Abstract
Silicosis is an irreversible lung disease resulting from long-term inhalation of occupational dust containing silicon dioxide. However, the pathogenesis of silicosis has not been clearly understood yet. Accumulating evidence suggests that miR-29 may have a significant anti-fibrotic capacity, meanwhile it may relate to Wnt/β-catenin pathway. The purpose of this study was to discuss the role of miR-29 in the progression of silicosis. A lentiviral vector was constructed, named Lv-miR-29c, which was overexpressing miR-29c. In vivo, intratracheal treatment with Lv-miR-29c significantly increased expression of miR-29c, and reduced expression of β-catenin, matrix metalloproteinase (MMP)-2, and MMP-9 in the lung and levels of transforming growth factor-beta 1 (TGF-β1) and interleukin-6 (IL-6) in bronchoalveolar lavage fluid, and notably attenuated pulmonary fibrosis as evidenced by hydroxyproline content in silica-administered mice. These results indicated that miR-29c inhibited the development of silica-induced lung fibrosis. Thus, miR-29c may be a candidate target for silicosis treatment via its regulation of the Wnt/β-catenin pathway.
Introduction
Silicosis is an irreversible lung disease with pulmonary diffuse fibrosis as main manifestation resulting from long-term inhalation of occupational dust containing silicon dioxide (SiO2). 1 Meanwhile, it is one of the most common and severe occupational diseases with no effective treatment, being caused by excessive epithelial injury, the formation of fibroblastic foci, extracellular matrix (ECM) accumulation, and epithelial–mesenchymal transition (EMT). 2 However, the pathogenesis of silicosis has not been clearly understood yet. This highlights the urgent need to explore the mechanism of silicosis by modern molecular biology techniques for the purpose of finding the effective treatments.
MicroRNAs (miRNAs) are a class of noncoding small RNAs containing 21–25 nucleotides, which regulate gene expression at the posttranscriptional level by completely or incompletely binding to the 3′untranslated region of its target mRNA to degrade the target mRNA or inhibit its translation. 3 MiRNAs play an important role in many biological processes, including growth and development, cell proliferation, differentiation, and apoptosis. 4,5 Recent researches suggested that miRNAs are important mediators in the regulation of lung fibrosis. 6 –8 MiR-29 is a newly discovered miRNA family, which is closely related to fibrotic diseases through inhibiting the expression of a variety of ECM proteins and mediating many signaling pathways associated with fibrosis. 9 Some evidences showed that miR-29c was the significantly downregulated miRNA in lung fibrosis and exerted its anti-fibrotic capacity on pulmonary fibrosis by regulating fibroblasts apoptosis sensitivity, collagen synthesis, and EMT. 10,11 To date, however, none of the study has systematically investigated the role of miR-29c in the progression of silicosis or the molecular mechanisms by which miR-29c exerts its function. Dysregulated Wnt/β-catenin signaling is a common feature in lung fibrosis, and an inhibition of this signaling leads to an alleviation of pulmonary fibrosis. 12 Our previous study described that blockade of the Wnt/β-catenin signaling pathway prevented the development of silica-induced lung fibrosis. 13 Some evidences showed that miR-29c suppressed Wnt/β-catenin signaling. 14 Thus, we hypothesized miR-29c regulates the activation of Wnt/β-catenin signaling pathway caused by β-catenin in silicosis.
In the present study, a lentiviral vector was constructed, named Lv-miR-29c, which was overexpressing miR-29c. To investigate whether upregulation miR-29c could suppress Wnt/β-catenin signaling pathway and prevent silicosis development, a mouse silicosis model was used to estimate the effect of Lv-miR-29c inhibition on silicosis. It was observed that Lv-miR-29c increased miR-29c expression, meanwhile the expression of β-catenin, matrix metalloproteinase (MMP)-2, MMP-9, and secretion of transforming growth factor-beta 1 (TGF-β1) and interleukin-6 (IL-6) in vivo. In addition, intratracheal of Lv-miR-29c attenuated lung fibrosis in the mouse silicosis model.
