Abstract
Background
One of the primary flavonoids found in the Glycyrrhiza glabra is called glabridin, which has anti-inflammatory, anti-bacterial, and antineoplastic effects. However, the insolubility of glabridin in water limits its application. Liposomes can increase the solubility of insoluble drugs and improve their bioavailability.
Objectives
We examined the potential for the treatment of glabridin liposomes on histamine-induced atopic dermatitis.
Materials and Methods
After GL treatment, histopathology, inflammatory cytokines, and atopic dermatitis-related proteins were used to evaluate the therapeutic effect of GL.
Results
Glabridin liposomes alleviated histamine-induced scratching behavior; reduced mast cell proliferation, infiltration, and degranulation; and restrained the expression of associated pro-inflammatory cytokines. Additionally, glabridin liposomes restored nerve growth factor to normal levels and enhanced the expression of filaggrin to promote cuticle growth and repair skin damage caused by atopic dermatitis.
Conclusion
Glabridin liposomes may relieve histamine-induced skin hypersensitivity and cortical hyperplasia by inhibiting the production of inflammatory cytokines, demonstrating their potential for the clinical treatment of atopic dermatitis.
Introduction
Atopic dermatitis (AD) is a type of inflammatory skin disease caused by exposure to allergens. Its clinical characteristics include dry and rough skin, itching, swelling, and epidermal hyperplasia (Barone et al., 2020; Bischoff, 2007). Notably, 10%–20% of people in advanced countries suffer from AD, which is one of the most prevalent chronic diseases in the world (Deckers et al., 2012; Williams et al., 2008). The etiology of AD is related to impaired skin barrier function and skin inflammation (Edslev et al., 2021; Paller et al., 2019). Many cell regulators and inflammatory factors play important roles in AD, such as interleukins (IL-1β, IL-1, and IL-6) and filaggrin (FLG) (Cabanillas et al., 2017; Han et al., 2020; Paller et al., 2019). Traditional drugs for AD include topical agents such as skin moisturizers and topical corticosteroids. However, regular use of corticosteroids can cause skin atrophy, wrinkle aging, capillary dilation, and other side effects (Dhar et al., 2014). At present, AD-targeted drugs remain in an early stage of development, and research has focused mainly on phosphodiesterase-4 inhibitors, interleukin monoclonal antibodies, Janus kinase inhibitors, and histamine antagonists. Due to the insufficient drug supply, costly biological agents, prolonged use, medication resistance, and severe adverse effects, treating AD is still difficult (Dhar et al., 2014; Hagenström et al., 2021; Narla et al., 2022; Tang et al., 2020). Moreover, in many cases, the curative effect is satisfactory. Numerous investigations have demonstrated that Chinese herbal medicine has good therapeutic effects on mild-to-severe AD, and its anti-allergic active ingredients cause few side effects and typically do not produce drug dependence (Dhar et al., 2014; Fan et al., 2021; Hussain et al., 2017; Kishimoto et al., 2021; Li et al., 2022).
The Glycyrrhiza glabra is a well-known Chinese herbal medicine with a long history of use and has a wide range of purposes, including the removal of heat and toxins, spleen and stomach tonification, as well as anti-inflammatory, antibacterial, and antineoplastic properties. This plant can also be used as raw material for synthetic food additives and cosmetics (Barone et al., 2020; Dogra et al., 2021; Goel et al., 2021; Kokoska et al., 2019; Vaillancourt et al., 2021). Glabridin (Figure 1), which is an extract and active component of G. glabra, exerts anti-inflammatory effects and can prevent the cleavage of basophils and mastocytes, thereby reducing the symptoms of allergic reactions (Chang et al., 2021). However, the barrier purpose of human skin tissue composition and insolubility of glabridin impede percutaneous infiltration and active absorption of glabridin. Liposomes, which can be used as drug carriers, increase the solubility of insoluble drugs, improve the bioavailability and stability of drugs, and increase local and systemic drug delivery (Gupta et al., 2012; Rattanapak et al., 2012). Glabridin liposomes (GLs) can promote the skin penetration of glabridin to enable active materials to be retained at high concentrations in the skin, which is conducive to its efficacy. Thus, GLs show potential for use in treating AD.
