Abstract
Background
Osteoporosis is a chronic metabolic bone disease caused due to the dysregulation in the functioning of osteoblast and osteoclast cells leading to increased bone resorption predominantly in postmenopausal women and the elderly, thereby affecting a significant global population.
Purpose
The present study aims to computationally screen the phytochemical constituents of Oldenlandia umbellata Linn against the unique molecular therapeutic targets of osteoporosis and validate the results using Molecular Dynamics (MD) Simulation.
Materials and Methods
The molecular docking analysis of the selected phytoconstituents against the molecular targets were performed using AutoDock 4.2.6 – AutoDock Tools 1.5.6. In addition, the drug likeliness, ADME, bioactivity, and toxicity were predicted using Molinspiration ADMELAB2.0, ProTox-II and Orisis DataWarrior online tools. Molecular Dynamic Simulation studies were performed using WebGRO macromolecular simulation server.
Results
Molecular docking results and data analysis revealed that deacetylasperuloside possesses good drug-likeness, ADME properties, and bioactive scores and did not indicate any toxicity compared to other phytochemicals exhibiting binding energies below –8.00 Kcal/mol against the targets. Together, the study emphasized that deacetylasperuloside could bind with the molecular targets of osteoporosis, and the lead is a potential therapeutic candidate for osteoporosis treatment. Further, molecular dynamic simulation analysis for 100 ns revealed that the ligand–protein complexes, including glycogen synthase kinase 3β (GSK3β)-deacetylasperuloside and cathepsin K (CTSK)-deacetylasperuloside complexes, were stable and highly compact assessed from their trajectories. Hence, it can be emphasized that deacetylasperuloside could be a potential therapeutic molecule that could inhibit the targets, including GSK3β and CTSK.
Conclusion
Oldenlandia umbellata L. is a potential candidate for identifying therapeutic leads for osteoporosis treatment. Further, in vitro and in vivo studies are needed as an output of this research to evaluate its therapeutic efficacy.
Introduction
Osteoporosis is a chronic metabolic bone disease caused due to an imbalance in bone formation and resorption affecting millions globally. In 1993, the World Health Organization (WHO) elucidated osteoporosis as a “progressive systemic skeletal disease characterized by low bone mass and microarchitectural deterioration of bone tissue, with a consequent increase in bone fragility and susceptibility to fracture” (Noh et al., 2020). Osteoporosis is highly predominant among the elderly, especially postmenopausal women, resulting in an increased risk of fracture and mortality (Sun et al., 2020). Generally, bone homeostasis is governed by orchestrated functioning of osteoblast and osteoclast cells regulating bone formation and resorption to aid the continuous remodeling of the bone to maintain its architecture (Sun et al., 2020). Increased osteoclast or decreased osteoblast activity leads to increased bone resorption, causing osteoporosis. Mesenchymal stem cells (MSC) are the precursor of osteoblasts, which differentiates from the latter depositing the bone-forming extracellular matrix proteins such as collagen Type I, subsequently mineralizing the bone (Noh et al., 2020).
The major signaling molecules that play a crucial role in the differentiation of osteoblasts include β-catenin, glycogen synthase kinase 3β (GSK3β) (Saad, 2020), runt-related transcription factors (Runx2), and activator protein 1 (AP-1) (Chau et al., 2009). On the other hand, osteoblasts produce receptor activator of nuclear factor-kappa-Β ligand (RANKL), osteoprotegerin (OPG), a decoy receptor for RANKL, and macrophage colony-stimulating factor (M-CSF), which stimulates osteoclast differentiation, thereby regulating osteoclastogenesis. Osteoclast cells are differentiated from the progenitor, that is, hematopoietic stem cells (HSC), upon M-CSF and RANKL stimulation. Various signaling molecules, including cathepsin K (CTSK), AP-1, tartrate-resistant acid phosphatase (TRAP), nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB), and TNF receptor-associated factor 6 (TRAF-6) and αvβ3 integrin contribute to osteoclastogenesis and osteoclast survival, thereby increases the bone resorption (Győri & Mócsai, 2020). Hence, in general, among various targets, the signaling molecules play crucial roles in aggravating the pathogenesis of the disease.
Currently, estrogen replacement therapy is the most effective method for treating osteoporosis in postmenopausal women, whereas it also has an adverse effect leading to uterine and breast cancer (Sharan et al., 2009). In addition, bisphosphonates, selective estrogen receptor modulators (SERMs), CTSK inhibitors (Odanacatib), and anabolic agents (Parathyroid hormone and analogues of its related proteins) are used for the treatment of osteoporosis. Unfortunately, conventional treatment methods induce side effects such as cancer, stroke, toxicities, and cardiovascular problems and are incapable of restoring normal bone metabolism (Noh et al., 2020; Wang et al., 2019). Considering the effects of osteoporosis, the economic burden that can be levied upon the global population and the side effects caused by the conventional treatment methods, there is an urgent need for alternate and effective therapeutics to treat osteoporosis. Various studies have reported that the protective effects conferred by the phytochemicals of natural sources can be an effective alternative option for the treatment of osteoporosis (Suvarna et al., 2018).