Materials and methods
Construction of the lentivirus vectors of miR-29c
Total RNA was extracted from MLE-12 cells (American Type Culture Collection (ATCC), Roskilde, Denmark) using TRIzol reagent (Invitrogen, Carlsbad, California, USA). The pre-miR-29c gene was amplified and cloned into the digested pLenti-CMV-EGF vectors by TaiLeng Biotech Co., Ltd (Shanghai, China). The correct sequences and insertions were confirmed by DNA sequencing. A negative control (NC) sequence cited by our previous study. 13 Lentiviruses were produced in HEK-293T cells (ATCC) by transient transfection of three plasmids (TaiLeng Biotech Co., Ltd): the transfer vector pLKD-miR-29c or -NC, the packaging vector psPAX2, and the VSV-G expression plasmid pMD2G, followed by purification by ultracentrifugation, as previously described. 15 The lentivirus vectors were named as Lv-miR-29c and Lv-NC, respectively.
Experimental animals and design
Seventy-two healthy BALB/c mice, male and female half, were purchased from SLAC Laboratory Animal Co. Ltd (Shanghai, China). The mice were drinking and feeding freely for 1 week with the temperature control at 20–25°C and relative humidity of 45–55% in the clean animal laboratory. SiO2 was purchased from Sigma (St. Louis, Missouri, USA). The silica content of SiO2 was >99%, the dust particle size was 0.5–10 μm, and 80% of the particles were 1–5 μm. All experiments were carried out in accordance with the ethical guidelines of the Animal Experimentation Committee of Tianjin Centers for Disease Control and Prevention and were compliant with the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Mice were randomly divided into the following four experimental groups (=18 mice per group): (1) saline control group: instillation of 0.1-ml sterile physiological saline; (2) silica group: instillation of a suspension of 3-mg silica dust in a total volume of 0.1-ml sterile physiological saline; (3) silica + Lv-miR-29c group: instillation of a mixed suspension of 3 mg of silica dust and 5 × 107 transducing units (TUs) of Lv-miR-29c in a total volume of 0.1-ml of sterile physiological saline; (4) silica + Lv-NC group: instillation of a mixed suspension of 3 mg of silica dust and 5 × 107 TU Lv-NC in a total volume of 0.1-ml of sterile physiological saline. Animals were anesthetized with an intraperitoneal injection of 0.1-ml per mouse with 10% chloral hydrate. The skin of the neck was opened and blunt dissection exposed the trachea. Either physiological saline, silica in physiological saline, or silica with Lv-miR-29c or Lv-NC in physiological saline was instilled into the lungs using a seven-gauge needle inserted into the trachea through the epiglottis of the larynx. The site of surgery was sutured, and the mice were allowed to recover until they were killed. Six mice of each group were anesthetized with anesthetic ether, killed by decapitation at 7, 14, and 28 days post-instillation. The lungs were removed, and bronchoalveolar lavage fluid (BALF) was obtained by cannulating the trachea, injecting, and retrieving 1-ml aliquots of sterile physiological saline. Then, BALF was centrifuged at 1000 r/min for 1 min at 4°C and stored at −80°C.
qRT-PCR analysis
Quantitative real-time Polymerase Chain Reaction (PCR) (qRT-PCR) analysis was performed as previously described. 15 Total RNA, including miRNAs, was extracted from groups of lung tissue using the TRIzol reagent (Invitrogen) according to the manufacturer’s protocol. The RNA concentration and the ratio of A260/280 were determined by a UV spectrophotometer (Shanghai mapada Instruments Co., Ltd., China). Expression levels of miR-29c were quantified using a miScript PCR System (QIAGEN, Germany). Small nuclear RNA U6 was employed for internal normalization.
An amount of 5-μg total RNA of each sample was reverse transcribed in a volume of 0.1-ml using a reverse transcription kit (Takara, Japan) following the manufacturer’s instructions. qRT-PCR analysis was performed to determine the mRNA level of β-catenin, MMP-2, and MMP-9 in lung tissue with PrimeScript RT-PCR kit (Takara, Japan). Table 1 lists the sequences of the primers used.
Primers used for qRT-PCR.

MMP: matrix metalloproteinase; qRT: quantitative real-time.
Each PCR reaction mixture (20 μl) contained 10 μl of 2 × SYBR Green Master Mix (Takara, Japan), 1 μl of forward and reverse primers (5 µmol/μl), 1 µl of cDNA product, and water. The PCR reactions were run on ABI 7500 (Applied Biosystems, California, USA) using the following program: 95°C for 15 s and 40 cycles of 95°C for 5 s and 60°C for 30 s. Analysis was performed using the 7500 system software (Applied Biosystems). The qRT-PCR data were analyzed using 2−ΔΔCt method.
