Abstract
Objectives
The aim of this study was to investigate the effect of Panicum miliaceum involved in the repair of femur bone fracture in rats.
Materials and Methods
In this study, 40 Wistar rats in five groups were studied as healthy, rats with lesion induced by surgery in femur (FX), FX rats treated with P. miliaceum extract (FX +100 and 200 P. miliaceum) and FX rats treated with Osteocare syrup (FX + OC). After 40 days, the serum levels of blood calcium (Ca), phosphorus (P), and alkaline phosphatase (ALP) along with activity of glutathione peroxidase (GPx), superoxide dismutase (SOD), and catalase (CAT) enzymes, and nitric oxide (NO) level were measured. The serum level of cytokines (IL-6, C-reactive protein [CRP], IL-10, IL-1β, and tumor necrosis factor-α [TNF-α]) was also measured by ELISA. The expression of OPG, RANK, RANKL, and BMP-2 genes and proteins were evaluated by real-time PCR and Western blotting techniques, respectively.
Results
The results showed that P. miliaceum was able to decrease the activity of ALP enzyme in addition to increasing serum Ca and P levels and GPx, SOD, and CAT activity. P. miliaceum reduces serum IL-6, IL-1β, and TNF-α, and NO levels. Also, the extract of this plant could increase the expression of OPG/RANK/BMP-2 pathway genes and proteins.
Conclusion
P. miliaceum can be used as a prodrug in traditional medicine to repair bone fractures and osteoporosis.
Introduction
One of the main challenges of surgeons, orthopedists and researchers is the reconstruction and repair of bone injuries caused by trauma, infection, inflammation and diseases related to bone defects. Time, volume of damage, inflammatory factors, level of essential microelements and stimulation of damaged cells in the area are effective in bone repair and regeneration processes (Johnson & García, 2015). Osteoblast/osteoclast ratio and their activity and interaction with each other and other cells in the area are important in bone repair. Various factors are involved in the level of activity of osteoblast cells and the proliferation and secretion of matrix vesicles containing collagen organic matrix and calcium and phosphorus minerals (Plotkin & Bruzzaniti, 2019).
OPG and RANKL are synthesized by osteoblast cells and are effective in regulating the activity of osteoclast cells and their differentiation from osteoclast precursors. In addition, RANK-L, like macrophage-colony stimulating factor (M-CSF), is effective in the maturation of osteoclasts (Udagawa et al., 2021). Meanwhile, OPG, along with stimulating osteogenesis, inhibits RANK-L and ultimately prevents the activity and differentiation of osteoclasts. OPG increases the stimulation of apoptosis and inhibits the activation of mature osteoclasts. BMP-2 is a member of the bone morphogenetic proteins (BMPs) family, which inhibits RANK and increases OPG. BMP-2 stimulates osteoprogenitor cells to become osteoblasts through the ERK1/2 pathway and inhibits the differentiation of osteoclasts (Lin et al., 2021).
Panicum miliaceum (P. miliaceum) L. belongs to the Poaceae family, which is cultivated in the countries of East and Southeast Asia (China, India, and Nepal), Eastern Europe (Russia, Belarus, and Ukraine), as well as the United States, and is used in human food and animal feed industries (Wang et al., 2016). P. miliaceum is a short-day plant and its height reaches 30−100 cm. Liquid chromatographic-tandem mass spectrometric (LC-MS/MS) analysis on P. miliaceum extract showed the presence of various polyphenolic compounds such as isoflavonoids, formononetin, daidzein, biochanin A, luteolin and genistein and flavonoids quercetin, apigenin, R-coumaric acid, caffeic acid, syringic acid, gallic acid, carotenoids, chlorogenic acid, and kaempferol along with oleic and linolenic fatty acids (Habiyaremye et al., 2017). Due to the presence of mineral elements and polyphenolic compounds in P. miliaceum, in the current study, the extract of this plant was used to repair bone fractures in rats using biochemical, antioxidant, and anti-inflammatory and gene/protein expression of the OPG/RANK/BMP-2 pathway.
Materials and Methods
Collection and Extraction of P. miliaceum
P. miliaceum fresh seeds were cultivated for 4 months, new seeds were collected, cleaned, and dried. Then, 3000 g of dry seeds of this plant were powdered by Humboldt soil grinder (Humboldt company, US, catalog number: H-4199.5F) and added to 70% alcohol (volume ratio of water/ethanol: 70/30). After 72 h of incubation (temperature of 25°C), the resulting mixture was passed through a paper filter (Whatman filter paper, Millipore, number 42, US, catalog number: 1442-125) and concentrated under the pressure of a BUCHI rotary evaporator (Rotavapor, Buchi company, Switzerland; model: 9230). The final extract (300 g: 10% extraction efficiency) was dried for 12 h at 40°C and then stored at −4°C until use (Bazm et al., 2020).
Liquid Chromatography-Electrospray Ionization-Mass Spectrometry Analysis
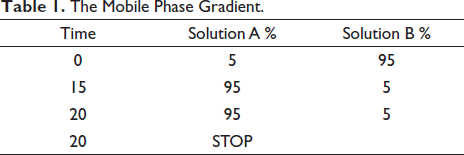
To analyze compounds with high molecular weight in P. miliaceum, we used liquid chromatography-electrospray ionization-mass spectrometry (LC-ESI-MS) with an Agilent chromatographic system (Agilent Technologies, Model No. 1290 series, USA) coupled to a HCT ultra ion trap MS detector (Shimadzu, Model No. 2010 A, Japan). In brief, 5 mg of P. miliaceum hydroalcoholic extract powder was dissolved in solvents A and B, respectively, containing acetonitrile 0.1% and formic acid 0.1%. The mobile phase gradient was run in the chromatographic system according to the following program (Table 1): total run time of 20 min, block temperature of 285°C, column temperature of 25°C, auxiliary gas of GS2 99% with 1.2 L/min, curtain gas of 25 psi, capillary voltage of 3.5 kV, and probe voltage of 3.5 kV (Bazm et al., 2020).
The Mobile Phase Gradient.