Oldenlandia umbellata (OU) Linn, also called Hedyotis umbellata Linn, is widely found in India, Burma, and tropical areas. It is an annual plant that finds its use as an expectorant. It has reported pharmacological properties such as anti-inflammatory (Mahibalan et al., 2008) and anti-pyretic (Padhy & Endale, 2014), anti-nociceptive (Saraswathy et al., 2008), anti-bacterial (Arun et al., 2010), anti-helminthic (Somnath et al., 2014), antioxidant, and hepatoprotective activities (Sivapraksam et al., 2014). In Siddha medicine, OU is used to treat asthma, bronchitis, and tuberculosis (Yoganarasimhan, 2000). Even though OU possesses potential pharmacological properties, it remains unexplored for its therapeutic potential against osteoporosis. Thus, the study aimed to computationally screen the reported phytoconstituents of OU against the potential therapeutic targets of osteoporosis to identify the potential therapeutic lead for osteoporosis treatment. The study also aims to validate the in silico results using molecular dynamics (MD) simulations.
Materials and Methods
Target Preparation
The three-dimensional (3D) structures of 10 molecular target proteins of osteoporosis, including GSK3β – PDB: 1UV5, CTSK – PDB: 4X6H, TRAP – PDB: 1QFC, TRAF6 – PDB: 3HCT, AP-1 – PDB: 2H7H, NF-κB (p65 and p50 – PDB: 1NFI, RANK – PDB: 3ME2, c-fms – PDB: 6N33, αvβ3 integrin – PDB: 1L5G are retrieved from The Research Collaboratory for Structural Bioinformatics – Protein Data Bank (RCSB-PDB,
Ligand Preparation
The 3D structures of five of the eight phytochemical compounds identified from OU Linn. (Mahibalan et al., 2016), such as asperulosidic acid (PubChem CID: 11968867), cedrelopsin (PubChem CID: 12302592), deacetylasperuloside (PubChem CID: 44593378), scandoside (PubChem CID: 21602023), and scandoside methyl ester (PubChem CID: 442433) have been retrieved from the PubChem Database in SDF format (
2D-Chemical Structures of Phytoconstituents of Oldenlandia umbellata.
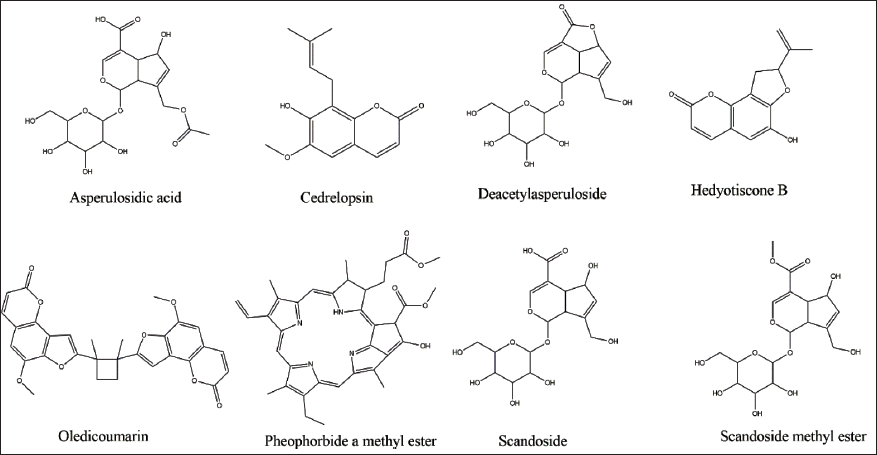
Molecular Docking Analysis
The molecular docking (rigid protein–flexible ligand) analysis was performed between the eight selected phytoconstituents of OU against the potential molecular therapeutic targets of osteoporosis using AutoDock 4.2.6–AutoDock Tools 1.5.6 (Morris et al., 2009). The native ligand-bound residues and residues predicted using CASTp 3.0 (
Evaluation of Drug-likeliness and ADME Properties
The physicochemical properties and drug-likeliness of the phytochemical compounds from OU were assessed using Molinspiration (
The ADME parameters were assessed using ADMETLAB 2.0 (
Bioactivity and Toxicity Prediction Analysis
The bioactivity scores of the selected phytoconstituents against G-protein-coupled receptor ligand (GPCR), ion channel modulator (ICM), kinase inhibitor (KI), nuclear receptor ligand (NR), protease inhibitor (PI), and enzyme inhibitor (EI) were also assessed using Molinspiration online tool and toxicity parameters were predicted using ProTox-II online tool (
