Abstract
Objectives
Rutin protects and stabilizes hepatocytes and is used to treat hepatitis, liver cirrhosis, and other diseases. However, there is little evidence to suggest that Rutin is associated with CCl4-induced hepatotoxicity. In this study, we investigated the effects of Rutin on CCl4-induced liver inflammation and discussed in vivo and in vitro mechanisms.
Materials and Methods
CCl4 was used to induce liver inflammation, and Rutin was used for intervention in mice. Then the body weight, liver weight, and liver inflammation of all mice were measured. Western blotting and RT-PCR were used to assess TLR4/MyD88/NFκB inflammation signaling pathways and inflammatory gene expression. Finally, TLR4 was activated in primary hepatocytes to verify the above signal pathways.
Findings
CCl4 mice developed liver function damage and liver inflammation. Further investigations showed that the inhibitory effects of Si on inflammation were mediated by TLR4/MyD88/NFκB inflammation signaling pathways inhibition. Primary hepatocyte studies also confirmed the participation of these signaling pathways and proteins.
Significance
Rutin improved CCl4-induced liver inflammation. Such mechanisms may be related to TLR4/MyD88/NFκB inflammation signaling pathways.
Introduction
The liver is the largest organ in the human body, with multiple functions, including material catabolism, biotransformation, and endocrine secretion, and it is vulnerable to various pathogenic factors and stimuli in vivo and abroad, leading to traumatic hepatitis (Trefts et al., 2017). Drugs, poisons, viruses, alcohol, fatty liver, and autoimmune diseases can all damage the liver (Jetter & Kullak-Ublick, 2020; Koppe, 2014; Lin et al., 2020;). If it progresses to cirrhosis or liver cancer, treatment is extremely difficult and mortality is extremely high (Chung et al., 2018). CCl4, an organic compound, is mainly used as an excellent organic solvent, dry cleaning agent, fire extinguishing agent, refrigerant, extract of spices, pesticide, and chemical raw materials (Meaden et al., 2020). However, CCl4 has been reported to have strong toxicity, which is recognized as a liver poison and a classic reagent for establishing liver injury models in basic research (Pessayre et al., 1982; Zhao et al., 2019). CCl4 caused liver damage in mice mainly by destroying the liver cells of mice, which led to the ALT and AST in the liver cells entering the blood through the liver cell membrane, leading to the increase of transaminase activity in the blood (Tuominen et al., 2021; Zhao et al., 2019). The relevant mechanism is mainly that CCl4 is metabolized into trichloromethyl radical by cytochrome P450 superfamily monooxygenase in the liver (Jeong et al., 2002). Subsequently, this free radical reacts with nucleic acids, proteins, and lipids, thereby damaging key cellular processes and inducing systemic liver injury characterized by liver inflammation, fibrosis, cirrhosis, and liver cancer (Lee et al., 2021).
Rutin, also known as vitamin P, is a natural flavonoid glycoside, which widely exists in plants and has anti-inflammatory, antioxidant, anti-allergic, anti-virus, and other effects (Chua, 2013). Rutin is a strong antioxidant that can scavenge free radicals, inhibit biofilm peroxidation, and inhibit the occurrence of inflammatory reaction and plays an important role in the body (Chua, 2013; Ghorbani, 2017). Inflammation is an immune defense reaction of living tissues with the vascular system against various inflammatory factors and the damage caused by them, and also a defense adaptive response of the body to eliminate various inflammatory factors and promote the repair of damage (Medzhitov, 2008; Muvhulawa et al., 2022). However, its occurrence process is often accompanied by blood circulation disorders, the release of inflammatory mediators, tissue degeneration, and necrosis, which will cause damage to the body to a certain extent (Arulselvan et al., 2016; Medzhitov, 2008). Research shows that Rutin can regulate the cytokines and inflammatory mediators in LPS-induced RAW264.7 cells by mediating TLR4-MyD88-TRAF6-NF-κB inflammatory signal pathway (Tian et al., 2021). In the study of liver diseases, it is also found that Rutin can effectively prevent and treat liver damage caused by doxorubicin, sorafenib, diabetes, and ischemia-reperfusion, and the main mechanism is that Rutin has strong anti-inflammatory and antioxidant capacity (Ahmed et al., 2022; Liang et al., 2018; Zhou et al., 2021). However, there is still no report on the liver inflammation caused by Rutin treatment of CCl4.
