Abstract
Background
Sanqi Xueshangning Capsules (SXC) is a famous Chinese patent medicine with a good curative effect on various gynecological pain diseases in China. However, its chemical composition and potential active ingredients were clearly unrevealed, which hindered the elaboration of its material basis functional mechanism and clinical application.
Materials and Methods
In the study, we developed an integrative ultra-performance liquid chromatography quadrupole time-of-flight mass spectrometry (UPLC-Q-TOF-MS) method coupled with SCIEX OS software for characterizing the chemical composition in SXC. The data collection was conducted using a Waters UPLCTM HSS T3 (2.1 × 100 mm, 1.8 µm) in a binary elution system. The SCIEX OS software coupled with multivariable processing techniques was used to screen absorbed prototypes in serum after oral administration of SXC. The structural identification of the components was performed by MS/MS fragmentation mechanism.
Results
A total of 113 components from SXC were tentatively characterized, including 38 saponins, 13 flavonoids, 10 phenylpropanoids, 9 alkaloids, 20 organic acids, and 23 others. Based on the in vitro composition results, 21 absorbed prototypes in rat serum were identified, mainly including alkaloids, flavonoids, phenylpropanoids, and saponins.
Conclusion
It was concluded that an integrative UPLC-Q-TOF-MS method coupled with SCIEX OS software could be successfully applied for the components’ characterization of SXC in vitro and in vivo, which laid a reliable scientific basis for promoting the discovery of material basis and the improvement of quality control of SXC.
Keywords
Introduction
In China, Traditional Chinese Medicine (TCM) prescriptions have been used to treat complex diseases for centuries due to their low toxicity and good therapeutic effects (Yuan et al., 2022). Moreover, it has unique drug theories, such as the tropism of nature and taste, functions and indications, and compatibility of prescriptions (Dong et al., 2022; Tang et al., 2021; Wurchaih et al., 2019). With the development of modernization and internationalization of TCM, it has become an imperative scientific problem to analyze the therapeutic material basis of TCM in clinical application (Qian et al., 2022). TCM, which contained multiple drugs and chemical components, was considered to be a complex chemical giant system (Dong et al., 2022). According to the holistic view of TCM theory, the traditional natural drug discovery method embedded with the active ingredient separation and screening technology was cumbersome and time-consuming and cannot reflect the characteristics of the synergistic effect of multitarget and multi-components of TCM, which will induce the development of drugs with low efficacy (Cao et al., 2019; Li et al., 2022). The establishment of a comprehensive elucidation method of the in vivo and in vitro components of TCM is conducive to the further discovery of its therapeutic material basis. Serum pharmacochemistry believes that only the chemical components that migrate into the blood after oral administration of TCM may be substances that directly affect the body (Wei et al., 2022; Yin et al., 2022). Meanwhile, ultra-high-performance liquid chromatography quadrupole flight mass spectrometry (UPLC-Q-TOF-MS) was an effective chemical component analysis tool for the characterization of trace components in Chinese herbal extracts and biological samples, due to its high sensitivity, high resolution, and wide qualitative range (Dong et al., 2022; Miah et al., 2019). Therefore, in vivo composition research integrating UPLC-Q-TOF-MS and serum pharmacochemistry has been extensively recognized as a powerful method to better reveal the potential material basis of TCM.
Sanqi Xueshangning Capsules (SXC) is a famous over-the-counter (OTC) Chinese patent medicine, consisting of seven herbs, namely, Notoginseng Radix ET Rhizoma, Paridis Rhizoma, Callicarpa macrophylla Folium, Aconiti kusnezoffii Radix Cocta, Dioscoreae Rhizoma, Veratrum japonicum, and Borneolum syntheticum, as recorded in the 2020 edition of the Chinese Pharmacopoeia. Following the theory of TCM, SXC has the effects of activating blood circulation, removing blood stasis, and relieving pain, and it is commonly used to treat gastric bleeding, duodenal ulcer bleeding, bronchiectasis bleeding, pulmonary tuberculosis hemoptysis, functional uterine bleeding, trauma, hemorrhoids, bleeding, and other diseases in clinical practice. However, the chemical composition of TCM prescriptions was complex, and the potential active constituents were not fully comprehended. Although many reports on the chemical composition of single herbal medicine (Jing et al., 2017; Liu et al., 2019), the chemical composition and pharmacodynamic material basis of SXC were still unclear, which inevitably restricted its pharmacological research, clinical application, and the improvement of its quality standards.
