chemotherapy, and the use of immunomodulation are the pillars of therapy. However, the onset of chemoresistance remains a significant challenge. The financial burden associated with ovarian cancer is also substantial. The high cost of treatment, including surgery, chemotherapy, and supportive care, can be crippling for patients and their families, especially in underserved communities with limited access to healthcare resources. Addressing these challenges will require a multidisciplinary approach, including the advancement of improved early-diagnosis methods and more effective therapies (Abdul-Aziz et al., 2025).
Cancer is a catastrophic disease marked by unregulated cellular growth, resulting in tumor formation that may disseminate to other bodily regions (metastases). This unbalanced cell growth and division result from a fundamental abnormality in the cellular mechanisms that regulate cell division and cell death, or apoptosis. Additionally, genetic deficiencies in the deoxyribonucleic acid (DNA) repair mechanisms can also contribute to the onset of cancer. While genetics can play an essential role in tumor susceptibility, with an estimated 5%–10% of human cancers showing a definite genetic component, epigenetic factors, such as DNA methylation patterns, also participate in the uncontrolled proliferation of tumor cells. In the quest to find effective cancer treatments, a key focus has been on understanding and manipulating the apoptotic pathways in cancer cells. This has resulted in the advancement of various drugs that target specific aspects of the apoptotic machinery (Schiliro & Firestein, 2021). A promising strategy involves the utilization of cytotoxic agents, specifically formulated to target and eliminate cancer cells while preserving healthy cells. The cytotoxic analysis of these drugs against cancer cell growth is crucial in determining their effectiveness and potential as cancer treatments. The uncontrolled proliferation of cancer cells is a complex process involving genetic and epigenetic factors, as well as the dysregulation of apoptotic pathways. Understanding these mechanisms is essential to developing potential cancer treatments, and the cytotoxic analysis of potential drug candidates against cancer cell growth is an essential step in this process (Contreras et al., 2022). The present results demonstrated that the treatment of scopoletin at different concentrations significantly inhibited the A2780 cell growth, which illustrates the cytotoxicity of scopoletin on the ovarian cancer cells.
Recent breakthroughs in cancer research have highlighted the critical role of ROS in the triggering of apoptosis in neoplastic cells. ROS are a collection of highly reactive molecules and free radicals generated during the partial reduction of oxygen (Verma et al., 2023). The intrinsic apoptotic cascade, which is dependent on the mitochondria, is often triggered by the accumulation of ROS within the cell. In this pathway, the pro-apoptotic Bax responds to the stress caused by ROS and gathers in the mitochondria, while the anti-apoptotic Bcl-2 becomes inactivated. This results in the release of cytochrome c into the cytosol, which then interacts with Apaf-1 to develop a complex that triggers the caspase signaling, ultimately inducing apoptosis (Nakamura & Takada, 2021). It has already been shown that various natural compounds possess the capacity to initiate apoptosis in tumor cells by the modulation of ROS levels. Furthermore, the extrinsic apoptotic cascade, which is activated by the binding of death receptors to their ligands, can also be triggered by ROS exposure (Dong et al., 2023). The analysis of the effect of sample drugs on the endogenous ROS levels in cancer cells is crucial for understanding their mechanism of action and potential therapeutic applications. This work shows that the scopoletin treatment considerably elevated the endogenous ROS production in ovarian cancer A2780 cells. This indicates that scopoletin can induce apoptosis in A2780 cells by enhancing the ROS stress.
Oxidative stress, defined by the disparity between the ROS generation and the antioxidant mechanisms, can result in DNA damage, protein modifications, and ultimately, programmed cell death. Tumor cells often exhibit an imbalanced redox status, resulting in the damage to DNA and proteins. This excessive oxidative stress can ultimately initiate apoptosis in tumor cells (Li et al., 2024). The analysis of oxidative stress biomarkers can offer useful insights into the role of oxidative stress in inducing apoptosis in tumor cells. Cancer cells often exhibit a hypermetabolic state, resulting in the overaccumulation of ROS, which can overwhelm the cellular antioxidant defenses, resulting in the activation of apoptotic mechanisms. The proliferation, dedifferentiation, and development of tumor cells rely on a complex interplay of disrupted cell cycle regulation, heightened growth factor pathway activation, and diminished apoptosis. These alterations in cellular mechanisms are often accompanied by an imbalance in the redox status of tumor cells, resulting in elevated oxidative stress (Domenicotti & Marengo, 2022). The capacity of tumor cells to adapt to and overcome these oxidative challenges is closely linked to their drug resistance and survival potential. The manipulation of the tumor microenvironment, including the modulation of redox-sensitive signaling pathways, is thought to participate in the survival of tumor cells. Various therapeutic agents have been investigated for their capacity to initiate oxidative stress-mediated apoptosis in cancer cells. The analysis of key antioxidants, as well as the levels of TBARS, can provide useful insights into the mode of action of these drugs (Kuo et al., 2022). TBARS are widely used as biomarkers of lipid peroxidation, a key consequence of oxidative stress. The assessment of these markers can shed light on the extent of oxidative damage induced by drug treatment and its impact on cellular integrity. The integration of these oxidative stress markers into the analysis of drug-induced apoptosis in cancer cells can provide a comprehensive understanding of the underlying mechanisms. Such insights can facilitate the advancement of potential and targeted therapies, ultimately improving the therapeutic outcomes for cancer patients (Aboelella et al., 2021). The present findings demonstrated a significantly decreased CAT, SOD, and GSH concentrations, and a subsequent elevation in TBARS in the scopoletin-treated A2780 cells. These findings suggest that scopoletin treatment effectively enhances the oxidative stress in A2780 cells, which may further facilitate apoptosis.