Therefore, the purpose of this study was to determine whether Rutin contributes to the anti-inflammatory effect in the CCl4-induced liver inflammation model in mice and determine its action target, so as to provide a strong experimental basis and theoretical basis for the clinical application of Rutin.
Materials and Methods
Experimental Animals
CCl4 was provided by Sigma-Aldrich (St. Louis, MO, USA), and Rutin was purchased from Med Chem Express. Male C57BL/6 mice (18–22 g, 4–6 weeks) were purchased from the Nanjing Junke Bioengineering Co., Ltd (Nanjing, China). Mice were induced through intraperitoneal injections of CCl4 (50%, 1% mL/100 g) weekly (Wu et al., 2019) and intragastric administration of Rutin every day (100 mg/1000 g) for 4 weeks (Ma et al., 2017). The mice are specifically divided into the following groups: (1) Control group (CON, intraperitoneal injection + gavage of an equal amount of physiological saline); (2) Rutin group (Rutin, intraperitoneal injection of an equal amount of physiological saline + gavage an equal amount of Rutin); (3) CCl4 group (CCl4, intraperitoneal injection of an equal amount of CCl4 + gavage an equal amount of physiological saline); and (4) Rutin + CCl4 group (Rutin + CCl4, intraperitoneal injection of an equal amount of CCl4 + gavage an equal amount of Rutin). All the animals were in a standard laboratory (12-h light/dark cycle) and had free access to drinking water. Body weight was measured every week.
Tissue and Blood Sampling Collection
The mice were euthanized. The blood and liver samples were removed. The serum was obtained immediately (2,000 rpm, 30 min). The liver was partially immersed in 4% paraformaldehyde and partially frozen in −80°C. Notably, 4% paraformaldehyde was purchased from Biyuntian Biotechnology Co., Ltd (Shanghai, China).
Histopathological Examinations
Liver samples in 4% paraformaldehyde were dehydrated in graded alcohol and embedded in paraffin. The liver sample was cut into 5-µM-thick sections. The sections were stained with H&E for histological examination. The specimens were placed on normal slides and examined under an optical microscope. H&E Staining Kit (C0105M) was purchased from Shanghai Biyuntian Biotechnology Co., Ltd (Shanghai, China).
Biochemical Studies
The ALP, ALT, AST, GGT, IL-1β, IL-6, IL-10, and LDH assay kits were purchased from Nanjing Jiancheng Bioengineering Institute (Nanjing, China). According to the protocols of the kits, the contents of ALP, ALT, AST, GGT, IL-1β, IL-6, IL-10, and LDH in serum were measured in the spectrophotometer.
Cell
Primary Hepatocytes Extraction
Male mice (4 weeks) were purchased from the Nanjing Junke Bioengineering Co., Ltd. (Nanjing, China). Primary hepatocytes were obtained from mouse liver by perfusion, digestion (collagenase IV), filtration, centrifugation, and inoculation. Collagenase IV was purchased from Sigma-Aldrich (St. Louis, MO, USA).
Hepatocytes Viability
First, CCl4 is divided into concentration gradients of 0%, 0.25%, 0.5%, 1%, and 2%. After determining the optimal cell treatment concentration through the CCK-8 kit, Rutin was divided into 25, 50, 100, and 200 µM according to concentration gradients.
CCK-8 was purchased from Shanghai Biyuntian Biotechnology Co., Ltd (Shanghai, China). According to the CCK-8 kit protocol, the reagent was added to each group of hepatocytes, the cells incubated at 37℃ for 1.5 h, and finally, the cell viability was measured in the spectrophotometer. The hepatocyte viability of the CON group was defined as 100%.
Cell Grouping
Primary hepatocytes were divided into six groups. All groups of liver cells were cultured using DMEM medium +10% PBS. Rutin is dissolved in DMSO solution in a ratio of 1 (µmol): 5000 (µL). The specific cell grouping is as follows: (1) CON group: DMSO; (2) CCl4 group: 0.5% CCl4; (3) Rutin + CCl4: 100 µM Rutin +0.5% CCl4; (4) Lv-EGFP:Lv-EGFP-Control; (5) Lv-EGFP-TLR4: Lv-EGFP-TLR4; and (6) Lv-EGFP-TLR4 + Rutin: Lv-EGFP-TLR4 + 100 µM Rutin.