In recent years, more and more commercial software and workstations have been matched with the UPLC-MS analysis system, and objective results were obtained by setting data processing signal parameters (Deng et al., 2020). As a powerful data processing platform, SCIEX OS software, an integrative MultiQuant™, MasterView™, and Libraryview™ intelligent software, can efficiently extract the mass spectrometry data of the compound by comparing with a database containing 800 reference standards under various acquisition modes, which automatically provides the possible cleavage pathway of the identified ingredient, and displayed its detailed information such as molecular formula, isotopic abundance, and additive form under the preset parameters (Li et al., 2022). In the current research, we established a rapid, sensitive, high-throughput, and highly selective analytical method in conjunction with SCIEX OS software to comprehensively characterize and identify the chemical ingredients in TCM, and explore its absorbed prototypes, which provided experimental data for the follow-up discovery of its material basis and the improvement of its quality standard.
Materials and Methods
Chemicals
Mass spectrometry grade formic acid, methanol, and acetonitrile were obtained from Fisher Scientific (MA, USA). Ultrapure water was purchased from Watsons (Guangzhou, China). SXC (Lot No.201101) was provided by Guilin Sanjin Pharmaceutical Co., Ltd (Guilin, China). Electrospray ionization (ESI) calibration solution was provided by AB SCEIX (MA, USA). The Oasis hydrophile lipophilic balance (HLB) 3cc solid phase extraction (SPE) column was provided by Waters (Milford, USA).
Preparation of SXC Extract
SXC powder is allowed to pass through a 30-mesh sieve. Further, 0.2 g of SXC powder was precisely weighed, placed in a conical flask with a stopper, and mixed with 5 mL of 50% methanol. The mixture was sonicated for 30 min, supplemented with solvent to compensate for weight loss, and centrifuged at 13,000 rpm for 10 min at 4°C. The supernatant was filtered by membrane for analysis.
Animals and Drug Administration
Specific pathogen-free (SPF) SD male rats (196 ± 7 g) were provided from Beijing Vital River Laboratory Animal Technology Co., Ltd. (Beijing, China). All rats were adaptively fed for one week in a controlled laboratory (25 ± 3°C with 55 ± 5% relative humidity room temperature, 12 h light/12 h dark). After one week of accommodation, rats were randomly assigned to two groups: a control group and a dosed group, with three rats in each group. The dosed group was given four times of the clinical dose SXC by gavage (2.16 g·kg−1), and the control group was given the same amount of distilled water. This research was supported by the Ethics Committee of Chengde Medical University (CDMULAC-20230306-001).
Biosample Preparation
After 1 h of administration, the abdominal cavity of anaesthetized rats was opened, and the blood was obtained by a vacuum blood collection tube from the abdominal aorta. The blood sample was placed for 30 min, centrifuged at 3,000 rpm at 4°C for 10 min, and the supernatant was obtained. The mixture containing 40 µL of 4% phosphoric acid solution and 2 mL of serum was treated with ultrasound for 1 min and vortexed for 30 s. The sample was added to the pre-activated SPE column. Then, the column was eluted with 2 mL of water and 2 mL of 100% methanol, respectively. The methanol eluent was dried with a nitrogen stream at 45°C. The residue was dissolved in 60 µL of 60% methanol (v/v), centrifuged at 4°C at 13,000 rpm for 15 min, and the supernatant was collected for analysis.
Chromatography and Mass Spectrometry Parameters
The analysis was performed on the ExionLCۛ UPLC system (AB SCEIX, USA) equipped with an ACQUITY UPLC BEH C18 (100 mm × 2.1 mm i.d.,1.7 µm; Waters Corporation, Milford, USA). The chromatographic column was eluted with a binary mobile phase. Mobile phases A and B were acetonitrile with 0.1% formic acid and water with 0.1% formic acid, respectively. The elution gradient was set as follows: 0.0–4.0 min, 0%–35% A; 4.0–8.0 min, 35%–71% A; 8.0–11.0 min, 71%–100% A; 11.0–11.1 min, 100%–0% A; 11.1–17.0 min, 100%–100% A. The flow rate of 0.3 mL/min, the injection volume of 4 µL, and the column temperature of 35°C were used. AB 5600 Triple TOF mass spectrometer equipped with an electrospray ionization (ESI) source (AB SCEIX, USA) was applied to perform ion data collection. The parameters were consistent with our previous research reports (Li et al., 2022). The ion source calibration solution was injected every 2 h to ensure the accuracy of the data.
Data Processing
Raw data were imported into the SCIEX OS software for data filtering and screening. The comprehensive score weights of mass deviation (<10 ppm), database matching degree (>70), and differential isotope ratios (<5) were set to 40%, 40%, and 20%, respectively. The possible element composition and chemical structure were reviewed by the SCIEX OS software with the literature report. For the screening and identification of absorbed prototype components, we imported the serum sample data of the control group and dosed group into the R 4.2.1 software for peak recognition, peak extraction, normalization, and so on. With the help of Ezinfo 3.0 software, multivariable pattern recognition analysis was conducted to screen out potential candidate ions that only existed in the dosed group. By comparing the results of in vitro components, the corresponding prototype components were identified.
