Abstract
Objectives
Bee pollen (BP) contains isoflavonoids and minerals that can be effective in bone repair, stimulation of osteoprogenitors, differentiation of osteoblasts, and inhibition of osteoclasts. For this purpose, in the present study, BP was used to stimulate osteogenesis in an animal model of a femur fracture.
Materials and Methods
After induction of the femur fracture model, 80 Wistar rats in eight groups (n = 10) were studied as follows. Normal control group (0.5 cc of distilled water, DW/gavage/90 days), BP group (200 mg/kg BP/daily/gavage/90 days), fracture (FX-0.5 cc of distilled water/gavage/90 days), FX group treated with 100 and 200 mg/kg BP (100 and 200 mg/kg BP/daily/gavage/90 days), FX group treated with osteocare (OC) syrup (1 mL/day/gavage/90 days) and combinatorial FX groups treated with BP and OC syrup (100 and 200 mg/kg BP plus 1 mL OC/daily/gavage/90 days). On the 30th, 60th, and 90th days, radiography of the lesion was done. Bone mineral density (BMD) was measured by dual-energy X-ray absorptiometry during the mentioned days. At the end of the study, blood calcium (Ca), phosphorus (P), and alkaline phosphatase (ALP), along with the activity of glutathione peroxidase (GPx), superoxide dismutase (SOD), and catalase (CAT) enzymes, and nitric oxide levels, were measured. Calcitonin and parathyroid hormone levels were measured with enzyme-linked immunosorbent assay (ELISA) kits. In order to evaluate the stimulation of osteoprogenitors, the expression of osteoprotegerin (OPG), receptor activator of nuclear factor kappa-B (RANK), receptor activator of nuclear factor kappa-B ligand (RANKL), and bone morphogenetic protein-2 (BMP-2) genes and proteins in bone tissue was measured.
Results
The evaluations of this study showed that BP could improve BMD parameters through stimulation of the OPG/RANK/BMP-2 pathway. BP also improved serum levels of biochemical factors (Ca, P, and ALP) and hormones related to osteogenesis.
Conclusion
BP can be used for bone fractures and disorders related to osteoporosis.
Introduction
Bone repair is a complex process that involves various cellular and molecular factors. The factors that influence bone repair include age, nutritional status, systemic diseases, smoking, alcohol consumption, medications, and mechanical stability (Houschyar et al., 2020). Nutritional status is also a crucial factor in bone repair, as adequate amounts of calcium (Ca), vitamin D, and other nutrients are required for bone growth and repair (Goodman et al., 2019). Systemic diseases such as diabetes, osteoporosis, and cancer can also affect bone repair, as can certain medications such as corticosteroids and chemotherapy drugs. Also, calcitonin and parathyroid hormone (PTH) are two hormones that play important roles in the regulation of bone metabolism and can be used in the treatment of osteoporosis. The balance between calcitonin and PTH is important for maintaining bone homeostasis and promoting bone healing. While the calcitonin/PTH ratio is typically used as a direct measure of bone fracture and repair, understanding the roles of these hormones in bone metabolism can help guide treatment decisions for bone disorders such as osteoporosis and fracture healing (Babić et al., 2022). The balance of the calcitonin/PTH ratio maintains the balance of the osteoblast/osteoclast ratio, which is effective in repairing bone tissue and the proliferation and secretion of matrix vesicles containing collagen organic matrix and Ca and phosphorus (P) minerals (Xi et al., 2022).
The osteoprotegerin (OPG)/receptor activator of nuclear factor kappa-B (RANK)/receptor activator of nuclear factor kappa-B ligand (RANKL)/bone morphogenetic protein-2 (BMP-2) pathway is a complex signaling system that plays an important role in bone repair. This pathway involves several different proteins, including OPG, RANK, RANKL, and BMP-2 (Xue et al., 2021). RANKL is a protein that is primarily produced by osteoblasts and is responsible for stimulating the activity of osteoclasts, which break down bone tissue. RANK is a receptor that is expressed on the surface of osteoclasts and binds to RANKL, activating osteoclast activity. OPG is a decoy receptor that binds to RANKL and prevents it from binding to RANK, thereby inhibiting osteoclast activity (Zhao et al., 2019). During bone repair, the OPG/RANK/RANKL pathway is activated to regulate the balance between bone resorption and bone formation. Initially, RANKL expression is increased to stimulate osteoclast activity and remove damaged bone tissue. As bone repair progresses, OPG expression is increased to inhibit osteoclast activity and allow for bone formation (Tobeiha et al., 2020). The OPG/RANK/RANKL/BMP-2 pathway is one of the important axes of the therapeutic target. RANKL:OPG ratio is an important index in the osteoblast–osteoclast activity system (Lv et al., 2019; Zhang et al., 2022).
Reactive oxygen species (ROS) and inflammatory cytokines such as interleukin-1 (IL-1), interleukin-6 (IL-6), and tumor necrosis factor-alpha (TNF-α) play important roles in bone repair, but they can also have detrimental effects if their production is excessive or uncontrolled. ROS are molecules that are generated during normal cellular metabolism, and their production can be increased in response to injury or inflammation. During the early inflammatory phase of bone repair, inflammatory cytokines and ROS are produced to stimulate the recruitment of immune cells to the site of injury (Marcucci et al., 2023; Wang & He, 2020). These cells are responsible for removing damaged tissue and debris and promoting the formation of new blood vessels. Therapies that target these molecules may be used to promote bone healing in individuals with bone fractures or other bone disorders (Arafa et al., 2023). Studies have shown that ROS can affect the RANKL/OPG ratio in bone cells. Excessive ROS production can increase RANKL expression and decrease OPG expression, leading to increased osteoclast activity and bone resorption (Ibarra et al., 2016). In contrast, antioxidant treatments have been shown to decrease RANKL expression and increase OPG expression, leading to decreased osteoclast activity and improved bone health. In addition, IL-1β/BMP-2, IL-6/RANKL, and TNF-α/OPG pathways, which play a role in the process of osteoblastic and osteoclastic system activity, are targeted in bone healing (Arafa et al., 2023; Lv et al., 2020).