Western blot analysis
Total protein was extracted from lung homogenates with T-PER Protein Extraction Reagents (Thermo, Rockford, Illinois, USA), containing 0.1 mM of phenylmethyl sulfonyl fluoride (Sigma) according to manufacturer’s protocol. The 20–40-µg proteins were separated by 10% Sodium dodecylsulphate polyacrylamide gel electrophoresis (SDS-PAGE) under a constant voltage of 120 V for 2 h and then transferred to a nitrocellulose membrane at a constant voltage of 300 V for 1 h using a Bio-Rad Mini-PROTEAN Tetra system (California, USA). After blocking with phosphate buffer saline containing 0.05% Tween-20 (PBST) and 5% non-fat dry milk, the membranes were incubated at 4°C overnight with PBST and 5% milk containing primary antibodies at the following dilution ratios: anti-β-catenin antibody (1:1000, Cell Signaling Technology, USA), anti-MMP-9 antibody (1:500, Proteintech, Chicago, USA), anti-MMP-2 antibody (1:500, Proteintech), and anti-GAPDH antibody (1:2000, Cell Signaling Technology, Massachusetts, USA). The membranes were then washed three times with PBST and incubated with horseradish peroxidase (HRP)-conjugated anti-rabbit IgG and HRP-linked antibody (1:5000, Cell Signaling Technology) for 1 h at room temperature (RT), followed by washing with PBST. Proteins were visualized by enhanced chemiluminescence (Amersham Biosciences, GE Healthcare, UK).
ELISA assay for TGF-β1and IL-6
The ELISA plate was coated with 100 µl of capture antibody in coating buffer per well of ELISA kit (eBioscience, San Diego, USA) and incubated overnight at 4°C. The plate was washed with 250 µl of wash buffer. Then, the well was blocked with 200 µl of assay diluent, incubated 1 h at RT. A volume of 100-µl BALF or the different dilutions of standard (for standard curve) was added to each well, incubated 2 h at RT. The well was incubated 1 h at RT with 100-µl detection antibody, followed by 100 µl of avidin-HRP for 30 min and 100 µl of substrate solution for 15 min. Before plate reading at 450 nm, 50 µl of stop solution was added. The ELISA assay was performed in triplicate.
Determination of hydroxyproline content
The lung tissue samples were measured for hydroxyproline content using a hydroxyproline kit from Nanjing Jian Cheng Institute (China) following the manufacturer’s instructions. The results were calculated as milligrams of hydroxyproline per gram of wet lung tissue weight.
Pathological examination
Following gross inspection of each mouse, small pieces of lung tissue from the middle of the lobes, along with the hilar lymph nodes, were fixated with 4% paraformaldehyde, embedded in paraffin, and sectioned at 5 µm. The tissue sections were stained with hematoxylin and eosin (HE) and Masson stain for collagen fibers. Silicotic nodules were graded as follows: cellular nodules as stage I; fibrotic cellular nodules as stage II; cellular fibrotic nodules as stage III; and fibrotic nodules as stage IV.
Statistical analyses
SPSS 19.0 software was used to conduct statistical analyses. The differences between values were evaluated through one-way analysis of variance using pair-wise comparison with the Student–Newman–Keulstest. A value of p < 0.05 was considered statistically significant.
Results
Lv-miR-29c increased the expression of miR-29c in the lung
The 293T cells were infected with Lv-miR-29c or Lv-NC; after 72 h, cell image was taken by fluorescence microscopy. The expression of green fluorescent protein was observed in a large number of 293T cells (Figure 1(a)). The results of qRT-PCR analysis demonstrated that expression of miR-29c in the silica + Lv-miR-29c group was significantly higher than that in the saline control, silica, and silica + Lv-NC groups (p < 0.05) at the three time points post-instillation (Figure 1(b)).

293T cells were infected with Lv-NC, or Lv-miR-29c, phase contrast and GFP expression under a fluorescent microscope were taken after 72 h (a). Expression of miR-29c (b) in lung tissue of silica-instilled mice was detected by real-time PCR by using −ΔΔCt method. Data are expressed as mean ± SD (n = 6). *p < 0.05, as compared to the saline control group; Δ p < 0.05, as compared to the silica model group; and # p <0.05, as compared to the silica + Lv-NC group. SD: standard deviation; GFP: green fluorescent protein.
