Abstract
Background
Modern strategies to alleviate the harmful effects of organophosphate pesticide diazinon (DN) abnormalities were focused mainly on using natural compounds or their derivatives. DN is an organophosphate compound that causes many health abnormalities in humans due to its usage as an insecticide in agriculture. TQ is one of the beneficiary active principles derived from plant sources that has pharmacological benefits.
Aim
This research reveals the therapeutic potential of thymoquinone (TQ) on DN-induced myocardial infarction (MI) in rats.
Materials and Methods
Male Sprague–Dawley rats were procured, acclimatized, and divided into four groups of six animals each with feed and water ad libitum. MI was induced in rats with a dose of 25 mg/kg DN by oral gavage and TQ in a dose of 20 mg/kg p.o. for the treatment. After animal sacrifice at the end of the experimental period, serum and heart tissue samples were collected and processed appropriately for various analyses such as changes in the body weight, heart weight, marker enzymes, oxidative markers, non-enzymatic and enzymatic antioxidants, inflammatory cytokines, histopathological studies, and cardiac-specific markers.
Results
DN-induced toxicity depicted decreased body weight (167.83 ± 4.62), heart weight (0.9 ± 0.06), and heart-to-body weight ratio (0.54 ± 0.03). Also, elevated marker enzymes (147.33 ± 20.85, 407.5 ± 31.3, and 110.67 ± 9.65 for CK-MB, AST, and ALT, respectively), elevated oxidative markers (12.87 ± 1.34, 125.17 ± 9.95, and 80.17 ± 5.78 for serum MDA, heart MDA, and heart GSSG, respectively), decreased enzymatic- and non-enzymatic antioxidants (3.15 ± 0.42, 12.23 ± 1.02, 5.75 ± 0.46, 2.02 ± 0.26, 0.72 ± 0.07, 18.05 ± 1.04, 8.62 ± 0.65, and 45.8 ± 2.43 for SOD, CAT, GST, GPx, heart GSH, serum GSH, vit.E, and vit.C, respectively), damaged cellular architecture, elevated inflammatory cytokines, and cardiac-specific markers were noticed.
Discussion
TQ significantly reduced the toxicities produced by DN in almost all the above parameters. The beneficial effect of DN could be attributed to the influential effect of DN on cardiac-specific Nrf2/HO1-related pathways.
Conclusion
These results suggest that TQ exerts protection against MI and could serve as a promising candidate for drug development.
Introduction
Application and exposure to pesticides are making serious threats to the environment and the living system in developing countries (Aktar et al., 2009). Organophosphate is one of the environmental pollutants causing renal failure, cardiovascular disease, neurological disorders, mutations, and many more abnormalities (Afsar et al., 2019; Figueiredo et al., 2018; Li et al., 2019). In agriculture, insecticides such as organophosphate were used to control the pests that cause cholinesterase inhibition (Colović et al., 2013). Diazinon (DN) (O,O-diethyl–O-[2-isopropyl-6-methyl-pyrimidin-yl]) is used to control the pests in vegetables, fruits, and lawn-crop insects in agriculture. Acute and chronic usage of DN causes stress in different tissues which was studied in humans and animals (Cobilinschi et al., 2022). Hepatic enzyme alterations, biochemical changes, and mitochondrial dysfunction in liver cells were noticed during DN intoxication. DN intoxication can also cause changes in blood-related parameters and induces DNA fragmentation (Cobilinschi et al., 2022).
Recent works of the literature indicated the accumulation of DN in various tissues and contribute to the biochemical and histological modifications. Various reports suggested that oxidative stress was an apoptotic inducer in diseases such as heart disease and ROS production (Nita & Grzybowski, 2016). Recently, organophosphate and DN at low dose was reported to be involved in the apoptotic pathway (Robb & Baker, 2020). The free radical scavenging mechanism plays a pivotal role in controlling oxidative stress by direct scavenging processes (Ahmadinejad et al., 2017).
