Abstract
Hepatoblastoma (HB) is a malignant liver tumor that occurs during childhood. The histone deacetylase SIRT6 functions as a tumor suppressor in diverse cancers. Quercetin, as activators and antioxidants of sirtuins, exhibits remarkable anticancer activity in many tumors. However, whether quercetin ameliorates HB is still unclear. In our study, we found that SIRT6 was downregulated in HB tissues and cell lines. Overexpression of SIRT6 observably suppressed cell proliferation and invasion, promoted cell apoptosis. Mechanistically, SIRT6 suppressed frizzled 4 (FZD4) transcription by deacetylating histone H3K9. Upregulation of SIRT6 reduced the protein levels of FZD4 and H3K9ac. Additionally, quercetin treatment could enhance the expression of SIRT6, repress FZD4 level, cell viability and invasion, and promote apoptosis. Overexpression of FZD4 signally reversed quercetin-treated the promotion effect on cell apoptosis, and the inhibition effects on FZD4 expression, cell viability, invasion and Wnt/β-catenin pathway related proteins. In addition, LiCl, an agonist of Wnt/β-catenin pathway, could recover the inhibition effects of quercetin on Wnt/β-catenin pathway related proteins, cell viability and invasion, and promotion effect on cell apoptosis. In vivo mouse xenograft tumor growth assay revealed that quercetin markedly suppressed tumor growth. In conclusion, these results demonstrated that the molecular mechanism of quercetin suppressing HB cell proliferation and invasion, promoting apoptosis was to promote the deacetylation of SIRT6 on FZD4 and inhibit the activation of Wnt/β-catenin pathway.
Introduction
Hepatoblastoma (HB) originates from the abnormal development of pluripotent stem cells or embryonic liver progenitor cells.1,2 It is the most common malignant in children accounting for nearly 80% of pediatric liver cancers. 3 In recent years, the incidence rate of HB has increased year by year, but chemotherapy drugs are limited, and clinical prognosis of HB is poor. 4 Therefore, it is urgent to explore the molecular mechanism of HB to improve the early diagnosis, treatment, and prognosis of HB patients.
Quercetin is a polyphenolic flavonoid found in various type of fruits and vegetables. 5 Quercetin has many pharmacological effects, including anti-inflammatory, antioxidant, anti-atherogenic as well as antitumor activities.6,7 Indeed, quercetin is a unique compound due to its potential to combat cancer-related diseases in a multi-targeted manner. Quercetin was reported to modulate various pathways to cause cell cycle arrest and promote apoptosis. 8 Quercetin also could induce cell apoptosis by regulating proteins related to apoptosis, such as p53, caspase-9 and caspase-3. 9 Recent studies manifested that quercetin exerted its anticancer properties by modulating ROS, AKT, and NF-κB pathways in prostate cancer. 10 Additionally, quercetin suppressed the migration and invasion of squamous carcinoma by inhibition of Src/Stat3/S100A7 signaling. 11 However, the effect of quercetin on HB has rarely been reported, and its potential signaling pathway remains to be explored.
Sirtuins (SIRTs) are NAD+-dependent protein lysine deacetylases that could regulate many cellular processes including controlling aging, obesity, insulin resistance, inflammatory response and cancer. 12 For example, SIRT2 acted as a cardioprotective deacetylase to promote AMPK activation by deacetylating liver kinase B1 in pathological cardiac hypertrophy. 13 SIRT7 depletion downregulated the androgen receptor signal pathway to inhibit cell proliferation and androgen-induced autophagy in prostate cancer cells. 14 As one of NAD+-dependent deacetylases, SIRT6 could have less research than SIRT2 or SIRT7 before, however, SIRT6 has been studied more and more now because of its role in genome stability, sugar metabolism and oxidative stress. 12 Some studies also have found that SIRT6 played a tumor inhibition role to affect tumor growth. In cancer stem cells, overexpression of SIRT6 suppressed PI3K signaling at the transcriptional level and antagonized tumor sphere formation independent of its histone deacetylase activity. 15
In this study, we investigated the anticancer effects of quercetin in HB and its potential mechanism in order to provide novel insights and strategies for HB treatment.