Scanning Electron Microscopy/Energy Dispersive X-ray Spectroscopy Analysis
Scanning electron microscopy/energy dispersive x-ray spectroscopy (SEM/EDS) was used to evaluate the mineral elements of P. miliaceum extract. Briefly, after coating 5 mg of hydroalcoholic extract of P. miliaceum with gold particles, Seron SEM microscope (Seron Technology’s, Model No. AIS2300C, Korea) was used with the following analysis conditions to identify mineral elements: X, Y beam rotation (200 µM), vacuum pressure (10−5 Torr), accelerating voltage (14 kV), and image rotation degree (360°) (Bazm et al., 2020).
Animal Care, Grouping, Treatment, Experimental Design, and Research Time Line
Animal Care
In this study, 40 Wistar rats were selected in five groups (n = 8/group) weighing 200 ± 20 g. The rats were kept under standard conditions for laboratory animals in propylene cages, at a temperature of 24 ± 4°C, a 12/12 night/day cycle, and a relative humidity of 45 ± 5%. The rats were fed using special pellets and drinking water connected to the city water system, and the rats had free access to water and food. The ethics committee of the Linyi Hedong Central Hospital, Linyi, Shandong, China supervised the maintenance, behavior, and sacrifice under national and international standard protocols according to NIH Publication, 1996.
Rat Fracture Model
After induction of pre-anesthesia/anesthesia with intraperitoneal injection of ketamine 10% (20 mg/kg)/xylazine 2% (90 mg/kg) and shaving and cleaning of the right thigh region, a closed fracture was created with special tweezers in the proximal femur region, 1 cm below the femoral head (Florence et al., 2017).
Animal Grouping, Experimental Design, and Research Time Line
After inducing fractures in the femurs of rats, five groups were studied as follows for 40 days:
Healthy group: normal rats without any lesion received deionized water (DW) (1 mL/40 days) by gavage. Rat with fractured femur without treatment (FX): FX rats received DW (1 mL/40 days) by gavage. FX rats treated with 100 or 200 mg/kg P. miliaceum extract (FX + 100, 200 P. miliaceum): FX rats received 100 or 200 mg/kg P. miliaceum extract dissolved in DW (1 mL) by gavage. FX rats treated with 1 mL/day Osteocare syrup (FX + OC): FX rats received OC dissolved in DW (1 mL) by gavage.
Rats received P. miliaceum extract and OC every day at a specific time (9:00 AM) and previous studies, pilot study, and calculation of LD50 were used to select the best therapeutic dose of extract and OC (Bornarodi et al., 2017; Nagareddy & Lakshmana, 2006).
Acute Toxicity Test (LD50)
LD50 was calculated with the help of Lork’s two-step method and was used to select the appropriate dose. In this method, first 10 rats in five groups received doses of 50, 100, 500, 1000, and 3000 mg/kg of P. miliaceum extract and after 24 h monitoring, any toxic symptoms (diarrhea, anorexia, vomiting, lethargy, and appetite) and possible death were recorded. However, four other rats in four groups received doses of 50, 100, 1000, and 5000 mg/kg of P. miliaceum extract, and after 24 h monitoring, any toxic symptoms and possible death were recorded. Then, the highest safe dose (L1) and the lowest lethal dose (L2) during monitoring in the groups were placed in the following formula.
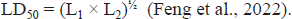
Serum Calcium (Ca) and Phosphorus (P) Levels and Alkaline Phosphatase Activity
On the 41st day of the study and after sacrificing the rats with the help of intraperitoneal injection of 10% ketamine (20 mg/kg) and 2% xylazine (90 mg/kg), blood was taken from the heart and serum was separated (centrifuge at 10,000g for 20 min). Serum levels of Ca (catalog number: ab102505) and P (catalog number: ab234579), and alkaline phosphatase (ALP) activity (catalog number: ab83369) were measured with the help of Abcam ELISA (enzyme-linked immunosorbent assay) (Abcam company, US) kit and the results were reported as mg/dl (Shishkova et al., 2019).
Serum Levels of Tumor Necrosis Factor-α (TNF-α), C-Reactive Protein (CRP), Interleukin-1β (IL-1β), IL-10, and IL-6
Serum levels of pro-inflammatory cytokines [TNF-α (catalog number: ab236712), IL-1β (catalog number: 255730), IL-10 (catalog number: ab214566), CRP (catalog number: ab256398), and IL-6 (catalog number: ab100772)] and were measured using commercial Abcam ELISA kits (Abcam company, US) according to the manufacturer’s recommended protocols. In short, after preparing the standard solution and diluting the primary antibody, 50 µL of serum samples and 50 µL of the standard solution were poured into 96 well plates, and then the diluted primary antibody was added to each well. After incubating the resulting mixture at 25°C (for 1 h), 100 µL of tetramethylbenzidine and 100 µL of tetramethylbenzidine streptavidin were added to each well. Finally, 100 µL of bovine serum albumin was added to the final mixture and incubated for 45 min at 37°C. The absorbance of the final mixture was read at a wavelength of 450 nm with a Milton Roy spectrophotometer (Milton Roy Company, Spain; model name: Spectronic 20) and reported as pg/dl (Chen et al., 2019).
Serum Nitric Oxide (NO) Levels and Glutathione Peroxidase (GPx), Superoxide Dismutase (SOD), and Catalase (CAT) Activity
Nitric oxide is one of the most important serum factors of oxidative stress, which occurs in various pathological conditions in addition to its physiological roles (neuronal communication, immune system modulation, and vasodilation). In the present study, Griess method was used to measure serum NO levels. Briefly, zinc oxide (ZnSO₄) (6 g) was added to the serum samples (300 µL) and after incubation (37°C for 10 min), it was centrifuged at 10,000g for 10 min. The supernatant of the resulting mixture was collected and stored in a separate tube. Then, 50 µL of Griess solution [a mixture of N-(1-naphthyl) ethylenediamine dihydrochloride 5 mM (50 µL), vanadium chloride 40 mM (40 µL) and sulfanilamide 20 mM (50 µL)] was added to the supernatant (50 µL) and incubated (1 h at 37°C). Finally, the absorbance of the resulting mixture at 540 and 630 nm wavelengths was read using a spectrophotometer with a Milton Roy spectrophotometer (Milton Roy Company, Spain; model name: Spectronic 20) and reported as µM/mL (Naseri et al., 2019).