Results and Discussion
Structural Identification of SXC Extract in vitro
According to the optimized method, mass spectrometry data of the SXC extract was swiftly collected using UPLC-Q-TOF-MS in the positive ion mode and negative ion mode. The basic peak ion chromatograms (BPC) of SXC extract in both ion modes are portrayed in Figure 1. According to the automatic matching function of SCIEX OS software and relevant literature reports, all compound structures were tentatively characterized. The results showed that the identified components mainly existed in the additive form of [M-H]− and [M+H]+. The SXC extract contained 113 compounds, including 38 saponins, 13 flavonoids, 10 phenylpropanoids, 9 alkaloids, 20 organic acids, and 23 others. Detailed information about each identified component including retention time, primary data, secondary data, and formula are provided in Supplementary Table S1.
Characterization of Saponins
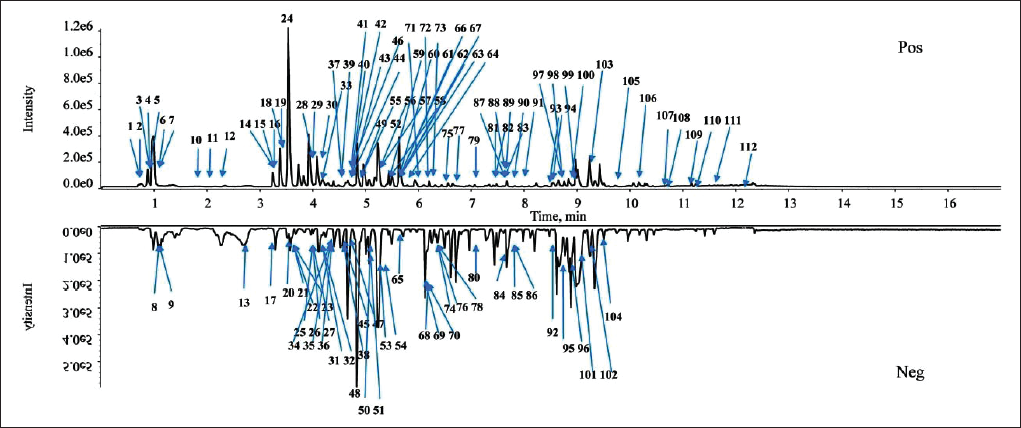
This research found that SXC contained many types of saponins. A total of 38 saponins from SXC were characterized, mainly including steroidal saponins, vinoretin, and diosgenin. Correspondingly, the adduct ions [M+H]+, [M+HCOO]−, and [M-H]− were mostly presented in the positive and negative ion modes. It was found that there were polyhydroxy substitutions on the carbon atom of the steroid nucleus, which were mostly linked to glucose (Glc), rhamnose (Rha), and arabinose (Ara). Concurrently, the type of steroid nucleus could be identified based on the characteristic diagnostic ions. For example, the characteristic diagnostic ions of dioscin-type ingredient and protodioscin-type ingredient were m/z 415, 397, 271, 253, and the characteristic diagnostic ions of vinoretin-type ingredient and protovinoretin-type ingredient were m/z 413, 395, 269, 251. Taking vinoretin-type ingredient as an example, peak 99 within the retention time of 9.00 min presented a high-intensity ion at m/z 855.4714 [M+H]+. Simultaneously, the mass spectrum of secondary fragments showed that the main fragments were m/z 723.4292, 577.3709, 415.3198, and 397.3086. The fragment ion at m/z 723.4292 [M+H-Ara]+ was produced after one molecule of arabinose from the excimer ion [M+H]+ was taken off, and then the removal of one molecule of rhamnose and one molecule of glucose, in turn, produced 577.3709 [M+H-Ara-Rha]+ and 415.3198 [M+H-Ara-Rha-Glc]+, and then, one molecule of H2O was removed to generate the fragment ion m/z 397.3086 [M+H-Ara-Rha-Glc-H2O]+, which was identified as polyphyllin I (Jin et al., 2021), as shown in Figure 2.
The Secondary Mass Spectrogram (A) and Possible Fragmentation Patterns (B) of Polyphyllin I.