Thymoquinone (TQ) is one of the active components of Nigella sativa, which has more pharmacological functions such as anti-inflammatory and analgesic functions. TQ safeguards the cell from chemical carcinogens attack by a mechanism that suppresses eicosanoid production (Khader & Eckl, 2014). TQ also suppresses the oxidative stress-induced cellular dysfunction in liver tissues intoxicated with carbon tetrachloride in cellular experimental models (Li et al., 2015). Nevertheless, TQ attenuates pro-inflammatory response in LPS-induced immune cells by modulating the synthesis of tumor necrosis factor-alpha (TNF-α) and NF-kB production. TQ also prevents membrane lipid peroxidation in liver cells (Liu et al., 2017).
With these earlier reports, this study was designed to evaluate TQ’s protective effects against DN-induced toxicities on body and heart weight changes, cardiac biochemical markers, oxidative stress markers, non-enzymatic and enzymatic antioxidants, histopathological studies, inflammatory cytokines, and cardiac-specific markers were studied.
Materials and Methods
Chemicals and Reagents
TQ (274666-5G), Aspartate aminotransferase kit, and Alanine aminotransferase kit were obtained from Sigma-Aldrich company (USA). Diazonin (DN) was purchased from Shijiazhuang Ageruo-Biotech Co. Ltd., Shijiazhuang City, China (CAS No.: 333-41-5; 1g/mL). All other reagents and chemicals used were of higher grade (analytical grade).
Study Protocol
Male Sprague–Dawley (SD) adult rats (200–250g) were procured from the central animal facility of the institution. Animals were maintained in the standard animal cage with pelleted normal animal feed at a room temperature of 25 ± 2°C with purified drinking water and food ad libitum. The study protocol was approved by the ethics committee of Shanxi Cardiovascular Hospital (Approval number 2022010) before the initiation of this research work. After acclimatization, rats were grouped into 4 groups of 6 animals each. Group I is considered as control and normal saline administered p.o. Group II rats were treated with TQ (20 mg/kg/day p.o.; dissolved in 80% ethanol) for 10 days. Group III rats received 25mg/kg b.wt DN by i.p. injection (each on days 7 and 8). Group IV rats treated TQ (as in group II) along with DN (as in group III). The above doses were decided by optimization study by estimating the levels of serum marker enzymes in an optimization study (Data are not shown in the manuscript).
Samples Collection
Blood
At the end of the experimental days, rats were weighed and sacrificed under ketamine anesthesia. Blood was collected by cervical decapitation and serum was prepared after 30 minutes by centrifuging the coagulated blood at 3000×g for 15 mins, which was stored at –20oC until further analyses.
Heart
Rat’s heart was quickly dissected out, cleaned in cold saline, weighed, and then stored at –20oC for the analyses of biochemical parameters. The left ventricle of the heart of all groups was fixed using 10% formalin for histopathological studies.
Measurement of Bodyweight (BW) and Heartweight (HW)
The body weights of the experimental rats were measured before sacrificing the animals at the end of the experimental period. Before weighing the excised heart tissues, fat and fibrous tissues were freed and dried using filter paper. The cardiac index was calculated by the ratio of HW to BW which was expressed numerically (Vasan, 2003).
Activities of Marker Enzymes in Serum
Serum creatine kinase isoenzyme-MB (CK-MB) activity was done using a biochemical kit (Stanbio, USA) (Piran et al., 1987). Serum-level biomarkers such as alanine transaminase (ALT), and aspartate transaminase (AST) were analyzed as per the standard protocol (Atiba et al., 2016).
Levels of MDA, Oxidized Glutathione (GSSG), and Glutathione (GSH)
Levels of MDA in cardiac tissues and sera were assessed using the method of Noeman et al. (2011). Briefly, the formation of MDA as a lipid peroxidation product that reacts with a chromogen (thiobarbituric acid reactive substances) was measured spectrophotometrically at 532nm and quantitated against the standard curve. Levels of GSH in cardiac tissues and sera were assessed using the method of Moron et al. (1979). Briefly, the reaction product of GSH with 5,5′-dithiobis(2-nitrobenzoic acid) (DTNB) was measured spectrophotometrically at 412nm.