To measure GPx (catalog number: E-EL-R2491), SOD (catalog number: E-EL-R1424), and CAT (catalog number: E-BC-K031-S) enzymes activity, the Elabscience commercial kit (Elabscience biotechnology company, Ltd, US) was used based on the recommendations of the kit protocol (Ustunova et al., 2020).
Femur Lipid Peroxidation Levels (TBARS) and Ferric Reducing Antioxidant Power Levels
After dissection of the rats, the left femur bone was separated from the surrounding skin, muscle, and fat tissues and kept in a nitrogen tank. To measure total antioxidant capacity of femur tissue samples, ferric reducing antioxidant power (FRAP) measurement and modified Benzie-Strain calorimetric method were used. Briefly, first the FRAP solution was prepared using hydrochloric acid (40 mM, 1 mL), 2, 4, 6-tripyridyl-riazine (TPTZ) (10 mM, 5 mL), ferric chloride (20 mM, 5 mL), and acetate buffer (0.1 M, 50 mL). Then, 200 µL of homogenized femur tissue was added to 10 µL of FRAP solution and 5 mL of ferric chloride (20 mM) and after incubation at 25°C for 15 min and observing the yellow colored-ferric chloride-TPTZ complex color change to blue colored-ferrous chloride-TPTZ complex, the absorbance of the resulting mixture was read at a wavelength of 593 nm with a Milton Roy spectrophotometer (Milton Roy Company, Spain; model name: Spectronic 20) and reported µmol/mg protein (Choi et al., 2019).
To measure the lipid peroxidation of femur tissue as an important index of oxidative stress, TBARS level measurement method was used. In this calorimetric method, first, 100 µL of homogenous femur tissue is added to 2 µL of butylated hydroxytoluene (BHT) 0.19 M, 50 µL of thiobarbituric acid (TBA) 0.02 M, 50 µL of phosphoric acid 20 mM, and to the resulting mixture after incubation (temperature of 37°C for 30 min) was centrifuged (10,000g for 5 min). The absorbance of the resulting mixture was read at a wavelength of 532 nm with a Milton Roy spectrophotometer (Milton Roy Company, Spain; model name: Spectronic 20) and reported nmol/mg protein (Ajiboye & Shodehinde, 2022).
BMP-2, OPG, RANK and RANKL Genes Relative Expression
Femur Tissue Total RNA Extraction
Real-time PCR technique was used to evaluate the expression level of genes involved in the proliferation and differentiation of femoral osteoblasts. To extract total RNA of femur tissue, a Norgen Biotek commercial kit was used according to the kit’s recommended protocols (Norgen Biotek company, US; catalog number: 25700). Briefly, first, 50 mg of femur homogenized tissue was added to RNA extraction mixture containing RL buffer (300 µL), proteinase K (20 µL), and β-mercaptoethanol (600 µL) and incubated for 15 min at −4°C. After centrifuging the resulting mixture at 12,000g for 15 min, it was added to the separation column and washed twice with 99.6% ethanol (500 µL). Finally, 50 µL of DW water was added to the formed RNA pellet, and 10 µL of it was used to evaluate the purity and quality of RNA extracted by nanodrop (BioNordika company, US, model: ND-OneC-W) and 2% agarose gel electrophoresis, and the rest was kept at −20°C (Vechetti et al., 2021).
cDNA Synthesis and Quantitative Real-Time PCR (qRT-PCR)
Takara kit (Takara Bio, Japan, catalog number: 6110A) was used to synthesize cDNA from RNA extracted from femur tissue according to the company’s recommended protocol. Briefly, the synthesis mixture containing 1 µL of oligo and random primers was added to 1 µg of total RNA, 10 µL of Takara Bio Mastermix, and 8 µL of DW. The synthesis temperature cycle included the resulting mixture including incubation for 10 min at 25°C, 45 min at 60°C, and 5 min at 70°C (Wan et al., 2020). Primers were designed using gene runner software and blasted using National Center for Biotechnology Information (NCBI) database (
The forward and reverse sequences of BMP-2, OPG, RANK, and RANKL primers are listed in Table 2, where β-actin is a housekeeping gene. However, 1 µL reverse and forward primers, 8 µL Thermo Fisher Master Mix (Thermo Fisher Scientific, US, catalog number: 4368577), 1000 ng cDNA were replicated with Applied Biosystems thermocycler (Applied Biosystems, Quantstudio 1, US) for 38 cycles as follows: 5 min at 50°C and 5 min in 95°C as holding stage, 15 sec at 95°C as denaturation stage, 1 min at 60°C as annealing stage, and 5 min at 70°C temperature as the extension stage. The melting curve stage was obtained at a rate of 1°C per sec from 50°C to 95°C. Finally, the threshold cycle (CT) of the sample genes (S) was recorded and according to the CT of β-actin gene and the healthy group, the fold change was calculated according to the following formula.
Primer Sequences.

Fold change of genes=2 −∆∆CT
∆∆CT = [(CT S − Ct β-actin) − (CT S –CT healthy)] (Wan et al., 2020).
BMP-2, OPG, RANK, and RANKL Protein Expression
Western blotting assay was used to evaluate BMP-2, OPG, RANK, and RANKL protein expression. For this purpose, first, 50 µL of homogenous tissue was incubated with 400 µL of lysate buffer containing proteinase inhibitors for 15 min at 4°C and then centrifuged (12,000g for 15 min). Then, 50 µg of the resulting mixture was transferred to polyvinylidene fluoride membrane and 10% SDS-polyacrylamide gel probed with monoclonal anti-BMP-2 (catalog number: E-AB-70352), anti-OPG (catalog number: E-EL-H1341), anti-RANK (catalog number: E-AB-60731), and RANKL (catalog number: E-AB-60724) (Elabscience biotechnology company, Ltd, US) and incubated for 1 h at 4°C. After washing twice with PBS and Tween-20 solution, HRP-labeled antibody was added and incubated for 60 min at room temperature. Further, 30 µL of Millipore solution (Millipore, UK, catalog number: WBKLS0100) was added as enhancer reagents with chemiluminescence detection system (e-BLOT company, China) and with the help of ImageJ software, the expression ratio of proteins to β-actin was calculated and checked (Jia et al., 2022).