Characterization of Flavonoids
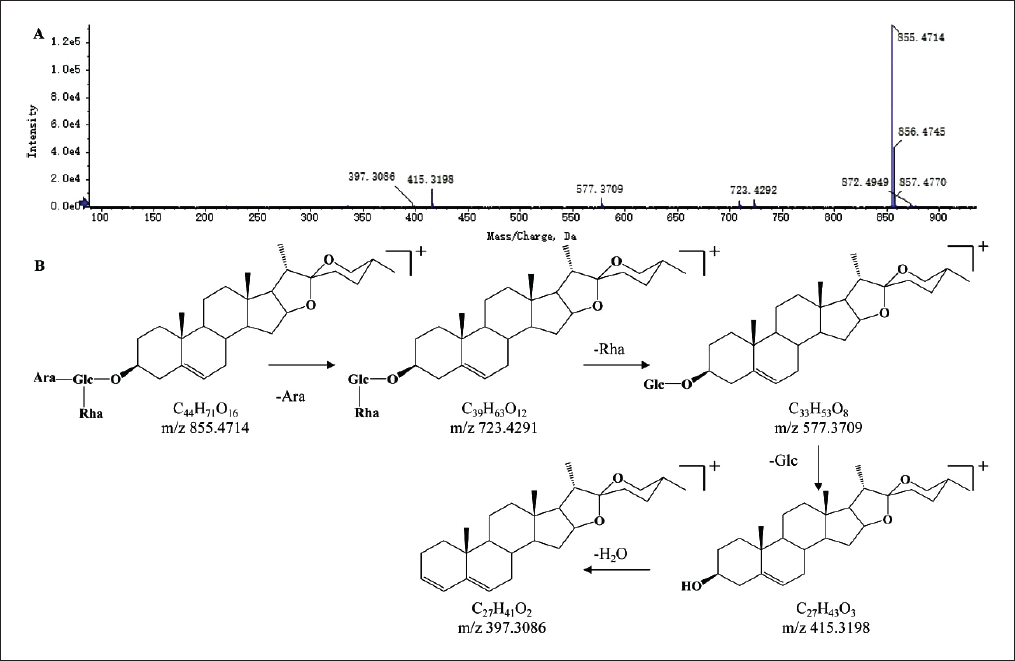
A total of 13 flavonoids were identified from SXC extract, mainly flavonoids and their glycosides. The structural characteristics of flavonoid glycosides were mainly the formation of oxygen glycosides connected with glucose or rhamnose. Under the high-energy collision of mass spectrometry, the breaking of continuous glycosidic bonds, and the neutral loss of characteristic fragments such as glucose C6H10O5 or rhamnose C6H10O4, produced high-intensity flavonoid aglycone fragments. The secondary flavonoid aglycone was mainly broken by the ring opening of the flavone mother nucleus and the loss of neutral ions such as CO2, CO, C2H2O, and H2O (Borges et al., 2001). Taking hesperidin as an example, the mass spectrometry behavior of the flavonoids was described. The primary mass spectrum information of peak 51 showed that the excimer ion peak was m/z 609.1895 [M-H]−, and its fragment ions were m/z 301.0737 [M-H-Rha-Glc]−, m/z 286.0506 [M-H-Rha-Glc-CH3]−, m/z 242.0606 [MH-Rha-Glc-CH3-CO2]−, m/z 151.0033 [M-H-Rha-Glc-C9H10O2]−. According to the fragmentation way and literature comparison (Zhang et al., 2022), it was inferred as hesperidin. The secondary mass spectrum and fragmentation pathway of hesperidin are shown in Figure 3.
The Secondary Mass Spectrogram (A) and Possible Fragmentation Patterns (B) of Hesperidin.
Characterization of Phenylpropanoids
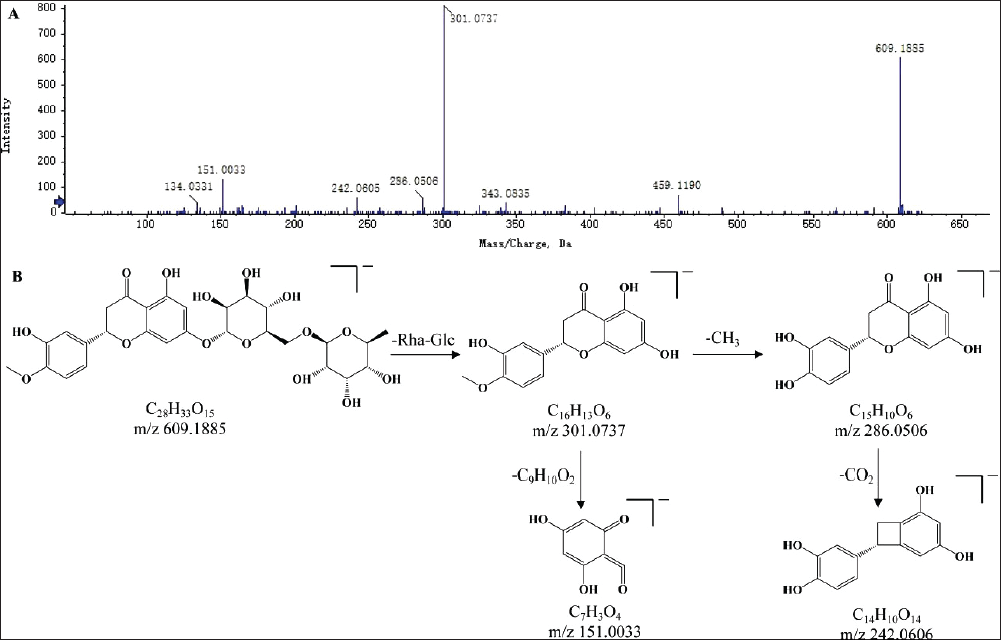
Phenylpropanoid is a naturally occurring compound composed of a benzene ring and three straight-chain carbon (C6-C3 group) (Liu et al., 2021). In this study, 11 phenylpropanoid compounds in SXC were discovered. Chlorogenic acid was a representative compound of phenylpropanoids. Taking chlorogenic acid as an example, in the negative ion mode, the parent ion was m/z 353.0891 [M-H]−. The Decaffeinato from the parent ion produced m/z 191.0565 [M+H-C9H7O3]− and