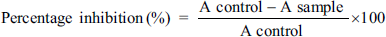Abstract
Background and Objectives
Blumea lacera (Burm.f.) DC. (Family—Asteraceae) is widely used for treating hemorrhoids by several ethnomedicinal practitioners and tribes in India. Thus, the main objective of the present investigation was to evaluate the potential of B. lacera leaf extract in the treatment of hemorrhoids using a croton oil-induced hemorrhoid rat model.
Materials and Methods
Ethanol extract of B. lacera (EBL) leaves was prepared using Soxhlet extraction, optimized using Box–Behnken design (BBD), and quantified using high-pressure liquid chromatography (HPLC). Furthermore, the vasoactive ions were estimated using ion-exchange chromatography. Hemorrhoids were induced in the recto-anal portion of experimental rats, followed by treatment with EBL (100, 200, and 400 mg/kg; par oral (p.o.)) and Pilex granules as a standard anti-hemorrhoid drug for 7 days. The anti-hemorrhoid potential was evaluated on the eighth day by assessing the severity of hemorrhoids, biochemical parameters, and histology of recto-anal tissue.
Results
Upon treatment with EBL and Pilex, there was a significant (p < 0.05) reduction in the inflammatory severity index, concentration of Evans blue dye, recto-anal coefficient (RAC), elevated cytokines level, and restoration of altered antioxidant status. Furthermore, the histopathological results revealed a marked reduction in the inflammatory zones along with minimally dilated blood vessels.
Conclusion
The present study confirmed the traditional claims of the plant B. lacera in the treatment of hemorrhoids, which may be attributed to its anti-inflammatory and antioxidant potential in EBL, where quercetin could be considered the main contributor.
Introduction
Hemorrhoids, also known as anal cushions, are clusters of vascular tissue, smooth muscle, and connective tissue lined by normal epithelial cells of the anal canal. When there is an enlargement and distal displacement of this anal cushion along with symptoms, it becomes a pathological condition termed hemorrhoids (Lohsiriwat, 2012; Mott et al., 2018). Though the pathologic condition lacks sufficient etiology and epidemiologic data to date, rectal bleeding linked with a bowel movement is the most common symptom of hemorrhoids (Gallo et al., 2018; Sandler & Peery, 2019). Evidence indicates that hemorrhoidal bleeding is arterial because of the bright red color and arterial pH of the blood. Besides constipation and prolonged straining, several other factors are involved in the pathogenesis of hemorrhoids. Connective tissue degeneration within the anal cushion, abnormal dilatation and deformation of the vascular channel, and specific inflammatory reactions are some of the main diagnostic findings of hemorrhoidal disease (Loder et al., 1994; Morgado et al., 1988). Reports suggest that hemorrhoids are a major problem of concern in developed and developing countries (36%–40%) (Ali & Shoeb, 2017; Mott et al., 2018; Sandler & Peery, 2019). The therapies available for treating hemorrhoidal disease involve laser treatment, cryotherapy, hemorrhoidectomy, sclerotherapy, bipolar diathermy, and infrared photocoagulation, which are associated with numerous side effects. Therefore, there is a need to assess medicinal plants for their anti-hemorrhoid potential, as plants are considered relatively safe as compared to conventional drugs (Dhaswadikar et al., 2022).
Blumea lacera (Burm.f.) DC. (Family—Asteraceae), commonly known as Kukundara, Tamrachuda, Mriducchada, Kukrondaa, or Kukursunga, is an annual herb with a strong odor. This herb is distributed throughout the plains of north-west India and is also found in tropical Africa, Southeast Asia, Bangladesh, Nepal, Indonesia, Malaysia, the Philippines, Thailand, and Vietnam (Khare, 2004). Traditionally, it is used internally and externally as a hemostatic and anti-inflammatory agent. The juice of leaves and black pepper (Piper nigrum) is given to cure bleeding piles (Dubey et al., 2021). This herb is also offered as an anthelmintic (especially for threadworm) (Pattewar et al., 2012). Leaves and roots are used as astringents, diuretics, febrifuges, and even in cuts and wounds (Kumar et al., 2007). For treating piles (hemorrhoids), the leaves of B. lacera have been used in ethnomedicinal practices. The Bhoxas tribes of Bijnor districts, Uttar Pradesh, India (Singh, 1988), the rural tribal communities of Tryambakeshwar hill region of Nashik, Maharashtra, India (Kakulte et al., 2014); the Bhilada tribe, Maharashtra, India (Korpenwar, 2012); and many more tribes extensively use the leaves of the plant for treating piles. The phytoconstituents mainly identified in the leaves of the plant include β-bourbonene, thymol methyl ether, E-α-bergamotene, β-caryophyllene, E-β-farnesene, α-humulene, β-himachalene, cadinene, thymoquinol dimethyl ether, β-caryophyllene epoxide, β-recocene I, 8-eudesmol, and quercetin (Dubey et al., 2019; Satyal et al., 2015). Despite having such extensive ethnomedicinal use in the treatment of hemorrhoids, there are no scientifically validated reports on the use of the leaves of the plant B. lacera in treating experimental hemorrhoids. Therefore, the present investigation was undertaken to scientifically justify the traditional claims of the leaves of the plant B. lacera in the treatment of hemorrhoids.