Statistical Analysis
SPSS version 16 software (SPSS, IBM Corp, Chicago, US) was used to analyze the results, and GraphPad Prism software version 8 (GraphPad Prism, San Diego, US) was used to design the graphs. Kolmogorov-Smirnov test (p > 0.05) was used to evaluate the normality of the data, as well as one-way analysis of variance (ANOVA) along with Tukey’s post hoc descriptive test to compare the differences of means between groups. Finally, the results were reported as “mean ± standard deviation (SD)”, and p < 0.05 was considered statistically significant.
Results
Acute Toxicity Test (LD50)
After 24 h monitoring of groups and evaluation of toxicity and possible death in rats, it was found that the highest healthy dose was 3000 mg/kg and the lowest death dose was 1000 mg/kg in the studied groups. According to Lorek’s formula, the LD50 of P. miliaceum extract was recorded as 1.732 g/kg. Therefore, doses lower than 1732 mg/kg of the extract of this plant can be used in animal studies.
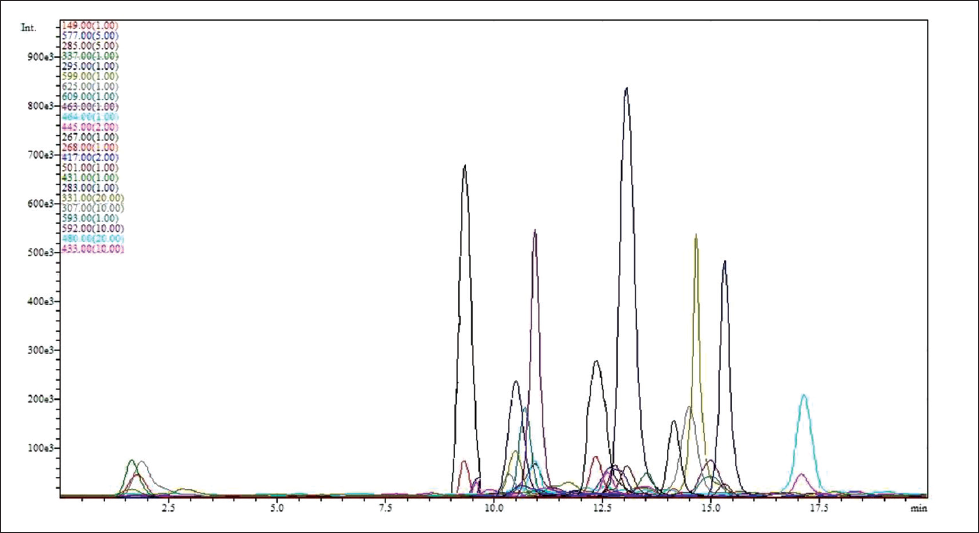
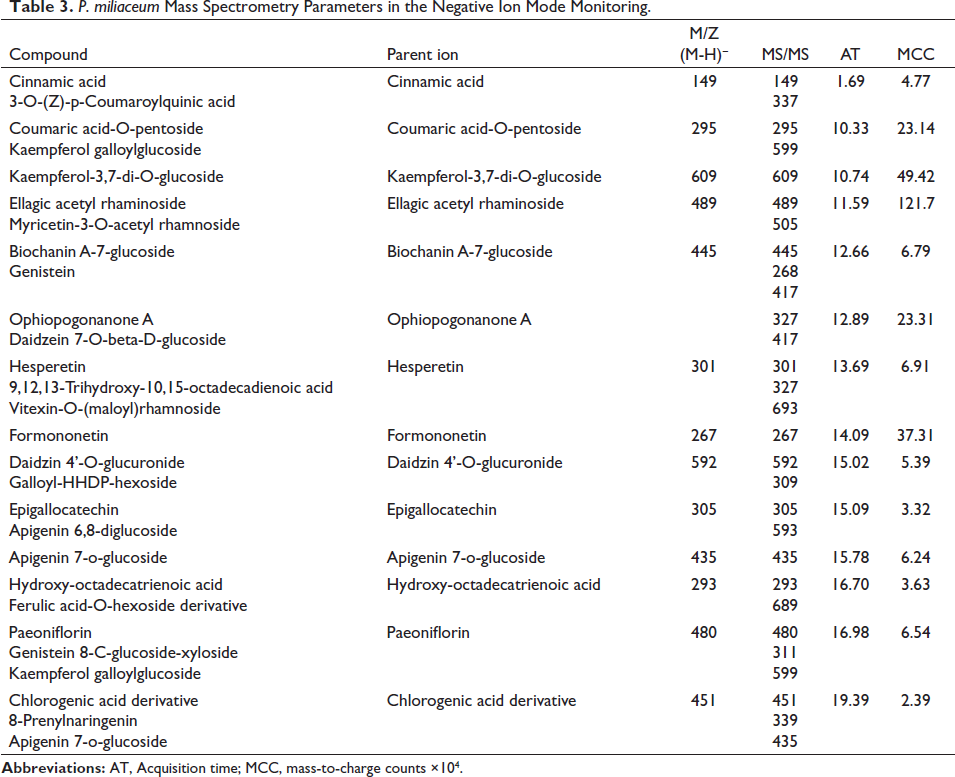
LC-ESI-MS Evaluation
After evaluating the MS graphs, with the help of the database
Representative Chromatogram of P. miliaceum Hydroalcoholic Extract.
P. miliaceum Mass Spectrometry Parameters in the Negative Ion Mode Monitoring.
SEM-EDS Evaluation
The analysis of mineral elements of P. miliaceum extract was detected with the help of SEM-EDS, the presence of 14 mineral elements with detectable intensity (>0.1 c/s) and concentration (>0.001 weight %). The results of this evaluation showed that after carbon (C) (599.28 c/s and 57.50 ± 3.11 weight %) and oxygen (O) (173.14 c/s and 39.05 ± 3.91 weight %), Ca (3.15 c/s and 0.167 ± 0.042 weight %) is found to be the most mineral element in this plant (Figure 2 and Table 4).
Mineral Elements in P. miliaceum Based on Concentration (wt. %) and Intensity (c/s) by Using an SEM-EDS.
