Abstract
Background
Schisandrin A (SchA) has multiple pharmacological features, and inflammatory bowel disease (IBD) represents a common digestive system disease mainly characterized by inflammation.
Objectives
To assess the anti-inflammatory effects and the mechanism of SchA in mice with enteritis and in MODE-K cells representing in vivo and in vitro models of inflammation.
Materials and Methods
DSS-induced IBD mouse models and MODE-K cells (an in vitro model of the intestine) were used to assess the effects of SchA on IBD inflammation and to determine the related signaling pathways.
Results
Our data showed that SchA exerted anti-inflammatory effects by reducing the general clinical symptoms and the pathological damage to the colonic mucosa in mice with IBD and by promoting the migration of H2O2-induced MODE-K cells, inhibiting apoptotic death, and reducing the release of inflammatory factors. Moreover, SchA downregulated Wnt/β-catenin in both enteritis mice and H2O2-induced cells.
Conclusion
SchA inhibits inflammation in DSS-induced IBD mice and H2O2-induced MODE-K cells by repressing Wnt/β-catenin signaling.
Introduction
Ulcerative colitis (UC), Crohn’s disease (CD), and some temporarily unspecified colitis forms are collectively referred to as inflammatory bowel disease (IBD), which constitutes a long-term atypical IBD. The incidence of IBD is increasing rapidly worldwide, posing a significant challenge to the physical and mental health of patients and imposing a huge burden on national healthcare resources. Additionally, IBD cases have a higher risk of anorectal dysfunction and impaired quality of life due to progressive disease features that cause proximal bowel extension stenosis, resulting in intestinal motility dysfunction and a greater risk of progression to colorectal cancer (CRC) later in life (Kobayashi et al., 2020; Rogler, 2014). The pathogenesis of IBD remains elusive, and it is generally accepted that the cause of IBD is multifactorial and includes genetic, host, and environmental factors (Flynn & Eisenstein, 2019).
Although UC and CD have their own characteristics (i.e., UC begins in the rectum and continues to expand into the colon, making it more likely to result in unformed and blood-like stools), CD shows patchy lesions that can diffuse anywhere in the gastrointestinal tract and are more likely to appear as watery stools and indeterminate symptoms, and so on; both pathologies often have inflammation as the main symptom (Seyedian et al., 2019). Thus, the main goal of IBD treatment is to diminish the inflammatory manifestations, enhance the patient’s health status, achieve complete clearance of the disease, or keep it stable to avoid invasive treatment (Seyedian et al., 2019).
Schisandrin A (SchA) (C24H32O6, Compound CID: 155256, Figure 1A; SchA) is one of the representative lignin components obtained from the fruit of Schisandra chinensis (Turcz.) Baill. It is widely used as a conventional Chinese medicine to prevent spontaneous sweating, chronic asthma, sleeplessness, and forgetfulness (Xu et al., 2019). The latest research findings show that SchA has multiple pharmacological effects, including anti-depressant, neuroprotective, anti-oxidative, anti-tumor, hepatoprotective, anti-diabetic, and musculoskeletal protective features (Chi et al., 2022; Fu et al., 2022; Ma et al., 2021b; Xu et al., 2019; Yu & Qian et al., 2021). Therefore, the pharmacological effects of SchA have gradually attracted clinicians’ attention.