Lv-miR-29c inhibited the expression of β-catenin, MMP-2, and MMP-9 in the lung
To determine the expression of β-catenin, MMP-2, and MMP-9 in the lung tissue, qRT-PCR and western blot assays were used to examine the mRNA and protein. The results showed that the expression of β-catenin mRNA and protein in the silica + Lv-miR-29c group were significantly lower than those in the saline control (p < 0.05), silica (p < 0.05), and silica + Lv-NC groups (p < 0.05) at the three time points post-instillation (Figure 2(a), (d), and (e)). The expression of MMP-9 mRNA and protein in the silica + Lv-miR-29c group were significantly lower than those in the saline control (p < 0.05), silica (p < 0.05), and silica + Lv-NC groups (p < 0.05) at the three time points post-instillation (Figure 2(b), (d), and (f)).The expression of MMP-2 mRNA and protein in the silica + Lv-miR-29c group were significantly lower than those in the saline control (p < 0.05), silica (p < 0.05), and silica + Lv-NC groups (p < 0.05) at the three time points post-instillation (Figure 2(c), (d), and (g)).The results indicated that Lv-miR-29c significantly suppressed the expression of β-catenin, MMP-2, and MMP-9 mRNA in lung tissue of silica-instilled mice.
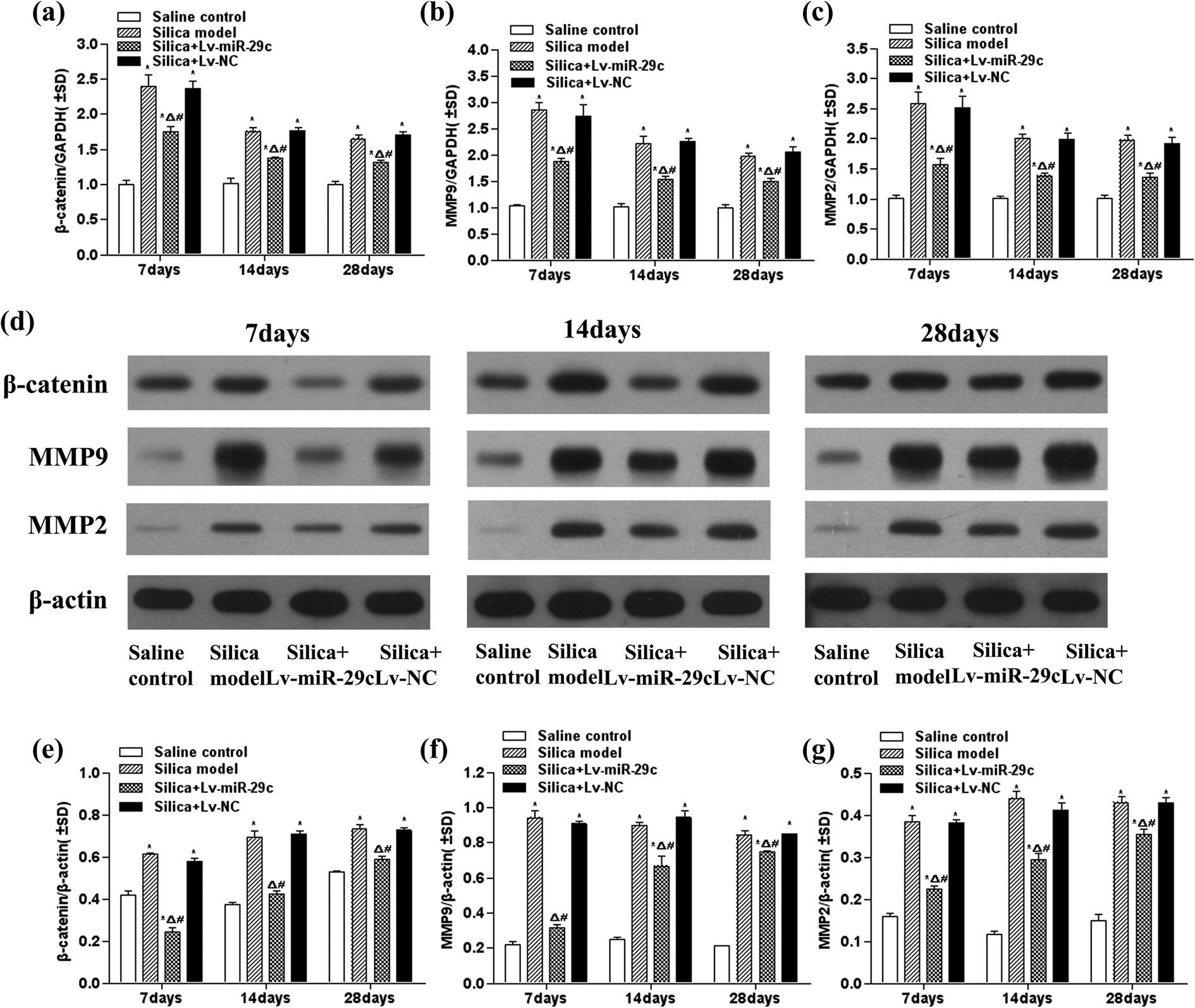
Expression of β-catenin (a), MMP-9 (b), and MMP-2 (c) mRNA in lung tissue of silica-instilled mice was detected by real-time PCR using −ΔΔCt method. Expression of β-catenin in the lung tissue of silica-instilled mice was assayed by western blot ((d) and (e)). Expression of MMP-9 in the lung tissue of silica-instilled mice was assayed by western blot ((d) and (f)). Expression of MMP-2 in the lung tissue of silica-instilled mice was assayed by western blot ((d) and (g)). Data are expressed as mean ± SD (n = 3). *p < 0.05, as compared to the saline control group; Δ p < 0.05, as compared to the silica model group; and # p < 0.05, as compared to the silica + Lv-NC group. MMP: matrix metalloproteinase; SD: standard deviation.