Western Blot
Liver tissue and primary hepatocytes were homogenized in standard protein extraction buffer at 4°C. The tissue homogenate was centrifuged at 12,000 rpm at 4°C for 15 min. The supernatant was collected for Western blot analysis. The total protein concentration was quantified by Bradford analysis (Bio-Rad, California, USA). Samples containing the same amount of protein were boiled in SDS sample buffer at 95°C for 10 min. Subsequently, protein samples were separated by sodium dodecyl sulfate polyacrylamide gel electrophoresis, transferred to PVDF membrane (Bio-Rad), sealed with 5% skimmed milk at room temperature for 1 h, and incubated with primary antibody at 4°C overnight. The membrane was then washed and incubated with secondary antibodies. A chemiluminescence detection system (Millipore Corp., Billerica, MA, USA) was used to detect protein expression. All secondary antibodies were purchased from Wuhan Proteintech Biotechnology Co., Ltd. (Wuhan, China).
PCR
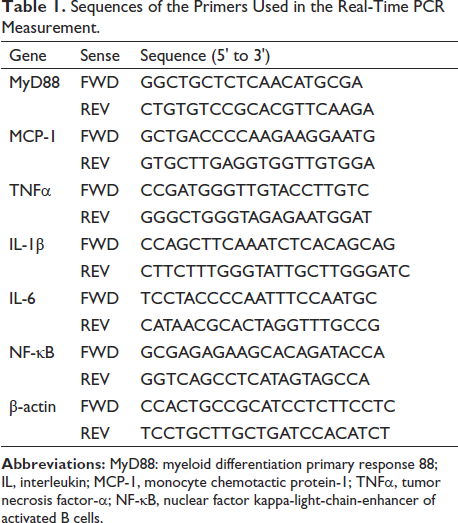
Using TRIzol reagent (Invitrogen, Shanghai, China), we isolated RNA from liver tissue according to the instructions. The gene expression level was quantified by qRT PCR. Adopt2−∇∇CT calculation is used to calculate the relative number of tested genes. GAPDH was used as an internal control. The primer sequences of these genes are shown in Table 1.
Sequences of the Primers Used in the Real-Time PCR Measurement.
Statistical Analysis
The normal distribution and homogeneity of variance of the data were detected using one-way or two-way (Figure 1) ANOVA, and Tukey’s multiple comparison post hoc test was used to analyze the significant differences between the groups. p ≤ 0.05 was considered statistically significant.
Result
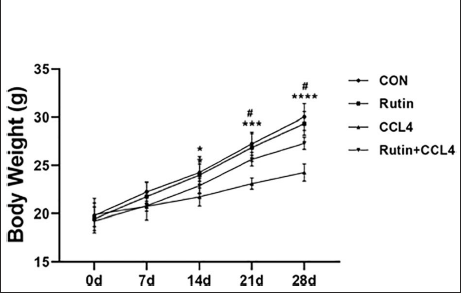
Rutin and CCl4 Affect the Body and Liver Weight of Mice
As shown in Figure 1, the body weight of mice in CCl4 group decreased significantly from the 14th day, while Rutin effectively prevented the weight loss from the 21st day.
As shown in Table 2, at the end of the model establishment, there was no significant difference in the liver weight of mice in each group, but the body weight of mice in the CCl4 group decreased significantly, and Rutin effectively reversed this change. At the same time, the liver/body weight was significantly increased in the CCl4 group, and Rutin effectively reversed this change.
Rutin Inhibited Liver Function Deterioration Caused by CCl4 (n = 3).