continued to dehydrate to produce m/z 173.0462 [M+H-C9H7O3-H2O]−. In addition, the daughter ions at m/z 179.0358 [M+H-C7H11O6]− and m/z 135.0459 [M+H-C7H11O6-CO2]− were generated by parent ion, presumed by the further continuous loss of a sugar group and CO2. Based on the fragmentation pattern combined with the literature, it was speculated as chlorogenic acid (Li et al., 2021), as shown in Figure 4.
The Secondary Mass Spectrogram (A) and Possible Fragmentation Patterns (B) of Chlorogenic Acid.
Characterization of Alkaloids
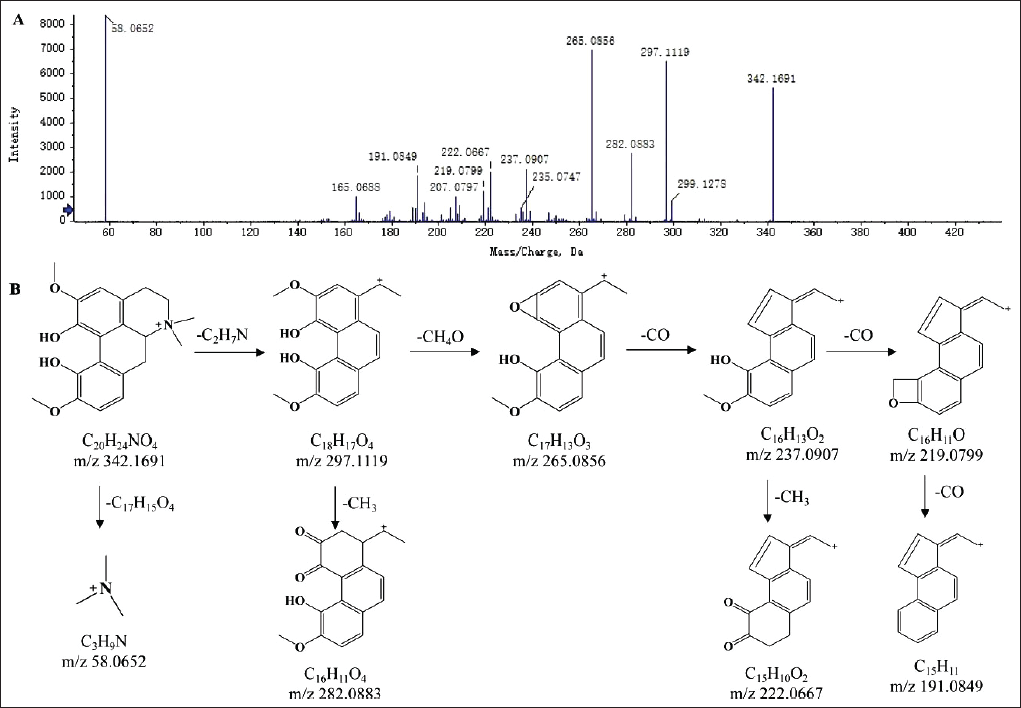
Alkaloid compounds from SXC included aporphine alkaloids, pyrrole alkaloids, and quaternary ammonium alkaloids. It was found that aporphine alkaloids were prone to side chain breakage and recombination, but not easily to form fragment ions with smaller m/z and larger abundance. If there were two methyl substituents on the nitrogen atom, it would lead to the preferential loss of one molecule of a dimethylamine group (C2H7N). Among them, magnoflorine was the representative of aporphine alkaloids, which were used to elucidate the cracking mechanism. The molecular ion peak at m/z 342.1692 [M]+ was found, and the molecular formula was C20H24NO4. Under the high-energy collision energy conditions, in the positive ion mode, the loss of a C2H7N group from the parent ion produced m/z 297.1119 [M-C2H7N]+ fragment ions, and further loss of one molecule CH3OH and CH3 was prone to produce m/z 265.0856 [M-C2H7N-CH3OH]+ and m/z 282.0883 [M-C2H7N-CH3]+ fragment ions, respectively. The other fragment ion peaks at m/z 237.0907 [M-C2H7N-CH3OH-CO]+, m/z 219.799 [M-C2H7N-CH3OH-CO-H2O]+, and 222.0667 [M-C2H7N-CH3OH-CO-CH3]+ were generally produced by removing neutral molecules such as CO, H2O, and CH3. The specific cleavage pathway is shown in Figure 5. Through literature search and SCIEX OS database matching, the compound was finally determined as magnoflorine (He et al., 2021).