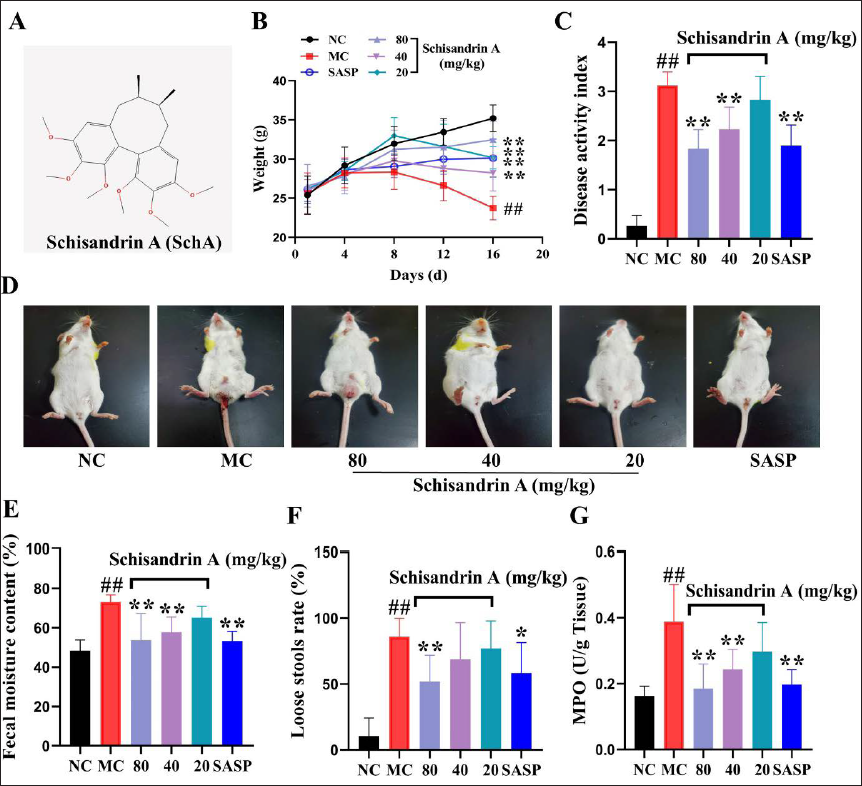
Although previous studies have confirmed that SchA has a noticeable therapeutic effect on IBD and can significantly inhibit the inflammatory response (Chi et al., 2022; Fu et al., 2022; Ma et al., 2021b; Xu et al., 2019; Yu et al., 2021), there is no clear conclusion on the mechanism and pathways involved in this anti-inflammatory effect.
In the present study, a mouse model of dextran sodium sulfate (DSS)-induced IBD and a H2O2-induced enterocyte murine (MODE-K) cell model were used to examine the inhibitory effect of SchA on the inflammatory response and to preliminarily explore whether this inhibition could be associated with Wnt/β-catenin signaling.
Materials and Methods
Materials
SchA was obtained from Nanjing DASF Bio-Technology (Lot No. W-005-151221; Nanjing, China). DSS was obtained from Sigma-Aldrich (Lot No. 11032-119; St. Louis, MO, USA). Mouse intestinal epithelium MODE-K cells were kindly provided by Shanghai Whelab Bioscience (Shanghai, China).
Animals
Totally 60 Kunming mice were provided by Liaoning Changsheng Biotechnology (Certificate No. SCXK [Liao] 2020-0001), weighing 17–23 g, including half male and half female. Animal housing was performed in the vivarium of the Liaoning University of Chinese Medicine, with adequate daily food and water supplies. All handling procedures in this experiment abided by the requirements of the Animal Experimentation Ethics Committee.
Establishment and Treatment of a Mouse Model of IBD
The animals were randomized into six groups (n = 10): normal control (NC), DSS-induced IBD model control (MC), sulfasalazine (SASP, 200 mg/kg), and SchA groups (80, 40, and 20 mg/kg).
MC group mice continuously drank 3% DSS solution for 16 days based on the method described previously with minor modifications (Wang et al., 2020; Wei et al., 2022; Zhou et al., 2022). Except for mice in the NC group that were provided pure water by gavage, the SASP and SchA groups were administered their respective drugs daily. Mice were weighed daily throughout the experiment. On the last day of the investigation, mice were placed in a closed environment, and fecal samples were taken for 3 h. Next, disease activity index (DAI), fecal water content, and loose stool rate were determined as described previously (Han et al., 2019; Ma et al., 2021a; Yang et al., 2022; Zhan et al., 2022). Mice were anesthetized with 20% chloral hydrate, and the myeloperoxidase (MPO) activity in extracted colon tissue was assessed based on the requirements of a specific kit.
Pathological and Immunohistochemical Examinations
The mouse colon was obtained after euthanasia, fixed with tissue fixative, and then made into 10 µm paraffin slides stained with hematoxylin and eosin (H&E) solution and photographed under an inverted microscope. Reference methods were used to score pathomorphology (Barrett et al., 2015). Additionally, routine immunohistochemical procedures were carried out to assess the expression of relevant proteins. After successive incubations with primary and secondary antibodies, color development and staining were performed with 3,3′-diaminobenzidine (DAB) solution, and the samples were imaged (Ren et al., 2022).
Western Blot
The protein samples of each group were adjusted to a consistent concentration with diluent and submitted to SDS electrophoresis. Then, routine membrane transfer, sealing, primary antibody (1:400) incubation, secondary antibody (1:800) incubation, and ECL development were carried out (Ma et al., 2021c; Yao et al., 2021).
