Abstract
Background
The thyroid is the prime organ of infection during the onset of autoimmune disease. The disease is known as Autoimmune thyroiditis (AIT). In recent times, AIT has been alarming worldwide, with growing attention towards pharmaceutical companies for finding new drugs for therapy. The present investigation aims to use a multi-targeting approach to identify the best compound from the root of Astragalus membranaceus for synergistic therapy of AIT.
Materials and Methods
To explore the mechanism of action of the compounds, the present study uses network pharmacology-based methods. Totally, 48 genes involved in AIT were collected from the Kyoto Encyclopedia of Genes and Genomes pathway (KEGG). Gene ontology (GO) annotation, disease ontology, and pathway enrichment analysis were conducted. The chemical structure of the compounds from A. membranaceus was retrieved from the PubChem database. To study the toxic and pharmacological efficiency of the chemical structure, absorption, distribution, metabolism, excretion, and toxicity (ADMET) screening was performed.
Results
The ononin and astroside were identified as suitable with all the pharmacokinetic properties can be employed to identify the disease targets. The intersection between the compound target (C-T-N) and the AIT-related genes was identified. Totally 16 genes among the C-T networks of ononin and astroside have shown interaction with thyroid disease-related pathways and signaling pathways. Consequently, the overlapping genes between the C-T network and AIT-related genes were determined as IL2. However, from the pathway enrichment, AIT-related genes have been involved in several viral infection pathways. The genes involved in infection pathways are also targeted by ononin and astroside.
Conclusion
The underlying mechanism of action of the compounds on AIT-related pathways was also determined. Therefore, a network pharmacology-based strategy will provide new insight into AIT therapy. However, further studies are required to validate the investigation that can prove bioactive as a promising candidate for the treatment of AIT.
Introduction
Autoimmune thyroiditis (AIT), is an organ-specific autoimmune disease that targest the thyroid compared to other organs of the human body (Boguslawska et al., 2022). AIT is the most common thyroidopathy observed in children and adolescents. The two main parts of AIT are Hashimoto’s thyroiditis and Graves’ disease. Hashimoto’s thyroiditis (Mincer & Jialal, 2023) is known as hypothyroidism, and Graves’ disease is hyperthyroidism (Segni, 2000). Hypothyroidism and hyperthyroidism are convertible with the existence of suitable conditions (Wong & Inder, 2018). The clinical conditions for this interplay include genetic, endogenous factors with the loss of thyroid antigens, and environmental factors. The main mechanism of the pathogenesis reported for AIT is cell-mediated and humoral-mediated immunity (Siano et al., 2021). The activation of T-cell and B-cell pathways, lymphocytic infiltration, and loss of immunological tolerance lead to the damage of the thyroid gland resulting from AIT. Consequently, environmental factors such as radiation exposure, stress, pregnancy, and iodine intake are key players of AIT disease (Didier-Mathon et al., 2021). Mostly, men, women, and children reported hypothyroidism in developed and developing countries. AIT occur during other autoimmune disorders such as Type1 diabetes mellitus (Vallianou et al., 2019), celiac disease, and vitiligo (Sandru et al., 2021). In children, the AIT is observed along with genetic syndromes such as Down syndrome Klinefelter, Turner, and Williams syndrome (Kyritsi & Kanaka-Gantenbein, 2020). The abnormalities observed in RAS/MAPK pathways related to the expression of the proteins, activation, and signaling process lead to the activation of inflammatory response and autoimmune mechanisms associated with AIT (Siano et al., 2021).
AIT patients have a vast array of remarkably adverse effects, including primary thyroid lymphoma, papillary thyroid cancer (Lee et al., 2017), other autoimmune disorders, cardiovascular diseases, and malignant neoplasms. Clinically, the most common manifestation is an enlarged thyroid gland (goiter), tiredness, weight gain, muscle weakness, thyroid eye disease, apoptosis of thyroid epithelial cells, follicular destruction, and the occurrence of antibodies that are specific to thyroid antigens (Frohlich & Wahl, 2017). Moreover, earlier reports state that humoral and cellular immune responses play critical roles in the pathogenesis of AIT through antibody, complement, and cytokine-mediated cytotoxicity (Ganesh et al., 2011). The thyroidal tissue exhibits specific selenoproteins, and the deficiency of selenium results mainly in the onset of thyroid autoimmunity, whereas its supplementation protects the thyroid gland from AIT disease (Pace et al., 2020). There is also a complex relationship between the liver and thyroid organ that balance health and disease. The liver regulates thyroid hormone activation/inactivation, transport, and metabolism. Hepatitis C viral infection in the liver has also resulted in AIT (Piantanida et al., 2020). Hence, the mechanism of etiology associated with AIT is still unclear. For any therapy, the diseased pathway and its mechanisms need to be understood. Hence, the present study aims to study the mechanism of action of the proteins involved in AIT and their activation pathways, which can be targeted by plant molecules.
