Abstract
Background
Asparagi Radix (AR) is one of the widely used Traditional Chinese Medicines (TCMs) for clinical applications, owning the effects of clearing the lungs and promoting body fluid, nourishing yin, and moistening the lung. To reduce the loss of active ingredients and improve production efficiency, the integrated processing technology of primary processing was used to obtain the decoction pieces of AR. However, there are no specific processing methods and index ingredients of AR in Chinese Pharmacopoeia (2020 edition).
Materials and Methods
This study aimed to establish a method of content determination of protodioscin and asparagine by high-performance liquid chromatography with charged aerosol detector (HPLC-CAD) as the index of process optimization. Furthermore, Box-Behnken design was used to optimize the integrated processing technology of primary processing.
Results
The result showed the contents of protodioscin and asparagine could reach 0.2678% and 0.4114%, respectively, both above the traditional process. After verification, the actual value (99.56) and predicted value (101.15) were similar, indicating that the integrated technology was feasible. In particular, the optimized process parameters of boiling time, drying time, and temperature were 25 min, 12 h, and 60°C, respectively.
Conclusion
In summary, these research findings may provide a reference for the primary processing and quality control of AR.
Keywords
Introduction
Asparagi Radix (AR) is the tuberous root of the Liliaceae plant Asparagus cochinchinensis (Lour.) Merr., which is one of the authentic herbal medicine in Guizhou (China). According to modern studies, it has anti-tumor (Zhang et al., 2021), anti-aging (Cho & Yang, 2018), anti-inflammatory (Lee et al., 2009; Samad et al., 2014) and immune regulation (Chen et al., 2011; Tang, 2014) for widespread using in bronchitis, pulmonary disease, and diabetes (Sung et al., 2017). According to the 2020 edition of Chinese Pharmacopoeia (Ch.P), the origin processing and the processing process of AR are disconnected from each other, which not only increases the production cost of AR processing but also leads to the loss of active ingredients. To reduce the loss of active components of AR and ensure clinical efficacy, it is necessary to establish the processing technology of AR.
The integration of processing and primary processing production of Traditional Chinese Medicine (TCM) was an organic combination of the processing links of the origin of Chinese medicinal materials and the processing and production process of decoction pieces, which not only reduced the duplication of production links, strengthens the quality control of the production process of TCM decoction pieces, but also reduced the loss of active ingredient during the processing of TCM (Yang et al., 2016). At present, the integrated processing research had been applied to TCMs (Feng, 2018a, 2018b; Zhang et al., 2022).
The response surface method (RSM), as a classical mathematical–statistical method, was used to simulate the real limit state curve by fitting the response surface through a set of determined tests. Using multivariate quadratic regression equations to investigate the relationship between factors and response values to find the optimal process parameters (Allaix & Carbone, 2011; Trinh & Kang, 2010). Protodioscin and asparagine, as the active components in AR. Protodioscin had a beneficial effect on inflammation and diabetic nephropathy (Guo et al., 2018; Liu et al., 2017), and asparagine had a good anti-tumor effect (Urazova et al., 2011). Both of those two ingredients had positive effects on the treatment of clinical diseases. As a consequence, the comprehensive score of the contents of these two components was used as the evaluation index in the experiment, and the Box–Behnken design-response surface method (BBD-RSM) was used to optimize the optimal process parameters of AR integration to provide reference and basis for the integrated production of AR.
Materials and Methods
Chemicals and Reagents
The medicinal materials (Batch No. 20211116) were purchased from Guizhou Fengtai Agricultural Development Co., Ltd. (Guizhou, China) and were identified by Professor Li (Guizhou, China) as the tuberous root of the Liliaceae plant Asparagus cochinchinensis (Lour.) Merr.
L-asparagine Standard (Batch No.107D021, Solarbio); protodioscin Standard (Batch No.20120414, Shanghai Yuanye); Methanol, acetonitrile, ammonium formate, and other reagents were high-performance liquid chromatography (HPLC) grade and analytical grade.