Materials and methods
Tissue specimens
Human hepatoblastoma tissues and their matched adjacent normal tissues were obtained from 23 patients (17 males and 6 females, average age 4.6 years old) who received the surgical treatment from June 2016 to March 2019 at Xi’an Children’s Hospital. Tissues were frozen in liquid nitrogen immediately after surgical resection and stored at −80°C. All patients had not received preoperative radiotherapy and/or chemotherapy and were obtained with informed consent before surgery. The protocols were approved by the ethics committee of Xi’an Children’s Hospital (XAETYY-2018-18).
Cell lines and culture
Human hepatoblastoma cell lines HepG2, HuH-6 and HepT1 (the photographs of three cell lines were provided in the Supplementary Figure 1) and nonmalignant liver cell line (QSG-7701) were purchased from American Type Culture Collection (ATCC, Manassas, VA, USA). All cells were cultured in Dulbecco’s minimal essential medium (DMEM) (Gibco, Rockville, MD), containing with 10% fetal bovine serum (FBS, HyClone, Salt Lake City, UT) and 1% penicillin-streptomycin (Sigma, St. Louis, MO, USA) at 37°C and 5% CO2 in a humidified incubator (Biobase, Jinan, China).
Quantitative reverse transcription-polymer chain reaction (RT-qPCR) analysis
Total RNA from tissues and cells was extracted using Trizol reagent (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s instructions. TaqMan microRNA Reverse Transcription Kit (Applied Biosystems, Foster City, CA, USA) was used to synthesized cDNA in a 25 µL reaction volume containing 12.5 µL SYBR Premix Ex Taq Ⅱ, 1.0 µL of RT primer, and 1 µL cDNA sample, and 10.5 µL double distilled H2O. QRT-PCR was performed at 95°C for 3 min, followed by 39 circles at 95°C for 10 s and 60°C for 30 s. GAPDH was acted as an internal reference. Data were analyzed using the 2−ΔΔCT method. The following primers were used: SIRT6 (forward 5′-3′: CAAGTGTAAGACGCAGTACG; reverse5′-3′: GATGGTGTCCCTC AGCTCTC).
Cell transfection
Lipofectamine 3000 Transfection Reagent was used for cell transfection. Plasmids (Vector, pcDNA-SIRT6 and pcDNA-FZD4), siRNA negative control (Scramble) and small interference RNA targeting SIRT6 (si-SIRT6) were constructed and purchased from Sangon Biotech Co., Ltd (Sangon, Shanghai, China).
Cell viability and cell proliferation
For cell viability assay, 5 × 103 HepG2 and HuH-6 cells were seeded into 96-well plates and incubated with 0, 60 and 120 μM quercetin for 48 h. For HepG2 and HuH-6 cell proliferation assay, 1 × 103 cells were seeded in 12-well plates for 0, 24, 48, 72 h of co-incubation. After incubation, 10 µL of CCK-8 solution (Bio-Rad, Hercules, CA, USA) was added to each well for another 2 h at 37°C. The absorption values were detected with a microplate reader (Molecular Devices, Sunnyvale, CA, USA) at the absorbance of 450 nm.
Invasion assays
24-well Transwell plates with Matrigel™-coated membranes (8-µm pore size, BD Biosciences, Bedford, MA, USA) were used to analyzed cell invasion. Briefly, 1 × 105 HepG2 and HuH-6 cells with free-serum medium were seeded into the upper 24-well plates and 600 µL of DMEM medium, containing 20% FBS, was added to the lower chamber. After incubation for 24 h, noninvaded cells were removed with a cotton swab while invaded cells were fixed in 4% paraformaldehyde for 30 min and stained with 0.1% crystal violet for 15 min. The stained cells were quantified from five randomly selected fields and photographed using a light microscope (Olympus, Tokyo, Japan).
Cell apoptosis
HepG2 and HuH-6 cells were collected and stained with 5 µL of Annexin V-fluorescein isothiocyanate (FITC) and 5 µL of propidium iodide (PI) at room temperature in the dark for 20 min. After washing in buffer, cells were examined using flow cytometry (BD FACS Verse, San Jose, CA, USA).