Levels of GSSG were done by the methods of Mcgill and Jaeschke (2015) in the heart tissues. Briefly, the sample was mixed with 2-vinyl pyridine and triethanolamine (to pH 7.0-7.5), incubated at room temperature for 1 hour, and measured. The ratio of GSSG/GSH was calculated manually and used as an oxidative stress indicator.
Levels of Non-enzymatic Antioxidants in Serum
Levels of vitamin E were determined by the method of Desai (1984) and vitamin C was determined by the method of Omaye et al. (1979).
Levels of Enzymatic Antioxidants in Heart Tissue
Superoxide dismutase (SOD) and catalase (CAT) were done by standard methods (Zhang et al., 2013). Activities of glutathione peroxidase (GPx) and glutathione-S-transferase (GST) were done by the method of Kuo et al. (2002) spectrophotometrically. BSA was used as a standard for protein determination (Redmile-Gordon et al., 2013).
Histopathological Study
Heart tissues were microtomed in a thickness of 5 µm, stained with hematoxylin and eosin (H&E), and examined under a compound microscope with high resolution at 40x magnification.
Specific Markers in the Heart by ELISA
Levels of specific markers in the heart were done by ELISA according to the manufacturer’s instructions. Levels of Nrf2, HO-1, NQO1, and SOD were analyzed using the samples. After the color development using chromogen, absorption was measured at 450nm using a plate reader. All the samples were done in triplicates and comparisons were made between groups as appropriate.
Statistical Analysis
Results were presented as mean ± SD. Statistical significance was considered at p≤ 0.05. Statistical evaluations between various groups were analyzed using SPSS statistical software and comparison was done by ANOVA followed by post hoc test.
Results
Effect of TQ on Body Weight and Heart Weight Changes
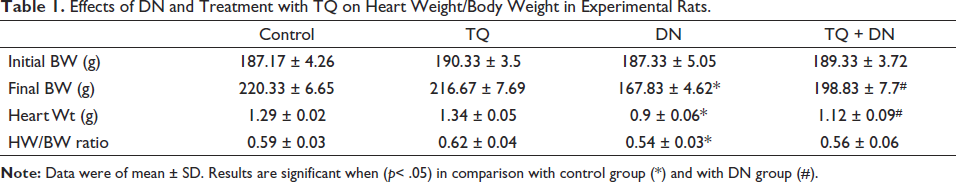
A noticeable change in the final body weight of DN-treated rats was observed when compared to control rats. Rats treated with TQ showed remarkable changes from DN-administered rats with significant statistical differences. Improvement in the HW to BW ratio was noticed (0.56 ± 0.06) in the treatment group of rats. Rats treated with TQ alone showed no significant change in the body weight or HW/BW ratio compared to the normal control. But an improvement in the heart weight was noticed (0.62 ± 0.04) without statistical significance. All these data are presented in Table 1.
Effects of DN and Treatment with TQ on Heart Weight/Body Weight in Experimental Rats.
Effect of TQ on Marker Enzymes
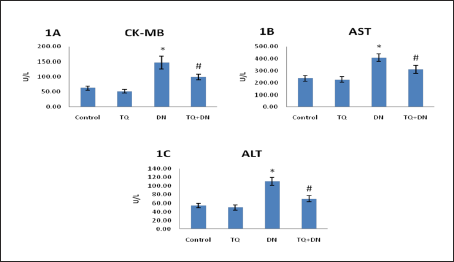
In Figure 1A–1C, serum cardiac marker enzymes (CK-MB, ALT, and AST) were shown. DN-treated rats showed significantly elevated enzyme activities as compared with control rats (147.33 ± 20.85, 110.67 ± 9.65, and 407.5 ± 31.3, respectively), and these abnormalities were alleviated in the TQ treatment. Noticeably lesser activities of these enzymes without statistical significance (52.17 ± 5.98, 49.83 ± 5.98, and 226.83 ± 23.63, respectively) were noticed in TQ alone administered rats.
