Abstract
The nasopharyngeal epithelium, which is frequently found in Southeast Asia and Southern China, gives rise to nasopharyngeal carcinoma (NPC). Despite improvements in diagnostic tools and remedial modalities, the prognosis of NPC remains meager. Thus, innovative and effective anti-cancer agents are desirable. Voacangine (VCG) is a recognized alkaloid sequestered from the plant Voacanga foetida. Hence, the current research assessed the anti-proliferative and apoptotic action of VCG on HK-1 human NPC cells and its underlying molecular actions. The results exposed that VCG (20 and 25 µ/ml) avert the HK-1 cells proliferation, which stimulates apoptosis by the amelioration of Bcl-2-associated X protein (Bax) and caspases, while it lessens cyclin-D1, B-cell lymphoma 2 (Bcl-2), c-Myc, survivin in a dose-dependent way. Furthermore, VCG alleviates inflammation, cell proliferation, and augmented cell death through the attenuation of Nuclear factor kappa B (NF-κB) facilitated Phosphoinositide 3-kinase/Protein kinase B/Mammalian target of rapamycin (PI3K/AKT/mTOR) signaling. This creates a Bax/Bcl-2 proportion imbalance, which triggers caspases cascade, Cyt-c, and induces apoptosis. Our findings deliver novel perceptions into exploring VCG as a beneficial bioactive alkaloid for the handling of NPC.
Introduction
Nasopharyngeal carcinoma (NPC) is a relatively infrequent tumor originating from the nasopharyngeal epithelium, and it forms a kind of head and neck malignancy. NPC is prevalently occurring in Southeast Asia particularly in Southern China (Dou et al., 2014; Huang et al., 2002). The incidence and mortality of NPC in China account for 38.29% and 40.14% respectively for the universal data (Xu et al., 2013). Also, men exhibited higher incidence and mortality of NPC than women; likewise, youngsters are more likely to grieve from NPC, whereas the elderly, exclusively those over 65 years, expose comparatively greater rates of occurrence and death. The leading contributing causes of NPC comprise Epstein–Barr virus (EBV) infection, genetic susceptibility, smoking, alcohol, dietary habits, and workplace exposure (Turati et al., 2017). Currently, radiotherapy and the combination of radiotherapy with chemotherapy are employed for the treatment of NPC. The treatment of radiotherapy may cause radiation harm in the brain (Li et al., 2013), while cisplatin-based medications are the widespread chemotherapeutics used to treat NPC, which are extremely lethal and are likely to drug resistance (Xie et al., 2008). Recently, natural, synthetic, or biological compounds have been revealed as active chemopreventive agents that can prevent, suppress, or delay tumorigenesis progression (Huang et al., 2007). Although traditional remedial approaches have generated favorable therapeutic benefits, the limitations, and non-specificity of these techniques reduce their ability to subdue NPC growth and development (Bossi et al., 2011). Thus, novel molecular therapeutic targets are being comprehensively explored.
Apoptosis is deliberated as a dynamic process comprising typical cell turnover, atrophy, embryonic development, immune function, and programmed cell death (Wang, 1998). An apoptosis commencement is firmly controlled by several activation cascades. Among them, the most recognized activation mechanisms are intrinsic and extrinsic pathways. A report stated that anticancer agents trigger apoptosis, and the distraction of apoptotic programs can lessen treatment effectiveness (Ravi et al., 2000). The family of nuclear factor kappa B (NF-κB) transcription factors stimulates tumorigenesis by inhibiting apoptosis (Shin et al., 2016). NF-κB may be a significant sign in the prompt diagnosis and treatment of cancers (Zheng et al., 2016). An inducible enzyme cyclooxygenase-2 (COX-2) exhibits a vital action in manifold processes of pathophysiological, inflammation, and carcinogenesis (Sheng et al., 2020), and its expression is compactly regulated by the NF-κB activators. COX-2 is recurrently hyper expressed in numerous premalignant, malignant, and metastatic human cancers; NF-κB regulates the transcription of a proliferation of genes chiefly those including inflammatory, immune, and anti-apoptotic reactions (Ghosh et al., 1998). Contemporary reports specified that NF-κB is frequently stimulated in naso pharyngeal (NP) and dealings with NF-κB inhibitors may constrain the progress and incursion of NPC (Thornburg et al., 2003; Wong et al., 2010), hence it is supposed that a possible molecular target is NF-κB for NPC remedy.