Preparation of Solution
Accurately weighed protodioscin (7.60 mg) and L-asparagine (8.06 mg) as reference substances, then dissolved them with 80% methanol by ultrasonic to prepare 3.80 mg/mL and 1.16 mg/mL reserve solutions.
Approximately 1.0 g of AR powder was taken into a 25 mL volumetric flask and diluted with 80% methanol solution, the compound was extracted in an ultrasonic bath with 100 W power for 30 min and then the 80% methanol was added to make up for the lost volume during the extraction. After centrifugation (12 000 rpm, 10 min), the supernatants were stored at 4°C and filtered through a 0.45 µm microporous membrane before being analyzed by the HPLC system.
Processing Method of Asparagi Radix
The traditional processing was divided into two parts. One part was the origin processing, the specific processing process was as follows: Wash the dust and remove impurities of AR, put in boiling water to cook through the core, remove the outer skin, wash and dry, and remove impurities. The other part was the processing, the operation process was as follows: After washing and cleaning quickly, cut into thin slices and dry (Pharmacopoeia of the People’s Republic of China, 2020).
The processing method was more complicated and time-consuming. Based on this, we adopt the integrated processing method to simplify the processing technology of AR, the concrete process was as follows: After washing and removing impurities, put it into a steamed pot, take it out, peel it, cut it into thin slices and put into the oven to dry.
Chromatographic Conditions
After optimization of column type, flow rate, column temperature, and elution gradient, the chromatographic conditions were as follows: Waters XBridge BEH Amide Column (4.6 mm × 250 mm, 5 µm) and maintained at 30°C; the mobile phase consisted of 10 mmol/L ammonium formate solution (pH 4.0) in deionized water (A) and acetonitrile (B) the gradient was as follows: 0–19 min, 85.5–84.5% B; 19–30 min, 84.5–81% B; 30–35 min, 81–81% B; 35–36 min, 81–75.5% B; 36–50 min, 75.5–75.5% B; 50–60 min, 75.5–72% B; 60–65 min, 72–70% B; Elution was performed at a solvent flow rate of 0.7 mL/min. The injection volume of each sample and the standard solution was 3 µL, nebulization temperature was kept at 50°C.
Design of Experiments
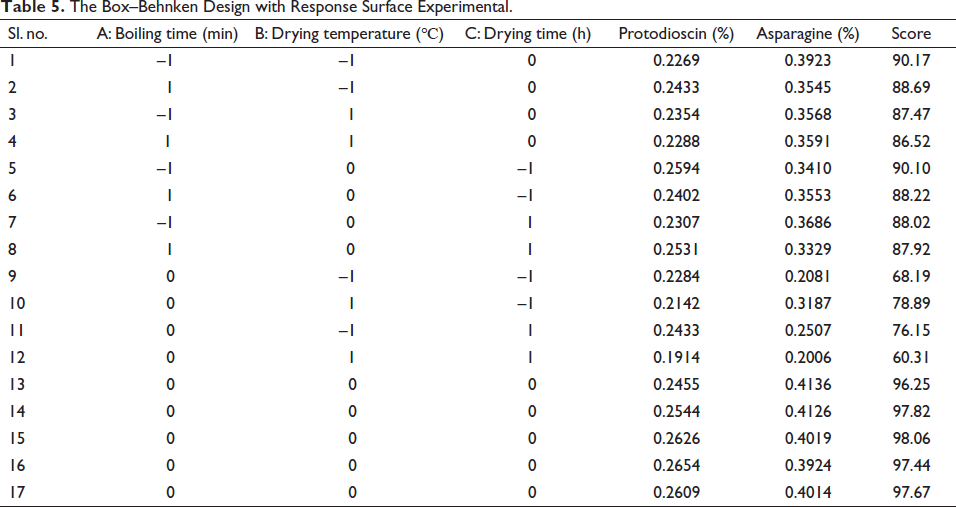
First of all, according to the results of the single-factor investigation and the experimental design principle of Box–Behnken (Ferreira et al., 2007). Taking the boiling time (A), drying temperature (B), and drying time (C) as independent variables. Three levels were taken for each independent variable to carry out the three-factor and three-level tests (Table 1). Second, the central point Design scheme in the Design Expert 10.0 software was used to arrange the process conditions of 17 groups of integrated processing of AR. Eventually, the optimal processing technology was predicted based on the analysis of variance (ANOVA), the level of each factor, and the interaction evaluation results.