Molecular Dynamics Simulation
MD simulation of the protein–ligand complexes were done using the UAMS WebGRO server for Macromolecular Simulations (
Results and Discussion
Molecular Docking Analysis
The molecular docking analysis between the target proteins and phytoconstituents was performed using AutoDock 4.2.6–AutoDock Tools 1.5.6. AutoDock offers multiple advantages in molecular docking of small ligands to target proteins due to receptor flexibility, blind-docking, precalculated grid maps on a binding site, free-energy scoring function based on linear regression analysis, good correlation between predicted inhibition constants and experimental data, whereas still it has a limitation that the unreliability of the results when the small molecules are docked to the flexible docking sites (Amjadi-Moheb & Akhavan-Niaki, 2019). The binding free energies of the docked target–ligand complexes are shown in Table 1. The phytoconstituents that exhibited higher binding affinity with the lowest binding energies below –8.00 kcal/mol against the targets were only considered significant in the present study. Among the eight selected phytoconstituents of OU, four compounds, including cedrelopsin, deacetylasperuloside, oledicoumarin, and pheophorbide a methyl ester exhibited the lowest binding energies below –8.00 kcal/mol, indicating higher binding affinity towards the molecular target proteins.
Binding Free Energies of the Target–Ligand Complexes.
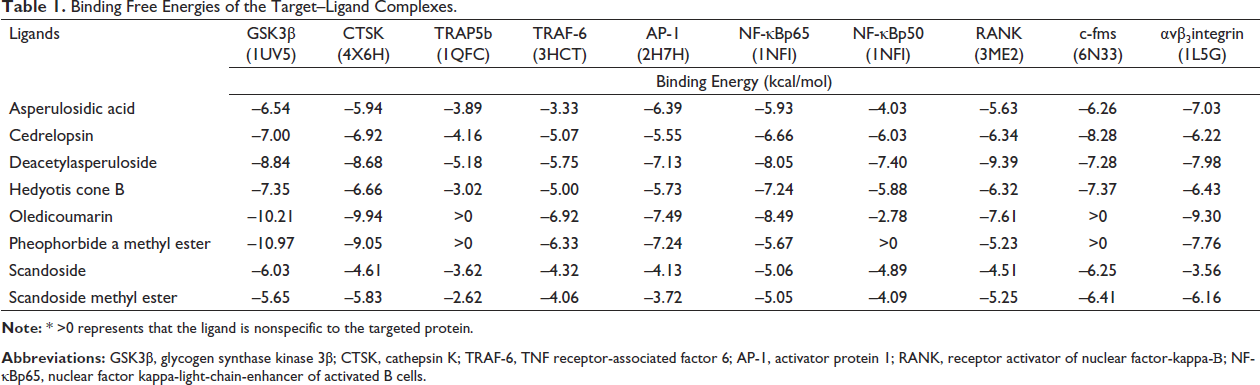
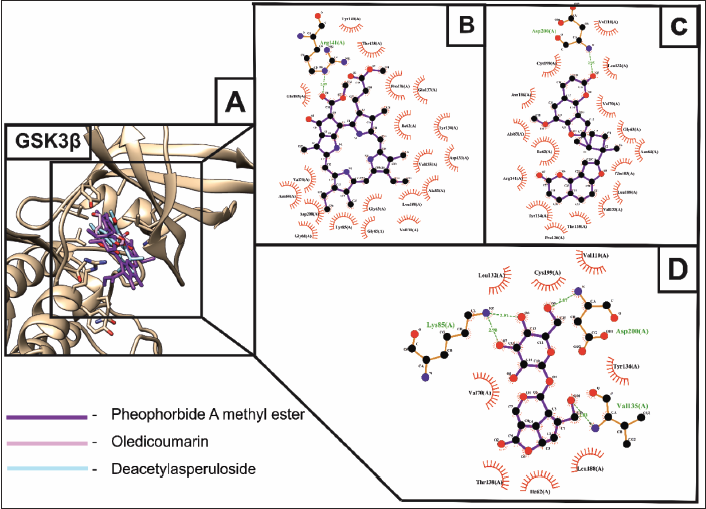
GSK3β, a serine/threonine kinase, is a central molecule of wnt/β-catenin signaling and has a crucial role in glycogen metabolism. GSK3β negatively regulates osteoblast differentiation and bone formation. Thus, overexpression of GSK3β in wnt/β-catenin signaling causes proteasomal degradation of β-catenin, preventing its nuclear translocation and activation of transcription factors, such as T-cell factor/lymphoid enhancer factor (TCF/LEF), Runx-2, and AP-1, which are essential for osteoblast formation (Amjadi-Moheb & Akhavan-Niaki, 2019). Thus, targeting GSK3β could be a potential therapeutic target for treating osteoporosis via stimulating osteoblast formation. Molecular docking showed that deacetylasperuloside, oledicoumarin, and pheophorbide a methyl ester exhibited the lowest binding energies of about –8.84 kcal/mol, –10.21 kcal/mol, and –10.91 kcal/mol, respectively, against GSK3β (Figure 2A). Among them, pheophorbide A methyl ester exerted the lowest binding energy of –10.91 kcal/mol towards GSK3β forming a strong hydrogen bond interaction (2.91 Å) with ARG141 (Figure 2B). On the other hand, oledicoumarin formed a hydrogen bond with the ASP200 (2.95 Å) residue of GSK3β with the binding energy of –10.21 kcal/mol (Figure 2C). Deacetylasperuloside formed 4H bonds against the residues of GSK3β, that is, LYS85 (2.91 Å and 2.98 Å), VAL135 (2.83 Å), and ASP200 (2.87 Å) exhibiting the binding energy of about –8.84 kcal/mol (Figure 2D).