The Secondary Mass Spectrogram (A) and Possible Fragmentation Patterns (B) of Magnoflorine.
Characterization of Organic Acids
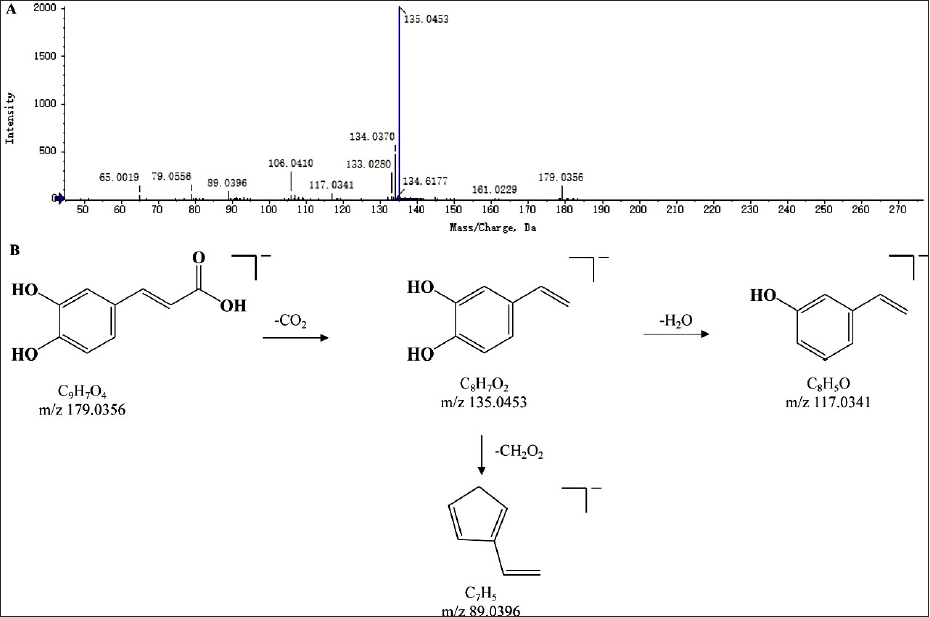
A total of 20 organic acids were identified from the SXC extract, and these compounds mainly existed in the form of the quasi-molecular ion peak of [M-H]− in the negative ion mode. Under the high collision energy condition, neutral molecules such as 18 Da (H2O), 45 Da (COOH), and 44 Da (CO2) were easily lost, resulting in the generation of characteristic fragment ion peaks. Taking caffeic acid as an example, in the negative ion mode, the MS spectrum of the compound showed the excimer ion was m/z 173.0358 [M-H]−, and its fragment ions were m/z 135.0453 [M-H-CO2]−, m/z 117.0341 [M-H-CO2-H2O]−, and m/z 89.0396 [M-H-CO2-CH2O2]−. According to the fragmentation rule, and comparison of literature (Liu et al., 2020), it was speculated as caffeic acid. The fragmentation patterns are shown in Figure 6.
The Secondary Mass Spectrogram (A) and Possible Fragmentation Patterns (B) of Caffeic Acid.