Factors and Levels of the Box-Behnken Experimental Design.
Preliminary studies (Du et al., 2020; Liu et al., 2016) had shown that when AR was cooked for 15 minutes, there was a white core on the section and the epidermis was hard to peel off. After about 25 minutes, the section has been cooked thoroughly and more easily stripped than 15 minutes. However, a small amount of AR was not easy to peel. when cooked for 35 minutes, the section was transparent and can be peeled off. Thus, the experiment used a cooking time of 35 ± 10 min to optimize the process. In the choice of temperature, due to AR contains polysaccharides, saponins, and amino acids, and the drying temperature should not be too high. The drying temperature of general medicinal materials did not exceed 60°C, we chose 60±5°C as the investigation condition. The preliminary experiment showed that the moisture content could meet the requirements of Ch.P (<16%), when dried at 60 °C for 11 hours. Based on the results of the pre-experiment and literature (Liu, 2017), we chose 12 h as the intermediate temperature investigation point. Because the drying time should not be too short or too long, we selected 12 ± 2h as the investigation time point of AR integrated processing.
Statistical Analysis
SPSS software (SPSS for Windows 26.0, SPSS Corporation, USA) was used for statistical analysis of experimental data. At the 95% confidence level, the factor significance was calculated using the ANOVA statistical tool (p < 0.05), and the results were presented as average values ± standard deviations (SD). Input the comprehensive scores of the marked ingredients obtained from the 17 tests into the Design Expert 10.0 software to analyze and process the test results.
Results and Discussion
Establish the Analytical Method
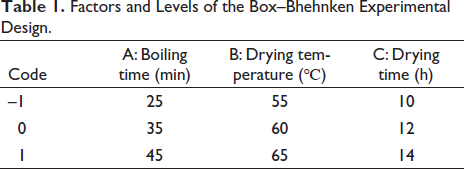
To establish a better analytical method, the column (Reversed-phase column, Normal phase column), column temperature (25℃, 30℃, 35℃), and flow rate (0.6 mL/min, 0.7 mL/min, 0.8 mL/min, 1.0 mL/min) conditions were investigated (Figure 1), respectively. Figure 1A showed that compared with the reverse phase column, the normal phase column separated more chromatographic peaks without a leading peak and trailing peak. Figure 1B showed that at 25℃ and 35℃ column temperatures, some peaks were not separated completely. The separation degree of target chromatographic peaks at 30℃ was better. Comparing the chromatographic peaks at four different flow rates (Figure 1C), it was not difficult to find that when the flow rate was 0.7 mL/min, the shapes of peaks and their separation degrees were better, the baseline was in a relatively stable state and the sensitivity was higher. In conclusion, the optimal chromatographic conditions were as follows: Waters XBridge BEH Amide column (4.6 mm × 250 mm, 5 µm); Flow rate: 0.7 mL/min; Column temperature: 30℃
Investigation of Chromatographic Conditions: (A) Investigation of Chromatographic Columns, (B) Optimization of Chromatographic Temperature, and (C) Investigation of Flow Rate.