In recent years, there has been a great deal of interest in newer bioactive compounds from the plant kingdom with health-promoting potential and being a remedy for most of the common diseases. Astragalus spp.., flowering plants, annual or perennial herbs, subshrubs, or shrubs grow throughout the temperature-arid regions (Sun et al., 2022). It is worthwhile mentioning that the main constituents of Astragalus spp., are Saponins, flavonoids, anthraquinones, alkaloids, polysaccharides, amino acids, and metallic elements (Veit, 2021). Astragalus is considered a medicinal food, which has a long history of medical use in the Traditional Chinese Medicines (TCM) and is also one of the most popular herbal medicines worldwide (Li et al., 2014).
Research on East Asian grown astragalus species dried roots reveals a number of analeptic properties as an antiperspirants, hepatoprotective, diuretics and tonics for a wide array of diseases including empyrosis, nephritis, diabetes mellitus, cirrhosis, cancer, hypertension, and leukemia (Balakrishnan et al., 2021). It has exhibit antioxidant, anti-inflammatory, immunomodulating, anticancer, and antiviral effects (Wang et al., 2022). Traditional herbal medicines play an important role in the body’s resistance to illness through effects on immune system components (Han et al., 2021). Thus, in the present study network pharmacology-based investigation will be performed to reveal the active ingredients of Astragalus for treatment of AIT and their molecular mechanism of action will also be revealed.
Materials and methods
Protein-protein Interaction (PPI) Network
The candidate targets of AIT were predicted through Kyoto Encyclopedia of Genes and Genomes pathway (KEGG) with the identification number as hsa05320. Later, the genes were imported into STRING database (
ADMET Properties
The in silico evaluation of absorption, distribution, metabolism, excretion, and toxicity (ADMET) is a very useful method to design and optimize lead molecules. For this, the molecules with different chemical structures were retrieved from the PubChem database, and the corresponding SMILES were loaded on the webserver to predict the ADMET properties (Table 1). The web server ADMETlab 2.0, available at
The Information of Compounds Such as Group, SMILES and PubChem ID of the Chemical Structures Identified From the Root of A. membranaceus was Represented.
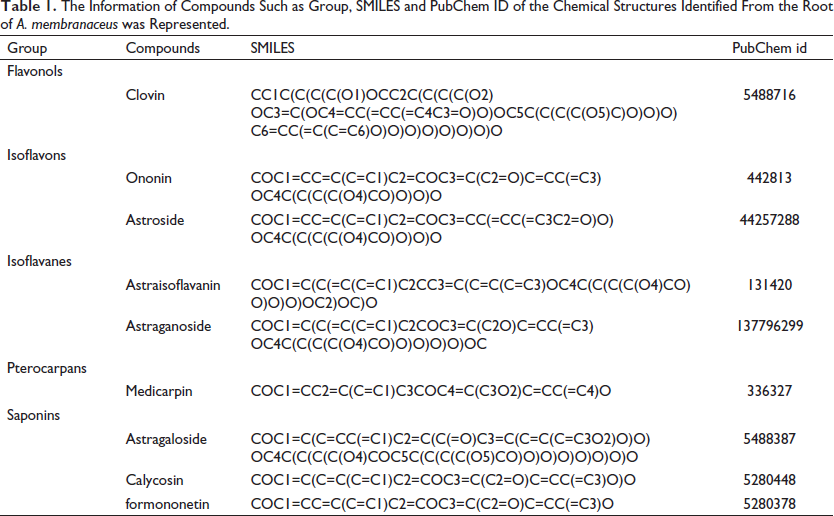
Construction of the Compound-target Network (C-T-N)
The network of component–target interactions was established between active compounds from Astragalus and their core target proteins. For this SwissTargetPrediction online server was used (
Gene Ontology (GO) Analysis
DAVID (database for annotation, visualization, and integrated discovery) is a functional annotation (biological process, cell composition, and molecular function) and pathway the proteins are integrated with the enrichment tool (Jiao et al., 2012). This public annotation database allows the sorting of the gene based on different categories, such as GO analysis (DAVID 6.8). EASE scoring method is used for sorting the genes and validating the enrichment. The hub genes of AIT were annotated using the GO terms, and distribution based on the hierarchy was explained.