Certain natural supplements, due to their richness in mineral elements and polyphenolic compounds (especially isoflavonoids and flavonoids), can be effective in promoting osteogenesis and bone repair through multiple mechanisms (Kushwaha et al., 2014; Ming et al., 2013). Studies have shown that flavonoids and isoflavonoids can decrease RANKL expression and increase OPG expression in bone cells, which leads to reduced osteoclast activity and increased bone formation. These effects may be mediated through the estrogen receptor pathway, as genistein has been shown to bind to estrogen receptors and activate downstream signaling pathways. Additionally, the potent anti-inflammatory properties of these compounds inhibit the release of proinflammatory cytokines, which in turn inhibit pathways related to osteogenesis (such as the IL-1β/BMP-2, IL-6/RANKL, and TNF-α/OPG pathways) (Chen et al., 2019).
Bee pollen (BP) is a mixture of nectar, flower pollen, enzymes, wax, and honey synthesized by bees and stored in the hive to feed the queen and worker bees. Studies have shown that this substance is rich in mineral elements such as Ca, magnesium, zinc, iron, and phosphorus, as well as polyphenolic compounds including apigenin, genistein, kaempferol, quercetin, and formononetin (Kasiotis et al., 2014; Komosinska-Vassev et al., 2015). Various studies have demonstrated its anticancer, antidiabetic, antioxidant, and anti-inflammatory effects (Shaldoum et al., 2021). Additionally, studies have shown that plant–animal derivative compounds containing apigenin, genistein, kaempferol, quercetin, and formononetin increase the expression of BMP-2 and OPG and decrease the expression of RANK-L, which ultimately stimulate osteogenesis by osteoblasts and inhibit the activity of osteoclasts (Ming et al., 2013). Therefore, the present study was conducted to evaluate the effect of BP on the bone healing process in an animal model of femur bone fracture using biochemical, molecular, and histopathological approaches.
Materials and Methods
Preparation of BP Extract
After preparing 3000 g of dry BP and grinding it into powder using a soil grinder (Cat. No. H-4199.5F; Humboldt Company, USA), the resulting powder was added to a solution of hexane and methanol (30:70 v:v) and incubated for 72 h at 40°C in a dark room. The mixture was then passed through a No. 42 paper filter (Cat. No. 1442-125; Millipore, USA), condensed with a rotary evaporator (model: 9230; Buchi Rotavapor, Switzerland), and the resulting BP extract (250 g: 20% extraction efficiency) was stored at −4°C (Shaldoum et al., 2021).
Animal Care, Grouping, Treatment, Experimental Design, and Research Timeline
Rat Fracture Model
To induce femur fracture in the rats, they were subjected to a standard protocol of sedation and anesthesia (ketamine 45 mg/kg and xylazine 30 mg/kg). After shaving the femoral area located 1 cm below the femur head, a closed fracture was created using scissors (Sun et al., 2015).
Animal Care
Eighty adult Wistar rats, 3 months old and weighing 220 ± 30 g, were housed in propylene cages at a temperature of 25 ± 2°C, with a relative humidity of 50 ± 3%, and a lighting cycle of 12:12 (dark/light). The rats were acclimated to standard laboratory conditions for 72 h before induction of the close femur fracture model. They had free access to sanitary tap water and standard laboratory pellets for rats and were kept in accordance with international standard guidelines and protocols, as well as the ethics committee of Wenzhou Medical University. The standard pellets used provided all of the essential nutrients necessary for proper nutrition. The manufacturer’s instructions and recommendations state that each kilogram of these pellets contains 4.5 Mcal of energy, 21 g of protein per 100 g, 3 g of total fat per 100 g, 0.8 g of calcium per 100 g, 0.4 g of phosphorus per 100 g, and 6 g of total fatty acids per 100 g (including palmitic acid, oleic acid, linoleic acid, and arachidonic acid) (Shaldoum et al., 2021).
Animal Grouping
After induction of a closed femoral fracture, rats were randomly grouped into eight groups according to Table 1.
Experimental Design and Animal Grouping.
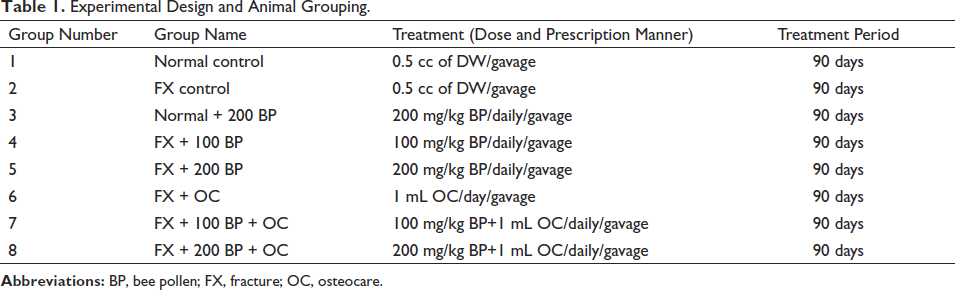
Throughout the 90-day study, the rats received BP every day at 9:00 a.m. and OC at 3:00 p.m. The appropriate dose of BP was chosen based on references, a pilot study, and the median lethal dose (LD50) technique to determine the most effective and nontoxic therapeutic dose (Shaldoum et al., 2021; Sun et al., 2015).
Acute Toxicity Test (LD50) for BP
To determine the most appropriate treatment dose with the least toxic effects, Lork’s two-step technique was utilized. Initially, nine rats were divided into three groups of three (n = 3/group) and administered doses of 10, 100, and 1000 mg/kg via gavage. Subsequently, three additional rats were divided into three independent groups and given doses of 50, 500, and 5000 mg/kg via gavage. After monitoring the groups for 24 h and using the following formula, the LD50 of BP was calculated, and the appropriate dose was selected for the study.
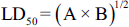
In this regard, (A) represents the lowest dose in which toxic symptoms (such as weight loss, diarrhea, nausea, and skin rashes) or death were observed, and (B) is the highest dose in which no death or toxic symptoms were observed in the group (John-Africa, 2019).
Serum Levels of PTH and Calcitonin Hormones
On the 91st day, at the end of the study, the rats were sacrificed using preanesthesia (100 mg/kg xylazine, 2%) and anesthesia (15 mg/kg ketamine, 10%) protocols. Blood was collected from the heart, and serum samples were separated through centrifugation of blood samples at 10,000 g for 15 min. The levels of serum PTH (Cat. No. NBP2-53170) and calcitonin (Cat. No. MAB-4614) hormones were measured using commercial Novus enzyme-linked immunosorbent assay (ELISA) kits (Novus Biologicals, USA) based on a colorimetric assay, following the manufacturer’s recommendations and protocol (Hu et al., 2022).