Materials and Methods
Plant Material and Its Authentication
Leaves of the plant B. lacera were collected from the local habitat of Banaras Hindu University, Varanasi, in October–November 2016 and authenticated by Dr. N. K. Dubey, Professor at the Department of Botany, Banaras Hindu University. A voucher specimen of the plant material with reference number Aster. 2016/3 has been deposited in the institute.
Extraction Process and Its Optimization and Screening
Shade-dried leaves of B. lacera were coarsely powdered (150 g), and the Soxhlet extraction process was used for extraction following optimization with the help of Box–Behnken design (BBD), where three astute factors, namely, mesh size, extraction solvent, and extraction time, were evaluated that affect the extraction process. Experimental boundaries were fixed using Design Expert (version 12.0.0 trial, Stat-Ease Ink.) for analyzing responses and, eventually, for process optimization (supplementary file). Each design factor was set to one of three levels, namely low, medium, or high, and the powdered leaves were extracted using a Soxhlet extractor at 70°C, as per the projected design. The resulting extract was then filtered and concentrated using a Rota evaporator (IKA, Germany) to obtain the crude extract (60 g) of ethanol extract of B. lacera (EBL). Furthermore, to examine the availability of various classes of phytoconstituents, preliminary phytochemical screening was done following standard procedures (Dubey et al., 2019; Khandelwal, 2008).
Standardization of EBL Using HPLC
High-pressure liquid chromatography (HPLC) analysis was carried out using a Waters HPLC system equipped with the Waters 1500-series pump attached to Waters 2998 photodiode array detector, and the data were analyzed using waters Breeze software (Waters, USA). Through a 0.45-µm membrane filter, the mobile phase and samples (EBL and standard quercetin) were filtered (Millipore, USA) and de-aerated before use. Analysis of the sample was carried out in a Lichro CART® 250-4 C18 column, and methanol:acetonitrile:water in the ratio of 40:15:45 (v/v/v) was used as the mobile phase for better peak shape and resolution at UV 368 nm. The flow rate was kept at 1.0 mL/min, the injection volume used was 10 µL, and all chromatographic operations were carried out at room temperature. Quantification of quercetin in EBL was done in triplicate by using the equation, y = 146949x + 230111.
Vasoactive Ion Estimation by Ion Exchange Chromatography
Powdered drug (1 g) in 10 mL of methanol was incubated (30°C/48 h) in a shaking water bath (Narang Scientific Works, New Delhi) and then centrifuged at 2500 rpm for 10 min (REMI, 24 PLUS, India). The supernatant was then analyzed by ion exchange chromatography (MagIC – 3.1, Metrohm, Switzerland) for determining its ion concentrations (Gulec et al., 2009).
In vitro Anti-Inflammatory Activity
The inhibition in albumin denaturation method was adopted for determining the in vitro anti-inflammatory potential of EBL following the procedure earlier described with minor modifications (Ruiz-Ruiz et al., 2017). Freeze-dried EBL and aspirin, which were used as anti-inflammatory standards, were diluted serially in dimethyl sulfoxide (DMSO) in concentrations ranging from 50 to 600 µg/mL. Initially, 1 mL of a 1% aqueous solution of bovine albumin fraction was mixed with 1 mL of EBL or Aspirin. Furthermore, the pH of the solution mixture was adjusted to 6.3 and then incubated at 37°C for 20 min, followed by heating it for 30 min at 51°C. It was then allowed to cool to room temperature, and its absorbance was measured at 660 nm on a UV-spectrophotometer (Agilent Technologies, USA). The percentage inhibition of protein denaturation was computed using the following equation. The findings were presented as IC50 values, where aspirin was referred to as the positive control and DMSO as the negative control.
where A control is the absorbance of negative control (i.e., DMSO) and A sample is the absorbance of the EBL.
