Abstract
Objective
To investigate the clinicopathological significance of NF-κB p65 and IKKβ protein and mRNA expression in nasopharyngeal carcinoma (NPC) patients from Guangdong Province, China.
Methods
Data and tissues from patients with NPC were retrospectively studied. Immunohistochemical staining and quantitative reverse transcription polymerase chain reaction were used to evaluate and compare NF-κB p65 and IKKβ protein and mRNA levels, respectively, in 60 NPC and 30 nasopharyngitis tissue samples. Statistical analysis was conducted to determine correlations between NF-κB p65 and IKKβ protein and mRNA levels with clinicopathological characteristics and prognoses of NPC patients.
Results
NF-κB p65 and IKKβ protein and mRNA expression in NPC were significantly correlated with tumor size, lymph node metastasis, and TNM stage. NF-κB p65 and IKKβ protein and mRNA levels were significantly increased in NPC patients with deep tumor invasion (T3–4), lymph node metastasis, and stage III/IV disease; high NF-κB p65 and IKKβ mRNA expression were associated with significantly shorter disease-free survival rates compared with cases showing low NF-κB p65 and IKKβ mRNA expression.
Conclusions
NF-κB p65 and IKKβ may affect the prognosis of NPC patients and could be potential therapeutic targets for this disease.
Keywords
Introduction
Nasopharyngeal carcinoma (NPC) is a malignant tumor with a distinct racial and geographical distribution that is highly prevalent in Southern China and Southeast Asia. 1 China is among the countries with the highest incidence and mortality rates of NPC. 2
Especially in Guangdong Province, the incidence rate of NPC has been increasing annually. Etiological factors of NPC include genetics, Epstein–Barr virus (EBV) infection, environment, smoking, and other factors. 3 Numerous studies have indicated that the occurrence of NPC is closely related to EBV infection. 4 Radiotherapy is the best therapeutic choice for NPC. Although radiotherapy techniques have become more advanced, approximately 20% of patients develop local recurrent disease after radiotherapy, and 15% to 30% of patients experience treatment failure because of distant metastases. 5 The molecular mechanisms underlying NPC recurrence and metastasis remain unclear; therefore, it is necessary to further explore these mechanisms to find molecular targets that can predict prognosis and/or guide targeted therapy.
Nuclear transcription factor-κB (NF-κB) is a pivotal multifunctional transcription factor that is involved in numerous biological functions including cell growth, survival, immunity, and inflammation; 6 NF-κB dimers are activated by IKK-mediated phosphorylation of IκBs, and the IKK complex (IKKα, IKKβ and IKKγ) plays an important role in activating the NF-κB pathway; IKKβ is a key catalytic subunit of the IKK complex. 7 Activation of NF-κB transcription factors can significantly increase cancer cell proliferation, inhibit apoptosis and autophagy, and facilitate the neovascularization that results invasion and metastasis. 8 Overactivation of NF-κB signaling has been shown to contribute to carcinogenesis and tumor development in various cancers, 9 but its roles in NPC cases from Guangdong Province have not been fully elucidated.
In this study, we analyzed the clinicopathological significance of NF-κB p65 and IKKβ in NPC patients to explore the relationship between NF-κB p65, IKKβ, and prognosis in NPC patients.
Materials and methods
Study subjects
This study included NPC biopsy and nasopharyngitis tissue samples from patients treated at the Traditional Chinese Medical Hospital of Zhongshan between January 2018 and July 2019. Tumor tissues were diagnosed as NPC by preoperative imaging and postoperative pathology. The mean age of the NPC patients was 49.1 ± 14.0 years, with a male-to-female ratio of 2.2 (41 men and 19 women). Clinical variables were studied from patient medical records including age, sex, tumor size and differentiation, tumor node metastasis (TNM) stage, and lymph node metastasis. The eligibility criteria for the study were: (1) age ≥18 years and Karnofsky performance score ≥70; (2) a NPC confirmed diagnosis via histopathological examination; and (3) no history of anticancer therapy. The exclusion criteria were as follows: (1) evidence of concomitant tumors at diagnosis; (2) insufficient heart, lung, liver, or renal function; and (3) severe anemia, acute infection, or autoimmune diseases.