Effect of TQ on Oxidative Stress Markers
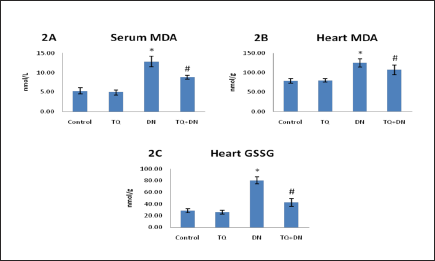
Oxidative stress marker MDA in the serum and heart were listed in Figure 2A and 2B. Stress-induced during DN administration significantly increased MDA content both in serum and heart compared with the control group. TQ treatment to DN intoxicated rats significantly decreased MDA levels when compared with the DN alone group of rats, recommending the protective antioxidant role of TQ in contrast with the myocardial infarction (MI) developed by DN alone. Figure 2C depicts the levels of heart GSSG that exhibit significant elevation in DN intoxication and normalized levels during TQ treatment. The level of increase in heart GSSG was ≈283% in contrast to heart GSH (Table 2) decrease of ≈48%. Approximately ≈5.9 fold change in the ratio of GSSG/GSH ratio in the heart was recorded in our study. No significant variations were noticed with TQ-only administered group of rats. These results correlated well with the marker enzymes results.
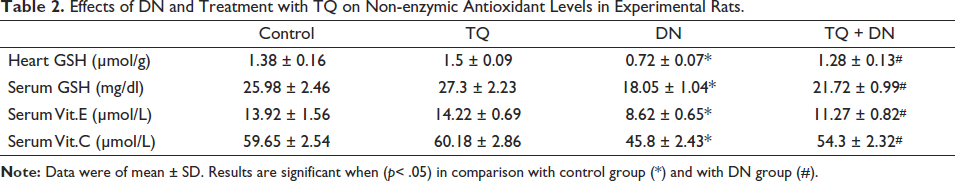
Effects of DN and Treatment with TQ on Non-enzymic Antioxidant Levels in Experimental Rats.
Effect of TQ on Non-enzymatic Antioxidants
Table 2 shows non-enzymatic antioxidants such as GSH (heart and serum), vit-E and vit-C in serum. After the experimental period, DN exhibited a significantly lower content of GSH in heart tissues and serum. Likewise, significantly lowered levels of vit-E and vit-C were also noticed in the serum of experimental rats. Administration of TQ with DN during the experimental period significantly increased GSH (heart and serum), vit-E and vit-C contents (1.28 ± 0.13, 21.72 ± 0.99, 11.27 ± 0.82, and 54.3 ± 2.32, respectively) to near normal levels in the heart tissues and serum. An improvement in the levels of all these parameters (1.5 ± 0.09, 27.3 ± 2.23, 14.22 ± 0.69, and 60.18 ± 2.86, respectively) were noticed in TQ alone administered rats that accounted a beneficial effect. These results correlated well with the marker enzymes and oxidative status of the groups in the experiment.