Natural products are rich sources of innovative constituents with cytotoxic activity that have been sequestered from numerous floras (Li et al., 2011). It is projected that over 2000 compounds are categorized as alkaloids, a few of them from the Apocynaceae family which are largely consumed as anticancer agents (Lu et al., 2012; Macabeo et al., 2009). Voacangine (VCG) is a renowned alkaloid isolated from the plant Voacanga foetida (Bl) K. Schum (Apocynaceae), which is mostly dispersed in Sumatra, Indonesia. It has been exposed that V. foetida leaves possess anti-inflammatory, antipyretic, and analgesic properties (Susanty et al., 2014). A recent study found that V. foetida leaves also exert promising anticancer and cytotoxic (Susanty et al., 2018) activity owing to their presence of alkaloid VCG (Hadi et al., 2019). Recently, VCG effectively suppresses oral cancer cell growth by activating apoptosis, cell cycle block at the G2/M phase, and subdual of the phosphoinositide 3-kinase/protein kinase B (PI3K/AKT) signaling (Xiao et al., 2020). Earlier studies have described that the PI3K/AKT pathway is crucial in the signal transduction of several growth factors, and is closely associated with the physiological functions of cells, incidence, and development of many cancers (Zheng et al., 2015). Thus, the PI3K/AKT signaling transduction pathway is concerned with growing attenuation.
Therefore, the current research examined VCG-induced cytotoxicity and apoptosis in NPC human HK-1 cells. Our findings established that VCG triggered apoptosis through NF-κB mediated P13K/AKT/mTOR signaling pathways in HK-1 cells.
Materials and Methods
Chemicals and Reagents
VCG, Roswell Park Memorial Institute (RPMI)-1640, fetal bovine serum (FBS), antibiotics, phosphate-buffered saline (PBS), dimethyl sulfoxide (DMSO), 3-(4,5-dimethylthiazolyl-2)-2,5-diphenyltetrazolium bromide (MTT), 4′,6-diamidino-2-phenylindole (DAPI), propidium iodide (PI), and sodium dodecyl sulfate (SDS) were acquired from Gibco (CA, USA). Enzyme-linked immunosorbent assay (ELISA) kits were bought from Abnova, China. The antibodies for western blot analysis were procured from Beyotime Biotechnology, USA.
Cell Culture
Human NPC cell lines HK-1 were gained from Shanghai Aiyan Biotechnology Co., Ltd., (Shanghai, China), and grown in the medium RPMI-1640 added with FBS (10%); penicillin (100 U/ml), and streptomycin (100 U/ml), maintaining CO2 (5%) at 37°C in a humidified chamber.
Cell Cytotoxicity Assay
Human HK-1 cell cytotoxicity was assessed by employing the MTT test (Mosmann, 1983). Concisely, NPC cells HK-1 were propagated into 96-wells (1,000 cells/well) and cultured at 37°C in a wet incubator ensuring 5% CO2. Once overnight preservation, the medium was separated, the cells were dipped with PBS, and incubated with diverse quantities of VCG (5, 10, 15, 20, 25, 30, and 35 µM) for 1 day. Successively, the preserved cells were treated with MTT (10 µL) solution and sustained for an additional 4 h to let the transformation of MTT into formazan insoluble crystals by the mitochondrial dehydrogenase. The subsequent formazan was dissolved by adding 150 µL of DMSO. Then, optical density (OD) was determined by employing an ELISA plate reader at 490 nm. Cell proliferation was determined as a % of proliferation against untreated HK-1 cells (100%). IC50 values were calculated and selected concentrations were used for further studies.