KEGG Enrichment Analysis
The functional attributes of the target protein and C-T-N proteins were allocated in the pathways using KEGG enrichment analysis (Chen et al., 2015). The score method for allocating the genes with default parameters (gene count >5 and p < 0.05) includes the threshold values to allocate target genes to distinct pathways to determine their functional attributes. The threshold values were gene count >2 and p < 0.05.
Results
PPI Network
PPI interaction reveals the biological mechanisms of the proteins that are crucial in disease. In absence of the detailed information on the proteins, a network-based approach predicts the interactome of the gene involved in AIT disease pathway. Thus, STRING analysis has revealed the 48 proteins involved in AIT (Figure 1). The representation of line color in the network indicates the protein-protein interaction. The attributes such as homology, text mining data, gene fusion, experiment and co-expression are shown by purple, light green, red, pink, and black color lines respectively. The string network has shown with 48 nodes, 356 edges, the average node degree was 14.8, average local clustering coefficient is 0.745, expected number of edges as 27, and PPI enrichment p-value as < 1.0e-16. From the clustering analysis, the network has revealed five different clusters (Table 2) occupied with the target proteins.
STRING Analysis for AIT-related Proteins. Totally 5 Clusters are Represented with Different Color Codes (Red, Blue, Green, Cyan, and Yellow).
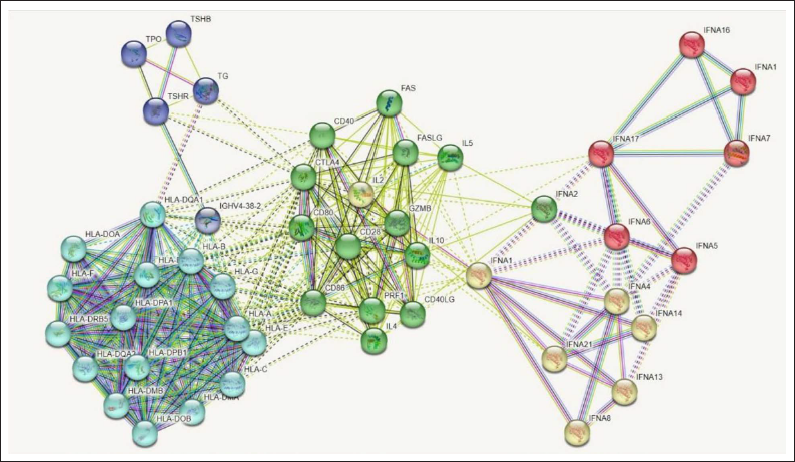
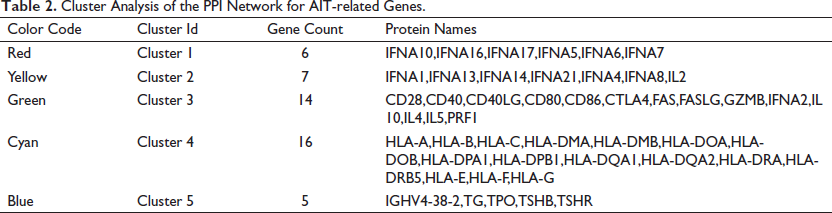
Cluster Analysis of the PPI Network for AIT-related Genes.