HepG2 xenograft model
BALB/c nude mice (age: 4 weeks old; weight: 17–20 g) were obtained from Laboratory Animal Center of Xi’an Jiaotong University. All mice were housed in a room with an ambient temperature of 25°C and had free access to food and water. Mice were subcutaneously injected with HepG2 cells (1 × 107 cell per 500 μL) suspended in PBS (200 μL) and then randomly divided into two group: control group (n = 12) and quercetin group (n = 12). Mice in the quercetin group were intraperitoneally injected with a DMSO solution of quercetin (10 mg/kg) for 7 consecutive days. Mcie in control group were injected with the same volume of saline. Tumor volumes were measured every 7 days with a vernier caliper and calculated with the following formula: tumor volume (mm3) = 0.5 × length × width 2 . After 28 days, mice were sacrificed by cervical dislocation and the tumors were resected, weighed, and processed for Western blotting analysis. All protocols performed in studies involving animals were approved by the Animal Experiments and Care Committee of the Xi’an Children’s Hospital (XAETYY-03-2019-10) and experiments were conducted according to the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Western blotting
Cells and tumor tissues were lysed in RIPA lysis buffer (Beyotime, Shanghai, China) for 30 min 4°C to obtain total protein lysates for immunoblot analysis. Protein concentration was quantified by using an enhanced BCA kit (Beyotime, Shanghai, China) and proteins were separated on 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to polyvinylidene difluoride membranes (Millipore, Bedford, MA, USA). After blocking with 5% skim milk at room temperature for 1 h, the membranes were incubated with primary antibodies against SIRT6 (1:1000 dilution, ab191385, Abcam, Cambridge, UK), H3K9ac (1:500 dilution, ab4441, Abcam, Cambridge, UK), H3K56ac (AKT; 1:5000 dilution, ab71956, Abcam, Cambridge, UK), Wnt5a (1:4000 dilution, ab179824, Abcam, Cambridge, UK), β-catenin (1:1000 dilution, ab32572, Abcam, Cambridge, UK), Cyclin D1 (1:100 dilution, ab16663, Abcam, Cambridge, UK), c-Myc (1:500 dilution, ab32072, Abcam, Cambridge, UK). Then membranes were washed and incubated with a horseradish peroxidase-conjugated secondary antibody (1:500 dilution, A32733, Abcam, Cambridge, MA, USA) for 2 h at room temperature. The protein bands were detected using enhanced chemiluminescence (ECL) solution (Pierce biotechnology, Inc.) and were visualized using a chemiluminescence imaging system (Bioshine ChemiQ 4800 mini, China, Oxiang, Shanghai).
Immunoprecipitation assay
HepG2 cells were transfected si-SIRT6 or pcDNA-SIRT6 to analyze the acetylated protein by performing acetylation immunoprecipitation. Cells were lysed, and proteins were obtained and immunoprecipitated with primary antibodies for 4 h at 4°C. Then 50 µL of protein A/G agarose beads were added. After centrifuging, the antibodies were added and then incubated at 4°C for 2 h, then bound proteins were eluted with SDS-PAGE buffer. Finally, the protein concentration was detected by Western blotting.
Statistical analysis
All data were reported as the mean ± SEM. ANOVA analysis was used for the comparison among groups. Student’s t-test was used to compare differences between two groups. P < 0.05 was considered to be statistically significant.
Results
SIRT6 was downregulated in HB tissues and cell lines
In order to investigate the role of SIRT6 in HB, we firstly analyzed the level of SIRT6 in HB tissues and adjacent normal tissues. Compared with adjacent normal tissues, the expression of SIRT6 was downregulated in HB tissues (Figure 1(A), P < 0.05). Furthermore, the mRNA and protein levels of SIRT6 in HepG2, HuH-6 and HepT1 cells were also lower than these in QSG-7701 cells (Figure 1(B) and (C), P < 0.05). The full blots of three cell lines were supplemented in Supplementary Figures 2 and 3. All results showed that SIRT6 was associated with the occurrence of HB.

SIRT6 was downregulated in HB tissues and cell lines. (A) The level of SIRT6 in HB tissues and adjacent normal tissues was detected using qPCR (n = 23). (B) The level of SIRT6 in HB cell lines (HepG2, HuH-6 and HepT1) and nonmalignant liver cell line (QSG-7701). (C) Western blot analysis of SIRT6 expression in HB cell lines and QSG-7701. Data are represented as mean ± SEM, n = 3. **P < 0.01 compared with adjacent normal tissues or QSG-7701.