Apoptosis Assessed by DAPI Staining
Human HK-1 cells were sowed 1 × 105 cells in 96 well plates and supplemented to VCG (20 and 25 µM) stationary with paraformaldehyde (4%) at 37°C for 10 min. These preserved HK-1 cells were stained with DAPI to assess the nucleus changes allied with apoptosis by the method described previously (Zheng et al., 2015). Then, the samples were mounted on a glass slip and perceived through a fluorescence microscope BX51 (Olympus, Japan).
Evaluation of PI Staining for Apoptosis
The apoptosis nuclei were explored by PI staining has performed. HK-1 human cells were preserved with diverse dosages of VCG (20 and 25 µM) and conserved for 48 h. Then, treated cells were gathered, and PI staining was allowed into the protocol (Zheng et al., 2015).
Measurement of Caspase-9 and Caspase-8
Apoptosis was measured by employing the ELISA kit rendering to the company’s provided protocol, and three independent replicates were performed.
Determination of mRNA Levels
Total RNA was isolated from human NPC cells HK-1 affording to the company’s techniques by employing TRIzol® reagent (Abcam, USA). The isolated RNA is transformed to cDNA through reverse transcription by consuming a cDNA Reverse Transcription high-capacity kit (Beyotime Biotechnology, USA) ensuring the company’s procedures. Then, the Fast Start master mix SYBR Green (Abcam, USA) was employed to explore the cDNAs rendering to the manufacturer’s procedures. The band intensity was scrutinized by using 1.5% agarose gels when they were exposed to electrophoresis; finally, the band intensity was measured by ImageJ 1.48 software. A technique of comparative threshold cycle (Ct) was employed to compute the fold variations in each gene of expression by formula 2−(∇∇Ct).
Statistical Examination
The statistical test was accomplished by GraphPad prism software version 8.0.1 was employed to perform an analysis of variance (ANOVA) and afterward Duncan’s trial. The results were stated as mean ± SD. p < 0.05 was measured as significant statistically.
Results
Anti-proliferative and Cytotoxic Effects of VCG on NPC Human Cells
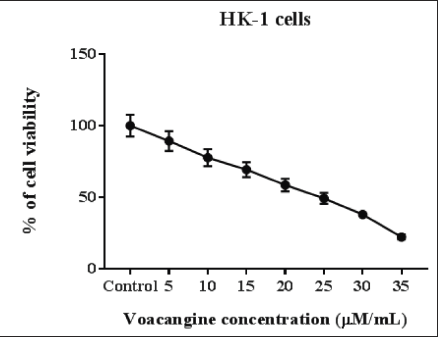
The cytotoxic action of VCG on human NPC cells HK-1 was assessed by MTT analysis with diverse concentrations of VCG (Control, 5, 10, 15, 20, 25, 30, and 35 µM/ml). The results exposed that VCG facilitates cytotoxic and anti-proliferative actions on HK-1 cancer cells in a dosage-reliant manner. VCG treatment at a dosage below 10 µM could not prevent NPC cell proliferation. However, 15, 20, 25, 30, and 35 µM concentrations of VCG considerably reduced (p < 0.05) HK-1 cells viability than untreated cells. At a higher concentration of 30 and 35 µM VCG impaired HK-1cells. From the MTT test, we noticed the IC50 value of VCG was 25 µM for HK-1 cells. Established on the figure of the inhibitory concentration data of 20 and 25 µM VCG has been chosen for auxiliary experiments (Figure 1).
VCG-triggered Apoptosis on HK-I Cells Evidenced by DAPI Staining
Human NPC cells HK-1 treated with DAPI staining unveiled the survival of usual viable cells with normal nuclei. VCG-administered HK-1 cancer cells triggered the apoptosis that enhanced the nuclear morphology and nuclear bodies’ disintegration against untreated HK-1 control cells. VCG (20 and 25 µM/ml) exposed to HK-1 cells perceived chromatin reduction, membrane blebbing, the devastation of the nuclear envelope, and cellular collapse. These outcomes highlight that VCG-triggered apoptosis in a dosage-dependent manner (Figure 2).