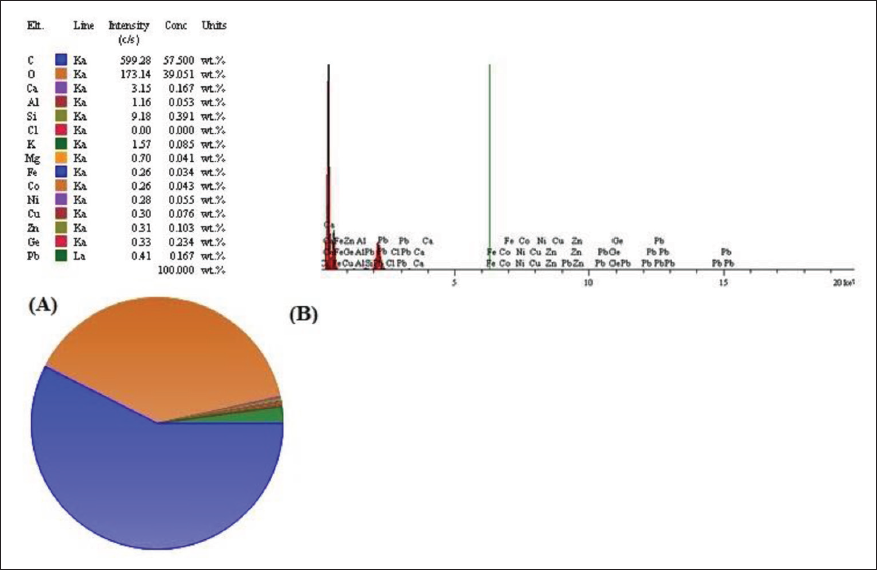
Mineral Elements Present in P. miliaceum Based on Intensity (c/s) and Concentration (wt. %).
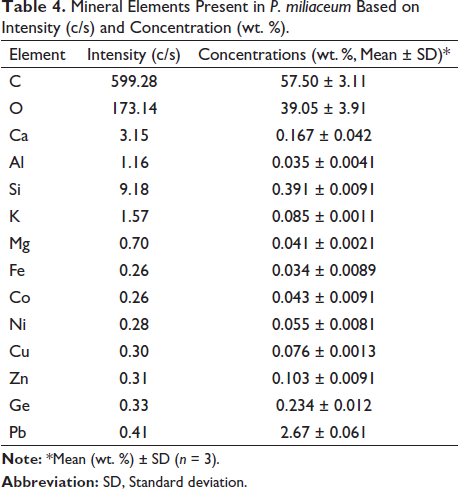
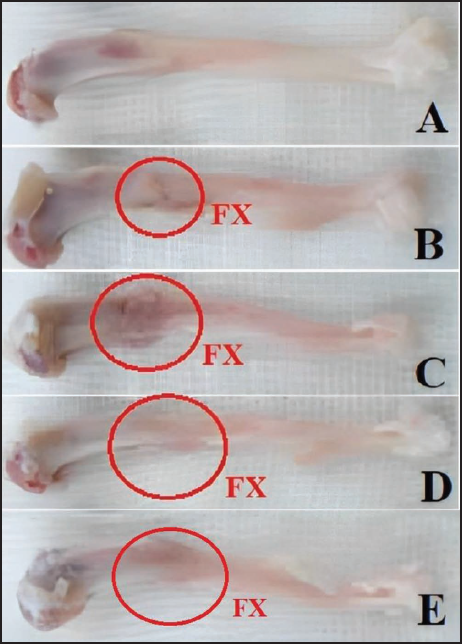
Rat’s Body and Femur Weights
The body weight and femur of rats decreased significantly after femur fracture compared to the healthy group (p < 0.05), while in the groups receiving P. miliaceum extract, both weight parameters increased in a dose-dependent manner compared to the FX group, and this increase was significant in the group receiving 200 mg/kg of P. miliaceum extract (p < 0.05). Also, the body weight and femur of rats in the group receiving OC showed a significant increase compared to the FX group (p < 0.05) (Figure 3).
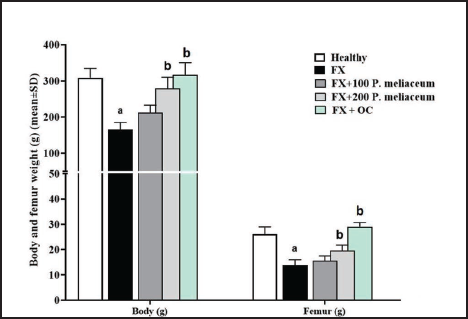
Serum Ca and P Levels, and ALP Activity
The analysis of serum markers of osteogenesis pathways showed that following the fracture, serum Ca and P showed a significant decrease compared to the healthy group (p < 0.05), while the serum levels of these two minerals in the groups receiving P. miliaceum extract compared to the FX group were increased in a dose-dependent manner, and this increase was significant in the group receiving 200 mg/kg of P. miliaceum extract (p < 0.05). Also, after measuring the activity of the ALP enzyme, it was found that the activity of this enzyme in the FX group has increased significantly compared to the healthy group (p < 0.05), while the activity of this enzyme in the groups receiving P. miliaceum extract has significantly decreased compared to the FX group (p < 0.05). After evaluating the results of the OC group, it is clear that the serum level of Ca and P in this group increased significantly and the activity of ALP enzyme decreased significantly compared to the FX group (p < 0.05) (Figure 4).