Effect of TQ on Enzymatic Antioxidants
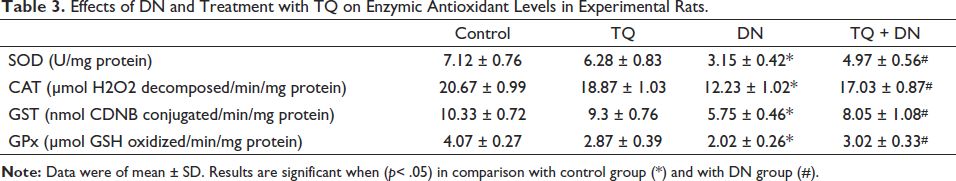
Activities of antioxidant enzymes such as CAT, SOD, GPx, and GST in the heart of the experimental groups are presented in Table 3. DN administration showed significantly decreased activities of CAT, SOD, GPx, and GST (18.87 ± 1.03, 6.28 ± 0.83, 2.87 ± 0.39, and 9.3 ± 0.76, respectively) in the heart tissue compared to the corresponding values of control rats. Treatment with TQ completely prevented DN-induced abrogation in the activities of these enzymes (17.03 ± 0.87, 4.97 ± 0.56, 3.02 ± 0.33, and 8.05 ± 1.08, respectively) in the heart tissues. However, TQ alone administered rats showed a slight decrease (without statistical significance) in the activities of these antioxidant enzymes (18.87 ± 1.03, 6.28 ± 0.83, 2.87 ± 0.39, and 9.3 ± 0.76, respectively) in the heart especially GPx activity. The decrease in the GPx activity could be due to the quenching of free radicals and the antioxidant effect of TQ leading towards its beneficial activity that correlated well with the oxidative status and non-enzymatic antioxidants of the groups in the experiment.
Effects of DN and Treatment with TQ on Enzymic Antioxidant Levels in Experimental Rats.
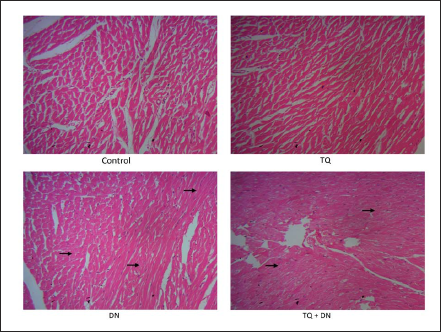
Effect of TQ on Histopathological Changes
Figure 3 shows the histopathological changes in experimental rats. Microscopic studies of DN-induced rats showed the penetration of inflammatory markers, necrosis and hyperemic condition in cardiac muscle cells. During treatment with TQ, restoration of inflammatory cells, lesser necrosis, and hyperemia were noticed. TQ alone treated group and control showed no architectural changes.
Effects of DN and Treatment with TQ on the Histopathological Injuries in the Heart of Experimental Rats. All images were at 40x magnification.
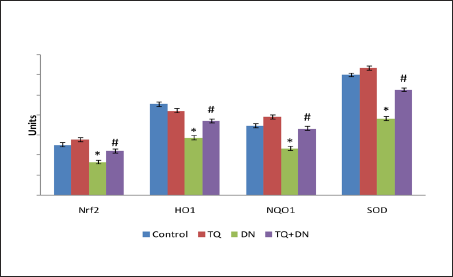
Effect TQ on the Levels of Nrf2, HO-1, NQO1, and SOD in the Heart
In Figure 4, levels of Nrf2, HO-1, NQO1, and SOD were presented. DN administration decreased all the levels of these markers significantly compared to control rats. During treatment with TQ, significantly increased Nrf2, HO-1, NQO1, and SOD were noticed (22.2 ± 1.41, 37.2 ± 2.6, 33.37 ± 4.28, and 52.7 ± 3.05, respectively), when compared to the DN intoxicated group. However, though an increase in the expression of SOD was noticed during treatment with TQ, data was not statistically significant. All these data correlated well with the status of the other parameters in the study.
Discussion
Cardiovascular disease is the foremost source of fatality (Daher et al., 2012). This research focused on TQ’s action on DN intoxicated cardiac damage in rats. DN intoxication caused myocardial damage and oxidative burst in various experimental models. A decrease in HW and HW/BW ratio in a significant manner indicated a myofibrillar loss and necrosis in myocardium. Reductions in body weight and heart weight were associated with the toxic induction of DN in animal models (Moris et al., 2017). Restoration of body weight and improvement in heart weight in TQ treatment showed beneficial effects of TQ treatment.