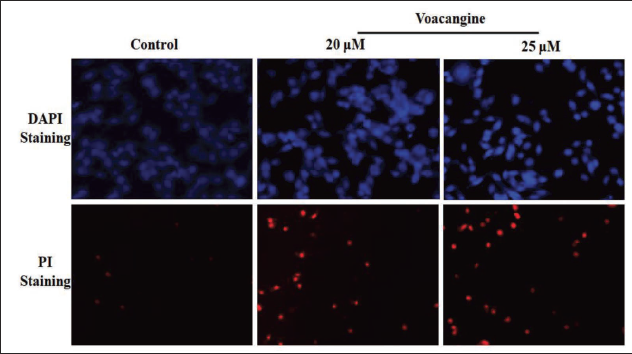
VCG-stimulated Apoptosis on HK-1 Cells Evidenced by the Staining of PI
Staining with PI was effective in differentiating the apoptotic nuclei of NPC cells HK-1. PI stain moves into the cells after lost membrane integrity, which is connected with the weakening of membrane polarization, pointing to cell death. The staining with PI revealed that VCG influenced apoptotic actions on HK-1 in a concentration-reliant way. VCG administration (20 and 25 µM/ml) augmented the effect of apoptosis counter to untreated HK-1 control cells. Hence, VCG-driven apoptosis could be one mechanism for averting NPC cell proliferation (Figure 2).
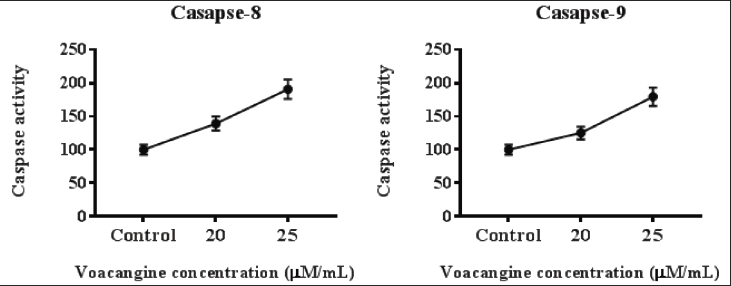
VCG-enhanced the Levels of Caspase-8 and Caspase-9 Detected by ELISA
Administration of VCG on human NPC cells HK-1 presented higher levels of caspase-8 and -9 in contrast to untreated cells. VCG at a concentration of 25 µM pointedly (p < 0.05) elevated the quantity of caspase-8 and -9 more than 20 µM VCG treatment. VCG enhanced the caspase activity in a quantity-reliant manner (Figure 3).
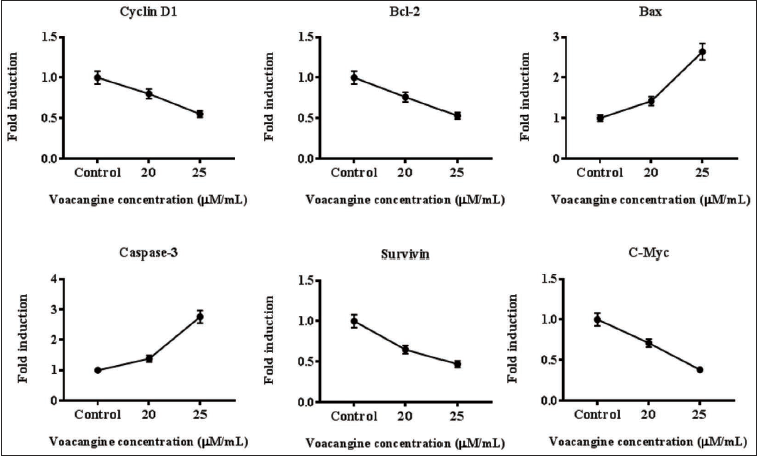
The Stimulus of VCG on the mRNA Expression of HK-1 Cells
Treatment of VCG (20 and 25 µM/ml) into HK-1 cells for measuring the levels of mRNA in apoptotic proteins. HK-1 control cells displayed amplified mRNA expression of cyclin-D1, B-cell lymphoma 2 (Bcl-2), c-Myc, and Survivin, however, Bcl-2-associated X protein (Bax) and caspase-3 were alleviated. VCG expressively mitigated Bcl-2, c-Myc, cyclin-D1, and survivin mRNA levels, whereas caspase-3 and Bax were ameliorated in an amount-reliant mode against HK-1 control cells (Figure 4).