Chemical Structure of Glabridin.
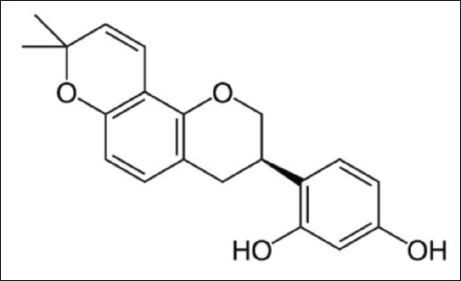
To study the effect of GL on histamine-induced AD, mice were used as an AD model to evaluate the anti-inflammatory effects of GL and investigate its method of action.
Materials and Methods
Materials and Reagents
Glabridin was purchased from Qinghai Lake Pharmaceutical Co., Ltd. (Qinghai, China). Soy lecithin, cholesterol, sodium stearate, butanediol, octanediol, hexanediol, and diphenhydramine hydrochloride were purchased from Aladdin Reagent (Shanghai, China). Saline (0.9%) was purchased from Shandong Qi Du Pharmaceutical Co., Ltd. (Shandong, China). Paraformaldehyde (4%) was purchased from Guangzhou Ding Guo Biology Co., Ltd. (Guangzhou, China). Propylene glycol, the hematoxylin-eosin (HE) staining kit, and enzyme-linked immunosorbent assay (ELISA) kits were provided by Jiangsu Meibiao Biotechnology Co., Ltd. (Jiangsu, China). Anti-FLG rabbit polyclonal antibody (pAb), anti-nerve growth factor (NGF) rabbit pAb, anti-IL-1β rabbit pAb, horseradish peroxidase-conjugated goat anti-rabbit IgG, and 3,3′-diaminobenzidine were purchased from Wuhan ServiceBio Technology Co., Ltd. (Wuhan, China).
GL was prepared using the film dispersion method as previously described (Zhang et al., 2021). First, soy lecithin, sodium stearate, and glabridin were dissolved in hot methanol, mixed with dichloromethane containing cholesterol, and dried. Finally, water, butanediol, and octanediol were added to the dried film and heated to dissolve them.
Particle Size and Morphology
The size distribution and zeta potential of GL were well characterized using Zetasizer (NANO ZS, Malvern Instruments, Malvern, UK) and transmission electron microscopy (JEM-2100, Jeol, Tokyo, Japan).
Animals and Experimental Design
This study used 24 four-week-old female KM mice, which were supplied from the Guangdong Experimental Animal Center. For feeding and connected experiments, the room temperature was kept at 25 ± 2°C, the humidity was maintained at 55%–65%, and the circadian rhythm was a 12-h cycle of day and night. The food and water for the experimental animals were always available. This experiment was carried out following the Guangdong University of Technology norms in terms of both techniques and operations.
The mice were adaptively reared for 1 week and then separated into a control group (n = 4), a group that was positive for diphenhydramine (DPH, n = 4), histamine group (n = 4), glabridin liposome low-dose group (GL-L, n = 4), glabridin liposome medium-dose group (GL-M, n = 4), and glabridin liposome high-dose group (GL-H, n = 4) at random. The day before the experiment, the mice were treated with a depilation agent to remove the hair from their back skin. The experiment was divided into a 1-week administration phase and an acute sensitization phase. At the beginning of the administration in the GL groups, propanediol solution containing GL was applied to the back of the mice where the hair had been removed (dosages in the GL-L, GL-M, and GL-H groups were 0.1%, 0.5%, and 0.9%, respectively, 200 µL). To the DPH group, we applied a 0.7% propanediol solution of hyaluronic acid (200 µL) to the same site on the mice. In the control and histamine groups, propanediol solution (200 µL) was applied to this site. All treatments were administered once daily for 7 days.