Overexpression of SIRT6 suppressed HB cell proliferation and invasion, promoted apoptosis
To examine the effect of SIRT6 on HB cells, HepG2 and HuH-6 cells were transfected with pcDNA-SIRT6. As shown in Figure 2(A), the level of SIRT6 was significantly increased after transfection with pcDNA-SIRT6 (P < 0.05). CCK-8 and Transwell assays showed that overexpression of SIRT6 could markedly suppress cell proliferation and invasion (Figure 2(B) to (E), P < 0.05). Moreover, overexpression of SIRT6 could promote HepG2 and HuH-6 cell apoptosis (Figure 2(F) and (G), P < 0.05). The percentage of cell apoptosis population in different quadrants was provided in supplementary tables. All these results suggested that overexpression of SIRT6 inhibited HB cell proliferation and invasion, promoted apoptosis.

Overexpression of SIRT6 suppressed HB cell proliferation and invasion, promoted apoptosis. (A) QPCR was used to detect the mRNA level of SIRT6 in HepG2 and HuH-6 cells after transfection with pcDNA-SIRT6. (B and C) CCK-8 assay was used to detect cell proliferation. (D and E) Transwell assay was used to detect the number of cell invasion. (F and G) Cell apoptosis was analyzed using Flow cytometer. Data are represented as mean ± SEM, n = 3. **P < 0.01 compared with Vector.
SIRT6 interacted with FZD4 and suppressed FZD4 by deacetylating H3K9
SIRT6 was overexpressed or silenced in HepG2 cells and immunoprecipitation was conducted to detect the relationship between SIRT6 and FZD4. The results showed that SIRT6 could directly bind to FZD4 (Figure 3(A) and (B), P < 0.05). Moreover, overexpression of SIRT6 could markedly deacetylate H3K9 and suppressed FZD4 expression (Figure 3(C) and (D), P < 0.05).

SIRT6 interacted with FZD4 and suppressed FZD4 by deacetylating H3K9. (A and B) The whole cell lysates were immunoprecipitated with anti-SIRT6 or anti-FZD4 antibodies, and western blot assay was used to detect the acetylation level of FZD4. (C and D) HepG2 cells were transfected with pcDNA-SIRT6, and the protein levels of SIRT6, FZD4 and H3K9ac were analyzed with western blotting. Data are represented as mean ± SEM, n = 3. **P < 0.01, *P < 0.05 compared with Scramble.
Quercetin inhibited HB cell viability and invasion, promoted apoptosis