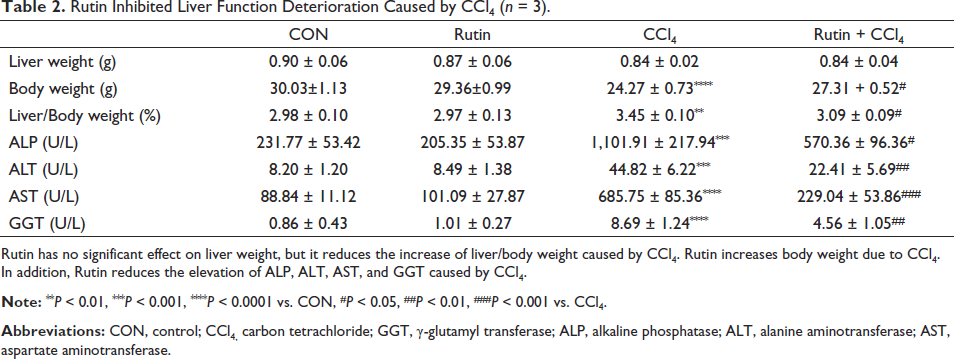
Rutin has no significant effect on liver weight, but it reduces the increase of liver/body weight caused by CCl4. Rutin increases body weight due to CCl4. In addition, Rutin reduces the elevation of ALP, ALT, AST, and GGT caused by CCl4.
Rutin and CCl4 Affect the Liver Function of Mice
As shown in Table 2, CCl4 successfully induced liver function damage in mice, specifically manifested as follows: blood concentrations of ALP, ALT, AST, and GGT increased significantly. However, the above abnormalities in the blood of the Rutin group mice were significantly reversed.
As shown in Figure 2A, CCl4 causes damage to hepatocytes, which is manifested by abnormal nuclear morphology, multinucleated cells, unclear or even disappeared cell membrane boundaries, and abnormal cytoplasm.
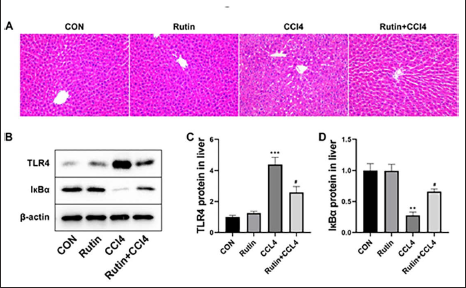
Rutin and CCl4 Affect the TLR4-induced Inflammation Signaling Pathway in Mice Liver
As shown in Figure 2B, TLR4 was significantly downregulated and IκBα was significantly upregulated in the liver of the CCl4 group mice compared with the CON group. However, Rutin significantly reversed the above changes. Figures 2C and D provide more intuitive data for the above results.
As shown in Table 3, the content of inflammatory factors in serum was significantly changed. Specifically, IL-1β, IL-6, and LDH were significantly increased, while IL-10 was significantly decreased in the serum of CCl4 mice. However, Rutin finally reversed the above changes.
Rutin Inhibited the Abnormality of Serum Inflammatory Factors Caused by CCL4 (n = 3).
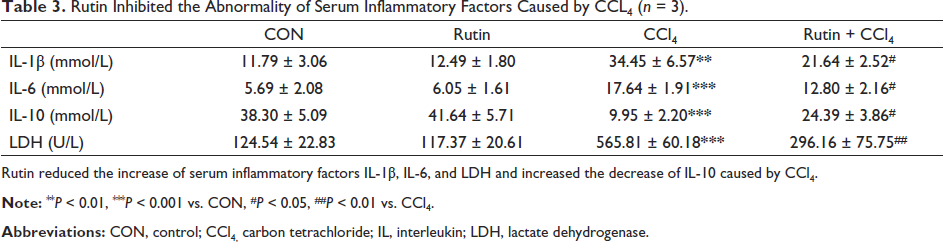
Rutin reduced the increase of serum inflammatory factors IL-1β, IL-6, and LDH and increased the decrease of IL-10 caused by CCl4.
As shown in Table 4, the expression of inflammatory genes in mice liver was significantly changed. Specifically, MyD88, TNF-α, IL-1β, IL-6, NF-κB, MCP-1, and VCAM-1 genes were significantly increased. However, Rutin finally reversed the above changes.
Effect of Rutin on Liver Inflammation-related Genes (n = 3).