Cell Culture and Cell Viability Assay
MODE-K cells underwent culture in Dulbecco’s modified Eagle’s medium (DMEM) containing 10% fetal bovine serum and 1% penicillin-streptomycin double antibody cocktail (P/S). Cells were inoculated in 5000 cells/well and incubated at constant temperature (37°C) with 5% CO2 and pre-protected with SchA for 24 h. Next, H2O2 was added at 1,000 µM for 5 h. For cell viability evaluation, 10 µL of CCK-8 solution was added to each well.
Scratch Wound Healing Assay
Cells inoculated into a 24-well plate underwent culture until confluency, and cell scratch wounds were made with a 100-µL pipette tip. After phosphate-buffered saline (PBS) washes, cells were administered with SchA at different concentrations. Finally, scratch wound healing was assessed after 0 and 24 h (Zeng et al., 2022).
Annexin V-FITC/PI Double Staining
MODE-K cells were administered H2O2 in 96-well plates, and a pre-configured staining solution was added. After incubation for 15 min in the dark, cells were imaged with a fluorescent microscope.
Immunofluorescence Staining
After model construction, the cells were fixed with 4% paraformaldehyde. After cell permeabilization, incubation was carried out with appropriate antibodies, followed by staining with DAPI solution. Finally, the treated cells were imaged using a fluorescent microscope (Wang et al., 2022).
ELISA
The cells were cultured and harvested, and ELISA was performed with specific kits as directed by the manufacturer, with absorbance measured at 450 nm.
Statistical Analysis
The data are mean ± standard deviation (SD). Multiple groups were compared by one-way analysis of variance (ANOVA). GraphPad Prism 9.0 (GraphPad Software, San Diego, CA, USA) was utilized for data analysis. P<0.05 indicated statistical significance.
Results
SchA Alleviates Common Clinical Symptoms in IBD Mice
At the end of modeling, IBD mice showed different degrees of diarrhea and blood in their stool, and their body weight gradually decreased. However, SchA alleviates DSS-induced weight loss and hematochezia in IBD mice (Figures 1B and D). Figure 1C shows that the DAI score markedly increased with damaged colonic mucosa; after treatment with SchA, the DAI score was significantly reduced (Figure 1C). In addition, SchA significantly decreased MPO content, fecal water content, and loose stool rate in IBD mice (Figure 1E–G).
SchA Attenuates Colonic Tissue and Immune Organ Damage in IBD Mice
As illustrated in Figures 2A and B, colonic tissue length was markedly shorter in DSS-induced IBD mice compared with non-DSS-treated mice, while colon length in IBD mice treated with SchA was recovered and was close to the normal value. The thymus and spleen indices in IBD mice were significantly reduced after SchA treatment (Figures 2C and D).
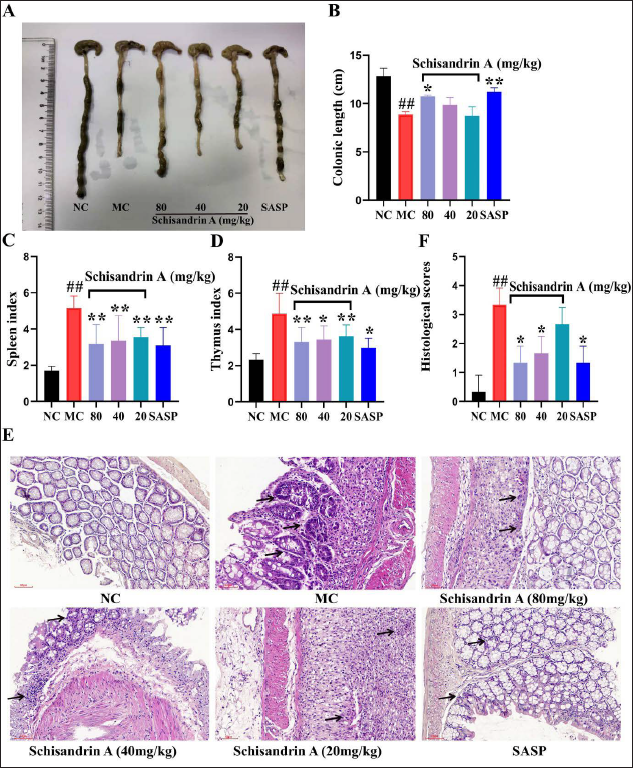
The intestinal mucosa of mice in the normal group was intact, without significant edema or inflammatory cell infiltration in any layers. In the model group, inflammatory cell infiltrates in the colon, ulcer foci in the intestinal mucosa, mucosal and submucosal necrosis, and histopathological score were remarkably elevated; in comparison with the model group, administration of 80 mg/kg SchA resulted in decreased inflammatory cell infiltration, ulcer foci, mucosal edema, and histopathological score (Figures 2E and F).