Lv-miR-29c reduced TGF-β1 and IL-6 content in BALF
TGF-β1 plays a critical role in the progression of silicosis. IL-6 maybe related to the form of fibrous connective tissue and the proliferation of smooth muscle cells. In this study, ELISA assay was used to measure the contents of TGF-β1 and IL-6 in BALF. The results demonstrated that contents of TGF-β1 and IL-6 in BALF of the silica group and the silica + Lv-NC group were significantly higher than those of the saline control group (p < 0.05) at 7 days post-instillation. TGF-β1 and IL-6 contents of the silica + Lv-miR-29c group were significantly lower than those in the silica group (p < 0.05) and the silica + Lv-NC group (p < 0.05) and significantly higher than those of the saline control group (p < 0.05) at 7 days post-instillation (Figure 3).
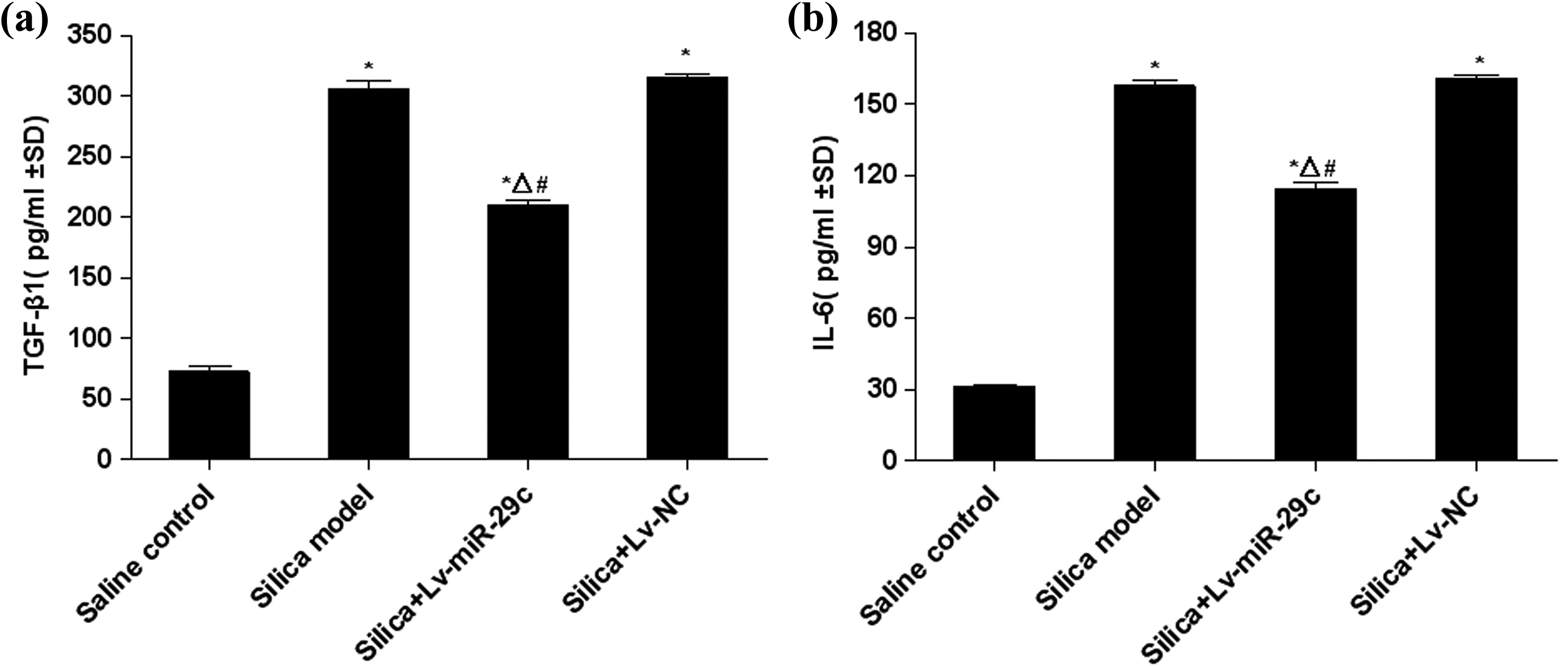
TGF-β1 and IL-6 level in the BALF of mice instilled with silica. The expression of TGF-β1 in the BALF was assayed by ELISA. (a) The expression of IL-6 in the BALF was assayed by ELISA. (b) Data are expressed as mean ± SD (n = 3). *p < 0.05, as compared to the saline control group; Δ p < 0.05, as compared to the silica model group; and # p < 0.05, as compared to the silica + Lv-NC group. BALF: bronchoalveolar lavage fluid; SD: standard deviation; TGF-β: transforming growth factor-beta; IL-6: Interleukin-6.
Lv-miR-29c reduced hydroxyproline content in the lung
Hydroxyproline content is an important indicator of determining the degree of pulmonary fibrosis. In this study, hydroxyproline content was measured using a hydroxyproline kit (Nanjing Jian Cheng Institute, Nanjing, China). The results showed that there was no significant difference in hydroxyproline content among the four groups at 7 days post-instillation. However, at 14 and 28 days post-instillation, the hydroxyproline