On day 7 of administration, an AD mouse model induced by histamine was established. All groups were administered 100 µL (1 mg/mL) of a histamine-saline solution via intraperitoneal injection 30 min after administration (the positive control group was given Benadryl Hydrochloride – saline solution 2 mg/mL/10 g by intraperitoneal injection), apart from the control group. The scratching times of the mice within 30 min were observed using video recording. The back skin tissue was preserved in paraformaldehyde for index assessment after the mice were killed by cervical dislocation.
Behavior Study
The AD mouse model was established after administration, and the scratching times of the mice within 30 min were recorded using a camera. Each time the mouse scratched its ear or back skin was counted as one scratching event (Han et al., 2018).
Pathology Section Analysis
Following the mouse sacrifice, slices of paraffin-embedded skin tissue (4 µm) were produced by removing the back skin tissue and fixing it in a 4% paraformaldehyde solution. The slices were stained with HE to evaluate epidermal hyperplasia and inflammatory cell infiltration and toluidine blue to evaluate mast cell infiltration (Zhang et al., 2017). The epidermal thickness was calculated, and the number of mastocytes was counted using Image Pro Plus (Media Cybernetics, Rockville, MD, USA).
Immunohistochemical Analysis
Paraffin-embedded mouse skin tissue slices (4 µm) were degreased with xylene and ethanol at a certain concentration gradient and then rehydrated. Ethylenediamine tetraacetic acid was used for the recovery of antigens. The sections were processed with 3% hydrogen peroxide to block endogenous catalase, followed by the addition of the primary antibody anti-IL-1β rabbit pAb at a ratio of 1:800. The primary antibody anti-NGF rabbit pAb at a ratio of 1:2000 was incubated overnight at 4°C in a humidified box and then with horseradish peroxidase-conjugated goat anti-rabbit IgG for 1 h. Finally, 3,3′-diaminobenzidine was used for chromogenic treatment, followed by reverse staining of the nucleus with hematoxylin (Xiang et al., 2017). The slices were photographed under a microscope, the integrated optical density was calculated using Image Pro-Plus, and morphometric analysis was performed to profile the contents of NGF and IL-1β in the skin tissue.
ELISA
Mouse dorsal skin tissue was homogenized in RIPA lysis buffer and centrifuged to extract total protein. After sodium dodecyl sulfate-polyacrylamide gel electrophoresis, the proteins were electrotransferred onto a methanol-activated polyvinylidene fluoride (0.45 µm) membrane, which was then incubated in 5% skim milk for 2 h. Anti-cytokeratin 17 rabbit pAb and anti-filaggrin rabbit pAb (1:3000) were inserted, and the samples were incubated overnight at 4°C. Then the samples were washed three times with Tris-buffered saline containing Tween 20, horseradish peroxidase-conjugated goat anti-rabbit IgG (H+L) (1:5000) was added, and they were incubated for 1 h. Finally, the secondary antibody was eluted with Tris-buffered saline containing Tween 20, a chemiluminescence system was used to visualize the specific waveband in a dark room, and data were examined using the ImageJ software (NIH, Bethesda, MD, USA) (Xiang et al., 2018).
Quantitative Real-Time PCR
Mouse skin tissue was mixed with the RNA extract and homogenized at 12,000 r/min for 10 min with a homogenizer. The RNA supernatant was extracted with chloroform, sedimented with isopropyl alcohol, laundered with 75% ethanol, and dissolved in RNase-free water.