Experimental Animals
Healthy and adult Charles foster rats (male; 8–10-week-old and weighing between 220 and 250 g) were obtained from the Central Animal House of I.M.S (Institute of Medical Sciences), B.H.U. Rats were acclimatized in groups of six in polypropylene cages at 25 ± 1°C (ambient temperature) and at a relative humidity of 45%–55%, with a 12:12 h of light/dark cycle. Animals were given a regular pellet diet and free access to water ad libitum and were acclimatized for at least 1 week before the commencement of the experiment. All experimental protocols were carried out in accordance with the IAEC (Institutional Animal Ethics Committee) guidelines and clearance (letter no. Dean/2016/CAEC/330).
Acute Oral Toxicity Study
Five female rats were used for the acute oral toxicity study (Anonymous, 2001). EBL (2000 mg/kg p.o.) was given to rats (fasted overnight), and observation of all the drug-treated rats (individually for 48 h) was done to assess any changes in behavior and neurological activity, viz., tremors, convulsions, salivation, diarrhea, sleep, and lacrimation, as a sign of acute toxicity (OECD guidelines 425).
Anti-Hemorrhoid Activity
Induction of Hemorrhoids
For induction of hemorrhoids, a croton oil preparation (COP) containing a 1:4:5:10 ratio of deionized water, pyridine, diethyl ether (S D Fine-Chem Limited, Mumbai, India), and 6% croton oil (Sigma Aldrich, St, Louis Mo. U.S.A.) in diethyl ether. Cotton swabs (sterile) of 4-mm diameter soaked in 100 µL of COP were inserted into the anus of rats (fasted overnight) up to the recto-anal portion (20 mm deep into the anal opening) and kept as is for 10 s. After the COP application, all animals were observed for 7–8 h for the development of edema. The edema lasting up to 12 h was considered hemorrhoids. Hemorrhoids were tested in two independent groups of rats: in the first set, plasma exudation of Evans blue dye was measured, while in the second, hemorrhoidal severity, biochemical, and histological parameters were investigated (Azeemuddin et al., 2014; Nishiki et al., 1988).
Grouping and Treatment
The rats were divided into six groups, namely, Group I: normal control (NC) group administered with 1% tween 80 (5 mL/kg, p.o.), Group II: hemorrhoid control (DC) group administered with 1% tween 80; Group III: hemorrhoid-induced group treated with EBL suspension prepared using tween 80 (100 mg/kg, p.o.), Group IV: hemorrhoid-induced group treated with EBL suspension (200 mg/kg, p.o.), Group V: hemorrhoid-induced group treated with EBL suspension (400 mg/kg, p.o.), and Group VI: hemorrhoid-induced group treated with standard Pilex granules (200 mg/kg, p.o., Himalaya Drug Company, Bengaluru, India). EBL and standard Pilex granules were administered once daily for 7 days.
Assessment of Evans Blue Exudation
The method described by Dey et al. (2016) and Azeemuddin et al. (2014) was used to determine COP-induced Evans blue dye plasma exudation in recto-anal tissue. Evans Blue dye (30 mg/kg; i.v.) was injected into the tail veins of the rats 30 min before the administration of COP (set 1 as discussed earlier) and retained for 24 h to induce hemorrhoids. The animals were then treated with different doses of EBL and Pilex granules for seven days. On the eighth day, the recto-anal tissue (20 mm) of animals was dissected and weighed. and Evans blue dye was extracted from the tissue using formamide. The quantification of Evans blue dye in the sample was done through a standard calibration curve, and λmax was recorded at 620 nm using a UV-spectrophotometer. Evans blue concentration was expressed in µg/mg of recto-anal tissue.
Evaluation of Hemorrhoidal Parameters
On the eighth day, blood (1.5 mL) was withdrawn through the retro-orbital plexus of the rats from the second set (Parasuraman et al., 2010) for estimating biochemical parameters. Later, recto-anal tissues (20 mm) were dissected and weighed after sacrificing the animals after anesthetizing them using thiopental sodium. After placing a small portion of the tissue in a 10% formaldehyde solution for histological examination, the rest was stored at –20°C to estimate biochemical parameters.
For determining the recto-anal coefficient (RAC), the recto-anal tissues of the rats were weighed and compared with the individual body weight of the rats, and the RAC was calculated using the formula: RAC = Weight of recto-anal tissue (mg)/Body weight (g).
The transverse section of recto-anal tissue was taken using a microtome and then examined for inflammatory cells, necrosis, congestion, hemorrhage, and vasodilatation (Azeemuddin et al., 2014; Dey et al., 2016). Furthermore, the macroscopic severity scoring of tissues was also carried out after mounting them on white paper, and the inflammation score (from 0 to 2) was noted as described by Azeemuddin et al. (2014).