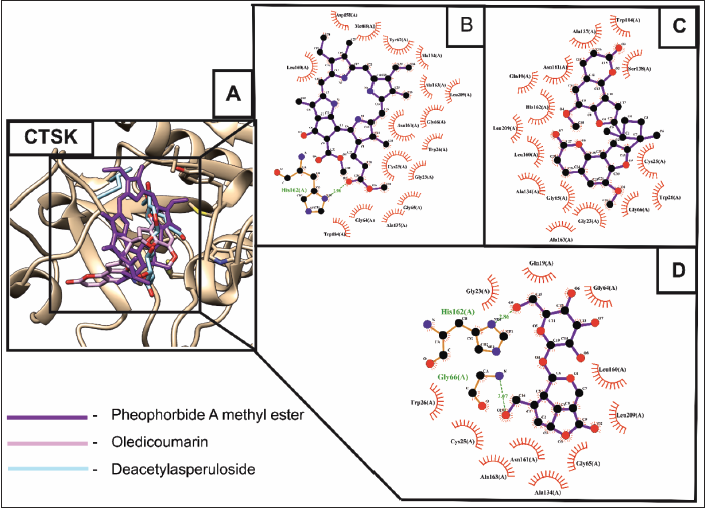
CTSK, a lysosomal protease enzyme abundant in osteoclasts, resorbs bone. RANK-RANKL signaling regulates CTSK expression and degrades bone matrix, primarily Type I collagen, and activates MMPs (matrix metalloproteases), which accelerate bone resorption. CTSK from mature osteoclasts induces actin ring formation, a sealing zone with an acidified milieu that dissolves the mineralized bone matrix, making it an attractive target for osteoporosis treatment (Dai et al., 2020). The molecular docking results showed that oledicoumarin exhibited the lowest binding energy of about –9.94 kcal/mol against CTSK. Subsequently, followed by pheophorbide a methyl ester (–9.05 kcal/mol) and deacetylasperuloside (–8.68 kcal/mol), as shown in Figure 3A. Pheophorbide A methyl ester formed an H-bonding with the residue of HIS162 (Figure 3B). In contrast, oledicoumarin did not show any hydrogen bond formation, which indicated that the higher binding affinity exhibited was due to the hydrophobic interactions towards the residues of target proteins (Figure 3C). At last, deacetylasperuloside formed two H-bonding with the HIS162 (2.84 Å) and GLY66 (3.07 Å) residues of CTSK (Figure 3D) exhibiting effective binding between the CTSK-ligand complexes.