To examine the effect of quercetin on HB cells, HepG2 and HuH-6 cells were treated with different concentrations of quercetin. As shown in Figure 4(A) and (B), quercetin increased the mRNA level of SIRT6, while reduced FZD4 mRNA expression (P < 0.05). The effects of 120 μM quercetin treatment on the promotion of SIRT6 and inhibition of FZD4 were better than these after 60 μM quercetin treatment. Compared with control group, 60 and 120 μM quercetin treatment could significantly reduce cell viability, suppressed cell invasion, and increased cell apoptosis (Figure 4(C) to (G), P < 0.05). Similarly, the effects of 120 μM quercetin treatment was superior to 60 μM quercetin treatment. These findings revealed that quercetin could suppress HB cell viability and invasion, and promote apoptosis.
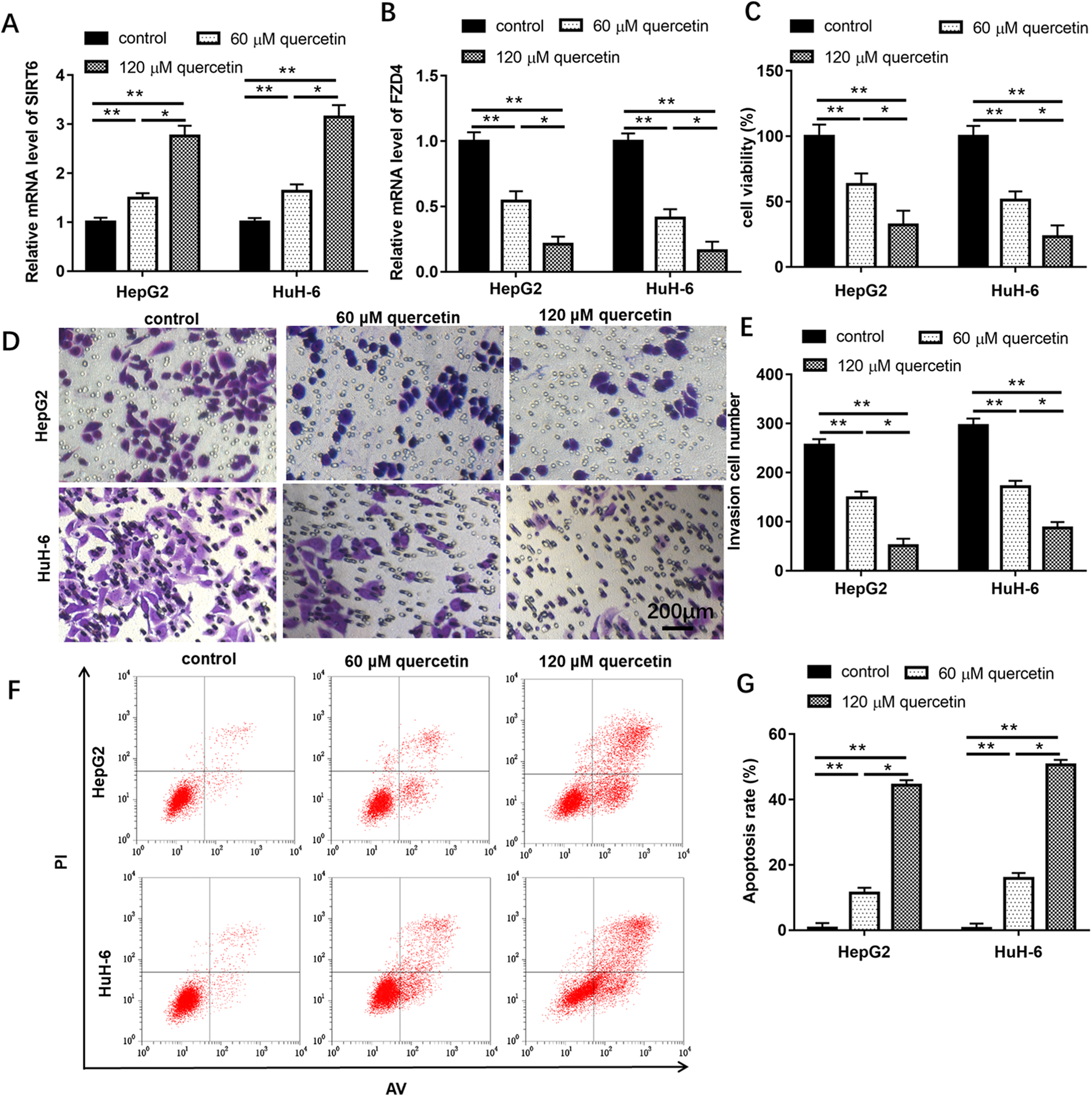
Quercetin inhibited HB cell viability and invasion, promoted apoptosis. HepG2 and HuH-6 cells were treated with 0, 60 and 120 μM quercetin. Relative expression of SIRT6 (A) and FZD4 (B) was measured by qPCR assay after treatment with 0, 60 and 120 μM quercetin for 48 h, respectively. (C) Cell viability were detected using CCK-8 assay. Transwell (D and E) and Flow cytometer (F and G) were used to detect cell invasion and cell apoptosis, respectively. The percentage of cell apoptosis population in different quadrants was provided in supplementary tables. Data are represented as mean ± SEM, n = 3. **P < 0.01, *P < 0.05 compared with control or 60 μM quercetin.
Downregulation of FZD4 reversed the inhibitory effect of quercetin on cell viability and invasion
To explore the relevance between quercetin and FZD4 in HB, HepG2 and HuH-6 cells were treated with 120 μM quercetin, or/and transfected with pcDNA-FZD4. In relation to control group, the level of FZD4 protein was decreased after treatment with 120 μM quercetin, while increased after treatment with 120 μM quercetin and transfection with pcDNA-FZD4 (Figure 5(A) and (B), P < 0.05). CCK-8 and Transwell assays revealed that 120 μM quercetin could repress cell proliferation and invasion (Figure 5(C) to (E), P < 0.05), while increase cell apoptosis (Figure 5(F) and (G), P < 0.05). However, in relation to the 120 μM quercetin and Scramble group, the abilities of cell proliferation and invasion were increased in 120 μM quercetin and pcDNA-FZD4 group, and cell apoptosis was inhibited. The percentage of cell apoptosis population in different quadrants was provided in supplementary tables. Moreover, 120 μM quercetin signally reduced the protein levels of Wnt5a, β-catenin, cyclin D1 and c-Myc, while overexpression of FZD4 could reverse all protein inhibition effects of 120 μM quercetin (Figure 5(H) and (I), P < 0.05). These findings showed that downregulation of FZD4 reversed the inhibition effects of quercetin on cell viability, invasion and Wnt/β-catenin related protein expression and the promotion effect of cell apoptosis.