ADMET Investigation
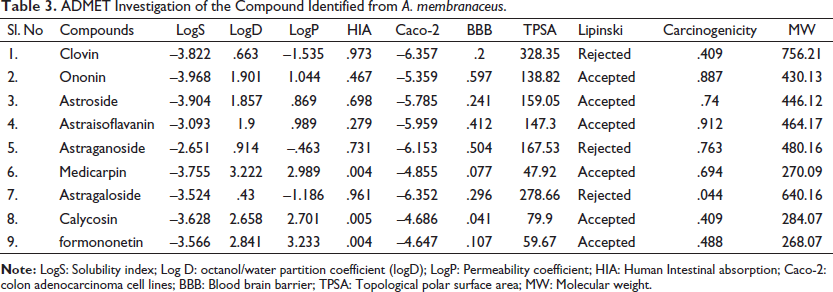
The compounds from root of Astragalus membranaceus were subjected to ADMET analysis to study different pharmacokinetic and toxicity properties. It was observed that all the nine compounds are in the range of solubility index (–4 to 0.5 log mol/L) and hence considered proper with LogS. The predicted octanol/water partition coefficient (logD) value of a compound from 1 to 3 log mol/L will be considered proper. The compounds such as clovin, Astraganoside, and Astragaloside are below the range of 1 and Medicarpin logD is 3.222 which is above the range. The predicted LogP value range from 0 to 3 log mol/L will be considered proper. The compounds clovin, Astraganoside, and Astragaloside have violated the range below LogP. From the absorption index in human intestinal absorption index Astraisoflavanin, Medicarpin, Calycosin, and formononetin are recorded as excellent for oral bioavailability. Caco-2 permeability of a given compound >–5.15 log cm/s is suitable for in vivo drug permeability. The compounds Medicarpin, Calycosin, and formononetin are recorded as poor with the effect on CNS that are required to cross the blood–brain barrier (BBB) to reach their molecular target. Therefore, the prediction of drug candidates showed Ononin, Astraisoflavanin, and Astraganoside have shown average BBB penetration. Other compounds have excellent BBB penetration. The compounds Ononin, Medicarpin, Calycosin, and formononetin are recorded as optimal for TPSA prediction. The other compounds have shown TPSA values above 140. The compounds Clovin, Astraganoside, and Astragaloside are rejected for the Lipinski rule. All nine compounds are predicted as non-carcinogens, and among them, Astragaloside is an excellent non-carcinogen. Among the compounds, Ononin and Astroside have excellent physicochemical, toxicity, and pharmacokinetic properties (Table 3).
ADMET Investigation of the Compound Identified from A. membranaceus.
C-T-N Analysis
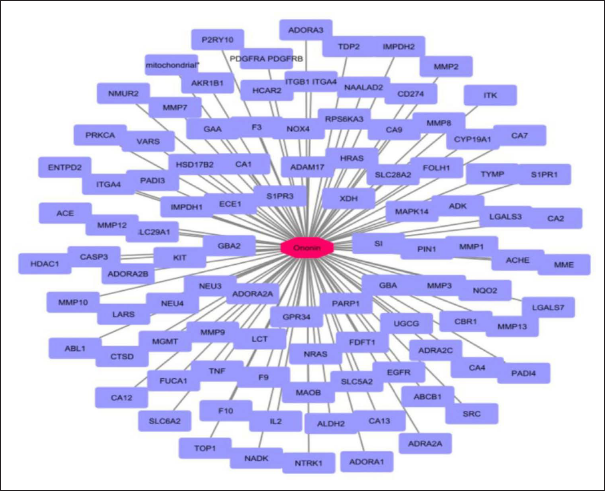
The best-hit compound Ononin was searched for its protein targets against the human target database. From the output, it was observed that there were 100 target proteins (Figure 2). The analysis of the ononin network revealed with 101 nodes, 100 edges, an average number of neighbors of 1.980, and network centralization as 1.0. Further disease enrichment analysis revealed from the 100 proteins are involved in the interaction. Totally, 88 proteins (89%) have been involved in different diseases. Among them, 12 proteins from the network was classified specifically for thyroid diseases. The ononin targeting the proteins HRAS, neurotrophic receptor tyrosine kinase 1 (NTRK1), LGALS3, KIT, SRC, ACHE, and MMP7 is involved in Thyroid cancer. The proteins NTRK1, SLC6A2, PDGFRA, EGFR, IL2, and PDFFRB are involved in hyperparathyroidism and hyperparathyroidism, secondary diseases. The proto-oncogenes such as NRAS, HRAS, and NTRK1 are involved in adenocarcinoma, thyroid neoplasms, and thyroid diseases. From the functional annotation, the biological process of the protein was reported as host-viral interaction. The cellular component details show proteins as secretory, cell membrane, and Golgi apparatus. The molecular functions of the proteins is tyrosine-kinase, kinase, and transferase activity.
Network of Ononin Showing Their Interacting Protein Partners (Purple Color).