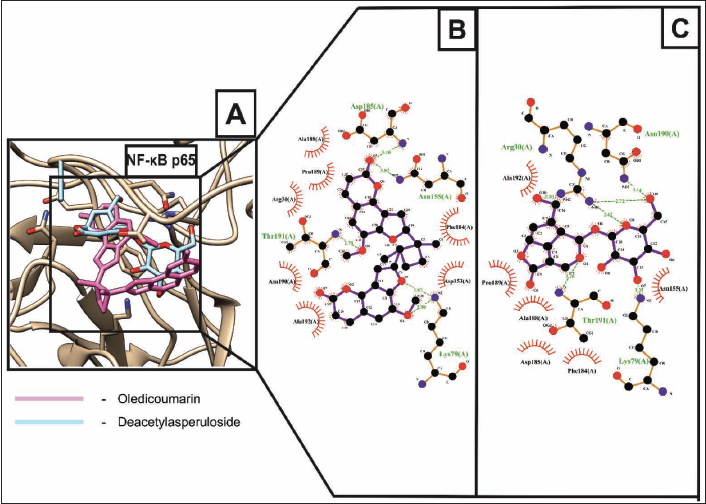
NF-κB is a transcription factor and a well-known master regulator of various physiological processes and pathways. Usually, in the inactive state, NF-κB exists in a hetero-dimer p65/p50 form. Upon activation through RANK-RANKL signaling, it is translocated to the nucleus leading to the activation of multiple transcription factors, including the nuclear factor of activated T-cells, cytoplasmic 1 (NFATc1), a master regulator of osteoclastogenesis facilitating bone resorption (Cheng et al., 2018). Thus, NF-κB is a potential molecular target of therapeutic value for osteoporosis treatment. The phytochemical constituents of OU were docked against NF-κBp65 and NF-κBp50, and they exhibited more binding affinity towards NF-κBp65. Among the phytochemical constituents, oledicoumarin and deacetylasperuloside exhibited higher binding affinity towards NF-κBp65 with the lowest binding energies of –8.49 kcal/mol and –8.05 kcal/mol, respectively, as shown in Figure 4A. Oledicoumarin formed five H-bonding with the NF-κBp65 residues, including ASN155 (3.07 Å), ASP185 (3.10 Å), THR191 (2.78 Å), and two H-bonding with LYS79 (2.87 and 2.90 Å) (Figure 4B,) whereas deacetylasperuloside formed six H-bonding with ARG30 (2.62, 2.72 and 2.84 Å), LYS79 (3.35 Å), ASN190 (3.14 Å), and THR191 (3.26 Å) residues of NF-κBp65 (Figure 4C).
The RANK belongs to the TNF receptor superfamily, which governs RANK-RANKL-OPG signaling that regulates osteoclast differentiation, survival, and function. On RANKL activation, RANK stimulates multiple downstream effectors such as MAPK, NF-κB, AP-1, c-FOS, and NFATc1, thereby leading to the pathogenesis of osteoporosis by inducing osteoclast differentiation.28 Thus, RANK is a potential target of osteoporosis. Our result showed that deacetylasperuloside has a higher binding affinity towards the molecular target RANK with a binding energy of –9.39 kcal/mol forming four H-bonding with target residues including GLU141 (2.95 Å), LEU156 (2.83 Å), LYS169 (3.06 Å), and CYS170 (3.02 Å) in the RANK-deacetylasperuloside complex (Figure 5A and B).
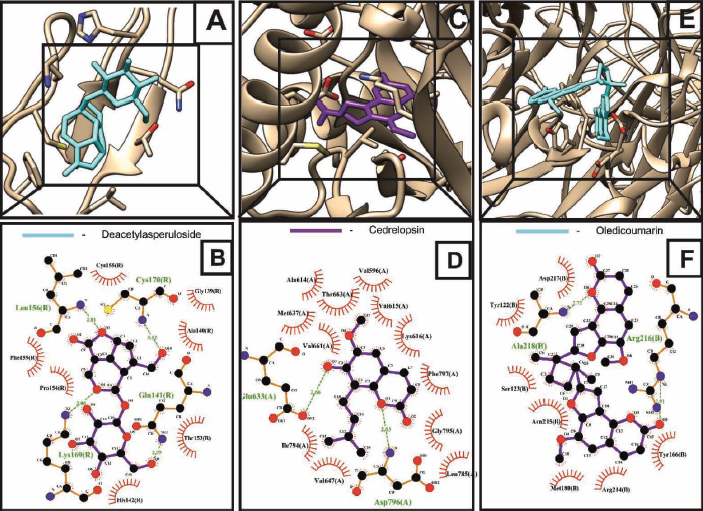
CSF1R (c-fms) is type III receptor tyrosine kinase (RTK) implicated in various diseases, from inflammation to cancer, with crucial physiological functions regulating cellular processes such as survival and differentiation. Until now, CSF1 and IL-34 are the known ligands of c-fms, and both tend to induce osteoclasts differentiation leading to osteoporosis. CSF1 plays a vital role in the differentiation of osteoclasts from its precursor implicating its crucial role in osteoporosis. In addition, C-fms mediated signaling tends to induce increased RANK expression in the osteoclast. Thus, making it a potential target for osteoporosis (Mun et al., 2020). Molecular docking analysis results implicated that cedrelopsin showed the lowest binding energy of –8.28 kcal/mol forming two H-bonding with c-fms residues, that is, GLU633 (2.66 Å) and ASP796 (2.83 Å) (Figure 5C and D).