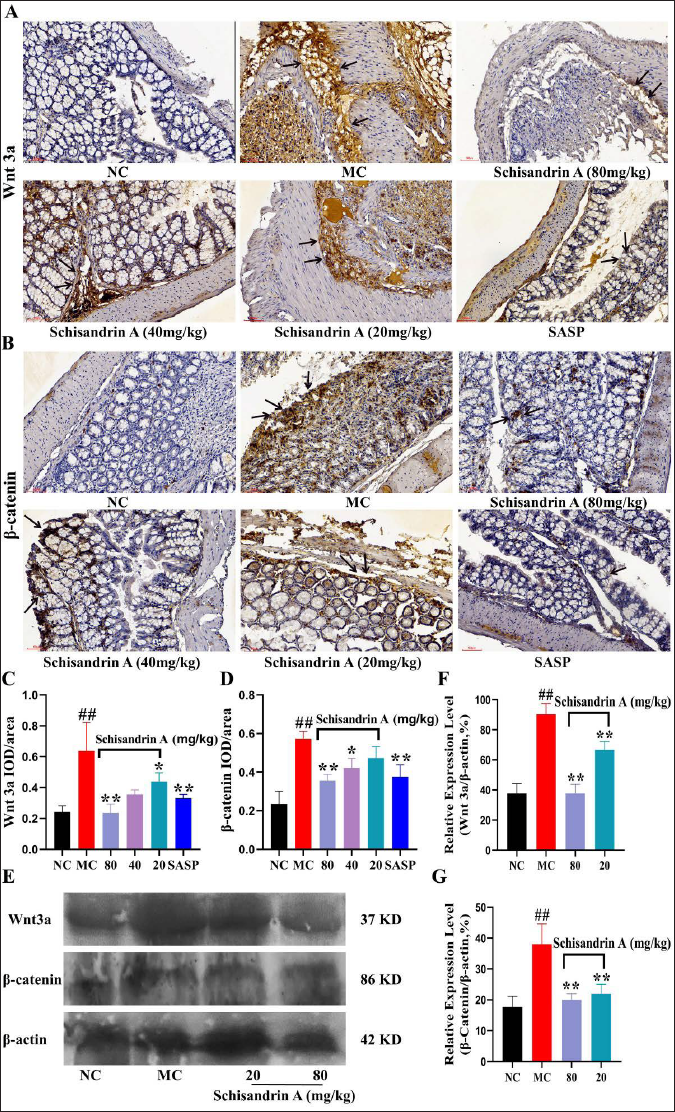
SchA Downregulates Wnt3a and β-Catenin in IBD Mice
As displayed in Figure 3, both immunohistochemistry and WB showed that SchA reduced the aberrantly elevated expression of Wnt3a and β-catenin in colon samples from DSS-induced IBD mice, bringing them to near normal values.