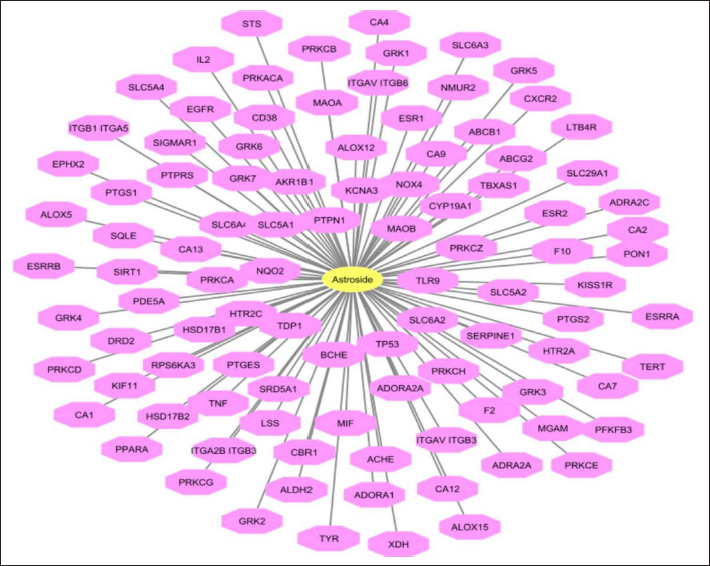
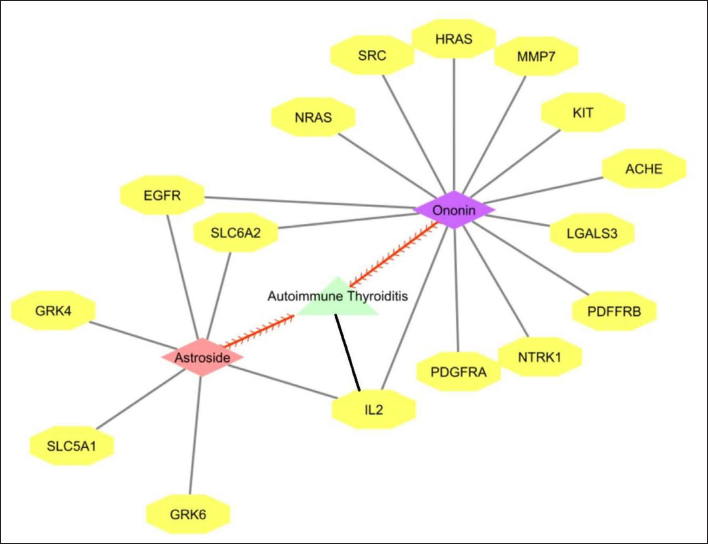
C-T-N for Astroside was observed with 100 target proteins (Figure 3). From the network analysis, using Cytoscape the number of nodes was identified as 101, edges as 100, with an average number of neighbors of 1.980, and network centralization as 1.0. Among the interacting partners, 6 proteins have been involved in hyperparathyroidism and hyperparathyroidism, secondary diseases targeted by Astroside. The functional annotation of the proteins have shown the biological process as symport. The functional role of the protein is kinase, transferase and serine-threonine protein kinase. The overall network of genes targeted by Astroside and Ononin in response to AIT is represented in Figure 4.
Network of Astroside Showing their Interacting Protein Partners (Pink Color).
Overall Network of Proteins From AIT-Compound-Target Network.
GO Analysis
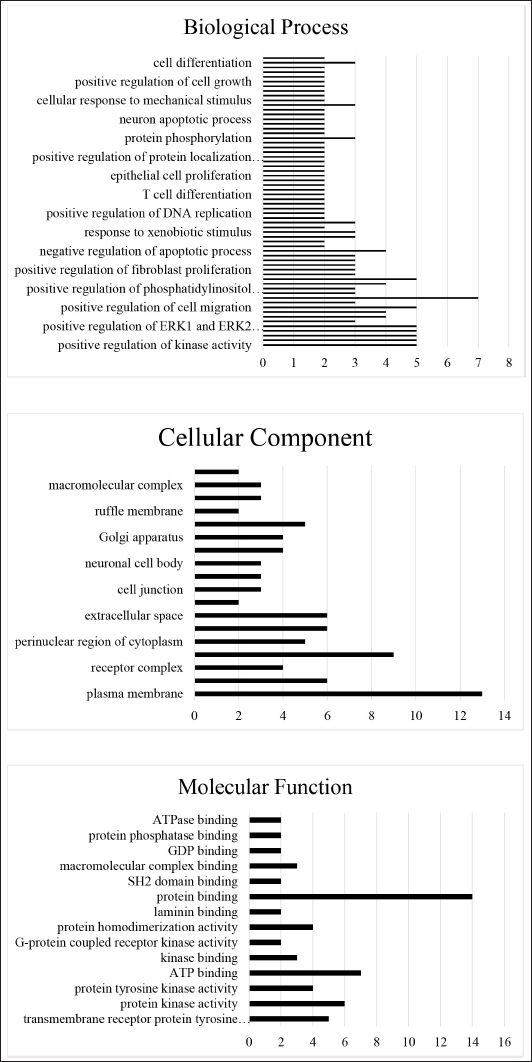
GO annotation was performed to enrich the involvement of protein in different functions such as biological process, cellular components, and molecular function (Figure 5). Ononin targeted proteins have enriched with a biological process such as “positive regulation of kinase activity,” “transmembrane receptor protein tyrosine kinase signaling pathway,” “peptidyl-tyrosine phosphorylation,” “protein auto-phosphorylation,” “positive regulation of ERK1 and ERK2 cascade,” “cell migration,” “cell proliferation,” and “signal transduction.” The enriched molecular function are “protein binding,” “complex binding,” “ATP binding,” “transmembrane protein kinase,” and “membrane activity.” The enriched cellular component revealed as “plasma membrane,” “cell surface,” “extracellular region,” and “integral component of the plasma membrane,” “Golgi membrane,” “mitochondria and Golgi apparatus.”