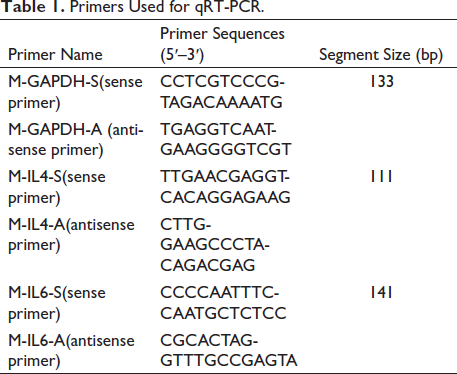
A reverse transcription system was used for polymerase chain reaction (PCR) amplification, and the results were evaluated using the ∇∇CT method (Li et al., 2018). SYBR Green was used to detect RNA expression using quantitative reverse transcription PCR (qRT-PCR). The PCR amplification conditions were pre-denaturation at 95°C for 10 min, followed by 95°C for 15 s and 60°C for 60 s for 45 cycles. The glyceraldehyde dehydrogenase gene was treated as an internal standard. Table 1 lists the primers used for qRT-PCR.
Primers Used for qRT-PCR.
Statistical Analysis
All data are expressed as the mean ± SD (n = 3). Statistical significance was tested using a one-way analysis of variance. *p ≤ 0.05 and **p ≤ 0.01 were regarded as statistically significant outcomes.
Results
Shape and Size of Liposomes
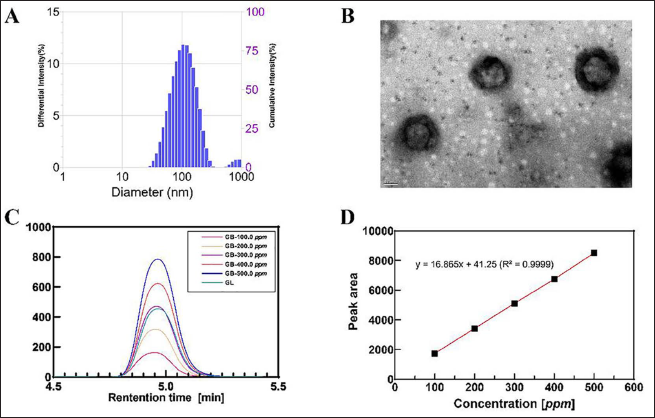
GL was encapsulated using the thin-film dispersion method. The polydispersity index of GL was 0.2 and its average particle size was around 100 nm, as shown in Figure 2A, suggesting that the liposomes’ particle size distribution was uniform. The morphology of GL was visualized using transmission electron microscopy, as shown in Figure 2B. The liposomes had a spherical morphology and were 150–200 nm in size. Glycyrrhizin in the liposomes was identified using high-performance liquid chromatography (Figure 2C); its concentration (2,907 ppm) was calculated using linear regression (Figure 2D). It was determined that the encapsulation efficiency was higher than 95%.
The Physical Properties of Glabridin Liposome. (A) Size Distribution, (B) TEM Image, (C) HPLC Curves, and (D) Linear Regression.
GL Relieved Histamine-induced AD Skin Injury and Scratching Behavior
Mice in the experimental groups were intraperitoneally injected with histamine, and the scratching status of mice in each group was recorded using a video recorder within 30 min. After 30 min, the mice’s skin on their backs was photographed (Figure 3A), and the scratching times were counted based on the video recordings (Figure 3B).
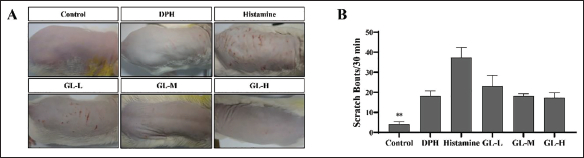
As shown in Figure 3A, histamine injection caused spontaneous scratching behavior because of skin itching in the mice as a typical clinical presentation of AD (Voisin & Chiu, 2018). Compared with the other administration groups, the histamine group showed obvious scratching, erythema, and bleeding in the skin test area. Compared with the histamine group, the DPH and GL groups showed noticeably reduced spontaneous scratching behavior. As shown in Figure 3B, the scratching behavior was induced by histamine, as the scratching frequency of the hind limbs in the histamine group was significantly higher than that in the control group (p < 0.01). When compared with the histamine group, however, the DPH and GL groups showed a significantly reduced number of spontaneous scratching events. Thus, GL exerted dose-dependent effects, and the GL-H group showed a better inhibitory effect on scratching.