Method Validation
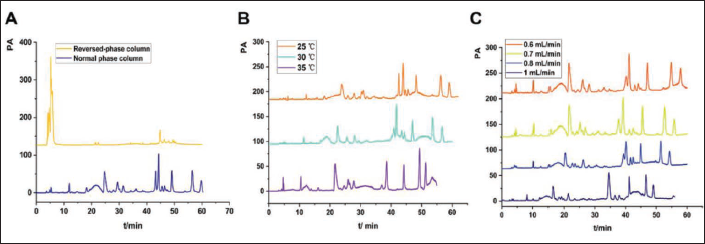
The specificity of the assay method was evaluated by comparing the chromatographic peak results of different solutions (Figure 2). It can be seen from Figure 2, the solvent peak of methanol in the blank solvent appears earlier and the baseline was stable, which did not affect the content determination. Besides, the chromatographic peaks of the two reference solutions were consistent with those of the AR sample solution, indicating that the chromatographic conditions could be used for the determination of the two components in the AR test solution. The two marker ingredients had a good linearity in a wide concentration range with correlation coefficients above 0.999 (Table 2), and LODs and LOQs were also listed in Table 2. The RSD values of precision, reproducibility, stability, and average recovery were less than 3.0% (Table 3), indicating that all of them can be used for the content determination of AR (Behrens et al., 2016).
AR Sample Solution, Protodioscin, L-Asparagine, Mixed Standard Solution and 80 % Methanol Solution HPLC: (A) Protodioscin and (B) Asparagine.
The Results of Precision, Repeatability, Stability, and Additional Recovery (n = 6).
Calibration Curves of Mark Ingredients (n = 6).
Results of Response Surface Optimization for Integrated Processing of AR
All chromatographic peak area data were obtained (Figure 3 and Table 4). According to the contents of two ingredients in 17 Box–Behnken design experiments (Table 5), the proportions of each factor (protodioscin 50%, L-asparagine 50%) were added to obtain a comprehensive score. The higher the value, the better the quality of AR processed under this condition. Using Design Expert 10.0 software, taking the comprehensive score as the response value, the multiple linear regression and binomial fitting were performed on the three factors of A, B, and C, respectively, and the regression equation was obtained: Y = 97.45 – 0.55A – 1.25B – 1.62C – 6.64BC + 4.22A2 – 13.46B2 –13.11C2 (R2 = 0.9938).
HPLC Chromatograms of 17 Groups of AR Samples.
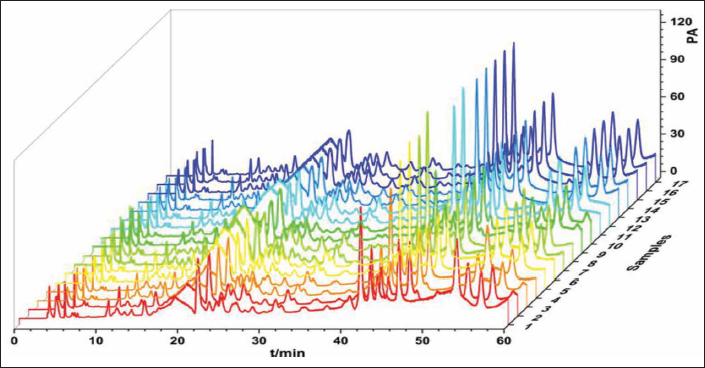
Peak Areas of Protodioscin and Asparagine in 17 Processed Products.
The Box–Behnken Design with Response Surface Experimental.
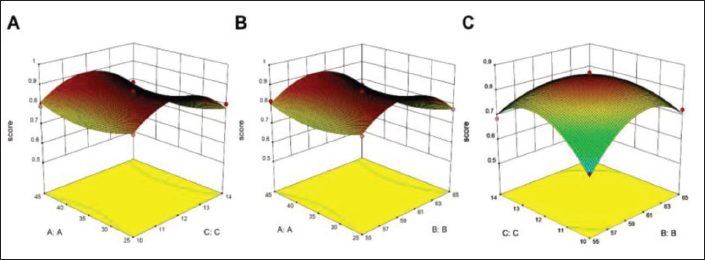
The ANOVA results of the model on the comprehensive score were shown in Table 6. As we all know that p < 0.05, which belongs to a very significant range, indicated that the model was effective and reliable (Liu et al., 2016). The lack-of-fit item of this model was p = 0.1106 (> 0.05), indicating the model difference was not significant, and explaining that other unknown factors had little interference with the experiment, which can better simulate the actual situation (Wang et al., 2020). The coefficient of determination and adjustment coefficient were 0.9938 and 0.9889, respectively, illustrating that the model had good stability and accuracy. Therefore, the model can be used to analyze and predict the integrated processing technology of AR origin. A2, B2, C2, B–B, C–C, BC items all satisfy p < 0.05, implying that these 6 items had a significant impact on the content of protodioscin and asparagine in AR, and three factors (A, B, C) on the contents of protodioscin and asparagine were B > C > A, namely drying temperature > drying time > boiling time.