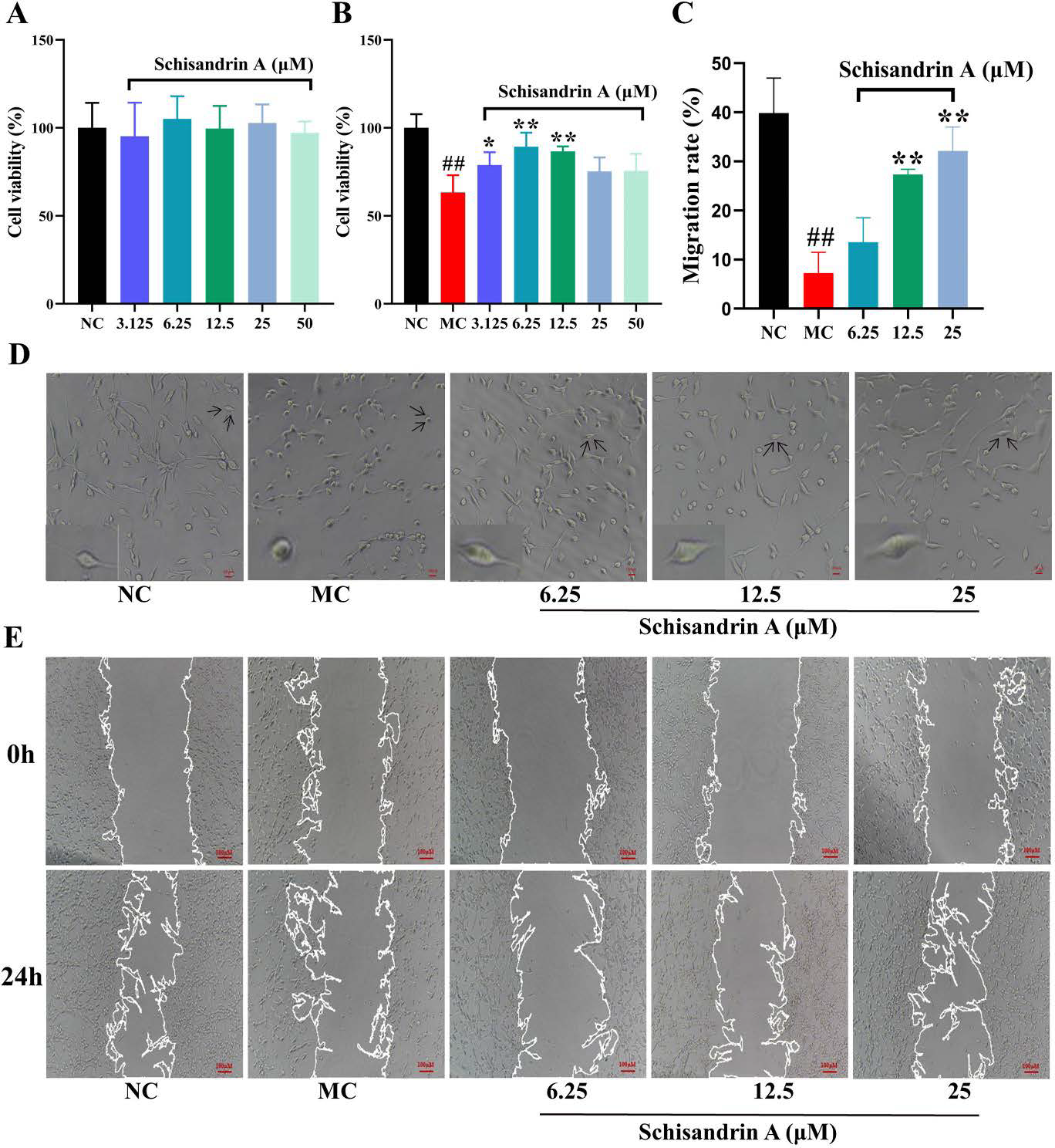
SchA Promotes the Proliferation and Migration of H2O2-treated MODE-K Cells
The results indicated that SchA (3.125–50 µM) exerted no significant effects on MODE-K cells, while it significantly increased the survival of H2O2-treated MODE-K cells (Figures 4A and B). After adding H2O2, MODE-K cells shrank to a round-shaped morphology, lost their original shape, or even lost adherence to the flask, leading to cell death. Besides, SchA restored the morphology of MODE-K cells to a certain extent (Figure 4D). Additionally, SchA accelerated healing in H2O2-treated MODE-K cells artificially scratched for 24 h (Figures 4C and E).
SchA Protects against H2O2-induced Apoptosis in MODE-K Cells
Caspase-3 is an apoptotic protein with typical properties. Immunofluorescence showed that caspase-3 expression was significantly upregulated in H2O2-induced MODE-K cells, and SchA inhibited the overexpression of caspase-3 (Figure 5A). Annexin V-FITC/PI double staining showed markedly elevated numbers of H2O2-induced apoptotic cells in the MC group, and SchA alleviated apoptosis in H2O2-induced MODE-K cells (Figure 5B).