GO Enrichment for Biological Process, Cellular Component and Molecular Function for the Target Genes.
The GO-enriched terms for the biological process of the Astroside targeted proteins are regulation of signal transduction, positive regulations of kinase activity, cell growth, and inflammatory response. The cellular component of the proteins is the intracellular vesicle, plasma membrane, an integral component of the plasma membrane, apical plasma membrane, and membrane raft. The molecular functions include G-protein coupled receptor kinase activity, protein kinase activity, and ATP binding.
Pathway Enrichment Analysis for AIT-C-T Networks
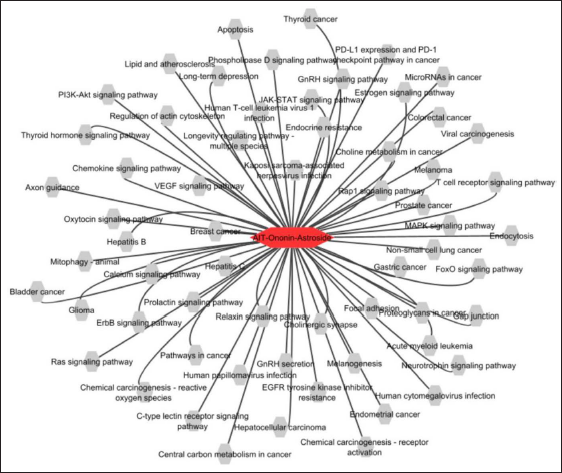
The compounds targeting the hub proteins involved in pathogenic mechanisms during the onset of AIT was revealed through KEGG pathway prediction (Figure 6A). Ononin has targeted the hub proteins involved in the regulation of the involved in the “Thyroid hormone signaling pathway (THSP).” The other pathways involved in associating with THSP are, “thyroid hormone synthesis,” “regulation of actin cytoskeleton,” “calcium signaling,” “MAPK signaling,” “PI3K-AKT,” “mTOR,” “HIF-1,” “p53,” “cardiac muscle signaling,” “WNT signaling,” “NOTCH signaling,” and “Glycolysis.” Similarly, astroside have targeted the hub proteins involved in the regulation of “Thyroid hormone signaling pathways,” “Metabolic Pathways,” “Serotonergic synapse,” “Pathways in cancer,” “PI3K-Akt signaling pathway,” “Chemokine signaling pathways,” “Focal adhesion,” “endocytosis,” “Chemical carcinogenesis - receptor activation,” and Rap1 signaling pathway.”
KEGG Pathway Enrichment Analysis of Proteins From AIT-Compound-Target Network.
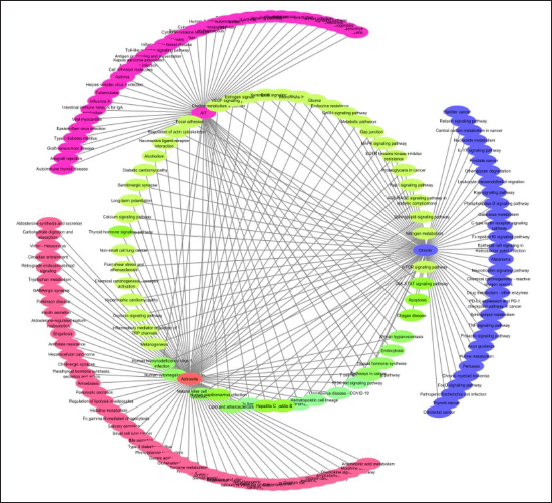
The intersection of Ononin and Astroside networks have resulted with 31 genes and the pathway enriched are “Nitrogen metabolism,” “Metabolic pathways,” “Coronavirus disease - COVID-19,” “AGE-RAGE signaling pathway in diabetic complications,” “Sphingolipid signaling pathway,” “Histidine metabolism,” “Proximal tubule bicarbonate reclamation,” “Alcoholic liver disease,” “Folate biosynthesis,” and “cGMP-PKG signaling pathway” (Figure 6B).