Serum Levels of P and Ca, and Alkaline Phosphatase (ALP) Activity
The serum was separated by centrifuging the blood at 10,000 g for 20 min, and the serum levels of P (Cat. No. ab234579) and Ca (Cat. No. ab102505), along with the serum activity of ALP (Cat. No. ab83369), were measured using Abcam ELISA kits, similar to the method for measuring PTH and calcitonin hormone levels. The measurements were performed according to the manufacturer’s instructions and protocol (Hu et al., 2022).
IL-1β, TNF-α, and IL-6 Serum Levels
After separating the serum by centrifuging the blood at 10,000 g for 20 min, the serum levels of IL-1β (Cat. No. 255730), IL-6 (Cat. No. ab100772), and TNF-α (Cat. No. ab236712) were measured using Abcam ELISA kits (Abcam Company, USA). The measurements were performed in a similar manner to the method for measuring PTH and calcitonin hormone levels, following the manufacturer’s instructions and protocol (Hu et al., 2022).
Serum Nitric Oxide Levels
Serum total nitric oxide (NO) levels were measured using the ZellBio commercial kit (Cat. No. ZX-44107-96; ZellBio GmbH, Germany). According to the manufacturer’s recommendations, 50 µL of a standard or serum sample was added to all wells of a 96-well plate, and then 50 µL of assay buffer was added as a zero standard. Next, 10 µL of nitric reductase solution was added to each well, and the plate was incubated for 20 min at 25°C. Then, 25 µL of solution A and 25 µL of solution B from the kit were added to each well and incubated for an additional 20 min at 25°C. Finally, the absorbance of the final mixture was read at a wavelength of 540–570 nm using a UV-visible spectrophotometer (Model No. V-630 Bio; JASCO, USA) (Afzali et al., 2020).
Serum Activity of Catalase, Superoxide Dismutase, and Glutathione Peroxidase
The serum activity of glutathione peroxidase (GPx) (Cat. No. E-EL-R2491; Elabscience, USA), superoxide dismutase (SOD) (Cat. No. E-EL-R1424; Elabscience, USA), and catalase (CAT) (Cat. No. ab100764; Abcam, USA) was measured using the ELISA-based commercial kits. The measurements were performed according to the manufacturer’s instructions and protocol Afzali et al., 2020.
Femur Lipid Peroxidation Levels (TBARS), Total Thiol, and Ferric-Reducing Antioxidant Power (FRAP) Levels
To measure the total antioxidant capacity of femur tissue using the FRAP method, the femur was dissected and a homogenous tissue mixture was prepared. Then, 200 µl of the homogenous tissue was incubated with the FRAP solution, which contained 5 mL of 2, 4, 6-tripyridyl-triazine, 50 mL of acetate buffer, and 3 mL of ferric chloride, at 37°C for 10 min. Finally, the absorbance of the final mixture was recorded using a Stat Fax ELISA reader (Stat Fax, USA) at a wavelength of 593 nm, and the total antioxidant capacity was calculated as µmol/mg protein Afzali et al., 2020.
To evaluate the lipid peroxidation level in femur tissue, the TBARS level measurement method was used. In this method, 100 µL of homogenous femur tissue was added to a TBARS solution containing thiobarbituric acid (50 µL), butylated hydroxytoluene (2 µL), and phosphoric acid (50 µL), and incubated for 30 min at 37°C. Finally, the absorbance of the final mixture was recorded using a Stat Fax ELISA reader (Stat Fax, USA) at a wavelength of 532 nm, and the TBARS level was calculated as nmol/mg protein Afzali et al., 2020.
To measure the total thiol level of femur tissue, 50 g of homogenized femur tissue was incubated with 250 µL of Tris-EDTA at 25°C for 10 min. The first absorbance (α) of the mixture was recorded at a wavelength of 412 nm using a Stat Fax ELISA reader (Stat Fax, USA). Then, 20 µL of 5,5-dithio-bis-(2-nitrobenzoic acid) (DTNB) was added to the mixture, and the resulting mixture was incubated at 25°C for 20 min. The final absorbance (β) of the mixture was recorded again at 412 nm. The absorbance of the blank solution (µ) (DTNB) was also recorded at 412 nm and placed in the following formula to calculate the tissue thiol level:
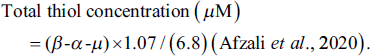
Real-Time Polymerase Chain Reaction (RT-PCR) Assay
Total RNA Extraction
To isolate total RNA from femur tissue, the Bio Basic column animal total RNA purification kit (Cat. No. EZ-10; Bio Basic Inc., Canada) was used according to the manufacturer’s recommendations. First, 50 mg of femur tissue was powdered by adding liquid nitrogen and added to RNase-Free 1.5 mL centrifuge tubes containing 350 µL of Buffer Rlysis-AG. After incubating the resulting mixture for 5 min at 25°C, 300 µL of ethanol was added and mixed gently. The resulting mixture was transferred to a spin column and centrifuged at 12,000g for 30 s at 25°C. Then, in two steps, 0.5 mL of kits GT and NT solutions were added to the mixture and centrifuged at 12,000g for 30 s at 25°C. Finally, 50 µL of RNase-free water was added to the mixture, and after incubating for 2 min at 25°C, the final mixture was centrifuged at 12,000g for 30 s at 25°C, and the extracted RNA was kept at −80°C Yang et al., 2013.
cDNA Synthesis and Quantitative Real-Time PCR (qRT-PCR)
To synthesize cDNA from the total RNA of femur tissue, the Takara cDNA synthesis kit (Cat. No. RTK0104; Takara Bio., Japan) was used according to the manufacturer’s recommendations. First, a mixture of 1000 ng total RNA, 1 µL of oligo and random primers, Takara Bio Mastermix (10 µL), and deionized water (8 µL) was prepared. The resulting mixture was transferred to a thermal cycler, and cDNA templates were synthesized from the extracted RNA by following the temperature protocol (10 min at 25°C, 45 min at 60°C, and 5 min at 60°C).