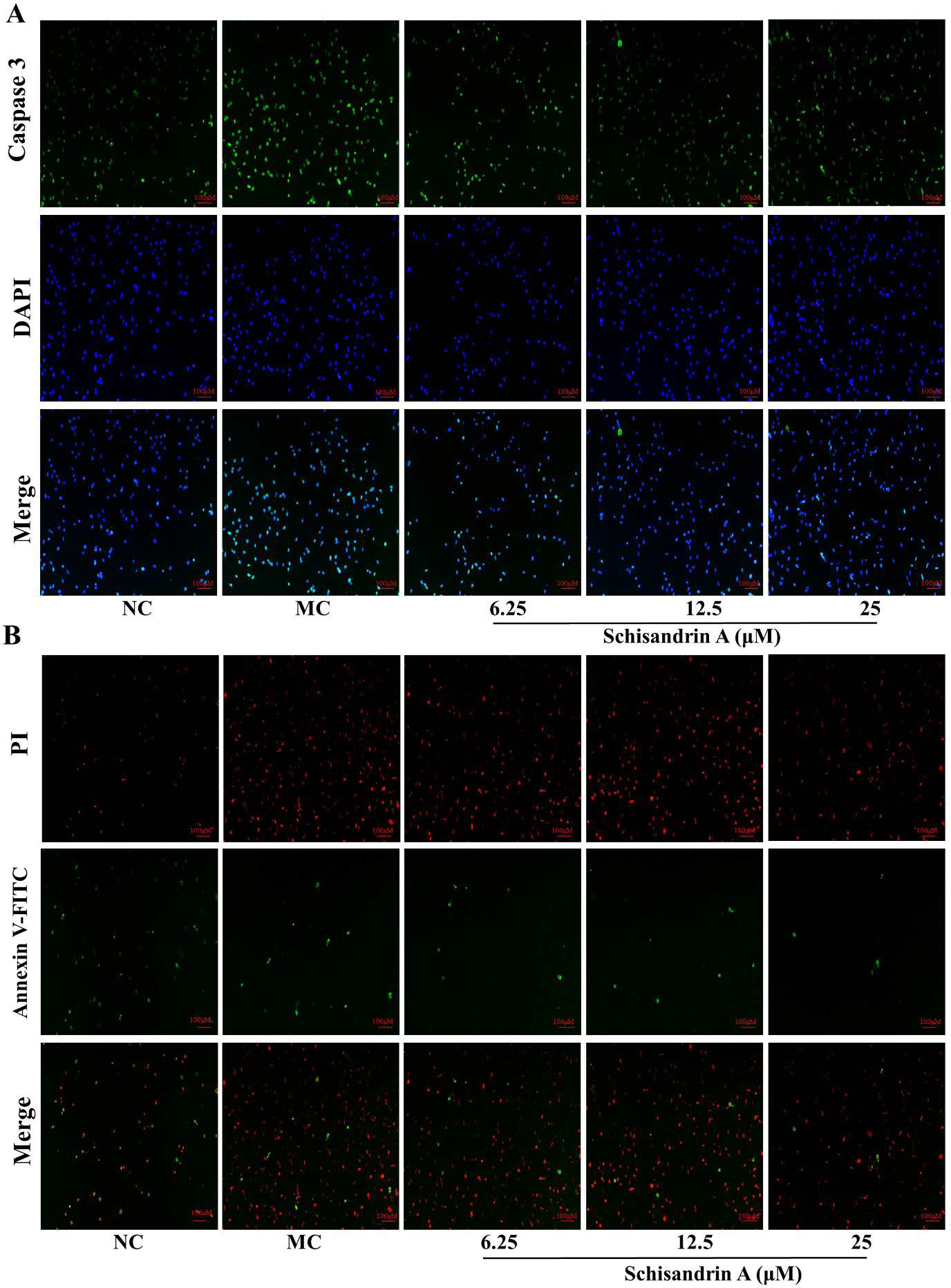
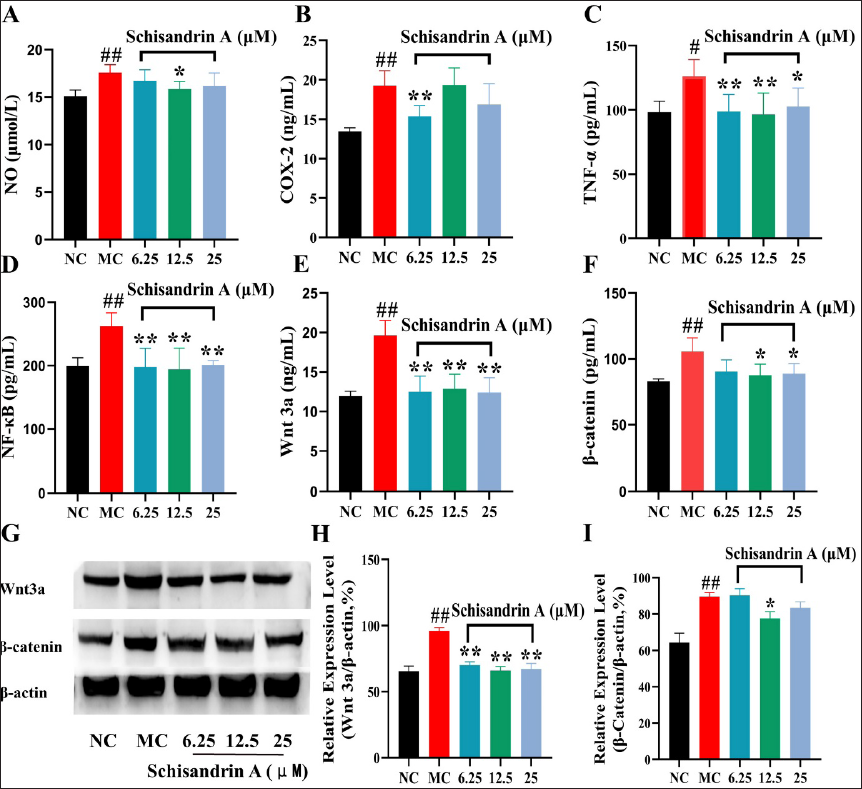
SchA Inhibits the Contents of Inflammatory Factors and Related Proteins in H2O2-induced MODE-K Cells
The results revealed that the contents of tumor necrosis factor-α (TNF-α), nuclear factor-κB (NF-κB), cyclooxygenase-2 (COX-2), and nitric oxide (NO), as well as Wnt3a and β-catenin amounts in H2O2-induced MODE-K cells, were significantly elevated. However, after administration of SchA, the contents of TNF-α, NF-κB, COX-2, and NO were markedly decreased (Figures 6A–D). Both WB and ELISA results revealed that SchA could downregulate Wnt3a and β-catenin (Figures 6E–I).
Discussion
A mouse model of DSS-induced IBD could mimic some critical disease features of IBD in humans. Therefore, researchers commonly use it to explore the intrinsic pathogenesis of IBD, underlying gut microbial changes, specific immune responses, and the developmental characteristics of IBD-associated colorectal cancer (CRC) (Chassaing et al., 2014; Perše & Cerar, 2012; Wirtz et al., 2017). We found that SchA significantly improved the general symptoms of IBD mice: prolonged colon transit time, reduced occult blood, decreased DAI score, and improved colon mucosal damage in IBD mice. In addition, MPO’s pro-inflammatory and pro-oxidant properties have often been considered an essential inflammatory indicator of the UC process. In this work, MPO content in the colon tissue of DSS-induced mice was much higher than that of normal mice. In contrast, MPO activity in mice administered SchA returned to near normal, suggesting SchA exerted a potential protective effect on IBD mice. Therefore, this work showed SchA had a definite alleviating impact on inflammatory responses in IBD mice induced by DSS.