Among the 60 NPC patients, 58 were treated with intensity modulated radiotherapy (IMRT) and two did not receive treatment. IMRT was delivered once daily, 5 fractions per week, for 7 weeks. No fewer than three cycles of cisplatin-based chemotherapy were given for patients with stage III–IV disease. The follow-up period was measured from the date of diagnosis to the time of the last clinical visit or death.
This research protocol was reviewed and approved by the Ethics Committee and Institutional Review Board of the Traditional Chinese Medical Hospital of Zhongshan, and written informed consent for study inclusion and publication was obtained from all patients included in the study. The reporting of this study conforms to the STROBE (STrengthening the Reporting of OBservational studies in Epidemiology) guidelines. 10
Immunohistochemistry (IHC) for NF-κB p65 and IKKβ
Formalin fixed paraffin-embedded (FFPE) NPC tissue specimens (N = 60) and nasopharyngitis tissue specimens (N = 30) were obtained from the Pathology Department of the Traditional Chinese Medical Hospital of Zhongshan. NF-κB p65 and IKKβ protein levels in the tissue specimens were assessed by IHC. The IHC staining procedures were as follows. Briefly, all specimens were sliced into 4- μm-thick sections, which were de-waxed in xylene and a graded alcohol series, hydrated, and washed with phosphate-buffered saline (PBS). Samples were then immersed in 3% H2O2. Subsequently, primary antibodies against NF-κB p65 and IKKβ (1:200 and 1:100 dilution, respectively, both from Abcam, Cambridge, UK) were incubated with the slices for 2 hours at 37°C. Secondary antibody (Maixin Biotech, Fuzhou, China) was then applied for 30 minutes. Samples were stained with 3,3-diamino-benzidine tetrahydrochloride (DAB; Maixin Biotech), and then rinsed with distilled water. Slides were counterstained with hematoxylin. IHC scores were calculated on the basis of multiplying the staining intensity (0, negative; 1, weak; 2, moderate; 3, strong) by the extent of immunoreactive cells (0%, 0; 1%–10%, 1; 11%–50%, 2; 51%–80%, 3; 81%–100%, 4). The interpretation of final scores was as follows: 0–1 (0, negative), 2–4 (1+, weakly positive), 6–8 (2+, moderately positive), and 9–12 (3+, strongly positive).
Quantitative reverse transcription polymerase chain reaction (RT-qPCR)
RNA was extracted from FFPE tissues using the RNA Maxi Kit (QIAGEN, Hilden, Germany) according to the manufacturer's protocol. RNA concentrations were measured using a NanoDrop ND-1000 spectrophotometer at 260nm and 280nm (Thermo Fisher Scientific, Waltham, MA, USA). Total RNA (5 μg) or poly(A) RNA (up to 500 ng) was converted into cDNA using the GoScript™ Reverse Transcription System (Promega, Madison, WI, USA). RT-qPCR was conducted to measure mRNA expression using a fluorescence quantitative PCR amplifier (AB71700 ABI; Thermo Fisher Scientific).
Amplification reactions were performed in a 30-μL volume comprising 1 μL of template cDNA, 1 μL of sense primer, 1 μL of antisense primer, and 15 μL of Master Mix (Takara, Dalian, China). The reaction conditions consisted of 94°C for 5 minutes followed by 40 cycles of 94°C for 30 s, 56°C for 30 s, and 72°C for 30 s, with a final extension at 72°C for 5 minutes. The cDNA was used as a template for PCR using SYBR Premix Ex Taq™ (Perfect Real Time) kits (Takara). Real-time PCR was performed with the following cycle profile: 30 s of preincubation at 95°C followed by 40 cycles of 95°C for 5 s and 60°C for 30 s. The specificity of the PCR amplification was validated by a single peak in the melting curves. GAPDH was used as the internal reference, and duplicate assays for each sample were conducted. The relative quantification (RQ) of target gene expression was calculated by the 2−ΔΔCT method. Primer sequences are listed as follows: NF-κB p65 forward: 5′-CGCGGATCCGCCACCATGGACGAACTG-3′ and NF-κB p65 reverse: 5′-CCGCTCGAGTTAGGAGCTGATCTG-3′; IKKβ forward: 5′-CCGACAGAGTTAGCACGACA-3′ and IKKβ reverse: 5′-GGCAATCTGCTCACCTGTTT-3′; GAPDH forward: 5′-TCGACCAGTCAGCCGCATCTTCTTT-3′ and GAPDH reverse: 5′-ACCAAATCCGTTGACTCCGACCTT-3′.