Estimation of TNF-α and IL-6 in Serum
The level of tumor necrosis factor-α (TNF-α) and interleukin-6 (IL-6) present in serum separated from the blood was determined using enzyme-linked immune sorbent assay kits following the manufacturer’s guidelines (Krishgen Biosystems, USA).
Histology and Histomorphological Scoring
For histological examination, previously fixed recto-anal tissue in 10% formalin was processed following a conventional procedure to prepare microscopic sections with a thickness of 5 mm and subsequently stained with hematoxylin (H) and eosin (E). To ascertain, if any pathological changes occurred in the tissues, the prepared slides were examined under a Nikon Eclipse E200 microscope. For describing the severity of lesions, the wound area was determined on an arbitrary scale (i.e., – corresponds to 0%–5%, + corresponds to 5%–10%, ++ corresponds to 10%–25%, +++ corresponds to 25%–50%, and ++++ corresponds to 50% and above) (Dey et al., 2016).
A histomorphological scale was validated to assess the severity of hemorrhoids compared to the recto-anal tissue of a normal rat. Lesions in the recto-anal tissue of rats in all groups were scored based on the histomorphological scale in a blinded fashion for evaluating the severity. The normal recto-anal tissue was considered the best possible outcome and scored as 28 (maximum total score).
In vivo Antioxidant Studies
On the eighth day, at the end of the study, a portion of recto-anal tissue isolated after sacrificing the animals was rinsed with ice-cold phosphate buffered saline (0.1 M, pH 7.4) thoroughly, the tissue was homogenized in 1.15% potassium chloride (KCl) and centrifuged at 16,000 g for 1 h at 0°C to prepare a 10% w/v suspension, which was utilized for the evaluation of in vivo antioxidant activity.
Evaluation of Lipid Peroxidation Activity
The thiobarbituric acid reactive substances (TBARS) were measured using the colorimetric method as per previous reports (Ohkawa et al., 1979) to estimate the lipid peroxidation (LPO) level in recto-anal tissue. For this purpose, 2 mL of 0.37% TBA, 0.25 M HCl, and 15% TCA in a 1:1:1 ratio (TBA-trichloroacetic acid-HCl reagent) were added to 0.1 mL of tissue homogenate and was incubated in a water bath for 15 min and then cooled. It was then centrifuged for 10 min at 3500g, and the clear supernatant was collected. Its absorbance was measured at 535 nm, and the final reading was expressed as mM/100 g-tissue.
Evaluation of Catalase Activity
For catalase (CAT) activity, 0.1 mL of tissue homogenate was treated with 1.0 mL of 0.01 M phosphate buffer (pH 7.0) and 0.4 mL of H2O2. Furthermore, a 5% dichromate-acetic acid reagent was prepared by using potassium dichromate and glacial acetic acid in a ratio of 1:3 and 2.0 mL of this reagent was added to the above reaction mixture to stop the reaction. Finally, the absorbance was noted at 620 nm, and the catalase activity was reported as µM of H2O2 consumed/min/mg protein (Sinha, 1972).
Estimation of Superoxide Dismutase (SOD) Activity
In this method, 0.5 mL of tissue homogenate was diluted with 1 mL of water, followed by the addition of 2.5 mL of cold ethanol and 1.5 mL of chloroform, and centrifugation at 4°C after 1 min of vigorous shaking. For initiation of the reaction, NADH solution was added to the mixture, which contained 1.2 mL of sodium pyrophosphate buffer (0.025 M, pH 8.3), 0.1 mL of phenazene methosulfate (PMS) (186 µM), 0.3 mL of nitroblue tetrazolium (NBT) (30 µM), enzyme preparation (diluted appropriately), and water (3 mL in a total volume). It was then incubated for 90 s at 30°C, followed by the addition of 1 mL of glacial acetic acid to stop the reaction. Furthermore, 4 mL of n-butanol was added to the mixture, which was stirred and shaken vigorously for a few minutes. Finally, the butanol layer was removed, and its absorbance was measured at 560 nm against the n-butanol blank. A sample without enzymes was taken as a blank. The enzyme reaction that inhibited NBT reduction up to 50% in 1 min under the same experimental environment was defined as one unit of the enzyme activity (Kakkar et al., 1984).
Statistical Analysis
The experimental results are represented as mean ± SEM (n = 6) followed by one-way ANOVA (Tukey’s test), whereas for analyzing score values, the Kruskal–Wallis (Dunn’s) test is performed. GraphPad Prism 5.03 software is used for analyzing the data statistically, and p-value < 0.05 was considered significant.