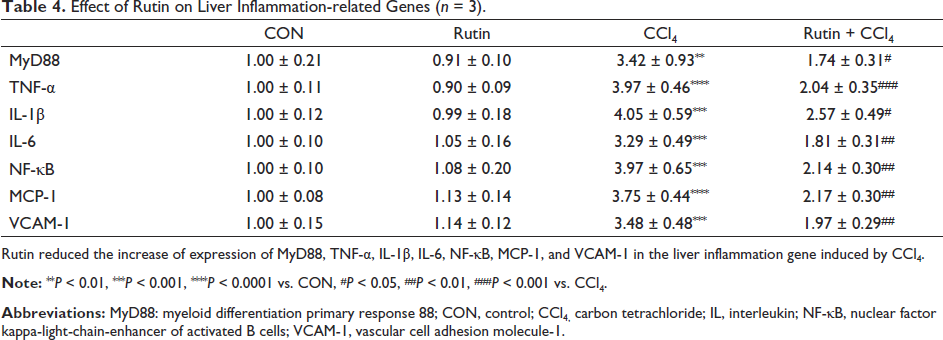
Rutin reduced the increase of expression of MyD88, TNF-α, IL-1ꞵ, IL-6, NF-κB, MCP-1, and VCAM-1 in the liver inflammation gene induced by CCl4.
Rutin and CCl4 Affect the Cell Viability in Primary Hepatocytes
As shown in Figure 3A, when the concentration of CCl4 is 0.25% and 0.5%, the primary hepatocytes have a certain degree of cell viability decline, whereas, when the concentration of CCl4 is 1% and 2%, the primary hepatocytes have a large number of deaths.
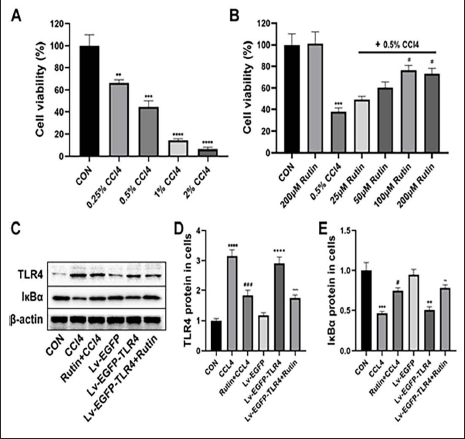
As shown in Figure 3B, under the condition that the concentration of CCl4 is 0.5%, when the concentration of Rutin is 25 and 50 µM, it does not significantly reverse the decline of primary hepatocyte viability caused by CCl4, but when the concentration of Rutin is 100 and 200 µM, the primary hepatocyte viability is significantly reversed, the difference is statistically significant.
Rutin and CCl4 Affect the TLR4-induced Inflammation Signaling Pathway in Primary Hepatocytes
As shown in Figure 3C, TLR4 was significantly downregulated and IκBα was significantly upregulated in the hepatocytes of the CCl4 and Lv-EGFP-TLR4 groups compared with the CON and Lv-EGFP groups. However, Rutin significantly reversed the above changes. Figures 3D and E provide more intuitive data for the above results.
As shown in Table 5, the expression of inflammatory genes in hepatocytes was significantly changed. Specifically, MyD88, TNF-α, IL-1β, IL-6, NF-κB, and VCAM-1 genes were significantly increased in the hepatocytes of the CCl4 and Lv-EGFP-TLR4 groups compared with the CON and Lv-EGFP groups. However, Rutin finally reversed the above changes.
Effect of Rutin on Primary Hepatocytes Inflammation Related Genes (n = 3).
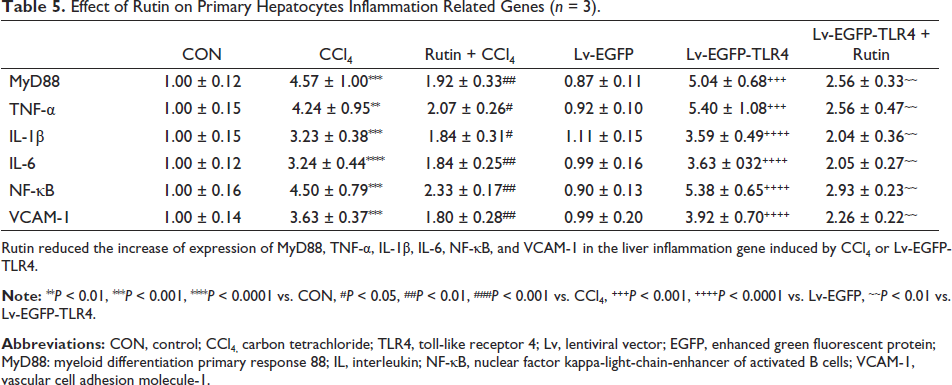
Rutin reduced the increase of expression of MyD88, TNF-α, IL-1β, IL-6, NF-κB, and VCAM-1 in the liver inflammation gene induced by CCl4 or Lv-EGFP-TLR4.