Analysis of Variance and Statistical Parameters of the Model.
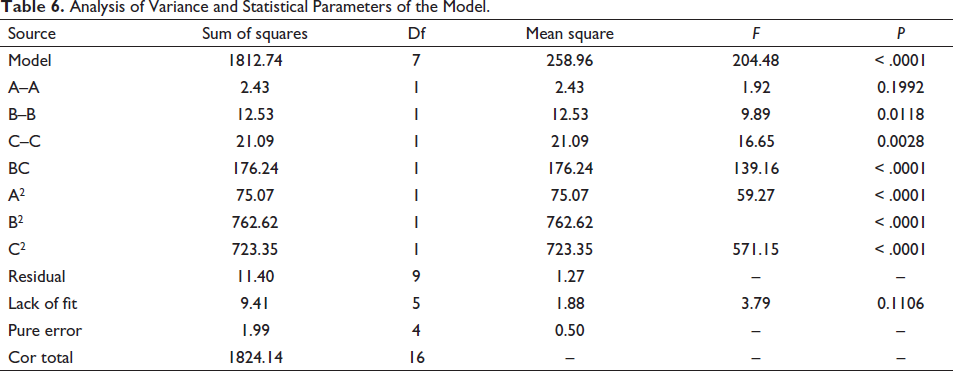
Fixing one of the factors, plot a 3D response surface of the effect of the other two factors on the composite score (Figure 4). It can be seen from Figure 4 that with the increase of boiling time, the content of active ingredients changed little; As the drying time increased, the content of active ingredients decrease after an increase; With the increase of drying temperature, the contents of protodioscin and asparagine increased significantly at first, and then decreased obviously. This implies the influence of three factors on the comprehensive score: drying temperature > drying time > boiling time. After software analysis and screening, the optimal processing parameters of AR were obtained as follows: boiling time: 25.26 min, drying temperature: 59.95℃, drying time: 11.75 h. Considering the actual situation comprehensively, the final determinations are that the boiling time was 25 min, the drying temperature was 60°C, and the drying time was 12 h.
3D Response Surface of Boiling Time, Drying Temperature, and Drying Time for Their Mutual Interaction.
Furthermore, the verification test was carried out according to the optimal process obtained by the response surface analysis. 1000 g of fresh medicinal materials of AR were washed with water, put into a steamed pot, boiled for 25 minutes, taken out, cooled in cold water, and peeled to obtain AR medicinal materials. The AR medicinal materials were rinsed and cut into oblique slices with a thickness of about 2 mm, which were dried at 60℃ for 12 h to obtain integrated decoction pieces (Figure 5). Three batches of samples were prepared in parallel, and the content of the index components was determined. The calculated actual OD value was 99.56, which was not much different from the original theoretical OD value of 101.15. It showed that the mathematical model established in this experiment is accurate and reliable, with a strong predictive ability and good reproducibility of the integrated optimal process conditions (Xie et al., 2015).
Process Flow Chart of AR.

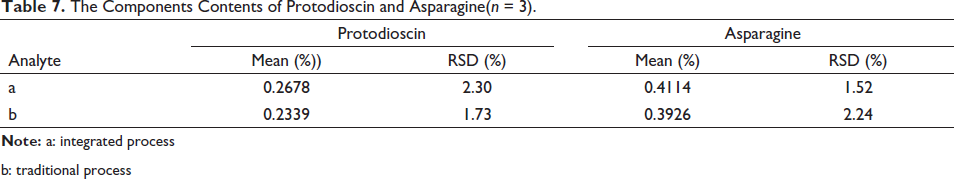
According to the above method, the traditional and integrated AR slices were processed, respectively, and then prepared the traditional and integrated test solutions. The contents of protodioscin and asparagine were measured three times in parallel, and further evaluation experiments were optimized. The effects of the obtained integrated processing and traditional processing on related components were shown in Table 7. Compared with traditional processing, integrated processing can reduce the loss of protodioscin and asparagine during the processing process.