Results
Extraction Process and Its Optimization and Preliminary Phytochemical Screening
The hydroalcoholic extract of B. lacera was obtained through Soxhlet extraction involving the powdered leaves. The process was optimized using BBD to increase the extraction efficiency and phenolic content to their maximum levels. The best response model was obtained by varying three astute factors, namely, time of extraction process, solvent blend, and mesh size, based on which an optimized extraction cycle and condition were predicted. The optimum conditions for highest extraction efficiency (39.24%) and total phenolic content (134.221 mg/g) were achieved when powder was screened through sieve no. 20 and extracted with hydroalcoholic solvent (90:10) for 15 h. Detailed results are provided in the supplementary file. The results of the preliminary phytochemical screening of EBL showed the presence of mainly phenol, phytosterols, flavonoids, tannins, and alkaloids as major constituents, along with carbohydrates, reducing sugars, proteins, and amino acids.
Standardization of EBL Using HPLC
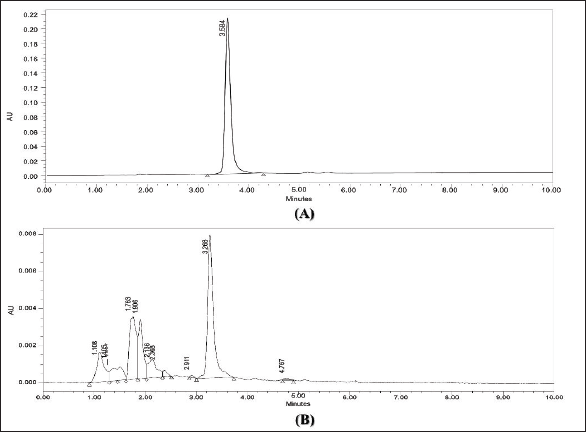
Figure 1 represents the chromatogram of the standard and confirmation of quercetin in EBL. Under optimum operational conditions, the HPLC method was applied for the identification of quercetin in EBL, and the presence of quercetin was confirmed by comparing the HPLC chromatogram of EBL with that of standard quercetin. A peak with a retention time of 3.468 min was confirmed as quercetin in EBL, and the quantity of quercetin in EBL was found to be 31.603 ± 1.141 mg/g.
Vasoactive Ion Estimation by Ion Exchange Chromatography
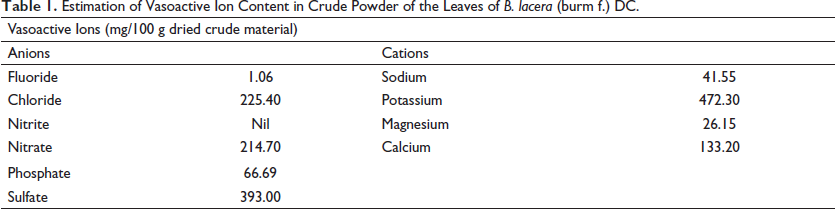
Anion and cation measurement results are represented in Table 1. EBL showed the presence of cations like potassium (472.30 mg/100 g), calcium (133.20 mg/100 g) and anions like sulfate (393.00 mg/100 g), chloride (225.40 mg/100 g), and nitrate (214.70 mg/100 g). The result showed that the concentration of vasoconstrictive ions was found to be higher than that of vasodilator ions.
Estimation of Vasoactive Ion Content in Crude Powder of the Leaves of B. lacera (burm f.) DC.
In vitro Anti-Inflammatory Activity
The in vitro anti-inflammatory activity of EBL was evaluated against protein denaturation, where EBL showed an IC50 value of 94.79 ± 4.32 µg/mL, which was quite comparable to the protein protection capacity of standard aspirin (IC50 value of 77.10 ± 5.26 µg/mL).
Acute Oral Toxicity of EBL
During the entire period of the acute oral toxicity study of EBL, there were no signs of behavioral or neurological toxicity in rats, which also showed a zero mortality rate up to 2 g/kg.
Anti-Hemorrhoid Activity
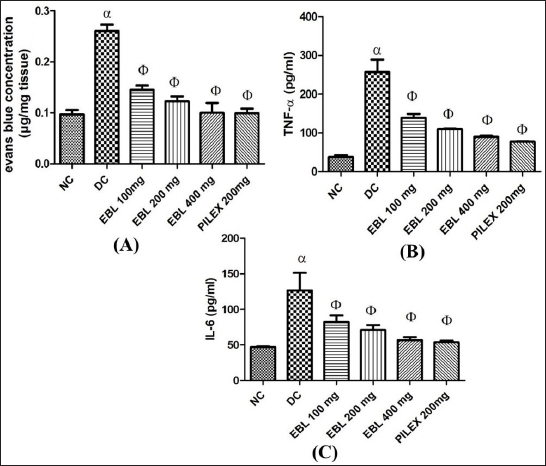
From the observations, COP caused a significantly (p < 0.05) increased extravasation of Evans blue dye as observed from the recto-anal tissues of the negative control as compared to the normal control. However, on treatment with EBL (100, 200, and 400 mg/kg; p.o.) and Pilex granules (200 mg/kg; p.o.), a significant (p < 0.05) reduction in Evans blue concentration was noted as compared to the negative control (Figure 2A). From the overall observation, EBL at 400 mg/kg; p.o. was found to be most effective, which was quite comparable with the standard Pilex granules.