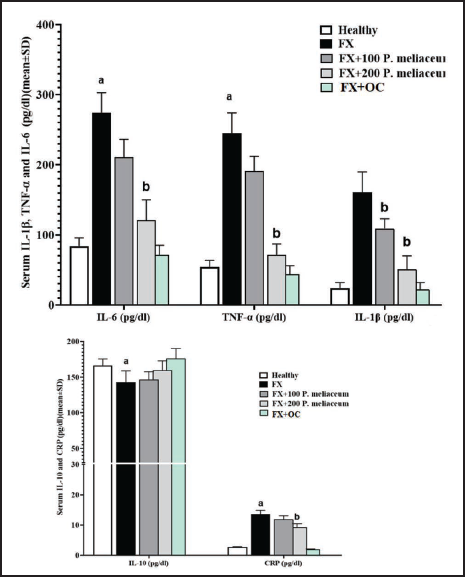
Serum Pro-inflammatory (IL-6, CRP, IL-1β, and TNF-α) and Anti-inflammatory (IL-10) Cytokine Levels
The levels of all four pro-inflammatory (IL-6, CRP, IL-1β, and TNF-α) cytokines effective in the process of proliferation and differentiation of osteogenic cells in the FX group were significantly increased compared to the healthy group (p < 0.05). Also, the level of anti-inflammatory cytokine (IL-10) in the FX group was significantly reduced compared to the healthy group (p < 0.05). While the levels of all four pro-inflammatory cytokines decreased in a dose-dependent manner in the groups receiving P. miliaceum extract compared to the FX group, this decrease was significant in the 200 mg/kg group (p < 0.05). Also, OC was able to significantly reduce the serum levels of all four pro-inflammatory cytokines compared to the FX group (p < 0.05). While the serum level of IL-10 in the treatment groups of P. miliaceum extract did not change significantly compared to the FX group (p > 0.05) (Figure 5).
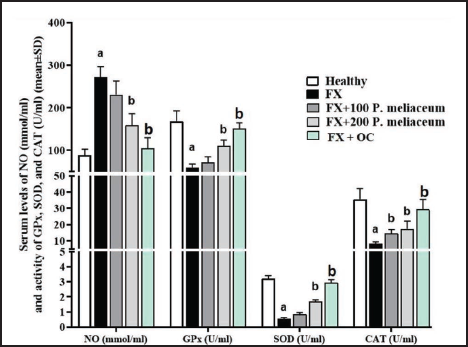
Serum and Femur Tissue Antioxidant Parameters
Serum NO Levels, Beside SOD, CAT, and GPx Activity
After a fracture in the femur, the serum level of NO increased compared to the healthy group, and the activity of antioxidant enzymes also showed a decrease (p < 0.05). This was despite the fact that P. miliaceum extract caused a decrease in NO and an increase in the level of all three antioxidant enzymes in a dose-dependent manner compared to the FX group, and these changes were significant at the dose of 200 mg/kg (p < 0.05). OC was also able to significantly increase the activity of all three antioxidant enzymes (p < 0.05) in addition to the significant reduction of NO levels compared to the FX group (p < 0.05) (Figure 6).
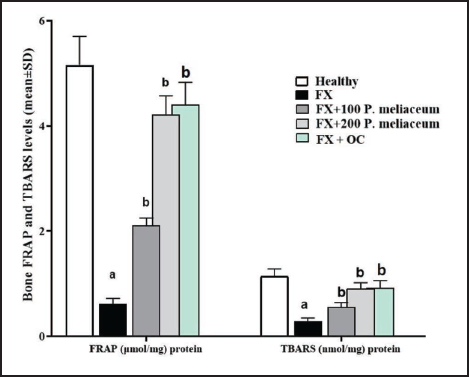
Femur Tissue TBARS and FRAP Levels
In the FX group, FRAP, and TBARS showed a significant decrease compared to the healthy group (p < 0.05). This was despite the fact that both doses of 100 and 200 mg/kg of P. miliaceum extract significantly increased both tissue antioxidant parameters (FRAP and TBARS) compared to the FX group (p < 0.05). FRAP and TBARS in the OC group also increased significantly compared to the FX group (p < 0.05) (Figure 7).
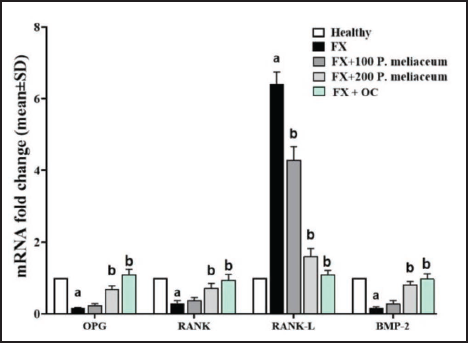
Femur Tissue OPG, RANK, RANK-L, and BMP-2 Genes Expression
The results of this study showed that the OPG/RANK/BMP-2 pathway that stimulates osteogenesis after fracture is inhibited, so that the expression of genes that stimulate this pathway showed a significant decrease compared to the healthy group (p < 0.05). This was despite the fact that P. miliaceum extract increased the expression of OPG, RANK, and BMP-2 and decreased the expression of the RANK-L osteogenesis inhibitor gene in a dose-dependent manner due to the presence of OPG/RANK/BMP-2 pathway stimulating compounds compared with FX group. The dose of 200 mg/kg extract of this plant significantly increased all three stimulatory genes (OPG, RANK, and BMP-2) and decreased RANK-L gene expression compared to the FX group (p < 0.05), while the dose of 100 mg/kg extract of this plant only caused a significant decrease in RANK-L gene expression compared to FX group (p < 0.05). OC was also able to significantly increase the expression of all three osteogenesis stimulating genes (OPG, RANK, and BMP-2) (p < 0.05) and significantly decrease the RANK-L gene expression compared to the FX group (p < 0.05) (Figure 8).
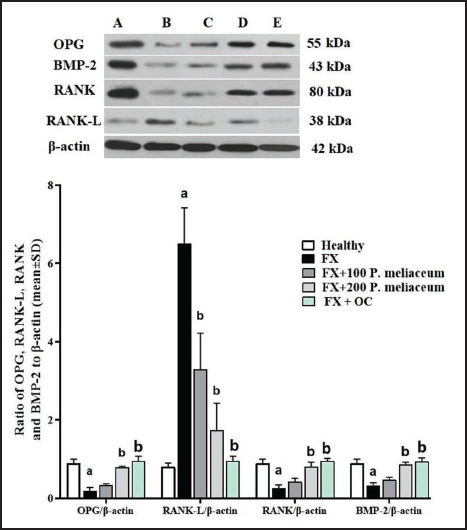
Femur OPG, RANK, RANK-L, and BMP-2 Protein Expression
After evaluating the expression of the proteins of the OPG/RANK/BMP-2 pathway, it was found that following the fracture in the femur, the expression of the proteins of this pathway (OPG, RANK, and BMP-2) decreased significantly compared to the healthy group (p < 0.05), and the expression of the inhibitory protein of this pathway (RANK-L) also increased significantly (p < 0.05). P. miliaceum extract at a dose of 100 mg/kg was able to significantly decrease RANK-L protein expression compared to FX group (p < 0.05). This was despite the fact that the dose of 200 mg/kg extract of this plant significantly increased the expression of all three proteins OPG, RANK, and BMP-2 compared to the FX group and significantly decreased the expression of RANK-L protein (p < 0.05). OC was also able to significantly increase the expression of all three osteogenesis stimulating proteins (OPG, RANK, and BMP-2) (p < 0.05) and significantly decrease the RANK-L protein expression compared to the FX group (p < 0.05) (Figure 9).
