Abstract
Background
Eucommia ulmoides Oliv. is a traditional Chinese medicine, and its flowers, leaves, and seeds are widely used as food. Currently, E. ulmoides seed oil is capable of improving glucose and lipid metabolism in type 2 diabetic mice, but E. ulmoides seed meal is not fully utilized, and the anti-photoaging effect of it is still unclear.
Objectives
We examined the anti-photoaging effect of the ethanol extracts of E. ulmoides seed meal (EUSM) in UVB-stimulated HaCaT cells and mice.
Materials and Methods
EUSM was analyzed using Q-Orbitrap-based high-resolution LC-MS/MS. In vivo and in vitro experiments were conducted using normal mice and HaCaT cells, respectively. Hematoxylin and eosin (H&E) staining and Masson’s staining were used to evaluate epidermal thickness and degree of skin fibrosis. The production of hyaluronic acid (HA) and total protein using ELISA kits. The expression of levels for collagen type I α 1 (COL1A1), hyaluronidase-1 (HYAL-1), MMP-3, and MMP-9 were assessed using semi-quantitative real-time polymerase chain reaction (qRT-PCR).
Results
EUSM contains all-trans-retinoic acid, medicagenic acid, and other chemical constituents. EUSM increased the level of total collagen by reducing matrix metalloproteinase expression, and it enhanced the content of HA. In addition, EUSM reduced the production of wrinkles in UVB-induced mouse skin and showed that EUSM decreased the level of malondialdehyde (MDA) and increased the contents of superoxide dismutase (SOD) and catalase (CAT) in the mouse skin tissue.
Conclusion
EUSM has therapeutic potential for UVB-induced skin photoaging and provided a rationale for the application of EUSM in the cosmetic field.
Introduction
Photoaging is a series of complex skin changes caused by long-term UV exposure on the surface of the skin (Geng et al., 2021). The most visible results of photoaging are primarily on the skin, resulting in changes in skin structure and appearance, such as deep wrinkles, sagging, telangiectasia, and skin pigmentation (Battie et al., 2014).
Light with wavelengths ranging from 100 to 400 nm is called ultraviolet light, which is divided into three types according to different wavelength ranges: UVA: 320–400 nm; UVB: 280–320 nm, and UVC: 200–280 nm. In particular, UVB is the primary factor in a number of photoaging processes, including skin dryness and collagen degradation (Mohania et al., 2017). Reactive oxygen species (ROS) produced by exposure to UVB irradiation could disrupt the homeostasis of oxidative stress by reducing antioxidant enzyme activity (Tanaka et al., 2019). Then, excessive free radicals cause the expression of matrix metalloproteinases (MMPs), and these processes may lead to the breakdown and structural damage of collagen, an important gene closely related to the strength and elasticity of the skin, which is interrelated with skin photoaging (Tang et al., 2013). The skin extracellular matrix (ECM) is made up primarily of hyaluronic acid (HA), which is essential for tissue hydration (Papakonstantinou et al., 2012). Also, the loss of skin moisture leads to dry skin, which is also one of the manifestations of photoaging, and adequate moisture can maintain skin health.
In the investigation of photoaging, histological and biochemical alterations in mice skin and human skin are comparable (Jia et al., 2023). The histological changes of mice skin thickening, epidermal hyperkeratosis, and hyperplasia in the UVB-irradiated mice photoaging model are strikingly similar to the histological changes of human skin that has been exposed to sunlight. At the same time, the mice skin exhibits numerous wrinkles and decreased collagen, which also mimic the signs of photoaging in human skin (Kim et al., 2022).