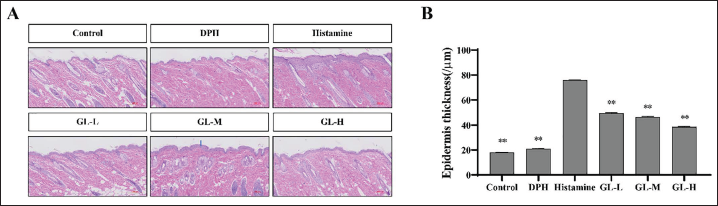
GL Alleviated Histopathological Changes
Epidermal hyperplasia is a trait of allergic reaction-induced skin injury that is frequently used to assess how well medications inhibit epidermal hyperplasia. Figure 4 shows that histological analysis of the skin showed that epidermal hyperplasia in the DPH and GL groups was lower than that in the histamine group, suggesting that the topical use of GL can prevent AD-induced epidermal hyperplasia. The high GL concentration strongly inhibited epidermal hyperplasia. The skin thickness was remarkably increased after histamine injection in comparison with the control group (p < 0.01) (Figure 4B) but significantly decreased in the DPH and GL groups compared with the histamine group (p < 0.01). These findings indicate that high concentrations of GL had a better anti-allergic effect on the skin and a better protective effect against skin inflammation.
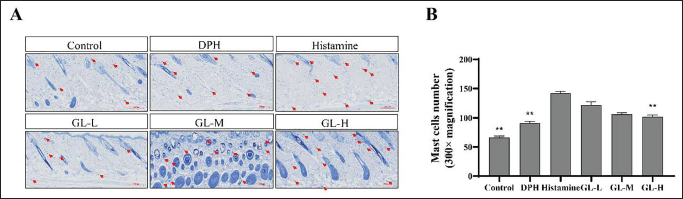
Mast cell infiltration is a characteristic feature of skin inflammation caused by allergic reactions. Toluidine blue staining was performed to analyze the effect of GL on mast cell infiltration. The infiltration of mast cells in the dorsal skin tissue was increased significantly in the histamine group (p < 0.01) (Figure 5). However, mast cell infiltration was markedly lower in the GL-H group than in the histamine group (p < 0.01). Therefore, high concentrations of GL inhibited mast cell infiltration in mouse skin tissue better than the other doses.
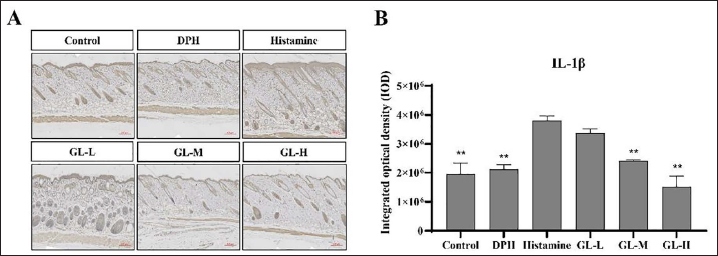
GL Inhibited Inflammatory Factors and Regulated the Expression of Related Channel Proteins
IL-1β is involved in immune activity and is a proinflammatory factor. NGF is a nerve cell growth regulator. However, in the pathological process of AD, mast cells release NGF, which induces the growth of axons of nearby toxic neurons and increases pain at the inflammatory site (Tuszynski et al., 2015). The ELISA was used to quantify IL-1β and NGF expression in the mouse skin tissues. As shown in Figure 6, IL-1β expression was found to be substantially higher in the histamine group (p < 0.01). IL-1β expression was notably lower in the GL-M and GL-H groups than in the histamine group (p < 0.01). The histamine group exhibits a considerable increase in NGF expression as compared with the control group (Figure 6) (p < 0.01), and its expression levels in the GL-M and GL-H groups were significantly lower than those in the histamine group (p < 0.01). These findings imply that GL decreased IL-1β and NGF expression in a dose-dependent manner.