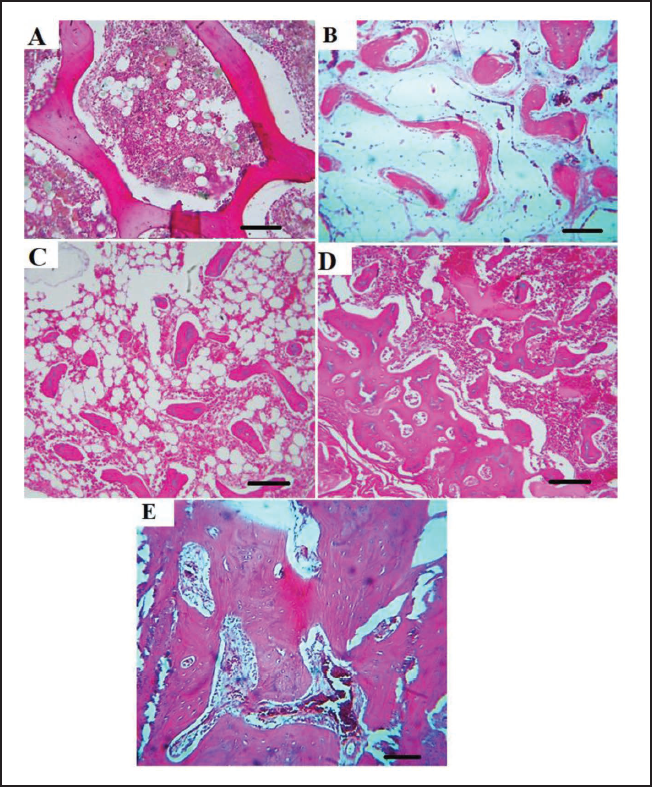
Histopathological Findings of the Femur Tissue
After the dissection of the rats and the separation of their femurs, it was also found that there was a fracture seam in the FX group without supportive treatment and acute inflammation was present in the area along with edema. In the FX group, no signs of periosteum and endosteum could be seen due to the acute inflammation, and the fracture seam had a small number of scattered repair fibers. While in the groups that received P. miliaceum extract, there was no evidence of inflammation and edema in the mineralized fibrous tissue repair at the fracture site, especially at the dose of 200 mg/kg. In the groups treated with the extract, callus formation occurred in the drill hole, and normal periosteum and endosteum arrangement with the presence of osteoblasts were seen linearly. The fracture seam in the OC group was also visible with fibrous connective tissue around, mineral fibrous tissue in the center and the arrangement of endosteum and periosteum around callus formation (Figure 10). The synthesis and secretion of bone matrix (BM) increased in the P. miliaceum treatment groups (especially in the dose of 200 mg/kg) so that the complete BM containing osteocytes and osteon formations (OF) could also be seen. While in the fracture group, very little secreted bone matrix and no OF were observed (Figure 11).
Discussion
In the current study, the healing effects of P. miliaceum extract on the fracture created in the rat femur were evaluated with biochemical, antioxidant, anti-inflammatory, and molecular findings. The results of the study showed that the extract of this plant has the same effects as Osteocare syrup; in addition to supplying minerals needed for osteogenesis, it stimulates osteoblasts and inhibits osteoclasts (increasing the expression ratio of OPG/RANK-L) through different routes.
After a fracture, the need for minerals, including Ca, is necessary to supply the mineral part of the bone, which mainly decreases during the fracture. In addition, osteoclastogenic cytokines such as IL-6, TNF-α, and IL-1β from damaged tissues, osteoclasts and other macrophages in the area stimulate pathways related to inhibiting the proliferation, differentiation, and activation of osteoblasts and also increase the activity of osteoclasts (Yang et al., 2022). These pro-inflammatory cytokines play a role in increasing the rate of RANK-L/OPG, which induces the signaling pathways related to the maturation and activation of osteoclasts. Studies show that IL-6 and TNF-α in interaction with RANKL increases MCSF in the bone marrow microenvironment for the maturation and activation of osteoclasts. In addition, TNF-α and IL-1β through increasing the expression of extracellular signal-regulated kinases (ERK1/2), c-Jun-N terminal kinase (JNK) and transcription factors p38 and mitogen-activated protein kinase (MAPK) increases osteoclastogenesis and their activation. Fracture and inflammation of the site also increased pro-inflammatory cytokines (TNF-α, IL-6, and IL-1β) in this study (Fan et al., 2018).
Reactive oxygen species (ROS) are unstable and active species (such as H2O2, •O2−, and •OH) that are created after the occurrence of fracture and subsequent inflammation, and in reaction with biological macromolecules of the cell, including lipid membrane, DNA, ribosomes, and mitochondria, induce apoptotic pathways by increasing the ratio of Bax/Bcl-2 and p53 are expressed in osteoprogenitor, osteoblast, and osteocyte cells (Shao et al., 2021). Studies show that ROS increases the apoptosis of osteoblast cells through signaling pathways dependent on MAPKs, ERK1/2, and JNK. Also, these free radicals increase the differentiation, maturation, and activation of osteoclasts by increasing the RANK-L/OPG ratio (Akbari et al., 2017). The results of the present study also showed that following the fracture, the level of oxidative stress increased and along with serum changes (NO levels), and the antioxidant capacity of the femur tissue decreased (FRAP and TBARS levels). The increase of pro-inflammatory cytokines, along with the increase of ROS, inhibits the pathway (OPG/RANK/BMP-2) that stimulates the differentiation and activation of osteoblast cells, and finally, the rate of RANKL/OPG increases, which subsequently increases the differentiation and activation of osteoclast cells (Azizieh et al., 2019). In the present study, the expression levels of OPG, RANK, and BMP-2 genes and proteins decreased after fracture compared to the healthy group, while the expression of RANK-L gene and protein increased.