The results revealed a significant increase in the RAC on administration of COP, as evident through the negative control group rats, and were found to significantly (p < 0.05) reduce on treatment with EBL in a dose-dependent manner, where EBL (400 mg/kg; p.o.) was found to be most effective and showed a similar effect as that of standard Pilex granules (Table 2).
Effect of EBL on RAC and Macroscopic Severity Score.
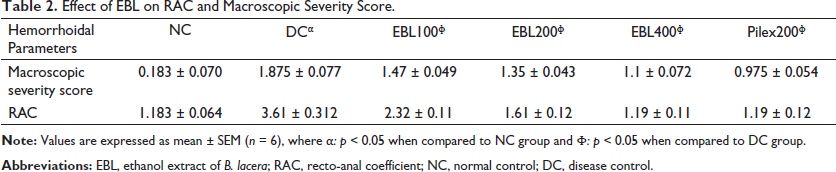
The macroscopic severity score was also observed to increase in the negative control group after COP administration when compared to the normal control group, which was also significantly (p < 0.05) reduced on treatment with EBL and standard, thus confirming minimal recto-anal damage as compared to croton oil control (negative) rats (Table 2) and is well supported by the results of the histopathological examination.
The results also suggested an increased level of pro-inflammatory markers such as TNF-α and IL-6, imparting inflammation on the recto-anal tissues. The increased level of these cytokines was in correlation with a high severity score in the negative control. However, a significant decrease in the levels of these cytokines after the treatment with EBL established the anti-inflammatory nature of the EBL (100, 200, and 400 mg/kg; p.o.) (Figures 2B and C).
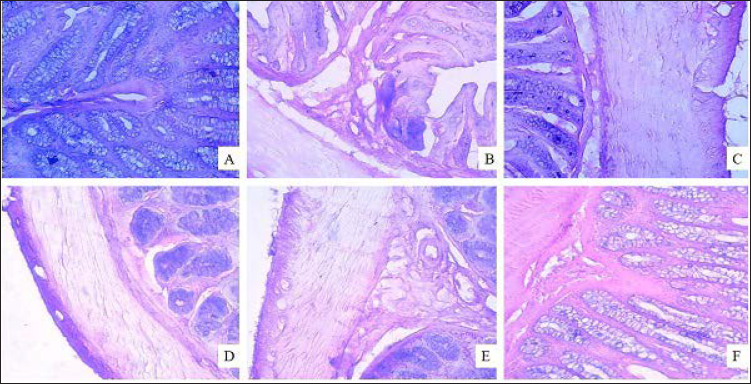
In a histopathological study, normal architecture of recto-anal tissue was observed in a normal control with all three layers intact, namely, muscular, mucosal, and submucosal layers (Figure 3A). Acute inflammatory reactions (Figure 3B) were seen in recto-anal tissue from COP-treated rats, along with necrosis, severe edema, and other degeneration of the mucosal layer. Dilated blood vessels and a focal area of congestion and hemorrhages were also seen as compared to normal control rats. Thickening of the mucosal layer along with fibroblast escalation in mild to moderate form was also observed in the inflamed portion. In treatment groups, less severe lesions in the recto-anal tissues as compared to the COP-treated negative control group were noted with minimal to mild score (Figures 3C and D). EBL (400 mg/kg; p.o.) and Pilex granules-treated groups exhibited marked to moderate protection, respectively, against histological damage (Figures 3E and F).
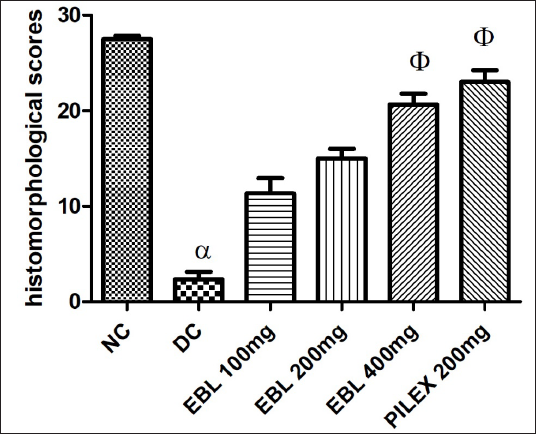
Treatment with EBL (400 mg/kg; p.o.) exhibited a higher total histomorphological score when compared with the croton oil control group (p < 0.05). EBL (100 and 200 mg/kg; p.o.) and Pilex (200 mg/kg; p.o.) did not show any significant recovery of lesions (Figure 4).