Using the cDNA templates (1000 ng), reverse and forward primers (1 µL), and Takara Bio Mastermix (8 µL), a RT-PCR mixture was prepared. The expression of OPG, RANKL, RANK, and BMP-2 genes was measured by running 42 temperature cycles in an Applied Biosystems thermocycler (Applied Biosystems, Quantstudio 1, USA). Each temperature cycle in the RT-PCR included a holding stage at 50°C for 5 min, a denaturation stage at 95°C for 15 s, an annealing stage at 60°C for 1 min, and an extension stage at 70°C for 5 min. After the temperature cycles, a melting curve was drawn by increasing the temperature from 60 to 95°C at a rate of 1°C/1 s. The threshold cycle (CT) of genes including control (C), sample (S), and β-actin was recorded and used in the fold change (2−∆∆CT) formula.
The primers used in the RT-PCR were designed using Primer-3 software (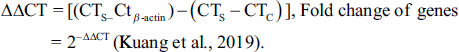
Primer Sequences.

Western Blotting Assay
To evaluate the expression of OPG/RANK/RANKL/BMP-2 pathway proteins, the Western blotting technique was used. First, femur tissue was washed with PBS (200 µL), and 50 mg of femur tissue was lysed with radioimmunoprecipitation assay buffer containing Tris-HCl (50 mmol/L), NaCl (150 mmol/L), and 0.1% NP-40 [w/v]. The resulting mixture was centrifuged at 12,000 g at −4°C for 10 min. Next, 50 µg of the resulting mixture was mixed with 10 µL of loading buffer containing Tris-HCl (62.5 mmol/L), β-mercaptoethanol (1 µL), 10% glycerol (5 µL), and 0.5% sodium deoxycholate (5 µL) [w/v] and incubated for 10 min. The mixture was then loaded onto a 10% sodium dodecyl sulfate polyacrylamide gel (Cat. No. WBKLS0100; Millipore Bio Inc., UK) and transferred onto a polyvinylidene fluoride (PVDF) membrane.
The PVDF membrane was incubated with GeneTex OPG (Cat. No. GTX55734; 1:1000), RANK (Cat. No. GTX11697; 1:100), RANK-L (Cat. No. GTX32834; 1:200), and BMP-2 (Cat. No. GTX14933; 1:500) antibodies (GeneTex Bio., USA) overnight at 4°C. After washing with Tris-buffered saline and Tween-20 (0.05%) [w/v] to block excess antibodies, a horseradish peroxidase-conjugated secondary antibody was added and incubated for 1 h at 37°C. Finally, using Bio-Rad software and an enhanced chemiluminescence reagent (e-BLOT company, China), the signals of protein bands were recorded and evaluated using Image J software (Liu et al., 2019).
Bone Densitometry
The rats were subjected to a sedation/anesthesia protocol using ketamine (45 mg/kg) and xylazine (30 mg/kg) to induce ventral recumbency. Dual-energy X-ray absorptiometry scans were performed on the 0th, 30th, 60th, and 90th days of the study using a specialized scanner. The scans were analyzed using Small Animal Analysis Software (LI-COR Biosciences) to measure bone mineral density (BMD) and other parameters (Wong et al., 2018).
Femur Tissue Histopathology
To evaluate the histopathological changes of the femur tissue, the bone tissue was dissected and fixed in 10% formalin. Decalcification of the femur tissue was performed using 10% formic acid and 5-µm sections were prepared and placed on slides. Hematoxylin and eosin staining was used to evaluate the thickness of trabeculae, the degree of mineralization of spicules, the density of osteoblasts, and the space of bone marrow cells. The evaluation was done at 100× magnification using an optical microscope (model No. BX61TRF; Olympus, Japan) connected to ImageJ software (Akbari et al., 2017).
Statistical Analysis Approach
To compare all quantitative results between the studied groups, a one-way analysis of variance followed by the Newman–Keuls post hoc test was used. A p-value less than 0.05 was considered statistically significant. The normality of the data was evaluated using the Kolmogorov–Smirnov test, and p-values greater than 0.05 were considered statistically significant for normal and homogeneous data. All results are expressed as means ± standard deviation. Data analysis was performed using SPSS software (Ver. 16; IBM Inc., USA), and graphs were drawn using GraphPad Prism software (Ver. 9; GraphPad Inc., USA).
Results
Acute Toxicity Test (LD50)
Based on the evaluation of the LD50 of BP after 24-h monitoring of rats, it was found that no dose caused the death of rats. Only at a dose of 5000 mg/kg (A), a decrease in appetite was observed in rats. The highest dose (B) at which no toxic symptoms were observed in rats was 3000 mg/kg. Using Lorke’s formula, the LD50 of BP was calculated to be 3.870 g/kg. This means that doses lower than 3870 mg/kg in rats are considered safe. However, it is important to note that the safety and toxicity of BP may vary in different animal species and in humans, and further studies are needed to determine safe dosages in humans.
Weights of Body and Femur in Rats
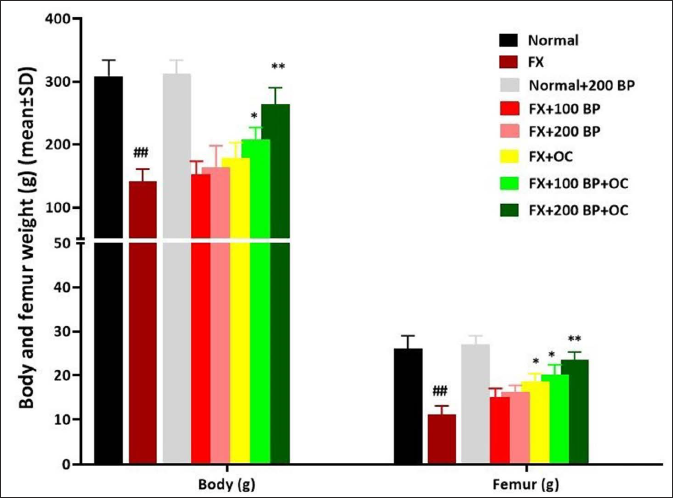
The results of the study showed that following the fracture of the femur, both body weight and femur weight at the end of the study decreased significantly compared to the normal control group (p < 0.01). However, BP was able to improve both weight parameters in the receiving groups, with significant weight gain observed in the BP and OC co-treatment groups [FX+100 BP+OC (p < 0.05) and FX+200 BP+OC (p < 0.01)] compared to the FX group (p < 0.05) (Figure 1).