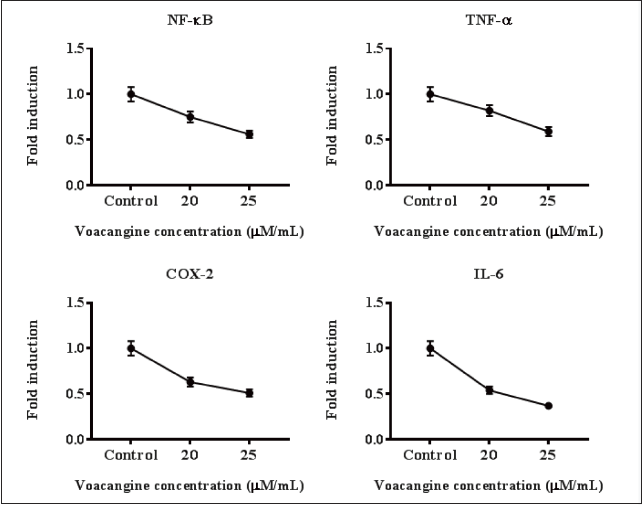
VCG Averted Inflammatory Markers Level on HK-1 Cells
To uncover the anti-inflammatory action of VCG on HK-1 cells the inflammatory cytokines’ mRNA expression was assessed. The levels of TNF-α, COX-2, interleukin-6 (IL-6), and NF-κB were augmented in HK-1 cells, while VCG-treated HK-1 cells reduced these mRNA levels in a quantity-reliant way. These outcomes emphasize that VCG attenuated inflammatory markers in a dosage-needy way (Figure 5).
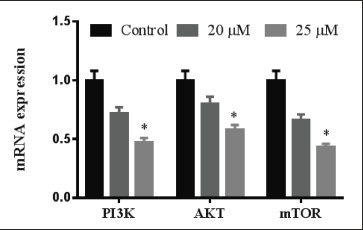
VCG Suppressed P13K/AKT/mTOR Pathway
P13K/AKT/mTOR signaling has a substantial action in cancer cell growth and development. Human NPC cells HK-1 have exhibited that P13K, AKT, and mTOR mRNA levels were enhanced. Treatment with VCG (20 and 25 µM/ml) in the HK-1 cells attenuated the P13K/AKT/mTOR levels. These levels of mRNA were mitigated by VCG in a quantity-needy way over HK-1 cells (Figure 6).
Discussion
NPC is an endemic cancer, that has racial and geographical dispersal inclination that is utmost communal in certain parts of Asia, predominantly Southeast China (Dou et al., 2014; Huang et al., 2002). Southern China and Hong Kong have a high incidence of NPC which is known as “Guangdong Tumor” (Dou et al., 2014; Huang et al., 2002; Xu et al., 2013). Although the precise cause of NPC is still unknown, it is thought to be caused by a viral infection, as well as by genetic, environmental, and nutritional factors (Ozoya et al., 2016; Turati et al., 2017). Herbal bioactive substances have been gradually described as an alternative to chemotherapy and its associated harmful side effects in the prevention and treatment of NPC. Data from the current experiments established that VCG treatment expressively repressed cell proliferation and triggered in HK-1 cells apoptosis over the inhibition of NF-κB-related cytokines and P13K/AKT/mTOR signaling pathways in a dosage-dependent mode.