Studies have shown that P. miliaceum extract is rich in minerals such as calcium, phosphorus, magnesium, and iron. In addition to mineral elements, this plant is a good source of polyphenolic compounds, including isoflavonoids and flavonoids (Saqib et al., 2022). Studies have shown that isoflavonoids such as genistein, daidzein, and biochanin A, along with flavonoids such as quercetin and kaempferol, can be effective in reabsorbing calcium, stimulating osteoblast activation pathways, and inhibiting osteoclast activity through different pathways (Şöhretoğlu & Renda, 2020). Ma et al. (2011) showed that genistein can stimulate the differentiation and mineralization of osteoblasts by increasing the expression of BMP-2 and RUNX-2 in in vitro and in vivo models in rat’s frontal and parietal bones (Ma et al., 2011). Zhou & Lin (2014) study on quercetin in the proliferation and differentiation of Mesenchymal stem cells (MSCs) isolated from adipose tissue revealed that this flavonoid compound increased the expression of BMP-2, Runx2, and OPN induce osteogenic differentiation in these cells into osteoblasts (Zhou & Lin, 2014). Studies have shown that daidzein and genistein increase the reabsorption of Ca by the intestinal microflora and proximal convoluted tubules of kidney and its absorption in the bone tissue, and hence consuming these isoflavonoids in postmenopausal women prevents osteoporosis (Lu et al., 2020). Also, studies have shown that isoflavonoids genistein, formononetin, and biochanin A stimulate the OPG/RANK/BMP-2 signaling pathway through the activation of ERK, p38, and AKT signaling and ultimately inhibit osteoclastogenesis and activate osteoblasts (Valizadeh et al., 2021). Noor et al. (2018) showed that extracts containing flavonoids rutin, apigenin, and kaempferol can prevent osteoporosis by regulating the RANK/RANK-L/OPG system and reducing the rate of RANK-L/OPG (Noor et al., 2018).
Also, the study of Kassi et al. (2011) showed that the extract containing genistein, daidzein, and kaempferol can increase the differentiation ratio and osteoclast/osteoclast activation through the reduction of IL-6 and RANK-L and the increase of OPG (Kassi et al., 2011). Ruiz et al. (2018) reported that flavonoids inhibit the differentiation and activation of osteoclasts by downregulating RANK-L through inhibition of the pro-inflammatory cytokines TNFα, IL-1, IL-6, IL-7, and IL-17 (Ruiz et al., 2018). In the present study, P. miliaceum was able to reduce the level of pro-inflammatory cytokines (TNFα, IL-1β, and IL-6), which subsequently strengthened the OPG/RANK/BMP-2 signaling pathway, effective in the osteoblastic process of osteogenesis. Studies have shown that P. miliaceum, due to its high content of polyphenolic antioxidant compounds, has protected various tissues against oxidative damage induced in them by increasing the activity of antioxidant enzymes (SOD, catalase, and GPx) and reducing free radicals (Sabir et al., 2011). Tian et al. (2017) showed that the inhibition of ROS-induced ERK/NF-κB/TNF/IL-6 pathway can stimulate osteoclast apoptosis and inhibit osteoclastogenesis. These researchers showed that polyphenol compounds activate and differentiate osteoblasts by increasing endogenous antioxidant enzymes (SOD, CAT, and GPx) and inhibiting the Nrf2/HO-1 pathway (signaling pathway that inhibits osteoblastic differentiation) (Tian et al., 2017). In the present study, P. miliaceum extract, due to its high content of antioxidant compounds, was able to increase the activity of endogenous enzymes (SOD, CAT, and GPx) and also increase the total antioxidant capacity (FRAP and TBARS) of the femur tissue. Therefore, it can be said that the extract of P. miliaceum plant by providing the necessary microenvironment for osteogenesis through the increase of minerals and also through its anti-inflammatory and antioxidant activities strengthens the path of osteoblastic proliferation and differentiation and also by reducing the rate of RANK-L/OPG, from to prevent the differentiation and activation of osteoclasts.
Conclusion
According to the results of this study, it can be said that some medicinal plants, including P. miliaceum, with their multifactorial activities, can accelerate osteogenesis and repair the process of bone tissue along with the surrounding soft tissue by preventing oxidative damage and suppressing pre-inflammatory pathways. It seems that with additional studies, it is possible to design effective prodrugs in the process of osteogenesis by purifying active ingredients or preparing commercial extracts from P. miliaceum plant used in orthopedics of bone lesions. Also, in future studies, the green synthesis of P. miliaceum nanoparticles could be explored to enhance its therapeutic effects at lower doses. It is recommended that other pathways involved in the process of osteogenesis, such as PI3K-Akt, BMP-2/Runx2, and BMP/Smad signaling, be evaluated in in vivo and in vitro models to pave the way for clinical trials involving patients with osteoporosis and other bone-related lesions.
Footnotes
Abbreviations
Ca: Calcium; P: Phosphorus; ALP: Alkaline phosphatase; M-CSF: Macrophage-colony stimulating factor; BMPs: Bone morphogenetic proteins; LC-MS/MS: Liquid chromatographic-tandem mass spectrometric; ELISA: Enzyme-linked immunosorbent assay; TNF-α: Tumor necrosis factor-α; CRP: C-reactive protein; IL-1β: Interleukin-1β; NO: Nitric oxide; GPx: Glutathione peroxidase; SOD: Superoxide dismutase; CAT: Catalase; FRAP: Ferric reducing antioxidant power; and TPTZ: 2, 4, 6-Tripyridyl-riazine.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Key Program of Natural Science Research of Anhui Provincial Education Department. Grant number: KJ2020A858.
Statement of Informed Consent and Ethical Approval
Necessary ethical clearances and informed consent was received and obtained respectively before initiating the study from all participants.