In vivo Antioxidant Studies
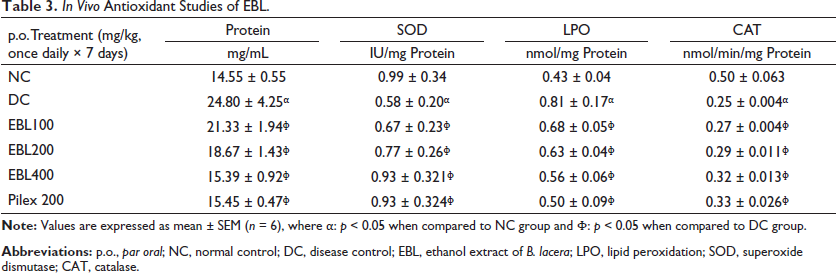
Results of the in vivo antioxidant evaluations revealed that the antioxidant capacity of EBL at 400 mg/kg; p.o. was significant and quite comparable with that of standard Pilex granules. This was significantly justified by increased levels of SOD and CAT with a concomitant decrease in the level of LPO on treatment with EBL (Table 3).
In vivo Antioxidant Studies of EBL.
Discussion
Process optimization helps in getting improved results in both qualitative and quantitative ways. The practical yield of extract (60 g in 150 g of crude leaves) was in accordance with the extraction efficiency (39.24%) calculated using BBD. The results of in vitro antioxidant and anti-inflammatory activity revealed that the EBL could be an effective agent in treating diseases involving oxidative stress and inflammation, such as hemorrhoids. To assess the safety of EBL upon oral administration, an acute oral toxicity study was performed, which showed that EBL could be safe up to 2 g/kg.
The present study involves croton oil as an inducing agent for the induction of hemorrhoid in rats as it contains active phorbol esters, 12-O-tetradecanoylphorbol-13-acetate, which exerts inflammatory reactions followed by polymorphonuclear leukocyte infiltration, vasodilatation that results in the development of edema in tissue. This active constituent from croton oil enables the release of inflammatory mediators like prostaglandins, leukotrienes, bradykinins, nitric oxide, chemokines, and cytokines, which accelerate inflammation. These factors together trigger the regulation of endothelial cells, mast cells, fibroblasts, and macrophages, along with newly assigned inflammatory cells like eosinophils, neutrophils, lymphocytes, and monocytes that contribute to severe inflammation (Azeemuddin et al., 2014; Dhaswadikar et al., 2022). In our study also, application of COP caused severe inflammation and significantly increased macroscopic severity scores and the RAC in negative control as compared to normal control, which confirmed the induction of hemorrhoids, complying with the earlier research findings (Azeemuddin et al., 2014; Dey et al., 2016). Significant amelioration of hemorrhoidal parameters after 7 days of treatment with EBL and Pilex is suggestive of the curative effects of EBL. Application of COP in the recto-anal portion causes extreme vasodilatation and subsequent inflammation, which is associated with bleeding and is a diagnostic feature for hemorrhoids (Brill et al., 2005; Kaidar-Person et al., 2007). Our study also revealed a significant increase in Evans blue concentration in recto-anal tissue of negative control, affirming severe vasodilatation by COP when compared with normal control in the Evans blue dye extravasation test, which was further justified by observations of recto-anal tissue histology (observed for bleeding, inflammation, and marked edema). However, treatment with EBL caused a significant reduction in the level of Evans blue dye, confirming a marked reduction in the inflammation caused in the recto-anal tissues. The lower scores of the histomorphological examinations and in vitro anti-inflammatory study also justified the protective role of EBL in hemorrhoidal conditions.
As discussed earlier, migration and subsequent release of many inflammatory cells and mediators are accelerated by COP application (Brill et al., 2005; Feghali & Wright, 1997). In the same manner, the present study also showed significantly higher levels of cytokines like TNF-α and IL-6 in serum on COP application, which compliments the earlier research outcomes (Azeemuddin et al., 2014; Gurel et al., 2013) and was also confirmed by severity score analysis. The decrease in the levels of these cytokines in serum after treatment with EBL affirmed the anti-inflammatory role of EBL, which is very essential for any candidate to have anti-hemorrhoid potential (Basnet et al., 2015).