The Components Contents of Protodioscin and Asparagine (n = 3).
b: traditional process
In this experiment, due to the quality standard of AR included in the Ch. P (2020 edition) without the content determination items. In addition, according to the literature review, the components of AR had no ultraviolet (UV) absorption. Therefore, when establishing the determination method of AR, the CAD detector was selected, which not only had the characteristics of high sensitivity, good repeatability, and wide linear range but also had a great advantage, especially for the detection of substances without UV absorption. Protodioscin was an anti-tumor active ingredient in AR, and studies had shown that it can inhibit the survival and proliferation of cancer cells (Liu et al., 2020); AR was rich in amino acids, which was mainly asparagine. Studies had shown that asparagine had anti-tumor and anti-tussive pharmacological (Jiang et al., 2021). These two substances were widely found in nature, so they were selected as indicator ingredients. Subsequently, an HPLC-CAD method for the determination of AR was established by investigating the factors of chromatographic column, flow rate, and column temperature. The methodology validation experiment showed that the method had the advantages of strong specificity, high precision, and good reproducibility. According to Box–Behnken’s design and response surface test, the best-integrated processing was selected as cooking for 25 min, and drying in an oven at 60℃ for 12 h. Experimental verification results showed that the integrated processing of primary processing in the AR origin had good reproducibility and simple operation. Compared with the traditional process, it can effectively shorten the processing cycle, improve production efficiency, reduce the loss of effective ingredients, and meet the needs of actual production. In previous studies, the content of total saponins, total polysaccharides, and total amino acids was determined by ultraviolet spectrophotometry. In this study, the HPLC-CAD method was used to determine the content of active ingredients in AR for the first time. However, there were some limitations in optimizing the processing conditions only from the content changes of the two active ingredients in AR. In subsequent experiments, multiple effective indicators can be used to optimize the comprehensive processing method to provide a reference for the quality standard research of AR.
Conclusion
To sum up, using the content of protodioscin and asparagine as indicators, a method for the determination of the content of AR was established, and then established the best-integrated processing method for AR, which can reduce the loss of active components in AR based on HPLC-CAD combined with BBD. However, this study only determined the content of protodioscin and asparagine, and the component categories and sample batches were less. Subsequent studies can increase sample batches and component categories to optimize the integration process of AR.
Summary
A quantitative method for the determination of protodioscin and asparagine in AR was established.
The composition content of integrated processing is more than that of traditional processing.
Provides a reference for the improvement of the quality standard of AR.
Footnotes
Abbreviations
AR: asparagi radix; ANOVA: analysis of variance; BBD: box–behnken design; Ch.P: pharmacopoeia of the people’s republic of China; HPLC-CAD: high performance liquid chromatography with charged aerosol detector; LOD: limit of detection; LOQ: limit of quantitation; OD: overall desirability; RSM: response surface methodology; TCM: traditional chinese medicine.
Acknowledgements
The authors thank Shenzhen Institute for Drug Control (Shenzhen, China) for providing technical support for the research.
Author’s Contribution
JZ and BW: design, data curation, investigation, and writing – original draft. WL, HS, and WW: investigation and validation. GZ, XS, and ZH: formal analysis and conceptualization. GF and X-AY: resources, supervision, funding acquisition, and writing – review and editing. All authors have read the final manuscript and approved of the submission.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This work was supported by the Guizhou Province Science and Technology Foundation Project (grant no. Qkhzc [2021]417), the Guiyang College of TCM Doctor Startup Fund Project (grant no. Guizhongyi Doctor Fund [2017] 1), the National training program for innovative backbone talents for TCM (grant no. Zjjh [2019] 128), and the “Thousand” level Innovative Talents Project in Guizhou Province (grant no. Qrlf [2020] 4).