Eucommia ulmoides Oliv., a single species of the Eucommia genus belonging to the Eucommiaceae family, is a precious medicinal material in China (Wang et al., 2019). E. ulmoides bark, a traditional medication, has antihypertensive, hypoglycemic, and anti-fatigue effects among its properties (Kwan et al., 2003). In addition, the aqueous extracts of E. ulmoides leaves and E. ulmoides flowers, namely, E. ulmoides tea and E. ulmoides flower tea, are popular folk drinks in China and Japan (Ding et al., 2014; Du et al., 2017). As functional foods, some studies have shown that E. ulmoides leaves improved hyperglycemia in streptozotocin-induced diabetic rats by preserving insulin-positive beta cells (Lee et al., 2005). Also, E. ulmoides seeds can be pressed into E. ulmoides seed oil, which is one of the common edible oils in China, and E. ulmoides seed oil is capable of improving glucose and lipid metabolism and intestinal dysbiosis in type 2 diabetic mice (Wu, et al., 2022). However, pressing the remaining E. ulmoides seed meal is currently underutilized. Therefore, in this study, based on the existing theories (Ho et al., 2005), we investigated a number of photoaging-related factors to estimate the effects of E. ulmoides seed meal ethanol extract on UVB-induced HaCaT cells and skin impaired hydration, wrinkle formation, and oxidative stress status in mouse skin tissue.
Materials and Methods
Chemicals and Reagents
All chemicals were obtained from Sigma Chemical Co. (St. Louis, MO, USA). The following kits were used in this study: kits for superoxide dismutase (SOD, A001-3-2), malondialdehyde (MDA, A003-1-2), and catalase (CAT, A007-1-1) were obtained from Nanjing Jiancheng Bioengineering Institute (Nanjing, China). ELISA kits for HA (ml1268526-J) and total collagen protein (ml179521-J) were purchased from Shanghai Enzyme Link Biotechnology Co., Ltd. (Shanghai, China). Culture reagents were purchased from Gibco BRL Co. (Grand Island, NY, USA). Sclareol (A0381) and ursolic acid (A0139) were purchased from Chengdu Must Biological Technology Co., Ltd. (Chengdu, China). Medicagenic acid (B28185) and palmitoleic acid (B27196) were purchased from Shanghai Yuanye Bio-Technology Co., Ltd. (Shanghai, China).
Preparation of the Plant Material
E. ulmoides seed meal was purchased from Shandong Beilong Eucommia Bioengineering Co., Ltd. (Weifang, China). E. ulmoides seed meal (90 g) was soaked in 80% ethanol (1800 mL) for 24 h. Then, the precipitate was removed using a Brucella funnel, and the filtered extract was then concentrated in a vacuum drying oven (DZF-6053; Shanghai Yiheng Technology Instrument Co., Ltd., China) to obtain the ethanol extract of E. ulmoides seed meal (14.24 g), the yield being 15.82%, and finally stored at –20°C. For the cell experiments, EUSM was dissolved in dimethyl sulfoxide; for the animal experiments, EUSM was dissolved in distilled water and added to a surfactant (Tween 80) to make a stable emulsion.
Characterization of EUSM
EUSM was analyzed using Q-Orbitrap-based high-resolution LC-MS/MS, and LC-MS/MS analyses were performed using a UHPLC system (Vanquish, Thermo Fisher Scientific, MA, USA). Chromatographic analyses of EUSM were performed using a UPLC HSS T3 column (2.1 mm × 100 mm, 1.8 µm; Waters®, Milford, MA, USA), maintained at 35°C. 5 mmol/L each of ammonium acetate and acetic acid in water (A) and acetonitrile (B) made up the mobile phase. Following are the steps taken to implement the gradient program: 0–0.7 min, 1% B, 0.35 mL/min; 0.7–9.5 min, 1%–99% B, 0.35 mL/min; 9.5–11.8 min, 99% B, 0.35–0.5 mL/min; 11.8–12.0 min, 99%–1% B, 0.5 mL/min; 12.0–14.6 min, 1% B, 0.5 mL/min; and 14.6–14.8 min, 1% B, 0.5–0.35 mL/min. The sample injection volume was 2 µL. An electrospray ionization source (ESI) was used to execute mass spectral studies. ESI conditions were as follows: capillary temperature of 350°C; 30 Arb for the sheath gas flow rate, and 25 Arb for the aux gas flow rate. The data gathered by high-resolution liquid and mass spectrometry were completed by CD2.1 (Thermo Fisher Scientific, MA, USA). After sorting, we searched the mzCloud, mzVault, and ChemSpider databases and compared the results.