Discussion
In recent years, network pharmacology has promising role in the drug and pharmaceutical industry, which is the multicomponent and multitarget systematic strategies. The approach assists in the exploration of effective drugs interactions with different proteins in the human system (Muhammad et al., 2018). Additionally, the approach is useful in drug discovery to reduce the time and cost of drug improvement by understanding the toxicity, metabolic properties, efficacy of the drug, and other modulatory effects (Poornima et al., 2016). The present study deals with the identification of key ingredients from the root of A. membranaceus using network pharmacology for potential treatment against AIT disease. The bioactive ingredients were collected based on the literature survey with remarkable properties such as anti-inflammatory, anticancer, and immunostimulant (Li et al., 2014). The chemical structures were classified as Flavonols, Isoflavons, Isoflavanes, Pterocarpans, and Saponins. Later, the bioactive compounds were investigated for their pharmacokinetic properties. The properties such as molecular weight, TPSA, logS, logP, logD, Caco-2 cells, HIA, carcinogenicity, BBB penetration and Lipinki’s rule were evaluated using the ADMETlab web tool (Xiong et al., 2021). According to Drug-Like Soft rule, the molecular weight of the compounds between 100 and 600 is determined as optimal for a drug. The first step of drug candidate prediction is Lipinski Rule. The basic sets of requirements are MW ≤ 450; logP ≤ 5; Hacc ≤ 10; Hdon ≤ 5. If any of the two properties of Lipinski’s rule was violated the compounds is expected with poor absorption. TPSA according to the Veber rule between 0 and 140 is optimal for a drug molecule. The logS determined the aqueous solubility value which is determined by the drug absorption process. Once the drug is administered the dissolving property of the drug enhances its easy absorption. This property should be determined for developing a good and complete oral drug. Thus, the early prediction of this property is more important in the drug discovery process. The predicted compounds with solubility ranging from –4 to 0.5 log mol/L are considered to be a proper drug. The logP represents the permeability coefficient of the drug candidates. This property determines the impact of the drug on both membrane permeability and hydrophobic binding to macromolecules (target receptor, plasma protein, transporter and metabolic enzymes). The compounds with a logP value between 0 and 3 log mol/L will be suitable as drug molecule. The logD represents the n-octanol/water distribution coefficient. For a therapeutic drug, mechanism includes the entering of the drug into the blood circulation and reaching necessary sites of action. Additionally, the drug candidate needs to balance the lipophilic and hydrophilic properties of the blood fluid and penetration through the biomembranes. Therefore, in drug development, the prediction of candidate molecules with 1–3 log mol/L will be considered suitable.
The next important feature used alternatively for oral bioavailability is intestinal absorption. The compound with less than 30% is considered poorly absorbed. The oral drug reaches the systemic circulation, and should cross the intestinal cells and membrane via active and passive diffusion. Caco-2 cells used as an alternative for the human intestinal epithelium to determine the in vivo drug permeability. The Caco-2 cell permeability index predicted value >–5.15log cm/s are determined as an eligible candidate for a compound as a drug molecule. The important step of the drug development is drugs blocked by the CNS effect. The molecules need to cross the blood-brain barrier (BBB) to reach the molecular target. The compound with predicted values in the probability within the range of 0–1 is suitable for drug discovery. The carcinogenic effect of the compound is of great concern because the compounds can have a destructive effect on the genome and cellular metabolic process. Hence, compounds with a damaging role can cause serious effects on human health. Therefore, this carcinogenic effect exhibiting molecules called carcinogens has an impact on drug discovery. Thus, molecules within the range of 0–1 are carcinogens and 0 are valued as non-carcinogens. Through the in silico investigation, the compounds predicted as suitable and satisfying all the criteria proceeded to the next level of investigation. Here, the astragalus compound such as astroside and ononin satisfying all these criteria are investigated further using a network pharmacology-based approach.