αvβ3 integrin is a heterodimeric adhesion receptor mediating cell–cell and cell–matrix interaction, where osteoclasts exhibit the highest αvβ3 integrin expression. αvβ3 integrin aids in the actin ring formation, forming a sealing zone around the bone matrix and where osteoclast erodes the bone matrix, thereby increasing the bone resorption. It also has been proved that inhibiting the αvβ3 integrin could reduce osteoporosis without affecting the number of osteoclast or osteoblast cells (Nakamura et al., 2007). Our results implicated that oledicoumarin, with the lowest binding energy of –9.30 kcal/mol, had the highest binding affinity towards the molecular target αvβ3 integrin. In addition, it also formed two H-bonding with the ARG216 (2.91 Å) and ALA218 (2.72 Å) residues of αvβ3 integrin (Figure 5E and F). From the molecular docking results, cedrelopsin, deacetylasperuloside, oledicoumarin, and pheophorbide a methyl ester, were considered for further evaluation.
Drug-likeliness and ADME Analysis
In general, the drug-likeliness of the compounds was assessed based on Lipinski’s-rule-of-five (Ghose et al., 1998). Lipinski rule states that a drug candidate should have an MW < 500, Log P < 5, HA < 10, and HD < 5 to be orally effective (Lipinski et al., 2001; Ali et al., 2012). The drug-likeliness and absorption, distribution, metabolism, and excretion (ADME) scores of the selected four ligands are shown in Table 2. The data suggested that cedrelopsin and deacetylasperuloside obeyed the Lipinski rules with 0 violations.
Drug Likeliness Scores and ADME Analysis of Selected Phytoconstituents of Oldenlandia umbellata.
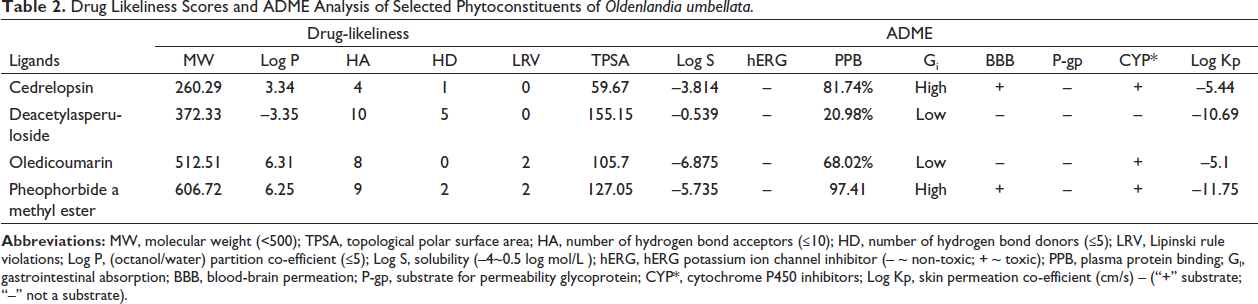
The ADME properties of the selected ligands are shown in Table 2. TPSA is an ideal parameter for characterizing the drug transport properties and correlating the hydrogen bonding, indicating the molecule’s bioavailability. The optimal limit of TPSA is suggested to be less than 160 Ǻ2 (Husain et al. (2016). The TPSA of four selected phytoconstituents were in the range of 59.67–155.15, Ǻ2, which is below the limit indicating good bioavailability. Besides, the solubility (Log S) of a drug greatly confers the facilitation of drug absorption, making it a crucial property to be evaluated. Compounds exhibiting aqueous solubility of about –4.0 to 0.5 log mol/L are considered to have considerable aqueous solubility. Thus, cedrelopsin and deacetylasperuloside exhibited solubility of about –3.814 and –0.539 log mol/L, signifying adequate aqueous solubility. The human Ether-à-go-go-Related Gene (hERG) encodes for the voltage-gated potassium channel capable of regulating the voltage potential in cardiac action. Thus, the inhibition of the hERG could cause repolarization of cardiac potential; thereby, it could induce sudden death (Jing et al., 2015). Thus, results indicated that pheophorbide a methyl ester is capable of inducing cardiac toxicity, as shown in Table 2.
Among the ADME parameters, Gi is a vital parameter to assess the extent of absorption of the drug in the gastrointestinal tract, as it directly affects the bioavailability of the drug. Results exhibited that cedrelopsin and pheophorbide a methyl ester showed the highest probability of gastrointestinal absorption. Distribution properties were assessed based on PPB, their ability to cross the BBB and to be a substrate for P-gp. PPB is an essential pharmacokinetic parameter as it influences the ADME properties and plays a crucial role in drug–drug interaction (Sun et al., 2018). Hence, the phytoconstituents exhibiting below 65% of PPB were considered significant. Results showed that deacetylasperuloside exhibited about 20.98% of PPB. Thus, the unbound deacetylasperuloside (70%) is available for exerting its bioactivity. Cedrelopsin and pheophorbide A methyl ester were shown to have the ability to cross the blood-brain barrier, whereas other constituents could not permeate through BBB. Hence, deacetylasperuloside and oledicoumarin will not induce any adverse effects affecting CNS. It is interesting to note that none of the selected phytoconstituents was a substrate for P-gp. The distribution properties of the phytoconstituents are represented in Table 2.