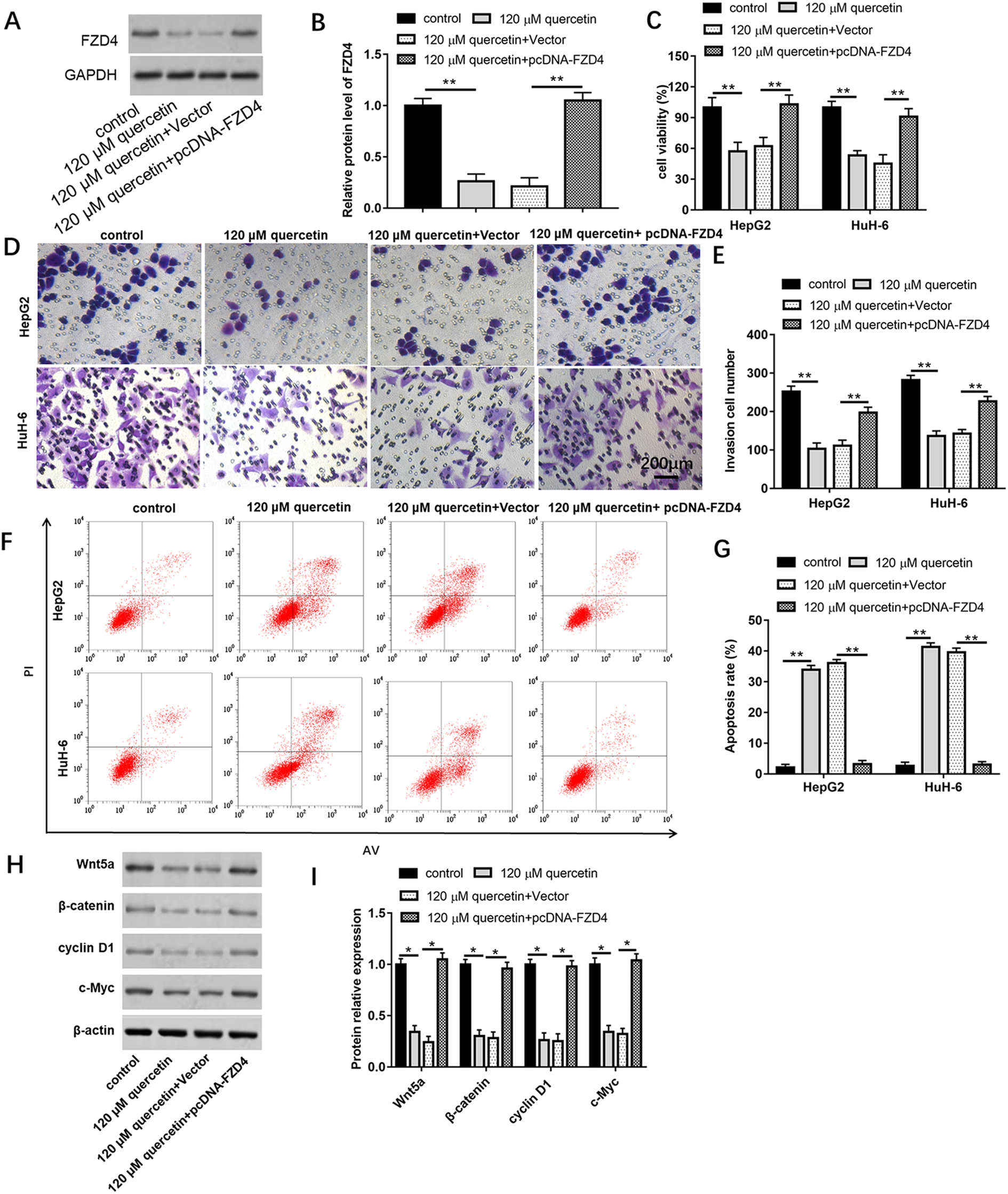
Downregulation of FZD4 reversed the inhibitory effect of quercetin on cell viability and invasion. (A and B) qPCR was used to detect the mRNA level of FZD4 in HepG2 and HuH-6 cells. (C) Cell viability was detected with CCK-8 assay. (D and E) Cell invasion was analyzed using Transwell assay. (F and G) Cell apoptosis was analyzed using Flow cytometer. (H and I) Western blot assay was conducted to detect SIRT6, H3K9ac, H3K56ac and H3 protein expression in different groups of HepG2 cell line. Data are represented as mean ± SEM, n = 3. **P < 0.01, *P < 0.05 compared with control, 120 μM quercetin +Scramble.