Currently, only a few cell culture models have been developed for use in lieu of an in vivo model that can replicate the in vivo condition closely. Human colon adenocarcinoma CaCo-2 cells are broadly utilized as an in vitro model (Luongo et al., 2020; Wu et al., 2022). Moreover, the resemblance of multiple crucial pathophysiological features (e.g., remarkable susceptibility to pro-inflammatory factors) makes MODE-K cells a novel and reliable in vitro model of the gastrointestinal tract for investigating signaling pathways (Luongo et al., 2020; Wu et al., 2022). The current work revealed that after addition of SchA to the MODE-K cell culture system, the survival rate of inflammatory cells was increased, the wound healing distance between cell scratches was markedly elevated, and the contents of pro-inflammatory factors, such as TNF-α and COX-2, were significantly decreased.
Wnt/β-catenin signaling, including the typical Wnt/β-catenin-dependent and non-typical Wnt/β-catenin-independent pathways, regulates proliferation and differentiation in intestinal stem cells, with critical roles in the proliferation and differentiation of intestinal epithelial cells (Dong et al., 2019; Koch, 2017; Wang et al., 2020). Although the Wnt/β-catenin pathway is not the mainstream inflammatory pathway, increasing evidence confirms it should be considered a regulator of inflammation in the pathogenesis and inhibition of pro-inflammatory pathways, and activation of Wnt/β-catenin signaling is a well-known characteristic of chronic IBD in experimental animals (Dong et al., 2019; Koch, 2017; Wang et al., 2020).