Biotransformation of the absorbed drugs occurs via metabolism involving various biochemical pathways and enzymes structurally modifying them to different molecules (Zhang & Tang, 2018). Among the multiple enzymes regulating metabolism, CYP 450 enzymes (Subfamilies: 1A2, 2C19, 2C9, 2D6 and 3A4) play a crucial role in metabolizing most of the drugs by oxidation. In addition, they can be inhibited or induced by drugs that can lead to adverse effects like toxicity or render the drug ineffective (Durán-Iturbide et al., 2020). CYP1A2 is a vital enzyme for metabolization in the liver, whereas CYP2C19 involves metabolizing several drugs and detoxifying potential carcinogens. On the other hand, CYP2C9 highly contributes to the metabolism, predominantly in the clearance of constituents with a narrow therapeutic index. The CYP2D6 isoform is vital in the hepatic metabolism of the drugs; inhibiting the CYP2D6 enzymes renders the drugs ineffective and leads to adverse effects (Durán-Iturbide et al., 2020). Cedrelopsin, oledicoumarin, and pheophorbide a methyl ester tend to be a substrate for CYP1A2. In addition, cedrelopsin tends to inhibit CYP2C19, whereas oledicoumarin is a substrate for CYP2C9. None of the phytoconstituents were substrates for CYP2D6 and CYP3A4. Fortunately, deacetylasperuloside is not a substrate for any CYP450 enzymes.
Permeability is an important parameter as it affects the ADME of the drug. Skin permeability (Kp) is the ability of the drug to permeate the skin. The more negative the Log Kp (cm/s) value, the drug is impermeable through the skin (Daina et al., 2017). The skin permeability (Log Kp) of the phytoconstituents ranged from –5.1 to –11, indicating that oledicoumarin is shown to have higher permeability, whereas pheophorbide A methyl ester shown to have low permeability.
Bioactivity and Toxicity Prediction
The bioactivity scores of the phytoconstituents were assessed using the Molinspiration tool and are represented in Table 3. Constituents with bioactivity scores higher than 0.00 are presumed to have significant bioactivity, whereas scores ranging from –0.50 to 0.00 and lesser than –0.50 are considered moderately active and inactive, respectively (Husain et al., 2016). Deacetylasperuloside showed bioactivity scores >0, that is, GPCR (0.34), ICM (0.11), KI (0.09), NR (0.21), PI (0.27), and EI (0.57), indicating inhibition of various receptors, ion channels, and enzymes involving multiple pathways and has significant bioactivities.
Bioactivity Scores and Toxicity Prediction Analysis of Selected Phytoconstituents of Oldenlandia umbellata.
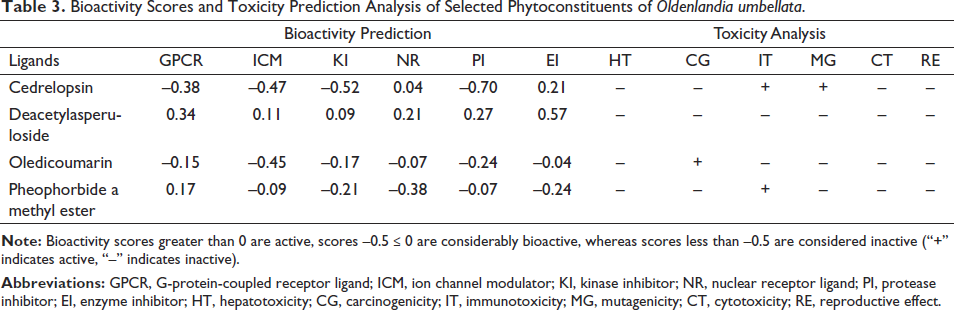
Toxicity is a critical parameter that should be considered in the drug development process as it confers to failure at the later stage of drug development. Acute toxicity, hepatotoxicity, cytotoxicity, carcinogenicity, mutagenicity, immunotoxicity, adverse outcome pathways (Tox21), and toxicity targets can all be predicted with the help of the free server ProTox-II, which uses molecular similarity, pharmacophores, fragment propensities, and machine-learning models (Banerjee et al. 2018). Table 3 depicts the toxicity assessment of the selected phytoconstituents of OU. Results showed that deacetylasperuloside did not show any hepatotoxicity, carcinogenicity, immunotoxicity, mutagenicity, cytotoxicity, and reproductive toxicity compared to other phytoconstituents. Overall results suggested that deacetylasperuloside did not violate the Lipinski rule and possess good ADME properties with good bioactive scores without significant toxicity. In this regard, deacetylasperuloside could be a good therapeutic lead for treating osteoporosis. Hence, the deacetylasperuloside complex with the lowest binding energies (≤8.0 kcal/mol) was subjected to MD simulation analysis.