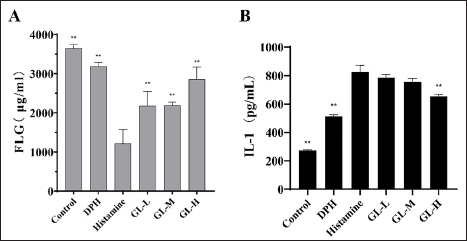
FLG and IL-1 are critical factors in the development of skin inflammation. The expression of FLG and IL-1 in mouse skin tissue was detected using ELISA. As shown in Figure 7A, the expression level of FLG in AD mice was higher in the control and GL groups than in the histamine group (p < 0.01), with the GL high-dose group showing the highest FLG expression. The expression of IL-1 in the skin tissue of control and GL-H group mice was significantly lower than that in the histamine group (p < 0.01) (Figure 7B). The expression of IL-1 in the mouse skin tissue in the GL group declined in comparison with the histamine group, with the GL-H group showing the lowest IL-1 expression.
IL-6 is rapidly produced during the inflammatory response. The expression levels of IL-6 in the mouse skin tissues were detected using qRT-PCR. As shown in Figure 8, IL-6 expression in the histamine group was higher than in the control group. Nevertheless, after GL treatment, the expression of IL-6 in the GL group was lower than that in the histamine group, with the lowest expression observed in the GL-H group.
Discussion
AD, mainly caused by immune imbalance, is among the most common chronic diseases worldwide, decreasing the quality of life of patients. Inflammatory factors and chemokines produced by skin cuticle cells significantly influence the development of AD (Lee et al., 2020; Pastore et al., 2006). Many drugs have been developed for AD, including antibody-based therapies (Sroka-Tomaszewska & Trzeciak, 2021; van der Schaft et al., 2019). However, the side effects of conventional drug therapy cause serious problems in patients (Paller et al., 2016). Therefore, safe and effective anti-specific dermatitis drugs must be developed.
In this study, GL showed anti-allergic activity by relieving skin itching and inhibiting the secretion of inflammatory factors such as tumor necrosis factor-α, IL-1β, IL-6, and NGF in a histamine-induced AD mouse model. AD has a long and repeated course, and frequent scratching can lead to skin damage and hyperplasia (Berke et al., 2012). Medium and high concentrations of GL had a greater effect on relieving histamine-induced itching in AD mice (Figure 3) and reduced epidermal hyperplasia (Figure 4) compared with the other evaluated doses. Mast cells release a series of allergenic mediators through a degranulation reaction, with histamine as one of the main inflammatory mediators (Kawakami et al., 2009; Thangam et al., 2018). During AD development, both mast cell infiltration and mast cell count rise. Application of GL can suppress the proliferation and infiltration of mast cells, reverse the degranulation of mast cells, and relieve skin allergy (Figure 5).
NGF not only provides nutritional support for nerve cells and regulates nerve injury but also performs various activities in the skin, where NGF is mainly produced by keratinocytes (Yamaguchi et al., 2009). However, in AD lesions, NGF levels in the skin tissue are elevated, and anti-NGF antibodies can alleviate skin damage caused by scratching (Dou et al., 2006; Takano et al., 2007). We found that GL relieved allergic skin pain and itching by inhibiting histamine-induced NGF expression (Figure 9).