Totally 200 genes were predicted in the C-T-N analysis of both ononin and astroside. The GO annotation for the target genes revealed that these genes mainly enriched in the plasma membrane and Golgi membrane. These genes are responsible for participating in the protein binding, ATP binding, and activation of several cell receptors for subsequent activation of inflammatory responses and signaling cascade. The pathway analysis was carried out to investigate the pathogenic mechanism involved in disease and several genes that are responsible for activation and destruction at the molecular level. From this study, AIT 48 genes are involved in the pathogenic mechanism of the disease. From the pathway study, the genes were involved in other critical pathways “cancer pathway,” “primary immune deficiency,” “T- cell receptor signaling, and thyroid hormone synthesis.” These findings have suggested the occurrence and development of AIT involve multiple targets. Several findings have also demonstrated that abnormalities in the immune environment and signaling process can result in cancer disease (Deichaite et al., 2022; Disis, 2010; Fang et al., 2022). Thus, the immune imbalance of AIT can lead to cancer progression; however, such evidence is limited. The disease analysis showed 48 genes related to AIT are linked to other diseases such as respiratory, primary immune disease, lymphatic disease, Graves’ disease, Goiter, thyroiditis, myxedema, and thyrotoxicosis suggesting the same treatment strategy for similar pathogenic mechanisms.
Further studies were performed using SwissTarget Prediction, to unravel the connection among the compound, gene expression, pathways and disease. A. membranaceus with potential anticancer, antioxidant and promising immunomodulatory effects were explored for the therapeutic potential for AIT. The two best-hit Ononin and Astroside are glycosides of simple O-substituted isoflavones, screened with potential pharmacological properties from astragalus was used for further investigation. Ononin have been reported as an anti-inflammatory agent to treat Rheumatoid arthritis. The compound has a significant role in reducing chronic inflammation (Meng et al., 2021). Totally 16 genes (HRAS, NTRK1, LGALS3, KIT, SRC, ACHE, MMP7, SLC6A2, PDGFRA, EGFR, IL2, PDFFRB, NRAS, GRK4, GRK6, and SLC5A1) from the C-T network of ononin and astroside were enriched. These genes have significant role in thyroid-related pathways also in other signaling and cancer pathways. Interestingly, there are several infection-related pathways are involved with AIT such as Human cytomegalovirus infection (HCV), Virion – Adenovirus, Human T-cell leukemia virus 1 infection, Human papillomavirus (HPV) infection, and Herpes simplex virus 1 infection. Recently, AIT have gain more attention with the possible involvement of the virus and it development of the disease. The investigation with eight autopsy samples have shown possible involvement of viruses (EBV, CMV, and HHV-6) in the development of autoimmune diseases (Mori & Yoshida, 2010). From the C-T network of the ononin and astroside have confirmed the compound also target genes involved in several infection-mediated pathways such as Coronavirus disease—COVID-19, HPV, HCV, Shigellosis, and Yersinia infection. Therefore, we hypothesize that bioactive (ononin and astroside) from astragalus as promising candidate that can significantly inhibit the viral infection that are involved in the progression of AIT as well as other immune-related pathway involved in AIT.
Conclusion
Recently, increase in AIT disease is of great concern and alarming worldwide. The incidence of AIT is reported with other diseases and the severity of AIT disease is still unclear. Therefore, identification of the best therapeutic strategy to overcome the AIT can complement during the treatment for other diseases. Hence, the present investigation is multicomponent and multitargeted, resulting in the involvement of several pathways that are associated with the AIT disease. Additionally, it was observed AIT progression is also associated with different viral pathway specifically human herpes virus. From the network-based approach the best hit identified from the root of A. membranaceus, such as ononin and astroside are targeting the pathways genes involved in different viral infection are been clearly elucidated. Apart, AIT-associated pathways are also been targeted by these compounds. Thus, from the study the pathogenies of AIT was not only deciphered, also the multiple drug targets of AIT were also identified. The compounds, immunomodulatory, antiviral, and anticancer mechanism was known suggesting the multi-target, multi-pathway approach. Therefore, the study revealed two compounds ononin and astroside as promising candidates for the treatment of AIT disease and their synergistic effect during the treatment of AIT-associated disease.
Footnotes
Author Contributions
All authors took part in the creation of this manuscript, whether it is in writing, conceptualization, or literature survey.
Abbreviations
AIT: autoimmune thyroiditis; KEGG: kyoto encyclopedia of genes and genomes pathway; GO: gene ontology; ADMET: absorption, distribution, metabolism, excretion, and toxicity; TPSA: topological polar surface area; Caco-2: colon adenocarcinoma cell lines; THSP: thyroid hormone signaling pathway; BBB: blood-brain barrier; HPV: human papillomavirus; HCV: human cytomegalovirus infection.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This work was funded by the National Science Foundation China (Project No. 81874441).