Discussion
Liver injury refers to liver cell injury and liver function change caused by various reasons (Trefts et al., 2017). The long-term existence of liver injury inducement such as external force damage, lipid metabolism damage, and drug and poison damage will lead to irreversible pathological changes in liver tissue (Ginès et al., 2021). CCl4 is mainly inhaled by the respiratory tract and has serious damage to the liver, kidney, and central system, among which the liver is the most (Zhao et al., 2019). In hepatocytes, CCl4 can be converted into free radicals in vivo, which can disrupt the metabolism of lipids on the hepatocyte membrane and cause hepatocyte necrosis (Unsal et al., 2021). The toxicity of CCl4 is mainly manifested by the increase of liver-specific enzyme activity, the decrease of pseudocholinesterase activity, fat accumulation, the significant increase of serum ALT, AST, and other markers, and the decrease of liver function (Lee et al., 2021; Zhang et al., 2021). In this study, we successfully induced liver injury in mice by CCl4, which was specifically manifested as weight loss and liver/body weight increase, abnormal liver tissue morphology, abnormal serum liver function indicators such as ALP, ALT, AST, and GGT, abnormal liver TLR4/MyD88/NF-κB signal pathway, and liver inflammation outbreak.
As a monomer of traditional Chinese medicine, Rutin has anti-inflammatory, anti-microbial, anti-oxidative stress, and other pharmacological effects, which can significantly protect multiple organs from damage (Chua, 2013; Ghorbani, 2017). One of the practical findings in this study is that Rutin can effectively alleviate the liver damage caused by CCl4 and play an anti-inflammatory role by regulating the TLR4/MyD88/NF-κB signal pathway. Serum levels of ALP, ALT, AST, and GGT are indicators to evaluate liver function and reflect the state of liver tissue cells (Albhaisi & Qayyum, 2022; Faris et al., 2021). Rutin effectively reverses the increase of their levels caused by CCl4, which is consistent with the findings in research on the treatment of liver damage caused by doxorubicin with Rutin (Ahmed et al., 2022). Serum levels of IL-6, IL-1, LDH, and IL-10 are representative indicators reflecting the inflammatory state of the body (Libby, 2021; Ridker, 2016; Saraiva et al., 2020). IL-6, IL-1, and LDH promote inflammation, while IL-10 plays an anti-inflammatory role (Libby, 2021; Ridker, 2016; Saraiva et al., 2020). The results showed that Rutin reversed the increase of serum ALP, ALT, AST, GGT, IL-6, IL-1, and LDH levels and the decrease of IL-10 levels in mice caused by CCl4. There are various opinions on how Rutin exerts liver protection through anti-inflammatory effects. Rutin can alleviate liver inflammation caused by doxorubicin by regulating NRF2, regulate autophagy by regulating the BANCR/miRNA-590-5P/OLR1 axis, and promote liver cell proliferation by promoting the insulin IRS-2/PI3K/Akt/GSK-3β signaling pathway (Ahmed et al., 2022; Liang et al., 2018; Zhou et al., 2021). In summary, Rutin may exert anti-inflammatory effects in liver injury through multiple pathways, but the TLR4/MyD88/NF-κB signal pathway may play an extremely important role in CCl4-induced liver injury.