Quercetin inhibited the HB cell proliferation and invasion through Wnt/β-catenin pathway
The protein levels of Wnt5a, β-catenin, cyclin D1 and c-Myc in HepG2 and HuH-6 cells were reduced after treatment with 120 μM quercetin compared with control group, while increased after cells were treated with 120 μM quercetin and LiCl (Figure 6(A) to (C), P < 0.05). As shown in Figure 6(D) to (G), cells treated with 120 μM quercetin and 20 mM LiCl could counteract the inhibitory effect of 120 μM quercetin on cell viability and invasion, and the promotion effect on cell apoptosis. Moreover, overexpression of FZD4 could further increase the promotive effects of LiCl on Wnt/β-catenin pathway related protein levels, cell viability and invasion, and suppress cell apoptosis (P < 0.05). The percentage of cell apoptosis population in different quadrants was provided in supplementary tables. These results demonstrated that quercetin inhibited cell proliferation and invasion of HB through Wnt/β-catenin pathway.

Quercetin inhibited HB cell proliferation and invasion through Wnt/β-catenin pathway. HepG2 and HuH-6 cells were treated with 120 μM quercetin, or/and treated with 20 mM LiCl, or/and transfected with pcDNA-FZD4. (A) Western blotting was used to detect the Wnt/β-catenin pathway related proteins in HepG2 and HuH-6 cell line. (B and C) Quantization of western result according to densitometry analysis in HepG2 and HuH-6 cells, respectively. (D) Cell viability was detected using CCK-8 assay. (E) Cell invasion was analyzed using Transwell assay. (F and G) Cell apoptosis was analyzed using Flow cytometer. Data are represented as mean ± SEM, n = 3. #P < 0.01 compared with control, **P < 0.05, *P < 0.01 compared with 120 μM quercetin or 120 μM quercetin and Vector and LiCl.
Quercetin suppressed tumor growth in xenograft mouse model
HepG2 cells (1 × 107 cell) were subcutaneously injected into nude mice. Half of the mice received 10 mg/kg quercetin, and the remaining mice did not receive any treatment as a control group. Compared with the mice in the control group, the tumors in the quercetin treatment group grew slower, were smaller in size, and weighed less (Figure 7(A) to (C), P < 0.05). Western blotting results revealed that the level of SIRT6 protein in quercetin group was higher than that in control group, while FZD4 protein expression was lower than that in control group.

Quercetin suppressed tumor growth in xenograft mouse model. HepG2 cells (1 × 107 cell) were subcutaneously injected into nude mice. Mice in the quercetin group were intraperitoneally injected with quercetin (10 mg/kg, n = 12). Mcie in control group were did not receive any treatment. (A) Tumor volume in each group were calculated every 7 days. (B) Representative images of tumors from control group and quercetin group. (C) Tumor weight was determined after mice were sacrificed by cervical dislocation. (D) Protein levels of SIRT6 and FAD4 in tumor tissues were analyzed with western blotting. (E) Schematic diagram was generated to demonstrate the mechanism of quercetin on hepatoblastoma progression. Data are represented as mean ± SEM, n = 3. **P < 0.05, *P < 0.01 compared with control group.
Discussion
Several reports have suggested that quercetin has multiple pharmacological effects, including effective anticancer. 16 In breast cancer, quercetin enhanced apoptosis and the protein level of multidrug resistant protein 1. 17 Quercetin had the cytotoxic and genotoxic impacts on human bladder T24 cells. 18 However, there are few studies on the role of quercetin in HB. In our study, we found that quercetin had the same toxic and growth inhibition effect on HB cells. Compared with control group, 60 μM and 120 μM quercetin could markedly suppress HepG2 and HuH-6 cell viability and invasion, promoted cell apoptosis. Therefore, quercetin had excellent antitumor effect in HB.