Apoptosis has developed as a crucial concept in the field of tumor treatment. The balance between cellular survival and death signaling is crucial in maintaining homeostasis and preventing disease progression (Shahar & Larisch, 2020). In the context of cancer, the loss of this balance leads to the uncontrolled proliferation of malignant cells. However, this imbalance can also be leveraged as a therapy, as inducing apoptosis in tumor cells has become a primary focus of cancer treatment. Understanding the intricate mechanisms that govern apoptosis has become increasingly important in the context of tumors, as the dysfunction of this process is a phenomenon of tumor cells. Two key protein families that play essential roles in the induction and regulation of apoptosis are the Bcl-2 family and the caspase family (Qian et al., 2022). In healthy cells, Bax is predominantly found in the cytosol, but in response to several apoptotic stimuli, Bax translocates to the mitochondria and the endoplasmic reticulum, where it can induce the release of cytochrome c into the cytosol. The release of cytochrome c then activates the caspase cascade, resulting in the activation of caspase-9 and, ultimately, caspase-3, which is the terminal shearing enzyme responsible for regulating apoptosis (Kaloni et al., 2023). In contrast, Bcl-2 is an anti-apoptotic protein that localizes to the mitochondrial membrane and acts to block the pro-apoptotic functions of Bax. However, under certain conditions, such as heat stress, Bcl-2 can become phosphorylated and inactivated, thereby allowing Bax to initiate the apoptotic pathway (Thomas et al., 2013). The caspase enzymes, particularly caspase-3 and caspase-9, are also critical players in the apoptotic process. Caspase-9 is an initiator caspase that is activated by the cytochrome c-mediated formation of the apoptosome. Once activated, caspase-9 then goes on to proteolytically activate the executioner caspase-3 (Araya et al., 2021). The alteration of apoptotic pathways is not only responsible for tumor development and progression but also contributes to tumor resistance to conventional therapies. Therefore, understanding the complex networks of signaling pathways that govern apoptosis is critical for improving the efficacy of cancer therapies and overcoming drug resistance (Hardwick & Soane, 2013). The interaction between the Bcl-2 family and the caspase family is complex and tightly regulated, with various signaling cascades and feedback loops governing the induction and progression of apoptosis in tumor cells. Understanding the specific roles of Bax, Bcl-2, caspase-3, and caspase-9 in this process is crucial for the advancement of therapies that can selectively induce or inhibit apoptosis in tumor cells, while reducing the impact on healthy cells (Warren et al., 2019). The current results exhibited increased Bax, caspase-3, and caspase-9 concentrations while reducing the Bcl-2 level in the scopoletin-treated A2780 cells. These results evidence that scopoletin can regulate pro- and anti-apoptotic proteins, thereby facilitating apoptosis induction in ovarian cancer.
Conclusion
The current study demonstrates that scopoletin has the capacity to enhance apoptosis in ovarian cancer cells. It can suppress cell proliferation by causing oxidative stress and facilitating apoptosis in A2780 cells. The apoptosis-inducing capacity of scopoletin may involve the increase in endogenous ROS buildup, oxidative stress, and the activation of pro-apoptotic proteins. Moreover, the present work is deficient in comprehensive molecular tests to investigate its participation in additional molecular pathways. These limitations need to be rectified in the future to advocate for scopoletin as a useful anti-cancer agent to treat ovarian cancer.
Footnotes
Abbreviations
AMPK: AMP-activated protein kinase; ANOVA: Analysis of variance; AO/EB: Acridine orange/ethidium bromide; Apaf-1: Apoptotic protease activating factor 1; ATCC: American Type Culture Collection; Bax: Bcl-2-associated X protein; Bcl-2: B-cell lymphoma 2; CAT: Catalase; CIS: Cisplatin; CO2: Carbon dioxide; DCFH-DA: 2′,7′-Dichlorodihydrofluorescein diacetate; DMEM: Dulbecco’s Modified Eagle Medium; DMSO: Dimethyl sulfoxide; DMRT: Duncan’s Multiple Range Test; DNA: Deoxyribonucleic acid; FBS: Fetal bovine serum; GSH: Glutathione; IC50: Half maximal inhibitory concentration; MTT: 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; PI3K/AKT: Phosphatidylinositol 3-kinase/protein kinase B; ROS: Reactive oxygen species; SOD: Superoxide dismutase; SREBP: Sterol regulatory element-binding protein; TBARS: Thiobarbituric acid reactive substances.
Data Availability
Data will be made available on request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable as there is no animal or human subjects involved in the experiments.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