Human peripheral blood basophils are often used as effector cells to study allergic reactions (Wang et al., 2021). Basophils have high-affinity immunoglobulin E receptors on their surface. The binding of antigen to immunoglobulin E has been reported to lead to the release of inflammatory mediators, the IL-4 and IL-6 receptor signaling pathways are key factors in the onset of disease, and knocking out the genes for these two molecules alone leads to an almost complete blockade of the disease (Molfetta et al., 2010; Lee et al., 2020). Therefore, this study tested the changes in the levels of relevant pro-inflammatory cytokines under the influence of GL. The findings demonstrated that the expression levels of both pro-inflammatory factors IL-4 and IL-6 decreased in the skin tissues of mice after GL treatment, but the differences were not significant (Figure 7), indicating that dermal administration of Glab improved the inflammatory status of the animals, suggesting that GL inhibited the expression of IL-6.
FLG is an important component of the cuticle that forms a physical barrier in the epidermis. During keratinocyte migration, FLG is gradually degraded by enzymes into small molecules required by the cuticle, such as natural moisturizing factors, which play important roles in moisturizing and barrier integrity (Zaniboni et al., 2016). The stability of the cuticle environment depends not only on FLG itself but also on the reaction of its degradation products with related structures and biological processes (Sroka-Tomaszewska and Trzeciak, 2021). GL treatment increased the expression levels of FLG (Figure 7A) and promoted the regular aggregation of keratin fibers, thus promoting the growth of skin keratinocytes and resistance to invasion by exogenous allergens.
Conclusion
In animal experiments, local application of GL alleviated histamine-induced AD-like skin lesions and reduced scratching times in mice, possibly by decreasing the number of mast cells in the skin tissue, leading to decreased secretion of pro-inflammatory factors such as IL-6 and IL-1β. As a neuroregulatory factor, NGF is abnormally expressed in AD models. However, it can regulate NGF to normal levels and maintain its balance in the internal environment. A normal and intact skin barrier is an important indicator of skin health. Local application of GL on the back of mice increased FLG levels in the epidermis, which relieved itching, promoted the healthy growth of cuticle cells, and restored the skin to normal levels. Within a certain concentration range, higher GL concentrations are more effective for treating AD, with a glabridin concentration of 0.9% showing the best efficacy. Therefore, GL shows potential as a drug treatment for AD.
Abbreviations
AD: Atopic dermatitis; GL-M: Glabridin liposomes medium dose group; DAB: 3,3’-diaminobenzidine; GL-H: Glabridin liposomes high dose group; DPH: Di-phenhydraminepositive control group; HE: Hematoxylin-eosin; EDTA: Ethylenediamine tetraacetic acid; IgE: Immunoglobulin E; ELISA: Enzyme linked immunosorbent assay; IL-l: IL-l, IL-6, interleukin; FLG: Filaggrin; IOD: Integrated optical density; GAPDH: Glyceraldehate dehydrogenase; NGF: Nerve growth factor; GL: Glabridin liposomes; PDI: Polydispersity index; GL-L: Glabridin liposomes low dose group; qRT-PCR: Quantitative real-time PCR.
Footnotes
Acknowledgments
The Innovation and Entrepreneurship Leading Team Project of Panyu District (Grant No. 2019-R01-6) and the Guangdong Provincial Key Laboratory of Plant Resources Biorefinery (No. 2021GDKLPRB02) are acknowledged by the authors for their financial support.
Authors’ Contributions
Conceptualization, L.Z.; methodology, Y.L., C.Z., and L.C.; investigation, D.C. and B.Z.; data curation, L.R. and S.Z.; visualization, S.G. and X.X.; writing—original draft, M.T. and Y.A.; writing—review and editing, T.H. The paper has been read and approved by all authors.
Declaration of Conflicting Interests
The authors declared no conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
The experimental procedure and protocol of this study were approved by the Animal Care and Use Ethics Committee of the Guangdong University of Technology.
Funding
This study was supported by the Guangdong Basic and Applied Basic Research Foundation (2020A1515110715) and Guangdong Provincial Key Laboratory of Plant Resources Biorefinery (2021GDKLPRB02).