Cardiotoxicity enzyme markers in the serum were the vital markers and their measurement in serum determines the extent of cardiac damage (Tian et al., 2014). Cardiac tissues are rich in enzymes such as CK-MB, AST, and ALT. Leakage of these enzymes to serum due to myocardial toxicities and/or damage leads to notably elevated levels in serum (McGill, 2016). As shown in our study, these enzyme levels were elevated in DN intoxication and diminished by TQ treatment that showed a cardioprotective effect due to TQ treatment.
In the current study, considerable damage in the heart might be due to oxidative stress created by DN. As a result, cardiac muscles showed increased levels of MDA and GSSG during cardiac damage caused due to DN administration. DN causes a rise in cardiac and serum MDA together with GSSG due to the higher formation of free radicals which results in oxidative stress-implicated tissue damage (McGill, 2016). GSH is a cofactor in numerous enzymatic and signaling pathways of different types of machinery, which may boost up the reaction of association of intracellular electrophilic conversion of oxidative products. Importantly GSH involves as a main cofactor for GPx and GST involved the removal of oxidative radicals (Desai, 1984). Our results depicted notable reduced GSH levels and a drastic increase in the GSSG/GSH ratio in the heart tissue, which can occur due to its usage as an antioxidant molecule. In this study, TQ treatment showed significantly decreased MDA and GSSG formation compared to control rats which accounted for the TQ’s beneficial effect. GSH usually exists in inconvertible form. However, during higher amounts of ROS formation, GSSG levels could enhance and the ratio of the formation of GSSG could be higher than the removal reaction (Forman et al., 2009). Perhaps, findings from our research indicated that TQ has a significant efficacy to control the levels of oxidized and reduced glutathione pools and able to decrease the GSSG/GSH ratio.
Non-enzymatic antioxidants (GSH, Vitamin C, and Vitamin E) and enzymatic antioxidant enzymes (SOD, CAT, GPx, GST) of myocardial tissue plays an important role in defense against free radicals and oppose the generation of oxidative radicals (Desai, 1984). Vitamin-C and Vitamin-E were proven to act together in synergy during the production of free radicals (Zingg & Azzi, 2004). Levels of Vitamin C and Vitamin E were severely affected during the administration of DN in our study, which is consistent with the previous study reported (Abdel-Daim, 2016). However, during TQ treatment, levels of these vitamins were restored to normalcy, which augmented the quenching and antioxidative effect of TQ. Treatment with TQ alone showed improvement in the antioxidant enzymes’ actions which indicated its role as a potent free radical scavenger. Management of SOD involved in the maintenance of mitochondrial ROS synthesis during metabolic pathways could secure the cells against oxidative stress (Snezhkina et al., 2019). DN intoxication significantly decreased SOD activity, which may be due to the occurrence of oxidative stress and impaired mitochondrial energy, needed for normal cardiac activity. Reduction in CAT function depicts its critical function in managing ROS in cardiac tissue. GPx were found to be extensively low in cardiac muscle after administration of DN. Generally, GPx gives protection to cell membranes from oxidative damage by initiating the elimination of hydrogen peroxide via loss of electrons in GSH by the action of glutathione reductase (Espinosa-Diez et al., 2015). In the current study, TQ enhances GSH and GPx activities in DN-intoxicated rats by a beneficial mechanism. From these findings, it was evident that TQ has a strong free radical scavenging effect and prevents oxidative stress-induced insult through enhancing antioxidant activity. Similarly, previous studies documented that TQ has strong antioxidant and free radical quenching activity (Khalife & Lupidi, 2007). Developmental levels of antioxidant enzymes could exist in myocardial stress-induced changes due to higher amounts of ROS (Rahman, 2007). Higher activities of antioxidant enzymes were observed in the treatment group of our study, which gave not enough room to cause oxidative stress leading to protection of the myocardium from lipid peroxidation. However, administration of TQ alone decreased the activities of SOD and CAT, which could be interpreted as a lesser expression of these enzymatic antioxidants due to the quenching effect of TQ in vivo that was reported earlier in the cardiac tissues (Li et al., 2015).