It was confirmed that the function of Wnt signaling depends on β-catenin expression, which is controlled by the phosphorylation amounts of protein complexes and their degradation. Moreover, Wnt 3a and β-catenin are activated and highly expressed in the colonic mucosa of untreated UC rats, followed by an upregualted Wnt/β-catenin pathway associated with upstream and downstream effectors (Zhao et al., 2019). Clinical data suggested that Wnt/β-catenin pathway activity and protein amounts in non-diseased, colitis, and CRC colon tissue specimens might parallel the normal-to-colitis-to-CRC transition (Shenoy et al., 2012). The expression of β-catenin was highly correlated with UC progression. In colonic mucosa damaged, β-catenin accumulates and is translocated into the nuclear compartment, which transcriptionally activates cell-proliferation-associated genes (c-Myc and cyclin D1), exacerbating UC (Li et al., 2021). As demonstrated above, Wnt3a and β-catenin amounts in DSS-induced IBD mice were starkly increased and then significantly decreased after administration of SchA, corroborating previously reported findings. Moreover, Wnt3a and β-catenin amounts were found to be reduced in H2O2-induced MODE-K cells after administration of SchA.
Several signal transduction cascades and transcription factors, such as NF-κB, mitogen-activated protein kinase (MAPK), and Janus kinase/signal transducer and activator of transcription (JAK-STAT), are associated with pathological inflammatory processes. At the molecular level, Wnt and inflammatory pathways highly interact (Moparthi & Koch, 2019). For instance, the components of Wnt/β-catenin signaling regulate inflammatory and immune responses by interacting with NF-κB, modulating Wnt/β-catenin signaling (Guan et al., 2021; Ma & Hottiger, 2016). NO represents a highly reactive free radical in animals, and TNF-α is a pro-inflammatory cytokine. TNF-α could activate NF-κB signaling and induce Wnt/β-catenin through the NF-κB pathway (Bradford et al., 2017). In the present study, SchA markedly reduced the contents of NO and TNF-α and downregulated NF-κB, Wnt3a, and β-catenin in H2O2-induced MODE-K cells.
Conclusion
In summary, SchA inhibits the inflammatory response in DSS-induced IBD mice and H2O2-treated MODE-K cells, likely by inhibiting Wnt/β-catenin signaling. However, the specific mechanism by which SchA affects the inflammatory response still needs further investigation.
Footnotes
Summary
SchA is one of the representative lignin components extracted from the fructification of S. chinensis (Turcz.) Baill, has multiple pharmacological effects. The present study showed that SchA had a definite alleviating effect on the inflammatory responses of IBD mice induced by DSS. In the meantime, the results of the present study revealed that, after adding SchA to the MODE-K cell culture system, the survival rate of inflammatory cells increased, the percentage of wound healing distance between cell scratches was significantly elevated, and the contents of pro-inflammatory factors significantly decreased. Additionally, the results of the present study indicated that the expression levels of Wnt3a and β-catenin in DSS-induced IBD mice significantly increased and then significantly decreased after administration of SchA.
Moreover, it was found that the expression of Wnt3a and β-catenin was inhibited in H2O2-induced MODE-K cells after administration of SchA. In summary, SchA could inhibit inflammation in DSS-induced IBD mice and H2O2-induced MODE-K cells, and these effects might be realized by inhibiting the activation of Wnt/β-catenin pathway. Certainly, the specific mechanism of SchA in the inflammatory reaction still needs to be further investigated.
Abbreviations
SchA: Schisandrin A; IBD: inflammatory bowel disease; UC: ulcerative colitis; CD: Crohn’s disease; CRC: colorectal cancer; DAI: disease activity index; MPO: myeloperoxidase; DSS: dextran sodium sulfate; PBS: phosphate-buffered saline; MAPK: mitogen-activated protein kinase; JAK-STAT: Janus kinase/signal transducer and activator of transcription; HE: hematoxylin and eosin.
Acknowledgments
The authors appreciate Professor Xuetao Li for his comments and express their gratitude here.
Authors’ Contributions
Zhili Xu performed the conception and design of study. Hong Xu completed the acquisition of data. Xi Chen and Xinlei Li performed the analysis and interpretation of data. Zhili Xu completed the drafting of the manuscript. Deqiang Dou and Zhili Xu made the revising the manuscript critically for important intellectual content.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All animal experiments in this study were recognized and approved by the Animal Ethics Committee of College of Pharmacy of Liaoning University of Traditional Chinese Medicine.
Funding
This study was supported by the Liaoning Natural Science Foundation (Grant No. 2019-ZD-0441), for which the authors express their gratitude.