TLR4/MyD88/NF-κB signal pathway is a signal transduction pathway closely related to cellular inflammation (Li et al., 2020). Recent studies have shown that the abnormal activation of TLR4/MyD88/NF-κB signaling pathway is closely related to the pathogenesis and pathological progress of liver injury (Tian et al., 2020; Yao et al., 2015). TLR4 is a pattern recognition receptor that plays an important role in innate immunity and is usually overexpressed in inflammatory cells (Tian et al., 2020; Vijay, 2018; Zamyatina & Heine, 2020). After TLR4 is stimulated by various reasons, it recruits its downstream MyD88 and other related cytokines, triggering the signal cascade, leading to the activation of downstream NF-κB, and ultimately leading to the occurrence of inflammatory reaction (Liu et al., 2014; Sun et al., 2021). Specifically, after TLR4 is activated, MyD88 protein is recruited to a specific binding site. Through phosphorylation reaction, IRAK and TRAF6 are combined to form a complex (Li et al., 2019; Roy et al., 2016). Finally, the downstream NF-κB inhibitor protein kinase is phosphorylated by the complex, making the I-κB ubiquitinated, resulting in the dissociation of NF-κB from the NF-κB/I-κB complex and living, translocation from the cytoplasm to the nucleus, and plays its pro-inflammatory activity (He et al., 2016). In this study, Rutin reduced the expression of TLR4 in experimental animals, increased the expression of I-κB, and decreased the gene expression level of MyD88 and NF-κB, revealing the role of TLR4/MyD88/NF-κB signaling pathway in the treatment of CCl4-induced liver inflammation by Rutin.
Currently, there are few clinical applications of Rutin, but there are also some preclinical studies (Choi et al., 2016; Hashizume & Tandia, 2020; Ragheb et al., 2020). For example, when cream containing or without Rutin is applied to human skin, it is found that Rutin increases the mRNA expression of type I collagen by clearing ROS, reduces the expression of Messenger RNA of matrix metallopeptidase 1, increases skin elasticity, reduces the length, area, and number of wrinkles, and delays skin aging (Choi et al., 2016). The combination of Rutin and vitamin C also strengthened the effects of blood glucose control, insulin resistance, lipid status, and oxidative stress markers in type 2 diabetes patients (Ragheb et al., 2020). In addition, the enzyme-modified form of Rutin, water-soluble α-glycosylated Rutin, can be applied to obese individuals to improve overall health by reducing abdominal visceral fat (Hashizume & Tandia, 2020). These all provide a reference value for the clinical application of Rutin, and in future clinical research on the treatment of liver diseases with Rutin, the following methods can also be used, but not limited to: (1) adding traditional drug compatibility, (2) combining with clinical medication, and (3) preparing new Rutin formulations. In summary, the application of Rutin in the clinical treatment of liver diseases is still in its early stages, and further research is needed to determine the safe dosage, specific mechanism of action, and therapeutic effect of Rutin in the human body. This is the current flaw and the direction of future work.
Conclusion
Our research shows that Rutin may be a natural inhibitor of TLR4, which can inhibit liver inflammation by regulating TLR4 and finally reverse CCl4-induced liver injury, highlighting its important potential mechanism. More importantly, it provides a more theoretical basis and clinical guidance for Rutin to regulate liver inflammation.
Abbreviations
CCl4: Carbon tetrachloride; ALT: Alanine aminotransferase; AST: Aspartate aminotransferase; ALP: Alkaline phosphatase; GGT: γ-glutamyl transferase; H&E: Hematoxylin & eosin; IL: Interleukin; CCK-8: Cell counting kit-8; CON: Control; Lv: Lentiviral vector; EGFP: Enhanced green fluorescent protein; TLR4: Toll-like receptor 4; MyD88: Myeloid differentiation primary response 88; NF-κB: Nuclear factor kappa-light-chain-enhancer of activated B cells; I-κB: NF-κB inhibitor protein; VCAM-1: Vascular cell adhesion molecule-1; LDH: Lactate dehydrogenase.
Authors Contributions
QH and KZ are responsible for the conception and design of the research, the key revision of the knowledge content, and the approval of the final manuscript version to be published and agree to be responsible for all aspects of the work. HH has made substantial contributions to data acquisition, analysis, and interpretation. QH is responsible for the drafting of the manuscript. QH and KZ confirm the authenticity of all original data.
Footnotes
Declaration of Conflicting of Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors did not receive no financial support for the research, authorship, and/or publication of this article.
Statement of Informed Consent and Ethical Approval
All experimental procedures in this study follow the guidelines of the Research Ethics Committee of Hubei University of Medicine.