Histopathological observations showed focal cell infiltrations and symptoms of edema in the heart tissues during the toxicities produced by DN. These findings were consistent with the previously reported studies (Mohafrash & Mossa, 2020). These abnormalities were attenuated during TQ treatment.
Nrf2 is the redox-sensitive master regulator and transcription factor which is considered to be involved in the oxidative stress mechanism. At normal physiological conditions, Nrf2 was found to bind with its inhibitor Keap1 in the cytoplasm. In oxidative stress, alteration of Keap1 conformation leads to forced dissociation of Keap1 from the bound Nrf2, leading to the entry of Nrf2 into the nucleus (Buendia et al., 2016). After entry into the nucleus, Nrf2 binds with antioxidant response elements (ARE) and enhances the expression of protective genes such as HO-1 and NQO1 (Hybertson et al., 2011). Our study correlated well with previous studies that depicted decreased levels of Nrf2, HO-1, and NQO1 during DN intoxication (Buendia et al., 2016). Nevertheless, earlier reports documented the involvement of BRG1 by its direct recruitment for the binding of Nrf2 with ARE during the induction of HO-1 gene expression (Garnier et al., 2013). Also, Nrf2 regulates numerous actions such as phosphorylation of Nrf2 by upregulated kinase and strong Nrf2 production via Keap1 cysteine changes (Zhang et al., 2013). TQ interacts with these signaling pathways, mainly through the association between the Nrf2/Keap1 pathways and regulating genes involved in cell secure molecules and antioxidant enzymes. An important antioxidant gene, SOD1, essentially occurs in the cytoplasm and mitochondria that involves in the mechanisms to remove superoxide radicals (Fukai & Ushio-Fukai, 2011). Induction of ROS production such as superoxide and hydrogen peroxide radicals could activate the expression of antioxidant enzymes (Pruchniak et al., 2016) that could increase the expressions of enzymatic antioxidant genes such as SOD and CAT (Zhang et al., 2013). Finally, it could be explained that the restoration of these gene expression levels was due to TQ treatment, modulated by Nrf2 gene networks that protected the heart from myocardial injury.
Conclusion
In this research, we documented DN-intoxicated severe abnormalities in the heart of rats. We reported the treatment efficacy of TQ on DN-induced MI, which showed improvement of enzymatic and non-enzymatic antioxidants, cellular oxidative status, histopathological alterations, and inflammatory markers towards cardioprotection. Further, we noticed that TQ modulated Nrf2-related cell protection and might have an effective prophylactic strategy in the suppression of DN-related abnormalities during MI.
Abbreviations
DN: Diazinon; TQ: Thymoquinone; MI: Myocardial infarction; SD: Sprague–Dawley; CK-MB: Creatine kinase MB; AST: Aspartate tramsaminase; ALT: Alanine transaminase; MDA: Malondialdehyde; GSH: Reduced glutathione; GSSG: Oxidized glutathione; SOD: Superoxide dismutase; CAT: Catalase; GST: Glutathione-S-transferase; GPx: Glutathione peroxidase; Vit E: Vitamin E; Vit C: Vitamin C; TNFα: Tumor necrosis factor alpha; NfkB: Nuclear factor κB; DTNB: 5,5′-dithiobis(2-nitrobenzoic acid; ELISA: Enzyme-linked immunosorbent assay; ANOVA: Analysis of variance; Nrf2: Nuclear factor erythroid 2 related factor; HO1: Heme oxygenase 1; NQO1: NAD(P)H quinone oxidoreductase 1.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author received no financial support for the research, authorship and/or publication of this article.
Ethics Approval and Consent to Participate
The study protocol was approved by the ethics committee of Shanxi Cardiovascular Hospital (Approval number 2022010) before the initiation of this research work.