P and Ca Serum Levels, and ALP Activity
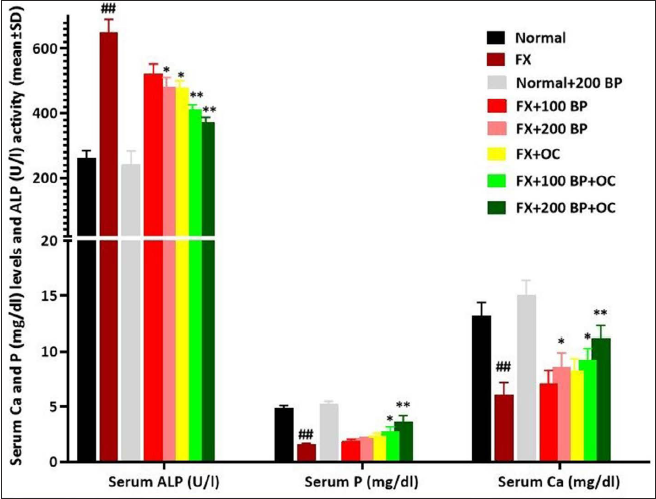
Following femur fracture, Ca-P homeostasis was disrupted, with the serum levels of phosphorus and Ca showing a significant decrease compared to the normal control group (p < 0.01) and the level of ALP activity showing a significant increase (p < 0.01). However, BP was able to stabilize bone mineral homeostasis, with BP alone significantly increasing (p < 0.05) the level of blood Ca and significantly decreasing (p < 0.05) the level of ALP activity compared to the FX group. The greatest effect of BP was observed in the co-treatment group with OC (especially in the FX+200 BP+OC group), wherein the co-treatment groups, in a dose-dependent manner, there was a significant decrease (p < 0.01) in ALP activity and a significant increase in the serum Ca and P levels (p < 0.01) compared to the FX group (Figure 2).
PTH and Calcitonin Serum Levels
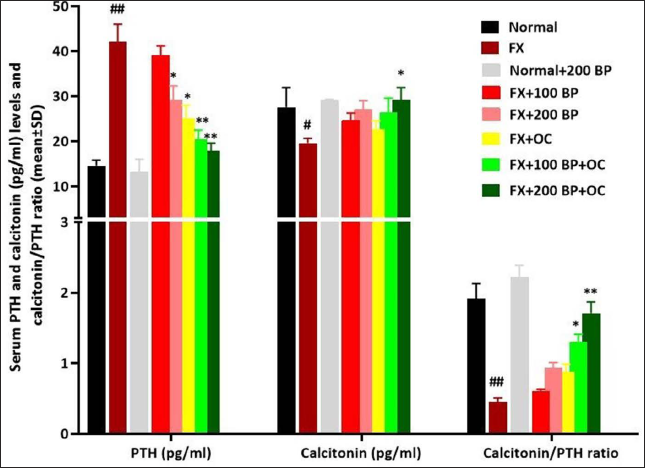
The results related to the serum level of hormones involved in the process of osteogenesis showed that in the FX group compared to the normal control group, the level of PTH increased (p < 0.01), the level of calcitonin (p < 0.05) decreased, and the calcitonin/PTH ratio decreased (p < 0.01) significantly. However, BP alone was able to significantly reduce (p < 0.05) PTH levels compared to the FX group. In the co-treatment groups, BP and OC caused a significant decrease (p < 0.01) in PTH level and a significant increase (p < 0.05) in calcitonin level in a synergistic and dose-dependent manner compared to the FX group (Figure 3).
IL-6, TNF-α, and IL-1β Serum Levels
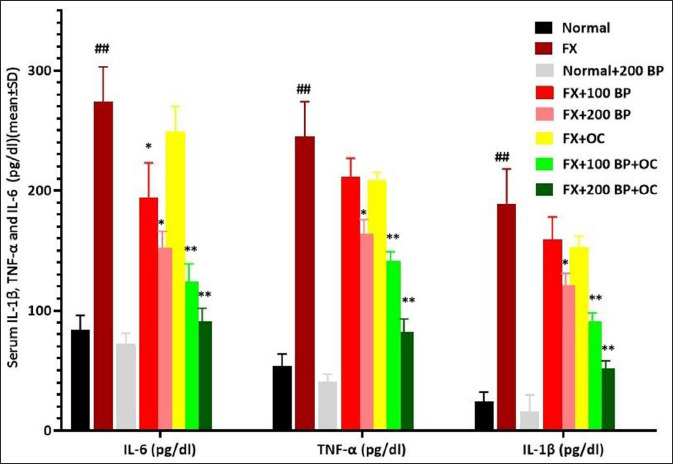
Following fracture and inflammation in the femur region, the levels of all three proinflammatory cytokines showed a significant increase (p < 0.01) compared to the normal control group. However, with its anti-inflammatory effects, BP alone was able to significantly reduce (p < 0.05) the levels of all three pro-inflammatory cytokines at a dose of 200 mg/kg compared to the FX group. The maximum reduction of all three cytokines was observed in the BP and OC synergistic groups, where all three cytokines showed a significant decrease (p < 0.01) in a dose-dependent manner compared to the FX group (Figure 4).
Serum Stress Oxidative Parameters
After femur fracture, the production of free radicals can cause a significant increase (p < 0.01) in the serum level of NO and a significant decrease (p < 0.01) in the activity of endogenous antioxidant enzymes compared to the normal control group. However, BP alone at a dose of 200 mg/kg was able to significantly reduce the serum NO level compared to the FX group and significantly increase (p < 0.05) the activity of all three endogenous antioxidant enzymes. The most significant antioxidant effects were observed in the co-treatment groups, where BP along with OC increased the antioxidant activity of all three enzymes in a dose-dependent and synergistic manner and decreased (p < 0.01) the serum NO level compared to the FX group (Figure 5).