HPLC Analysis of Medicagenic Acid and Ursolic Acid in EUSM
HPLC system (Agilent 1220 Infinity LC System, Agilent Technologies, Inc., State of California, USA) with BDS Hypersil C-18 column (4.6 × 250 mm; 5 µm, Thermo Fisher Scientific, MA, USA) were used to determine EUSM and its active ingredients. EUSM (1 mg/mL) is dissolved in methanol, ultrasonically dissolved, and passed through a 0.45 µm filter membrane to obtain a sample. Acetonitrile (A) and water with 0.1% phosphoric acid (B) make up the mobile phase. The steps for gradient elution are as follows: 0 min, 50% B; 1 min, 50% B; 25 min, 5% B; and 30 min, 50% B. The flow rate is 0.8 mL/min, the detection wavelength is 211 nm, and the sample injection volume is 20 µL. Methanol was used to dissolve the standard stock solutions of medicagenic acid (50 µg/mL) and ursolic acid (50 µg/mL). By measuring the peak area, determine the content of medicagenic acids and ursolic acid in EUSM.
Experimental Animals and Treatment
A total of 30 male Kunming mice (18–22 g) were purchased from the Qingdao Institute for Food and Drug Control (approval number: SYXK (Lu) 2021 0109). All animals were kept in a room with a 12-h cycle between day and night at a temperature of 22 ± 2°C. They had unlimited food and water. All associated facilities and experimental techniques were given the seal of approval by the Ethical Committee’s Institutional Animal Care and Use Committee at Qingdao University of Science and Technology (approval number: ACQUST-2021-082). Three groups of mice (n = 10 per group) were formed: the control group, UVB group, and UVB + EUSM group. Before the experiments, the mice were anesthetized with ether to shave hair from their back skin (2 × 3 cm2), and the residual hair was removed with a depilatory cream. After UVB irradiation, EUSM (100 mg/mL) was administered (120 µL/mouse) to the dorsal hair removal region.
UVB Irradiation
Five lamps (20 W; maximum wavelength 313 nm; Huaqiang Co., Nanjing, China) were tied to a cage side by side to test the anti-photoaging ability of EUSM. The distance between the mice and the lamps was 10 cm. In a preliminary experiment, we established the minimum erythema dose (MED), which was about 100 mJ/cm2. Mice received UVB irradiation for four weeks. Throughout the study, the amount of UVB exposure gradually increased: all mice, except the control group, were irradiated for 3 consecutive days in the first week at a dose of 1 MED and the other 4 days were not exposed to UVB irradiation. Mice were exposed to UVB irradiation every other day from the second to the fourth week at a dose of 2 MED. After the fourth week’s final irradiation, euthanize the mice in each group, and the exposed skin tissue on the back of the mice is taken and the excess fat is removed. The gathered skin tissue is separated into three parts, with the first part being fixed with 4% paraformaldehyde for histopathological observations, the second being stored at –80°C for kit determination, and the third being immediately pulverized using a frozen grinding instrument (Shanghai Jingxin Industrial Development Co., Ltd., China) for the detection of semi-quantitative real-time polymerase chain reaction (qRT-PCR).
Histopathological Evaluation
Skin tissue that had been fixed for 24 h in 4% paraformaldehyde was eluted with water and ethanol gradient, the xylene was dealcoholized, and paraffin was embedded. Sliced paraffin blocks were transparentized with xylene before being eluted in a reverse gradient with ethanol and water. The degree of skin fibrosis and epidermal thickness were measured using hematoxylin and eosin (H&E) staining and Masson’s staining. Tissue sections were viewed by an inverted optical microscope, and the percentage of collagen in the skin tissues was calculated using ImageJ software (National Institutes of Health, USA).
Immunohistochemical Staining
Immunohistochemical analysis was demonstrated using paraffin-embedded mice skin tissue sections. Sections were placed in a sodium citrate solution and microwave-boiled for antigen retrieval. After dripping the specific primary antibody diluted with phosphate-buffered saline (PBS) on the target tissue sections, they were incubated at 4℃ overnight. The sections were observed using green FITC-conjugated anti-mouse IgG. Sections were analyzed using a light microscope (OLYMPUS, Japan).
Cell Culture
HaCaT cells were acquired from the China Center for Type Culture Collection in Wuhan, China, and were cultured in DMEM with 10% FBS at 37°C with 5% CO2. Cells were collected by treating them with 0.05% trypsin-EDTA after they reached roughly 80% confluence. Cells were passaged every 2–3 days.