Molecular Dynamics Simulation
The protein–ligand complexes including deacetylasperuloside and GSK3β, CTSK. NF-κBp65, and RANK were subjected to 100 ns MD simulation to assess the stability of the complex.
RMSD is used to assess the stability of the complex via assessing the trajectories of the protein backbone coordinates, which is calculated by rooting the average value of the square of the atomic coordinates. Thus, the lower the fluctuation, the higher the stability of the complex (Abraham et al., 2015). Figure 6A shows that the GSK3β-DA complex has an average RMSD of 4 Å encompassing rigid structure attaining equilibrium after 60 ns. Besides, the CTSK-DA complex formed a rigid, stable complex with the average RMSD of 2 Å attaining equilibrium at 30 ns and maintained throughout the 100 ns simulation as shown in Figure 6B. The NFκBp65 and DA complex showed average RMSD >5 Å but equilibrating after 60 ns (Figure 6C). Compared to forementioned complexes NFκBp65-DA complex showed higher fluctuations but equilibration after 60 ns depicted the stability of the complex during simulation. From Figure 6C, it can be observed that from 50 ns, the protein–ligand complex has achieved equilibrium with the RMSD ranging from 3 to 5 Å, but a significant fluctuation in the RANK-DA complex indicates lesser stability of the complex. Hence, the protein–ligand complexes, that is, GSK3β and CTSK, were intact through the MD simulation, exhibiting stable conformations of the complexes. The experimental data for RMSF, Rg, and HBs are provided in the supplementary information.
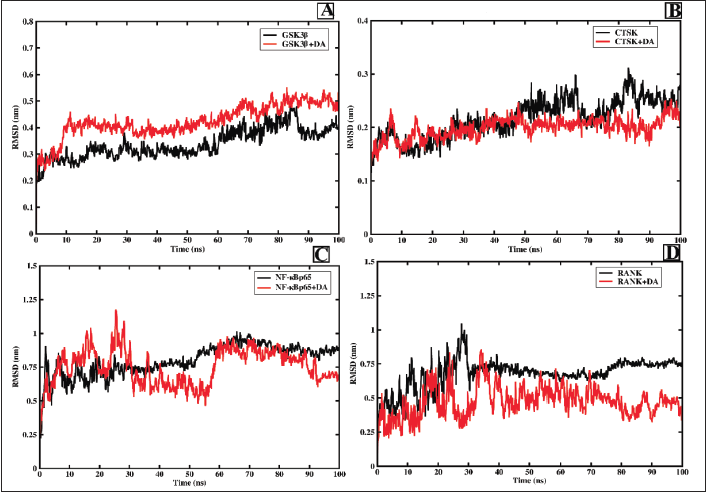
In conclusion, ligand complexed with GSK3β and CTSK has shown good stability, compactness, and rigidness compared to NF-κBp65 and RANK. Though pharmacokinetic and toxicity parameters of the phytoconstituents are computationally analyzed, it is imperative to analyze its safety and efficacy via in vitro and in vivo experimentation to assess its correlation with computational analysis. Further, in vitro evaluation in appropriate cell models and in vivo evaluation in animal models are needed to assess the therapeutic efficacy of the OU for the treatment of osteoporosis.
Conclusion
Conclusively, from the results, deacetylasperuloside exhibited significant binding affinity towards the molecular targets of osteoporosis, that is, GSK3β, CTSK, NF-κBp65, and RANK. Further, MD simulation for 100 ns revealed that GSK3β and CTSK complexes were highly stable and maintained their conformation till the end of the simulation. Hence, it can be anticipated that deacetylasperuloside could be a potential therapeutic molecule, which may inhibit the potential therapeutic targets.
Since all the phytoconstituents of OU showed good and considerable binding affinity towards the therapeutic targets of osteoporosis; further, in vitro and in vivo studies are needed to assess the therapeutic efficacy and safety profile of OU for the treatment of osteoporosis.
Author Contributions
Data curation, analysis, and preparation of the manuscript were done by Sivasakthi Paramasivam. Conceptualization, supervision of the study, and critical revision of the manuscript were done by Senthamil Selvan Perumal.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