Others
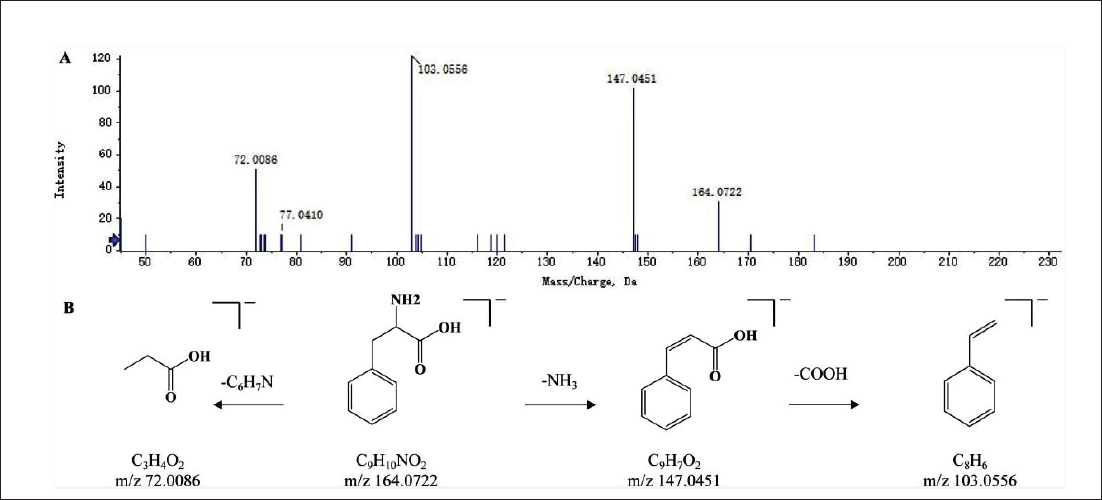
A total of 23 other compounds were determined in SXC extracts, most of which were amino acids. Amino acid compounds were prone to lose neutral molecules NH3 and COOH under high-energy collisions, thus forming characteristic fragment ions. We took L-phenylalanine as an example to illustrate the identification process. In the negative ion mode, the parent ion peak at m/z 164.0722 [M-H]− was observed, and the fragment ions at m/z 147.0451 [M-H-NH3]− and 103.0556 [M-H-NH3-COOH]− were produced by losing one molecule of NH3 and COOH in turn. Additionally, the molecular group (C6H7N) of the parent ion was removed to yield m/z 72.0086 [M-H-C6H7N]− fragment ion. Referring to the database and literature (Shi et al., 2021), it was inferred as L-phenylalanine. The MS/MS spectrum and possible cleavage pathway are shown in Figure 7.
The Secondary Mass Spectrogram (A) and Possible Fragmentation Patterns (B) of L-Phenylalanine.
Characterization of Prototype Components in SXC
Serum pharmacochemistry was a powerful means for exploring the blood components of TCM, which can reveal the potential pharmacodynamic material basis in the complex TCM system. In the current experiment, UPLC-Q-TOF-MS combined with SCIEX OS workstation was used to realize the data acquisition and analysis in the positive and negative ion modes. Ezinfo software was applied to conduct multivariable pattern recognition analysis of the dosed group and the control group to obtain the prototype candidate ions that only existed in the dosed group. For the BPC and PCA score plot (Figure 8), it can be seen that the profile of the dosed group was far from that of the control group, implying that the overall chemical profile was disturbed after administration. Ions that only existed in the dosed group and VIP > 1 (Figure 8) were taken as candidate variables, and then they were matched with the standard database in SCIEX OS software and in vitro component data for the screening of prototype components in vivo. A total of 21 prototype components were determined, including one organic acid, three alkaloids, five flavonoids, four phenylpropanoids, four saponins, and four other components. The base peak ion chromatogram and details of prototype components are displayed in Figure 8 and Supplementary Table S1, respectively.
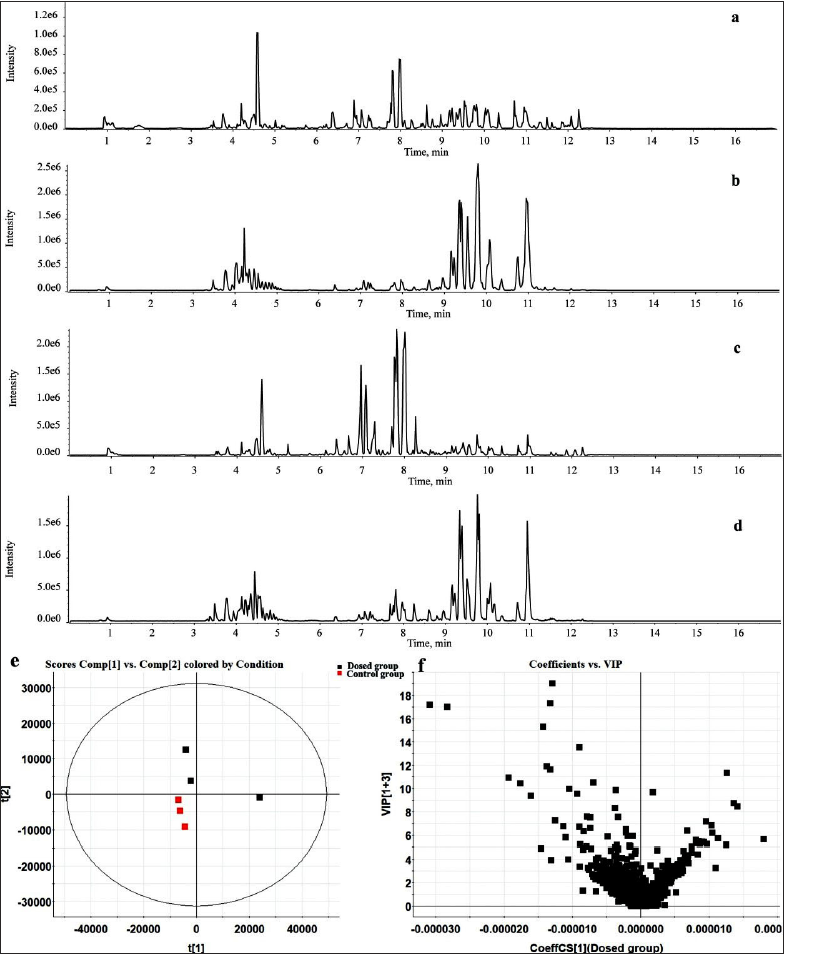
It was reported that the absorbed blood components from TCM have multiple pharmacological activities, which may be the material basis for the clinical efficacy of TCM formula. In the current study, 21 potential bioactive ingredients including hypaconitine, hesperidin, chlorogenic acid, forsythiaside A, and so on were observed. These compounds may be the therapeutic material basis that SXC exhibited many pharmacological actions. Some literature has reported that alkaloid compounds like hypaconitine have excellent pharmacological effects on chronic heart failure protection, anti-cancer, and pulmonary protection (Feng et al., 2017; Gao et al., 2020). Hesperidin and other flavonoids, as the active ingredients of many TCMs, have exerted extensive pharmacological effects such as anti-osteoporosis, anti-oxidation, anti-inflammatory, anti-bacterial, anti-tumor, improving myocardial damage, preventing and treating diabetes and its