Cell Treatment
HaCaT cells were irradiated with a UVB lamp (peak emission wavelength: 311 nm; Shanghai SIGMA High-tech Co., Ltd., China). Before UVB irradiation, PBS was used to replace DMEM in each well. After that, the cells were exposed to radiation at a dose of 340 mJ/cm2 (the cell radiation dose was based on our previous experiments). Then, PBS was replaced with DMEM containing the corresponding compound, and follow-up measurements were performed after 24 h.
Cell Viability Assay
HaCaT cells (8 × 103 cells/well) were treated with EUSM (12.5–100 µg/mL) for 24 h in 96-well plates. Then, UVB irradiation (340 mJ/cm2) was applied to the cells using a UVB lamp after EUSM treatment. Then, the cells were cultured with 50 µL MTT (2.5 mg/mL) for 4 h. By adding 150 µL of DMSO to each well, the absorbance was measured at 490 nm to assess cytotoxicity.
Measurement of HA and Total Protein Production
HaCaT cells were treated with EUSM for 24 h and then received UVB treatment for 24 h in 6-well plates (2 × 106 cells/well). The production of HA and total protein from the culture supernatant was examined using ELISA kits.
RNA Extraction and qRT-PCR
Total RNA was extracted from UVB-irradiated mice skin and HaCaT cells using the MolPure TRleasy® Plus Total RNA Kit (Shanghai Enzyme Link Biotechnology Co., Ltd.). Expression levels of collagen type I α 1 (COL1A1), hyaluronidase-1 (HYAL-1), MMP-3, and MMP-9 were determined via qRT-PCR using the BeyoFastTM SYBR Green One-Step qRT-PCR Kit (Beyotime Biotechnology, China). The 2–∇∇Ct method would be used to determine the mRNA’s relative expression level, and each sample was tested in triplicate. qRT-PCR was performed, and Table 1 contains the primer sequences used in this study.
Primer Sequences Used for qRT-PCR in This Study.

Biochemical Assays
10% skin tissue homogenates were obtained by grinding skin samples (0.1 g) using a frozen grinding instrument after they had been immersed in 9 mL of normal saline (4°C). The kits were used to measure the levels of MDA, SOD, and CAT in the skin, respectively. The levels of total collagen protein and HA in the skin were examined using ELISA kits.
Statistical Analysis
All data were presented as the mean ± SD. The difference between groups was determined using a one-way analysis of variance (ANOVA) followed by Tukey’s post hoc tests. GraphPad Prism 7 was used to conduct statistical analyses (GraphPad Software, San Diego, CA, USA). A p-value < 0.05 was considered statistically significant.
Results
Chemical Constituents of EUSM
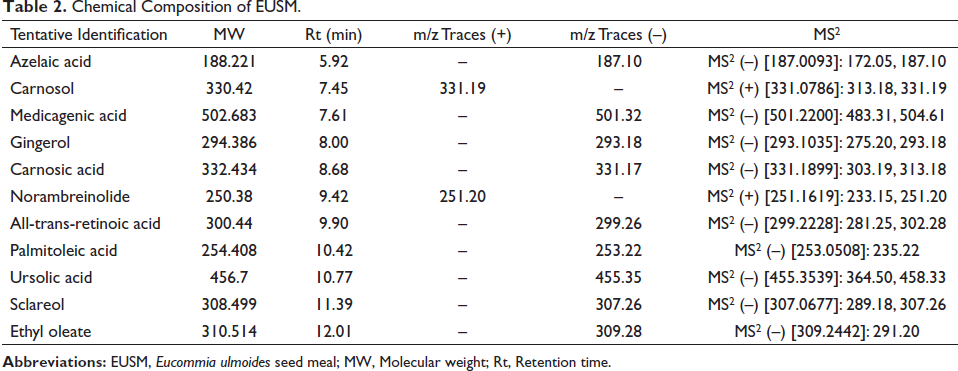
As shown in Table 2 and Figure 1, EUSM contains azelaic acid, carnosol, medicagenic acid, gingerol, carnosic acid, norambreinolide, all-trans-retinoic acid, palmitoleic acid, ursolic acid, sclareol, and ethyl oleate.
Chemical Composition of EUSM.