Alkaloids comprise an assorted collection of herbal metabolites with well-testified anti-tumor activities. These alkaloids have been exposed to employ anticancer actions by various molecular mechanisms including cytotoxic, apoptosis, autophagy, and blockage of the cell cycle (Lu et al., 2012; Macabeo et al., 200). VCG (12-methoxyibogamine-18-carboxylic acid methyl ester) is an alkaloid found predominantly in the root bark of the Voacanga africana tree, as well as in other plants such as Tabernanthe iboga, Tabernaemontana africana, Trachelospermum jasminoides, Tabernaemontana divaricata, and Ervatamia yunnanensis. VCG is an iboga alkaloid and a frequently used precursor for the semi-synthesis of ibogaine. In trials on animals, VCG was found to have similar anti-addictive properties to ibogaine itself. Barbiturates’ effects are also amplified by it. It is soluble in ethanol and its crystals glow a blue-green color when exposed to UV-A and UV-B light. Herein, we explored the anticancer activity of an alkaloid VCG against human NPC cells HK-1. Prior research revealed that V. foetida leaves exert auspicious anticancer and cytotoxic (Susanty et al., 2018) activity owing to their presence of alkaloid VCG (Hadi et al., 2019). Figueiredo et al. (2010) isolated an alkaloid, VCG from Tabernaemontana salzmannii and demonstrated cytotoxic effects on human leukemia cells (THP-1) over the induction of apoptosis. Wolter et al. (1997 have reported the cytotoxic and genotoxic potential of VCG against laryngeal epithelial carcinoma human cells (Hep-2). Recently, VCG alkaloid has been found to effectively inhibit the proliferation of oral cancer cells by triggering cell death and the G2/M phase cell cycle block through the regulation of PI3K/AKT signaling (Xiao et al., 2020). In the current study for the first time, VCG unveiled selective proliferative reticence against the NPC human cells HK-1, such selective inhibitory potential has been detected for further natural bioactive compounds also (Shamas-Din et al., 2013; Wolter et al., 1997). This research confirmed that VCG repressed the HK-1 cells’ viability in a concentration-reliant way evidenced by the MTT assay. The preventive potential of VCG on the HK-1 cell proliferation was owing to the stimulation of apoptosis. This finding is in conforms to preceding research wherein numerous plant-derived VCG have been exhibited to avert the growth of malignant cells through the induction of apoptosis (Rizo et al., 2013; Zou et al., 2019).
Apoptotic generation in malignant cells is a vital beneficial approach for tumor management. After administration of VCG, the apoptosis induction was confirmed by DAPI and PI staining, that the morphological variations of the HK-1 cells proved the hallmarks of cell death. The number of early and late apoptotic HK-1 cells was elevated in VCG-given cells than in untreated control; hence the results proposed that VCG was able to prompt apoptosis in HK-1 cells. Our outcomes are consistent with those in the aforementioned trials, where VCG was exposed to prompt apoptosis in tumor cells (Zou et al., 2019). The Bcl-2 family proteins contain pro-apoptotic Bax and anti-apoptotic Bcl-2, which are intricate in the control of mitochondrial-mediated cell death (Wu et al., 2015). Bcl-2 is an apoptosis controller that has been affecting the usual apoptosis of impaired DNA cells, abnormal cells, as well as cancer cells (Sun et al., 2012). In NPC cells, the Bcl-2 proteins are greatly expressed, which mentioned that Bcl-2 proteins may aid a significant action in the early phase and the hyperexpression of Bcl-2 may also be interrelated to the restrain of apoptosis (Lo et al., 2006). The present study confirmed that VCG enhanced caspase-3, caspase-8, caspase-9, and Bax levels, whereas reducing the mRNA level of Bcl-2 in HK-1 cells.