Statistical Analysis
Statistical analyses were performed using SPSS version 18.0 software (SPSS Inc., Chicago, IL, USA). Measurement data with a skewed distribution are expressed as median (interquartile range) and were analyzed by the Mann–Whitney U test. Comparisons of the frequencies between cases and controls were performed using the χ2 test. Disease-free survival (DFS) rates were analyzed using Kaplan–Meier survival curves and single-factor log-rank tests. P < 0.05 was considered statistically significant.
Results
NF-κB p65 and IKKβ protein expression in NPC and nasopharyngitis tissues
This study included 60 NPC biopsy tissue samples and 30 nasopharyngitis tissue samples. We evaluated NF-κB p65 and IKKβ protein expression in NPC and nasopharyngitis tissues by IHC. We found that NF-κB p65 and IKKβ staining were primarily observed in the cytoplasm of NPC cells. Cancer tissues were found to be strongly positive for NF-κB p65 and IKKβ, whereas negative or weak staining was observed in nasopharyngitis tissues (Figure 1a–1d). The positive expression rate of NF-κB p65 protein was 88.3% in NPC and 60% in nasopharyngitis, the positive expression rate of IKKβ protein was 56.7% in NPC and 33.3% in nasopharyngitis. These results showed that NF-κB p65 and IKKβ protein expression were significantly higher in NPC tissues than in nasopharyngitis tissues (P < 0.05, Table 1).

Immunohistochemical staining (IHC) showing (a) positive NF-κB p65 expression in nasopharyngeal carcinoma (NPC) tissue, (b) negative NF-κB p65 expression in nasopharyngitis tissue, (c) positive IKKβ expression in NPC tissue, and (d) negative IKKβ expression in nasopharyngitis tissue (magnification: 100× for all images).
NF-κB p65 and IKKβ protein expression in nasopharyngeal carcinoma (NPC) and nasopharyngitis tissues.

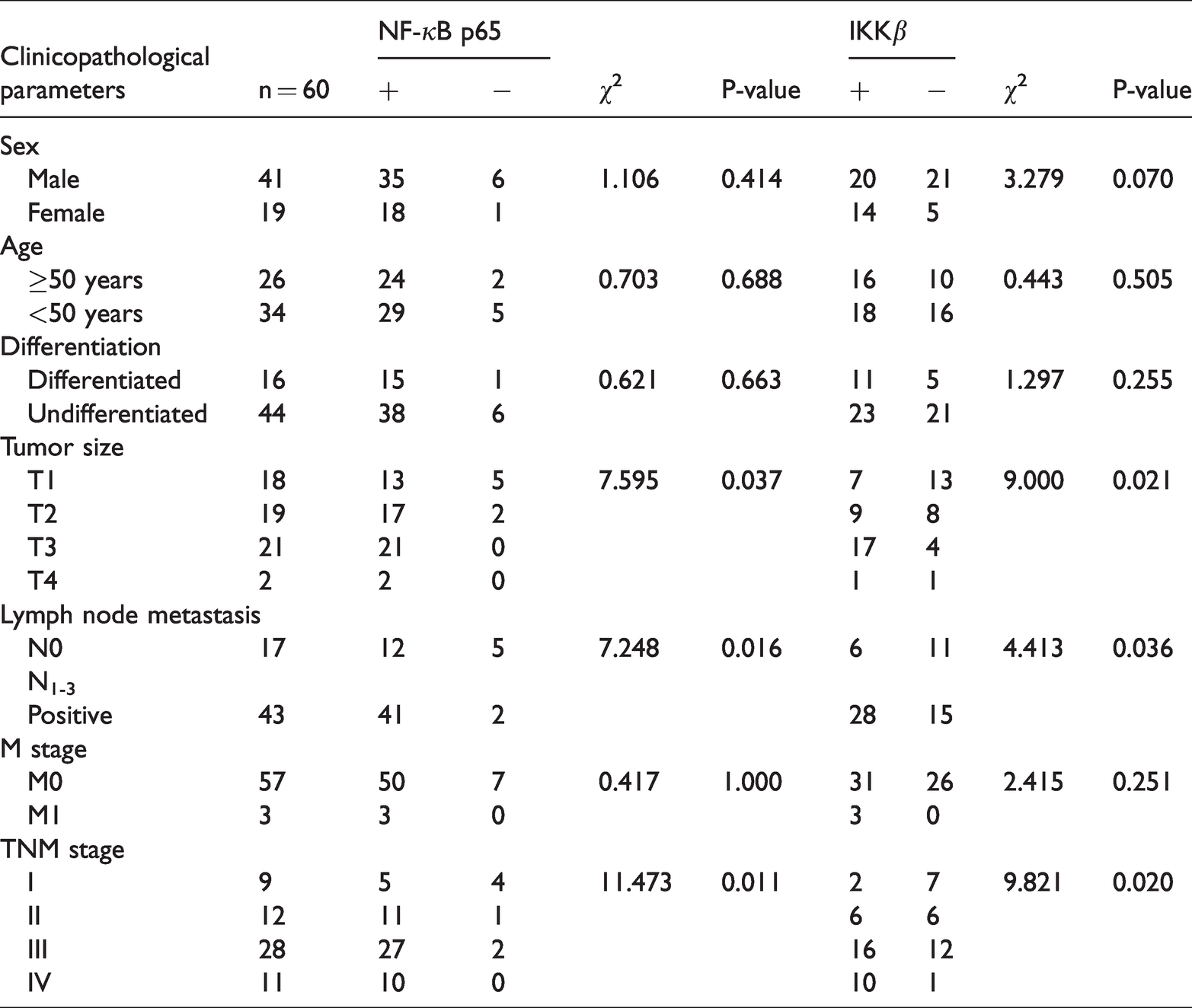
Relationships between clinicopathological characteristics and NF-κB p65 and IKKβ protein expression in NPC