complications, and regulating blood lipids (Sun et al., 2022). Phenylpropanoid compounds like chlorogenic acid have good therapeutic effects such as neuroprotection and cardiovascular protection (Tirapelli et al., 2005). Forsythiaside A, as an effective ingredient widely studied at present, has strong anti-bacterial activity, broad anti-bacterial spectrum, strong anti-virus and antioxidant activities, anti-pyretic, analgesic, and immunomodulatory effects (Gong et al., 2021). Because of this, it was not difficult to find that the pharmacological actions of these bioactive compounds were aligned with the efficacy of SXC in clinic practice, such as gastric bleeding, duodenal ulcer bleeding, bronchiectasis bleeding, and pulmonary tuberculosis hemoptysis (Ning et al., 2021). It should be noted that not many saponins and flavonoids were determined in rat serum. We speculated that it may be attributed to gastrointestinal degradation reaction or metabolic reaction in vivo after oral administration of SXC. The identification of related metabolites was still needed later.
Conclusion
In this study, the in vivo and in vitro chemical composition of SXC was systematically characterized using a rapid and effective data analysis strategy based on UPLC-Q-TOF-MS method and SCIEX OS software. By detailed analysis of their precise mass, neutral loss, possible fragment pathway, standard database, and literature, a total of 113 compounds were tentatively characterized in SXC. Serum pharmacochemistry analysis exhibited 21 identified prototypes in SXC, including one organic acid, three alkaloids, five flavonoids, four phenylpropanoids, four saponins, and four other components, which were considered as potential active components. As far as we know, it was the first time to comprehensively investigate the chemical compounds in SXC and its absorbed prototypes, which may lay a solid basis for further research on the pharmacodynamic substances, functional mechanisms, and quality markers of SXC.
Footnotes
Abbreviations
UPLC-Q-TOF-MS: Ultra-performance liquid chromatography quadrupole time-of-flight mass spectrometry; SXC: Sanqi Xueshangning Capsules; TCM: Traditional Chinese Medicine; OTC: Over-the-counter.
Authors’ Contributions
HZ, NL: Formal analysis and Writing – original draft. XC: Writing – original draft and Investigation. YY: Methodology, Conceptualization, and Formal analysis. YY: Conceptualization and Investigation. ZL, JC, YT: Data curation and Validation. CZ: Data curation and Supervision. HX: Funding acquisition, Project administration, Conceptualization, and Writing – review and editing.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest concerning the research, authorship, and/or publication of this article.
Funding
This work was supported by the Natural Science Foundation for the Youth funded by the Natural Science Foundation of China (82104384), Special project for Basic Scientific Research Business expenses of Chengde Medical University (KY202316), the S&T Program of Hebei (216Z2501G, H2022406026, and H2022329001) , Chengde Medical University Discipline Construction Funds (No. 2023LJ-1) and Pharmacodynamic Material Basis Team of traditional Chinese medicine of the Youth PI (principal investigator) Science and Technology Innovation Team Project of Chengde Medical University.