content of the silica group was obviously higher than that of the saline control group (p < 0.05). The hydroxyproline content of the silica + Lv-miR-29c group was significantly lower than that in the silica group (p < 0.05) and significantly higher than that of the saline control group (p < 0.05) at 14 and 28 days post-instillation (Figure 4).

Hydroxyproline content in the lung of mice instilled with silica. Data are expressed as mean ± SD (n = 6). *p < 0.05, as compared to the saline control group; Δ p < 0.05, as compared to the silica group; and # p < 0.05, as compared to the silica + Lv-NC group. SD: standard deviation.
Lv-miR-29c inhibited silica-induced lung fibrosis
The lung tissues of mice after HE staining and Masson staining, respectively, were observed by light microscope to monitor pathological changes. No obvious abnormalities were observed in the lung tissues of mice that received normal saline. In the silica and silica + Lv-NC groups, the histologic analysis showed a large infiltration of inflammatory cells and alveolar septal thickening in the lung, and occasionally a small amount of cellular nodules (stage I; Figure 5, Table 2) and tiny collagen fibers (Figure 6) were observed at 7 days post-instillation. Primary cellular nodules (stages I and I+) and tiny collagen fibers were observed in the silica and silica + Lv-NC groups at 14 days post-instillation, and primary cellular nodules and fibrotic cellular nodules (stages I+ and II) were observed in the silica group and silica + Lv-NC group at 28 days post-instillation (Figure 5). Masson staining revealed that the collagen deposition (blue areas) was significantly higher in the silica group compared with the saline group. Compared to the silica group, the numbers of nodules were fewer, and the size was smaller in the lung of mice in the silica + Lv-miR-29c group. Masson stain was weakly positive for collagen fibers (Figure 6).