NF-κB p65 and IKKβ protein expression in NPC were significantly correlated with tumor size, lymph node metastasis, and TNM stage (P < 0.05). NF-κB p65 and IKKβ protein expression were significantly higher in NPC patients with advanced T stage (T3–4), lymph node metastasis, and higher TNM stage (stage III/IV) (P < 0.05); NF-κB p65 and IKKβ protein expression were significantly higher in NPC patients with stage T3 disease than in those with stage T1 malignancies (P < 0.05); however, NF-κB p65 and IKKβ protein expression did not differ significantly in NPC patients with stages T1 and T2 disease. No correlation was observed between NF-κB p65 and IKKβ protein expression and tumor differentiation, M stage, patient age, or sex (Table 2).
Relationships between NF-κB p65 and IKKβ protein expression and clinicopathological characteristics in nasopharyngeal carcinoma (NPC).
Differences between NF-κB p65 and IKKβ mRNA levels in NPC and nasopharyngitis tissues
NF-κB p65 mRNA expression was calculated by 2−ΔΔct method using a relative quantification approach. NF-κB p65 mRNA expression did not differ significantly between NPC 0.2832 (0.1252, 0.7740) and nasopharyngitis 0.4538 (0.1833, 1.4667) tissues. IKKβ mRNA expression also did not differ significantly between NPC 1.0497 (0.7631, 1.9318) and nasopharyngitis 1.4340 (1.2295, 1.6358) tissues.
Relationships between clinicopathological characteristics and NF-κB p65 mRNA expression in NPC
RT-qPCR results showed that NF-κB p65 mRNA expression was significantly higher in NPC patients with deep tumor invasion (T3–4), lymph node metastasis, and stage III/IV disease (P < 0.05). NF-κB p65 mRNA levels were correlated with tumor size, lymph node metastasis status, and TNM stage in NPC. NPC patients with stage T3 disease had significantly higher NF-κB p65 mRNA levels than those with stage T1 disease (P < 0.05); however, NF-κB p65 mRNA levels in NPC did not differ significantly in cases with T1 and T2 disease. NF-κB p65 mRNA levels showed no correlation with tumor differentiation, M stage, patient age, or sex (Table 3).