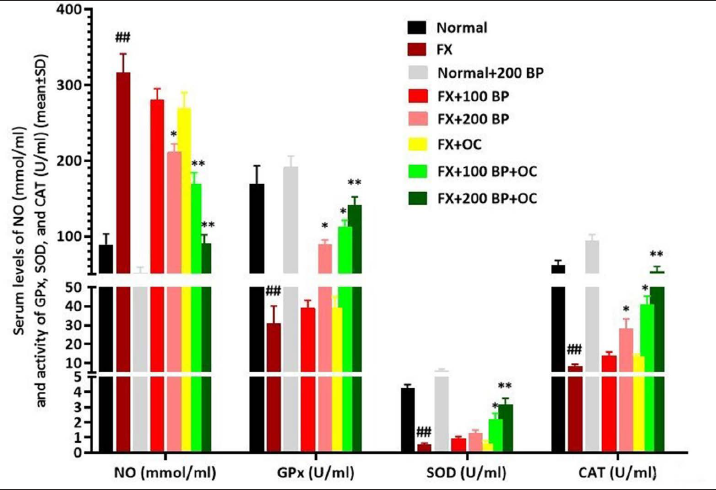
Femur Tissue Stress Oxidative Parameters
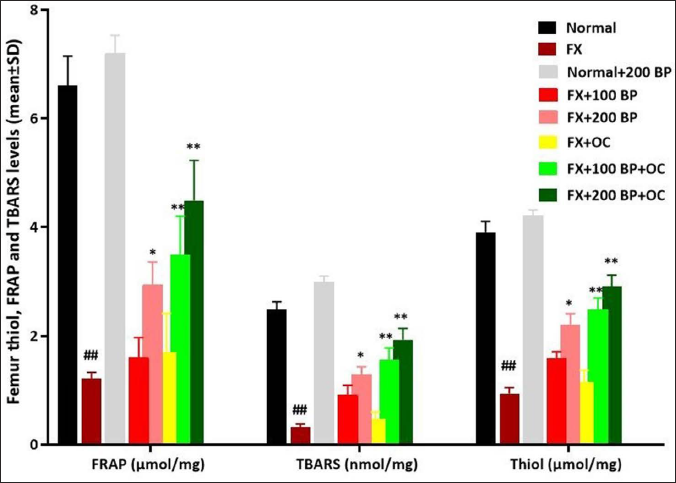
After femur fracture, the total antioxidant capacity (FRAP) of the femur decreases significantly (p < 0.01) compared to the normal control group, resulting in increased lipid peroxidation (measured by decreased TBARS) and total thiol levels in the femur tissue (p < 0.01). However, BP alone was able to increase (p < 0.05) FRAP compared to the FX group, resulting in decreased lipid peroxidation (measured by increased TBARS) and total thiol levels (p < 0.05). The antioxidant parameters of the femur tissue also improved in the BP and OC co-treatment groups in a dose-dependent and synergistic manner, where in both co-treatment groups, the tissue FRAP and TBARS levels increased significantly (p < 0.01) compared to the FX group, and total thiol levels decreased significantly (p < 0.01) (Figure 6).
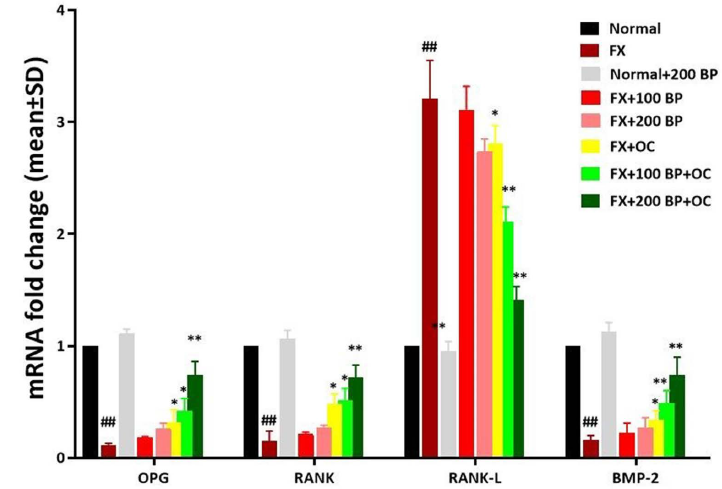
Expression of RANK, RANKL, OPG, and BMP-2 Genes
After femur fracture, the activity of osteoclasts increases, leading to bone destruction, and the expression of RANK, OPG, and BMP-2 genes has a significant decrease (p < 0.01) compared to the normal control group. In contrast, the expression of the RANK-L gene shows a significant increase (p < 0.01). However, BP and OC can synergistically improve the expression of osteoblast-stimulating genes. In the FX+100 BP+OC group, the expression of all three genes (RANK, p < 0.05; OPG, p < 0.05; and BMP-2, p < 0.01) significantly increased, and the RANKL (p < 0.05) gene expression decreased significantly compared to the FX group. The greatest effect of BP was observed in the FX+200 BP+OC group on the OPG/RANK/RANKL/BMP-2 pathway, where all three genes (RANK, OPG, and BMP-2) were significantly increased (p < 0.01) compared to the FX group, and RANKL gene expression decreased significantly (p < 0.01) (Figure 7).