The anti-inflammatory role of EBL was also supported through histopathological observations in which animals treated with EBL demonstrated a lower number of inflammatory cells and lowered degenerative changes, hypertrophy, necrosis, and vasodilatation with a few hemorrhagic spots as compared to the recto-anal tissue of the negative control group. A moderate reduction in histological damage after treating with Pilex granules was also seen; however, treatment with EBL showed marked reduction in the histological damage.
Studies have revealed that croton oil causes an increase in the expression of reactive oxygen species, free radical scavengers, and leukocytes infiltration, involving various neurotransmitters, which hampers the antioxidant status and was also confirmed through the observations of our negative control group rats (Dubey et al., 2018; Majed et al., 2015; Nishiki et al., 1988). Treatment with EBL significantly decreased LPO and increased SOD and CAT levels, suggesting inhibition of peroxidative tissue damage, possibly because of the antioxidant properties of EBL. This suggests that the anti-inflammatory as well as antioxidant properties of the EBL played a crucial role in exerting the anti-hemorrhoid effect; the same has been exhibited by the standard drug Pilex granules (Azeemuddin et al., 2014; Dey et al., 2016).
The vasoactive ions were also estimated for the first time in this plant, which showed the presence of various vasoactive ions, especially the vasoconstrictive ions. It is observed that vasoconstrictive ions can play an important role in the reduction of swelling, itching, and discomfort caused by blood vessel constriction (Hodes, 1996; Sause, 1995). However, the systemic effects of these ions are limited due to strict internal homeostasis. On the contrary, vasoconstrictor agents show synergistic effects, as shown previously; therefore, it might be possible that the vasoconstrictor ions in B. lacera could contribute to the curative effects of the plant in the treatment of hemorrhoids (Gul & Yildiz, 2002).
The results of preliminary phytochemical screening confirmed the availability of phenols, alkaloids, tannins, and flavonoids as major components in EBL. These phytoconstituents have been well reported to have anti-inflammatory and antioxidant activity (Dhaswadikar et al., 2022; Sengar et al., 2015; Yang et al., 2013), which directly or indirectly contributed to the anti-hemorrhoidal potential of EBL. Furthermore, EBL was standardized with the help of HPLC using quercetin as a marker. The marker quercetin has been reported to have potent antioxidant and anti-inflammatory activity and has specifically been proven to prevent gastrointestinal inflammation (Dey et al., 2016). A clinical study also confirmed the role of quercetin, along with four other flavonoids, in treating I–III degree hemorrhoids (Corsale et al., 2018). Thus, we may presume that quercetin could play a significant role in the observed anti-hemorrhoidal potential of EBL.
Conclusion
From the overall observation, we have successfully justified the traditional claims of the plant B. lacera in the treatment of hemorrhoids, which may be attributed to its potent anti-inflammatory and antioxidant effects. The role of quercetin in combination with other phytoconstituents could be the main contributor to the above therapeutic efficacy. However, for elucidation of the mechanism of action, further studies like the isolation of lead molecules are needed. Furthermore, clinical and formulation studies are also required to properly validate the therapeutic potential of B. lacera for human use.
Supplementary Material
Supplemental material for this article is available online.
Supplemental Material for Optimization of Extraction Process and Anti-Hemorrhoidal Activity of Blumea lacera (Burm.f.) DC. Leaves in Croton Oil-induced Hemorrhoid Model by Tarkeshwar Dubey, Kancharla Bhanukiran, Satyendra K. Prasad and Siva Hemalatha, in Pharmacognosy Magazine
Footnotes
Summary
B. lacera is used for treating hemorrhoids by several traditional healers. EBL was chemically characterized and standardized through HPLC using quercetin as a standard. The study showed that EBL possessed significant in vitro anti-inflammatory activity. Furthermore, EBL had significant anti-hemorrhoidal and cytokines inhibitory action at the molecular level. Therefore, the traditional claims of the plant were validated, which were attributed to its anti-inflammatory and antioxidant potential.
Abbreviations
EBL: ethanol extract of Blumea lacera; BBD: Box–Behnken design; HPLC: high-pressure liquid chromatography; COP: croton oil preparation; KCl: potassium chloride; PMS: phenazene methosulfate; NBT: nitroblue tetrazolium; p.o: par oral; RAC: recto-anal coefficient; DMSO: dimethyl sulfoxide; TNF-α: tumor necrosis factor-α; IL-6: interleukin-6.
Acknowledgments
The authors would like to thank the Indian Institute of Technology (Banaras Hindu University), Varanasi for Teaching Assistantship.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All the experiments were carried out in accordance with the IAEC (Institutional Animal Ethics Committee) guideline and clearance (letter no. Dean/2016/CAEC/330).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.