Relationships between NF-κB p65 mRNA expression and clinicopathological characteristics in nasopharyngeal carcinoma (NPC).

Relationships between clinicopathological characteristics and IKKβ mRNA expression in NPC
IKKβ mRNA expression was significantly higher in NPC patients with deep tumor invasion (T3–4), lymph node metastasis, and stage III/IV disease (P < 0.05). IKKβ mRNA expression was correlated with tumor size, lymph node metastasis status, and TNM stage in NPC. IKKβ mRNA levels were significantly higher in NPC patients with stage T3 compared with those with stage T1 disease (P < 0.05); however, IKKβ mRNA expression levels did not differ significantly in stage T1 and T2 NPC. IKKβ mRNA levels showed no correlation with tumor differentiation, M stage, patient age, or sex (Table 4).
Relationships between IKKβ mRNA expression and clinicopathological characteristics in nasopharyngeal carcinoma (NPC).

Correlations between NF-κB p65 and IKKβ mRNA expression and the prognosis of NPC patients
The follow-up time was between 25 and 32 months, with a median follow-up time of 29.0 months and a mean of 27.9 months. As of the last follow-up, 59 patients survived and 1 had died. Next, the cases were divided into high and low expression groups according to the median NF-κB p65 and IKKβ mRNA expression levels in NPC. DFS curves of the NF-κB p65 and IKKβ high and low expression groups in NPC patients were drawn for 1 to 32 months after radiotherapy. The DFS rates of the high and low NF-κB p65 expression groups were 70.0% (21/30) and 92.9% (26/28) respectively. The DFS rates of the high and low IKKβ expression groups were 69.2% (18/26) and 90.6% (29/32), respectively. These results showed that patients with high NF-κB p65 and IKKβ expression had significantly shorter DFS rates than those with low NF-κB p65 and IKKβ expression (Figure 2a–b, P = 0.037 and P = 0.042, respectively).

Relationships between NF-κB p65 and IKKβ mRNA levels and disease-free survival (DFS) rates in nasopharyngeal carcinoma (NPC). (a) DFS curves for patients with high and low NF-κB p65 mRNA expression; (b) DFS curves for patients with high and low IKKβ mRNA expression.
Correlations between treatment modalities and the prognosis of NPC patients
Fifty-eight NPC patients were treated with IMRT, and 25 received systemic therapy based on concurrent chemoradiotherapy (CCRT) (including 17 stage III cases and 8 stage IV cases). The follow-up time for these patients was between 25 and 32 months. The results showed that the DFS rate of stage III/IV patients receiving CCRT was significantly better than that of stage III/IV patients receiving IMRT only (Figure 3, P = 0.021).

Kaplan–Meier survival curves between the intensity modulated radiotherapy (IMRT) and concurrent chemoradiotherapy (CCRT) groups of stage III/IV nasopharyngeal carcinoma (NPC) patients.
Discussion
Compared with other cancer types, NPC is uncommon, albeit with a unique geographical distribution. NPC generally originates from nasopharyngeal epithelial squamous cells. There is no specific clinical manifestation in early stages, so it is not commonly noticed by patients. Thus, most patients have mid- to late-stage disease at diagnosis. 11 NPC has a high degree of invasiveness and metastasis, which makes treatment difficult and negatively affects the quality of life of patients. Because NPC is highly sensitive to radiation therapy, patients with stage III/IV disease need to undergo both radiotherapy and chemotherapy. 5 However, according to relevant data, radiotherapy and chemotherapy for NPC only achieve short-term local control and are not effective for long-term survival. 12 Related data showed that the 5-year survival rate of NPC patients is only 60%. 13 Metastasis is the major cause of treatment failure in NPC; therefore, preventing, predicting, and inhibiting metastasis is critical to improving treatment outcomes. 14
NF-κB refers to a family of evolutionarily conserved transcription factors that regulate a wide range of biological processes, including cell growth, survival, immunity, and inflammation. 6 The NF-κB transcription factor family comprises five structurally related subunits: NF-κB1/p50, NF-κB2/p52, RelA/p65, RelB, and c-Rel that homodimerize or heterodimerize to form NF-κB complexes. 6 When the NF-κB pathway is inactivated, p65/p50 and p65/p52 dimers are usually bound to inhibitory IκB family proteins to form a complex, which prevents them from shuttling into the nucleus. Therefore, the NF-κB complex presents as a transcriptionally inactive state in the cytoplasm. The NF-κB pathway can be triggered by diverse stimuli including inflammatory cytokines, immune signals and antigens, microbial products, and stress signals. 15 Upon stimulation, either the canonical or the non-canonical pathway is activated. The canonical pathway, through adapter proteins and kinases, activates IKK complexes (IKKα, IKKβ and IKKγ), which phosphorylate IκBα, leading to its degradation and the release of NF-κB dimers for nuclear translocation, where NF-κB binds to specific gene regulatory regions, promoting target gene transcription.