NF-κB belongs to a tumorigenesis proteins family, which are numerous functional proteins encoded by NF-κB that might stimulate cancer growth. There are two NF-κB binding sites existing in the cyclin-D1 promoter region that the stimulation of NF-κB upholds the cyclin-D1 expression and G1/S, which speed up cell cycle development (Zheng et al., 201). NF-κB may also stimulate tumorigenesis by preventing apoptosis. Contemporary reports point out that NF-κB is generally triggered in NPC, and several indications specify the connection between NPC tumorigenesis and NF-κB. According to earlier studies, NF-κB hyperexpression predicts the absence of an NPC diagnosis. The designation NF-κB indicates that it may be a healing mark in NPC (Cui et al., 2001). Presently, it is supposed that the EBV contagion and pro-inflammatory cytokines including COX-2, TNF-α, and IL-6 are powerful incitements of NF-κB stimulation in patients having NPC (Faqing et al., 2005; Wang et al., 2002). Numerous arguments have been made that the transcriptional factor NF-B is essential for the NPC tumor-associated genes cyclin-D1, survivin, and c-Myc (Hay, 2005; Thippeswamy et al., 2008). Our findings demonstrated the attenuation of transcriptional NF-κB action by VCG’s cause had reduced NF-κB target genes expression containing cyclin-D1, survivin, and c-Myc. Hence, the anti-proliferation action of VCG was probably related to the mitigation of survivin, cyclin-D1, and c-Myc in NPC cells, while an anti-inflammatory activity of VCG may be owing to the alleviation of cytokines such as NF-κB, COX-2, TNF-α, and IL-6. These findings were reliable to the preceding reports on the inhibition of NF-κB in diverse cancer cells (Engelman et al., 2006).
The signaling PI3K-AKT-mTOR is the main path involved in tumor development (Ocana et al., 2014), and it is recurrently transformed as well as hyper-activated in various human malignancies (Choe et al., 2003). The PI3K-AKT-mTOR signaling stimulates survival by constraining the pro-apoptotic and initiation of anti-apoptotic elements. Through the phosphorylation of route constituents, this pathway suppresses the activity of pro-apoptotic members while inducing anti-apoptotic proteins (Choe et al., 2003). Therefore, PI3K/AKT/mTOR signal suppression has been identified as a target for cancer therapeutic strategies. According to a report, VCG prevents oral cancer cell lines from undergoing apoptosis by inhibiting PI3K/AKT signaling. In this study, our results established for the first time that VCG suppresses PI3K/AKT/mTOR activity in HK-1 cells, which may be associated with attenuation of NPC cell proliferation and triggering of apoptosis, suggestive of its anticancer potential.
Conclusion
In summary, in the current research, we have established that VCG could expressively avert cell viability and promote apoptosis in HK-1 human cells. VCG ameliorated the activities of Bax, caspase-8, caspase-3, and caspase-9 while reducing the Bcl-2, cyclin-D1, survivin, c-Myc, and cytokines mRNA levels in HK-1 cells through the regulation of NF-κB facilitated PI3K/AKT/mTOR pathway. Hence, these findings may convey VCG as an innovative candidate for the development of potential NPC remedies.
Abbreviations
NPC: nasopharyngeal carcinoma; VCG: voacangine; NF-κB: nuclear factor kappa B; PI3K: phosphoinositide 3-kinase; AKT: protein kinase B; mTOR: mammalian target of rapamycin; Bax: Bcl-2-associated X protein; Bcl-2: B-cell lymphoma 2; NPC: nasopharyngeal cancer; EBV: epstein–barr virus; COX-2: cyclooxygenase-2; RPMI: roswell park memorial institute; FBS: fetal bovine serum; PBS: phosphate-buffered saline; DMSO: dimethyl sulfoxide; MTT: 3-(4,5-dimethylthiazolyl-2)-2,5-diphenyltetrazolium bromide; DAPI: 4′,6-diamidino-2-phenylindole; PI: propidium iodide; SDS: sodium dodecyl sulfate; CO2: carbon dioxide; ELISA: enzyme-linked immunosorbent assay; ANOVA: analysis of variance; IL-6: interleukin-6; NP: naso pharyngeal.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Summary
VCG inhibits cell proliferation of nasopharyngeal carcinoma cells. VCG-induced apoptosis through suppressed PI3K/AKT/mTOR signaling pathway.
Statement of Ethical Approval and Informed Consent
Necessary ethical clearances and informed consent were obtained before initiating the study from all the participants.