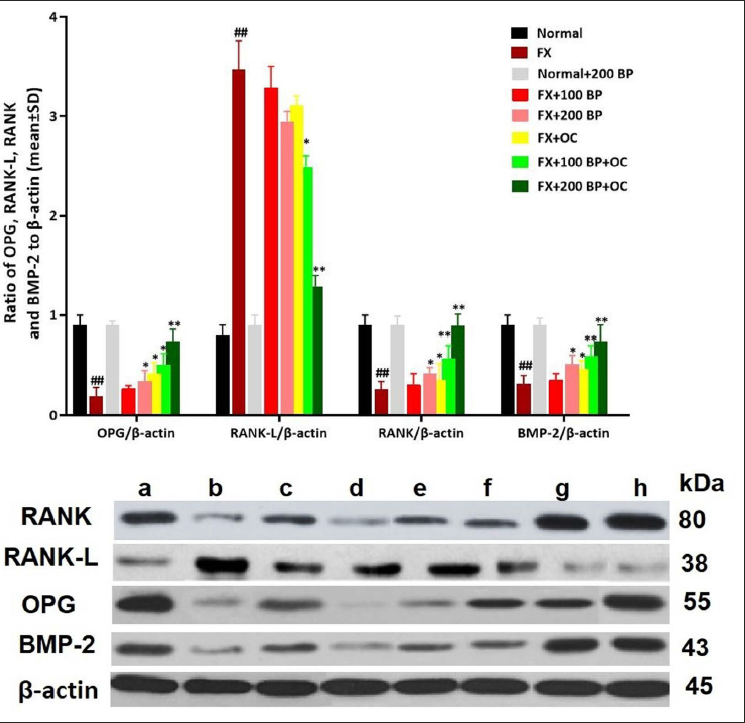
Expression of RANK, RANKL, OPG, and BMP-2 Proteins
After evaluating the results related to the expression of OPG/RANK/RANKL/BMP-2 pathway proteins, it was found that the stimulatory proteins of osteoblasts (RANK, OPG, and BMP-2) had decreased significantly (p < 0.01) compared to the normal control group, while the expression of RANK-L protein had increased significantly (p < 0.01). However, BP and OC could synergistically improve the expression of osteoblast-stimulating proteins. In the FX+100 BP+OC group, the expression of all three proteins (RANK, p < 0.05; OPG, p < 0.05; and BMP-2, p < 0.01) significantly increased, and the RANKL (p < 0.05) protein expression decreased significantly compared to the FX group. The greatest effect of BP was observed in the FX+200 BP+OC group on the OPG/RANK/RANKL/BMP-2 pathway, where all three proteins (RANK, OPG, and BMP-2) were significantly increased (p < 0.01) compared to the FX group, and RANK-L protein expression decreased significantly (p < 0.01) (Figure 8).
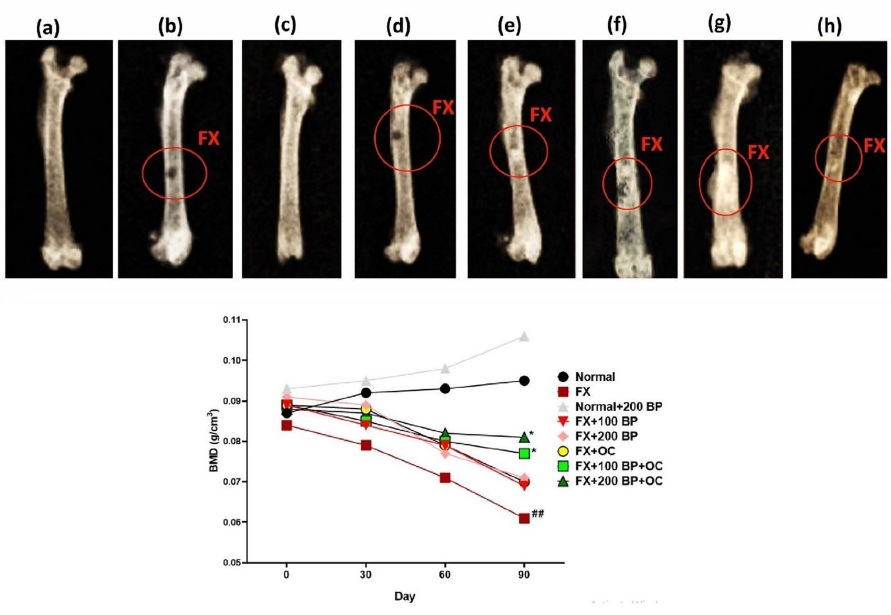
Bone Mineral Density
The evaluation of BMD showed a decrease in all groups of rats with fractured femurs compared to the normal control group. This reduction was significant (p < 0.01) in the FX group. However, on the 90th day of the study, there was a significant increase (p < 0.05) in BMD in the co-treatment groups of BP and OC (FX+100 and 200 BP+OC groups) compared to the FX group (Figure 9).
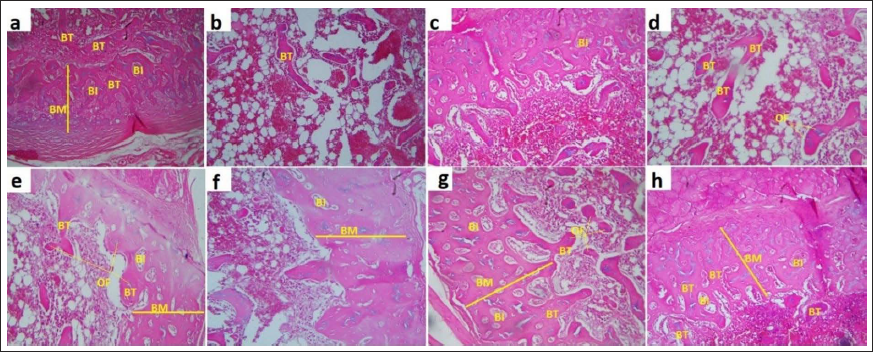
Histopathological Findings of the Femur Tissue
After femur fracture, inflammation and edema in the area, along with the destruction of the endosteum and periosteum, can inhibit mineralization and the formation of spicules and trabeculae in the fracture area. The absence of new osteon formation by osteoblasts and the presence of fibrous tissue can also be observed. However, in the treatment groups with BP alone and the synergistic groups of BP and OC, the formation of new osteons, trabeculae, and mineral spicules was observed. The healing areas showed the organization of bone islets (BI) centered on osteocytes and surrounded by osteoblasts, as evident in Figure 10. These findings suggest that BP and its synergistic effect with OC can promote bone repair and the formation of new bone tissue, leading to better healing outcomes.
Discussion
The results of the present study suggest that BP can improve the bone healing process through multiple mechanisms. Due to its high mineral content, BP can help maintain the serum calcium level within the appropriate range (20–30 mg/dL) and increase the calcitonin/PTH hormone ratio. Additionally, the high polyphenol content of this natural substance can increase the OPG/RANKL ratio, promoting the proliferation, differentiation, and activity of osteoblasts. Furthermore, BP can prevent the release of free radicals and inflammatory cytokines that can hinder the bone healing process, thereby supporting bone repair.