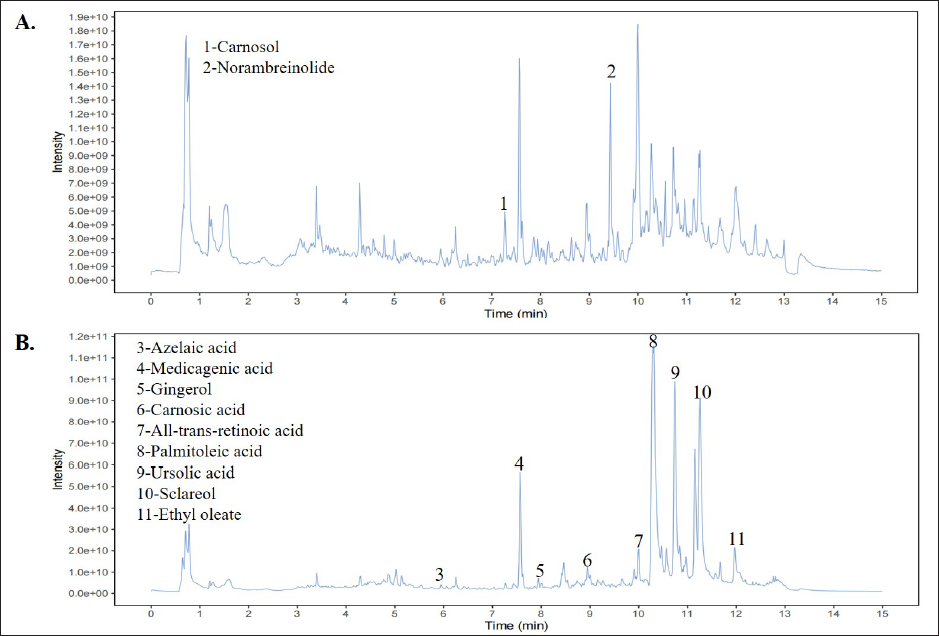
HPLC Analysis of Medicagenic Acid and Ursolic Acid in EUSM
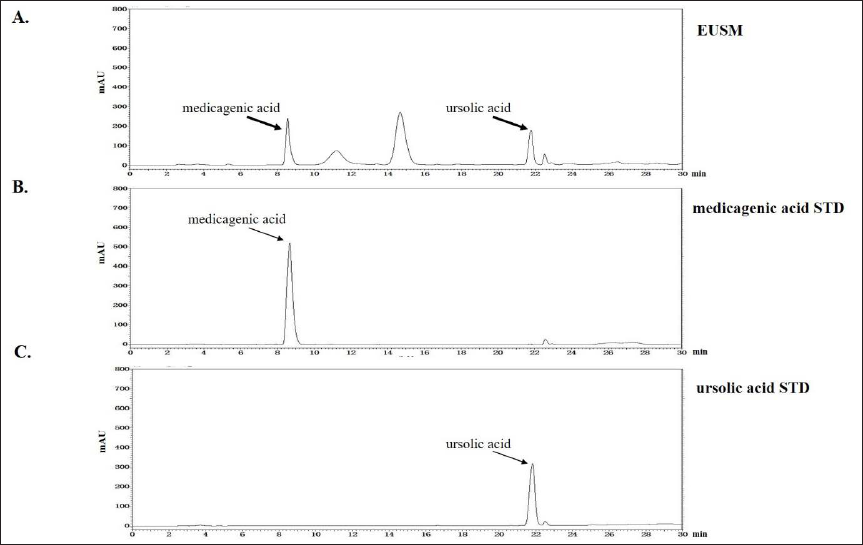
As shown in Figure 2, medicagenic acid and ursolic acid were detected in EUSM at 8.66 min and 21.77 min, respectively. The contents of two components were analyzed and found to be 0.66% medicagenic acid and 0.38% ursolic acid.