NF-κB controls the expression of more than 400 genes including genes involved in cellular redox metabolism and its regulation as well as those that promote cancer cell survival, proliferation, angiogenesis, epithelial to mesenchymal transition, and metastasis. 16 In addition to the central role of IKKβ as an activator of the NF-κB pathway through the phosphorylation of IκBα, IKKβ also phosphorylates a number of other components of the NF-κB pathway. 17 This includes NF-κB p105, which is phosphorylated by IKKβ at its C terminus to promote its limited proteasomal degradation to generate the p50 NF-κB subunit. NF-κB p65 is also phosphorylated by IKKβ at S468 and S536, 18 which serves to regulate p65 transcriptional activity. Increased expression of IKKβ and its association with an aggressive phenotype has been reported in several cancer types including head-and-neck, ovarian and liver cancers. 19
NF-κB pathways have been shown to be excessively or constitutively active under disease conditions. Dysregulation of NF-κB leads to the pathogeneses of cancer, inflammation, diabetes, and autoimmune disorders. 20 Aberrant NF-κB activation is associated with increased invasion and other cancer phenotypes. Several human tumors show constitutively elevated levels of NF-κB caused by genetic perturbations. 21 Blocking NF-κB activity decreases tumorigenicity. 22 Among the tumor types evaluated, NF-κB expression is associated with worse 3- and 5-year OS rates in colorectal cancer and esophageal carcinoma, while NF-κB is not significantly associated with either 3- or 5-year OS rates in gastric and ovarian cancers. This suggests that the prognostic significance of NF-κB depends on tumor type. 23 In our study, the positive expression rate of NF-κB p65 and IKKβ protein were found to be markedly higher in NPC tissues than in nasopharyngitis tissues (P < 0.05); which suggested that high NF-κB p65 and IKKβ expression play a role in NPC carcinogenesis. While NF-κB p65 and IKKβ mRNA levels did not differ significantly between NPC and nasopharyngitis tissues, the number of samples in this study was small, so this question needs to be further studied in the future. In our study, the clinical characteristics of patients were analyzed to determine correlations with NF-κB p65 and IKKβ expression. High levels of NF-κB p65 and IKKβ protein and mRNA expression in NPC were notably correlated with deeper tumor invasion (T3–4), lymphatic metastasis, advanced clinical stage (stage III/IV) (P < 0.05), and poor prognosis. DFS curves showed that NPC patients with high NF-κB p65 and IKKβ expression had a significantly lower DFS rate. These results suggested that NF-κB p65 and IKKβ could serve as novel prognostic biomarkers for NPC patients. Additionally, targeting NF-κB could be a potent mode of pharmacological interference against tumors. It has been reported that cancer cells with NF-κB pathway activation are resistant to chemotherapeutics and ionizing radiation, while inhibition of NF-κB activity effectively increased the sensitivity of cancer cells to these agents. 24 Therefore, the IKKβ/NF-κB pathway has been one of the most popular anti-tumor targets. Our study showed that among stage III/IV NPC patients, the CCRT group achieved a higher DFS rate compared with the IMRT-alone group, suggesting that stage III/IV patients could benefit from CCRT compared with IMRT.
In conclusion, this study showed that NF-κB overexpression in NPC tissues, as measured by IHC and RT-qPCR, was associated with a worse prognosis, suggesting that targeting NF-κB could be a promising therapeutic approach for NPC.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and publication of this article: This work was supported by the Zhongshan City Social Welfare Science and Technology Research Project of China (No. 2018B1055).