After a bone fracture, if the calcium level in the blood is low (usually falling below 20–30 mg/dL after the fracture), osteoclasts are activated and osteoblasts are inhibited. Furthermore, an increase in the rate of PTH/calcitonin occurs, and the formation of collagen aggregates along with matrix vesicles is inhibited. This process interferes with bone repair, especially in people who normally have blood calcium levels below 20–30 mg/dL, such as postmenopausal women and elderly men. Studies have shown that the consumption of chemical and natural supplements containing mineral elements, especially calcium, is effective in activating osteoblasts and inhibiting osteoclasts, promoting bone repair (Patel et al., 2016; Shuid et al., 2010). Xu et al. (2020) demonstrated that calcium, as a key microelement in bone repair, is effective in increasing the concentration of calcitonin hormone during bone fracture and increasing its level, thereby promoting bone repair (Xu et al., 2020). Studies have also shown that the serum calcium level can influence the OPG/RANK-L pathway and bone metabolism. Low serum calcium levels have been associated with increased RANKL expression and decreased OPG expression, leading to increased osteoclast activity and bone resorption. In contrast, high serum calcium levels have been shown to decrease RANKL expression and increase OPG expression, leading to decreased osteoclast activity and improved bone health (Tobeiha et al., 2020). In the present study, BP increased the serum calcium level and, in addition to increasing the calcitonin/PTH rate, also increased the OPG/RANKL rate at the gene and protein levels, thereby improving bone healing and increasing BMD in the BP recipient groups. Furthermore, the results of the present study showed that BP had synergistic effects with drugs and routine supplements prescribed for bone repair, including OC.
Free radicals such as H2O2, •O2−, and •OH are unstable molecules that can cause oxidative damage to cells and tissues. They are produced during normal cellular metabolism, and their production can be increased in response to injury or inflammation. Excessive free radical production can cause oxidative stress, which can impair cellular function and damage DNA, proteins, and lipids (Marcucci et al., 2023; Wang & He, 2020). Shao et al. (2021) demonstrated that these active species can increase mitochondrial apoptosis (Bax/Caspase-3/p53/Cytochrome C pathway) in osteoprogenitor cells and osteoblasts. Moreover, these molecules can inhibit the proliferation and differentiation of osteoblasts through apoptotic signaling pathways related to the proliferation of osteoblasts, including MAPKs, ERK1/2, and JNK, and interfere with the process of bone repair. These molecules increase the RANKL/OPG ratio and ultimately increase the activity of osteoclast cells (Prasadam et al., 2010). When a bone is fractured, a cascade of free radicals is generated, overwhelming the tissue’s endogenous antioxidant system. Studies have also shown that the ERK/NF-κB/TNF/IL-6 pathway, dependent on free radicals, induces apoptosis in osteoclast cells (Herrmann et al., 2011). In the present study, the activity of endogenous antioxidant enzymes and the total antioxidant capacity decreased during bone fracture, and the amount of lipid peroxidation caused by the destruction of the membrane of osteoblastic and osteoprogenitor cells increased. The presence of various polyphenolic compounds in BP, including apigenin, genistein, kaempferol, quercetin, and formononetin, could enhance the antioxidant system and improve indicators related to serum and tissue oxidative stress.
Valizadeh et al. (2021) demonstrated that isoflavone compounds such as genistein, formononetin, and biochanin A inhibit the differentiation and activity of osteoclasts through ERK, p38, and AKT signaling. These compounds can reduce the RANKL/OPG ratio by strengthening the RANK/OPG system and ultimately increase the proliferation, activity, and differentiation of osteoblasts (Valizadeh et al., 2021). Studies have also shown that levels of pro-inflammatory cytokines, including TNFα, IL-1, IL-6, IL-7, and IL-17, increase during bone damage. These cytokines inhibit the OPG/RANK/BMP-2 and BMP-2/Smad/Wnt signaling pathways, leading to the dominance of osteoclastogenesis and inhibition of osteoblast differentiation. In addition, IL-6 acts as a BMP-2 or OPG/RANKL inhibitor, interfering with the activity of osteoblasts. In the present study, after a bone fracture, the level of proinflammatory cytokines increased, but BP was shown to regulate them due to its polyphenolic compounds, thereby strengthening the process of osteogenesis (Arafa et al., 2023; Sahin et al., 2021). Based on the results obtained, it appears that BP may be useful for treating bone fractures and even for preventing fractures in individuals who are prone to them, particularly those who are elderly, have low serum calcium levels, postmenopausal women, or have absorption disorders. In such cases, BP may be prescribed along with calcium supplementation to improve bone health.
Conclusion
The findings of this study suggest that BP has the potential to increase bone density and promote bone repair in individuals with fractures through various antioxidant and anti-inflammatory mechanisms, regulation of essential minerals for bone repair, and strengthening of the OPG/RANK/BMP-2 pathway. Therefore, BP could be used as a preventive measure for individuals who are prone to fractures. Moreover, BP may be prescribed to individuals with fractures to support bone healing and improve bone health. Future studies could investigate other potential signaling pathways of BP in bone healing as well as explore its effects in other in vivo and in vitro models. It may also be necessary to develop a prodrug containing the pure extract or active ingredients of BP for use in future studies.
Footnotes
Acknowledgments
None.
Abbreviations
BMD: Bone mineral density; DXA: Dual-energy X-ray absorptiometry; Ca: Calcium; P: Phosphorus; ALP: Alkaline phosphatase; NO: Nitric oxide; PTH: Parathyroid hormone; OPG: Osteoprotegerin; RANK: Receptor activator of nuclear factor kappa-B; RANKL: Receptor activator of nuclear factor kappa-B ligand; BMP-2: Bone morphogenetic protein-2; ROS: Reactive oxygen species; IL: Interleukin; TNF-α: Tumor necrosis factor-alpha; CAT: Catalase; SOD: Superoxide dismutase; GPx: Glutathione peroxidase; FRAP: Ferric reducing antioxidant power; PCR: Polymerase chain reaction; qRT-PCR: Quantitative real-time PCR.
Authors’ Contribution
Conception and design: Y.L. Performing the experiments: X.Z. Analyzing the data and wrote the manuscript: C.W. Drafting the manuscript. All authors read and approved the final manuscript.
Availability of Data and Material
All data generated during this study are included in this published article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Informed Consent and Ethical Approval
The authors read and proved the final manuscript for publication.
The care and use of laboratory animals in this study were conducted in strict accordance with the Guide for the Care and Use of Laboratory Animals (NIH Publication No. 85-23, revised 1996), and the experimental design was approved by the Use Committee of Wenzhou Medical University.
Approval number: wydw2021-0580.
Funding
The authors did not receive any financial support for the research, authorship, and/or publication of this article.