SIRT6 is an important member of Sirtuin family and functions as a regulator of many cellular processes. 19 However, the role of SIRT6 in various cancers remains controversial, and it may show to play a role as either a tumor suppressor or a promoter, or both.20,21 In pancreatic cancer, SIRT6 promoted cytokine production and cell migration. 22 In renal cancer, SIRT6 was downregulated in renal cancer tissues and predicted to be a tumor suppressor. 23 SIRT6 protected the apoptosis and inflammation of podocytes by increasing autophagic flux. In our study, the level of SIRT6 was downregulated in HB tissues and cell lines. Overexpression of SIRT6 inhibited cell proliferation and invasion, while promoted cell apoptosis.
Sirtuins not only participate in many important biological processes, such as apoptosis, senescence and longevity, but also control the circadian clock and mitochondrial biogenesis. 24 Small molecules were reported to regulate sirtuins activity and have potential for the treatment of many human diseases, such as resveratrol and quercetin. For example, quercetin regulated rhinovirus-induced inflammation in chronic obstructive pulmonary disease airway epithelial cells by increasing SIRT1 expression. 25 As activators and antioxidants of SIRT, resveratrol and quercetin increased cell viability in Alzheimer’s disease. 26 Quercetin also could improve the decrease of maturation factor activity and the occurrence of cell apoptosis during postmenopausal aging, prevent the decrease of SIRT expression and histone methylation, and then alleviate the deterioration of oocyte quality. 27 Quercetin also was reported to activate SIRT6 via the isoform-specific binding site for pyrrolo[1,2-a] quinoxalines. 28 Hence, when cells were treated with quercetin, the mRNA and protein levels of SIRT6 was significantly increased. Studies had reported that quercetin-based compounds can activate Sirt6-dependent deacetylation through binding to the Sirt6-specific acyl binding channel. 28 It was reported that SIRT6 selectively deacetylated H3K9ac and H3K56ac to regulate downstream genes. 29 SIRT6 suppressed the transcription of Wnt target genes by interacting with transcription factor LEF1 and deacetylating histone 3 at lysine 56. 30 SIRT6 reduced Notch1 and Notch4 transcription by deacetylating histone H3K9. 31 Therefore, overexpression of SIRT6 could reduce the levels of H3K9ac and FZD4.
HB originated from dysregulation of pluripotent or stem cells or embryonic liver progenitor cells, and Wnt/β-catenin pathway played an important role in the activation and expansion of progenitor cells/stem cells during embryogenesis and liver regeneration, and abnormal activation of progenitor or stem cells activated the Wnt/β-catenin pathway. Therefore, the Wnt/β-catenin pathway was abnormally activated in HB.3,32 As a number of unconventional G protein-coupled receptor family, FZD4 was reported to active diverse intracellular signaling pathways, including Wnt/β-catenin signaling. 33 GATA6 antisense RNA 1 downregulated the expression of FZD4 to inactivate the Wnt/β-catenin signaling pathway to inhibit gastric cancer progression. 34 Hence, overexpression of FZD4 increased the protein levels of Wnt5a, β-catenin, cyclin D1 and c-Myc.
In conclusion, our findings provided strong evidence that quercetin inhibited the growth and invasion of HB and promoted apoptosis. The mechanism of quercetin is mediated by upregulating the expression of SIRT6 to reduce FZD4 expresion and inhibiting the Wnt/β-catenin pathway. The available results of quercetin indicated its great potential application on HB.
Supplemental material
Supplemental Material, sj-pdf-1-het-10.1177_09603271211030558 - Quercetin inhibited the proliferation and invasion of hepatoblastoma cells through facilitating SIRT6-medicated FZD4 silence
Supplemental Material, sj-pdf-1-het-10.1177_09603271211030558 for Quercetin inhibited the proliferation and invasion of hepatoblastoma cells through facilitating SIRT6-medicated FZD4 silence by T Liu, Z Li and F Tian in Human & Experimental Toxicology
Footnotes
Author contributions
Tao Liu performed the experiments and prepared the manuscript; Zhanhu Li prepared the figures and helped improving the manuscript; Fei Tian designed the study. All authors have reviewed and approved the final manuscript.
Data availability
The data supporting the conclusions of the study are available from the corresponding author upon request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
All patients had not received preoperative radiotherapy and/or chemotherapy and were obtained with informed consent before surgery. The protocols were approved by the Ethics Committee of Xi’an Children’s Hospital.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