HE staining for histopathologic changes inmicelungs (×400). HE: hematoxylin and eosin.
Silicotic nodule grade of the mice lungs in each group.


Masson staining for histopathologic changes in mice lungs (×400).
Discussion
Pulmonary fibrosis is the most important pathological change in silicosis. 16 An increasing amount of evidences showed that miRNAs play an important role in lung fibrosis. 17,18 However, only a few studies have demonstrated the efficacy of miRNAs in silicosis. According to the published data, miR-29 is a major regulator of genes associated with lung fibrosis, and the downregulation of miR-29 expression could be associated with the pathophysiology of silicosis. 1 Thus, a lentiviral vector was constructed, named Lv-miR-29c, to investigate the molecular mechanism of miR-29c in the development of silicosis using a mouse silicosis model.
Wnt/β-catenin signaling pathway is involved in the development of silicosis, and β-catenin is a key regulator in this procession. 13 The recent literature proved that miR-29c directly targeted the expression of the following β-catenin upstream regulators, that is, frequently rearranged in advanced T-cell lymphomas 2, low-density lipoprotein receptor-related protein 6, Frizzled-4, and Frizzled-5. 19 Thus, β-catenin may be the indirect target gene of miR-29c. An experimental mouse model of silicosis was induced by an intratracheal administration of silica dust in this study. The results showed that Lv-miR-29c effectively increased the expression of miR-29c in the lung tissue of silica-administrated mice accompanied by β-catenin downregulation.
The MMPs are important downstream target genes of the Wnt/β-catenin signaling pathway. 20,21 MMPs modulate a range of biological processes, especially process-related immunity and tissue repair or remodeling. Recent studies showed that MMPs have emerged critical to the pathogenesis of lung fibrosis by modulating the synthesis and degradation of ECM. 21 Our results showed that Lv-miR-29c effectively suppressed the elevated expression of MMP-2 and MMP-9 in the lung of silica-administrated mice.TGF-β is the most important fibrotic factor, which is mainly secreted by pulmonary epithelial cells and macrophages in lung tissue. 22,23 Following silica-induced lung injury, alveolar macrophages were stimulated and secreted large quantities of biologically active TGF-β1. Meanwhile, IL-6 is a multifunctional cytokine with various biological activities, such as increased collagen deposition, inhibition of ECM degradation, and stimulation of fibroblast proliferation, which plays an important role in the formation of fibrous connective tissue and the proliferation process of smooth muscle cell. 24 In accordance with results in vivo, attenuation of TGF-β1 and IL-6 secretion in BALF by Lv-miR-29c was observed in mice of silicosis. The experimental model of silicosis was proved to result in varying degrees of fibrotic silicosis. 25 Lung fibrosis was alleviated by Lv-miR-29c as evidenced by HE and Masson staining, hydroxyproline content accumulation in the lung. Therefore, Lv-miR-29c could prevent the development of silica-induced lung fibrosis.
To summarize, overexpression of miR-29c following treatment with Lv-miR-29c in silica-induced lung fibrosis may lead to the decreased expression of β-catenin, MMP-9, and MMP-2, and levels of TGF-β1 and IL-6 in BALF, and notably attenuated pulmonary fibrosis in silica-administered mice. Therefore, miR-29c inhibited the development of silica-induced lung fibrosis via its regulation of the Wnt/β-catenin signaling pathway. Our study may provide a new insight for understanding the mechanism of silicosis. The miR-29c may represent a novel target for prevention and treatment of silicosis.
Footnotes
Author Contributions
XW carried out the experiments, participated in the experimental design and in the interpretation of data, and drafted the manuscript. KX conceived of the study and participated in the molecular biology experiments. JL and XY participated in the animal instillation and in the histological experiments. QZ and FW initiated the project, participated in the design of the study and in the interpretation of data, and revised the manuscript critically.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Foundation of China (81102106), the China Postdoctoral Science Foundation (2014M560189), and Tianjin Municipal Natural Science Foundation (15JCQNJC10500).