Effects of EUSM on Cell Viability
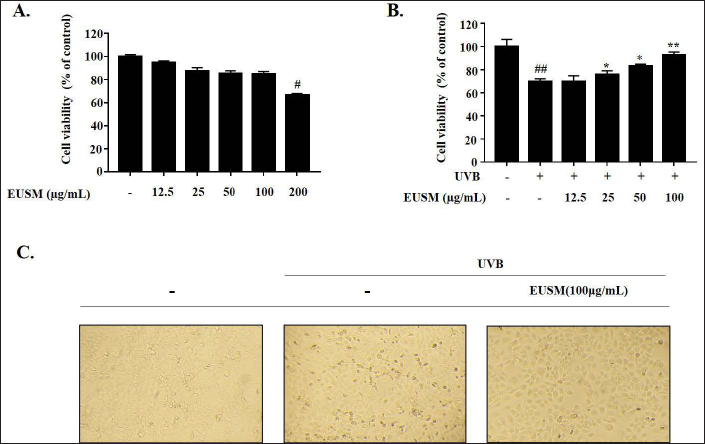
We evaluated EUSM’s cytotoxicity against HaCaT cells and its cytoprotective effect against UVB-irradiated HaCaT cells using the MTT assay. As shown in Figure 3A, the results demonstrated that EUSM was non-cytotoxic at concentrations between 12.5 and 100 µg/mL. Therefore, EUSM was applied to the cells at doses ranging from 12.5 to 100 µg/mL in all the following tests. Figure 3B showed that pre-treatment of HaCaT cells with EUSM for 24 h protected them from UVB-mediated damage. HaCaT cells treated with EUSM were irradiated with or without UVB, and their morphology was observed under a microscope. Figure 3C reveals that treatment with EUSM (100 µg/mL) significantly reduced UVB-induced HaCaT cell death.
Effects of EUSM on Active Molecules in UVB-treated HaCaT Cells
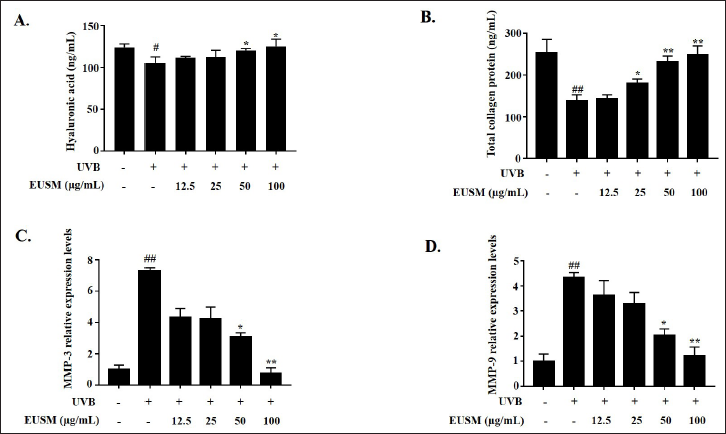
UVB radiation (340 mJ/cm2) was used to irradiate HaCaT cells for 24 h before being treated with EUSM (12.5, 25, 50, and 100 µg/mL) to investigate the effects of EUSM on the photoaging process. We confirmed the content of HA and collagen, and the expression levels of the mRNA for MMP-3 and MMP-9 were illustrated by qRT-PCR. As shown in Figure 4, the production of HA and total collagen was inhibited by UVB irradiation, while the expression levels of the mRNA for MMP-3 and MMP-9 were upregulated. EUSM (12.5–100 µg/mL) dose-dependently reversed these changes.
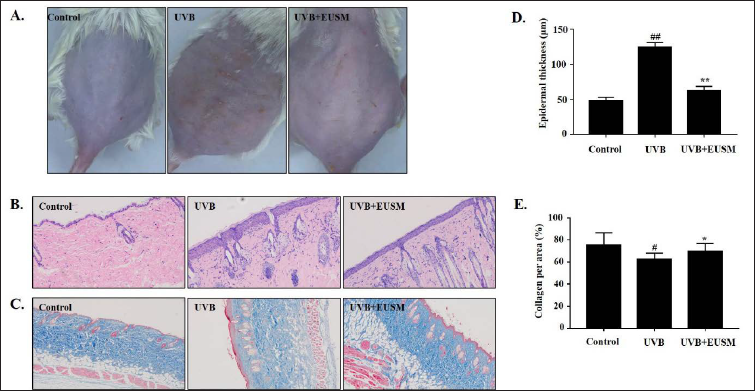
Effects of EUSM on the Prevention of Skin Photoaging in UVB-irradiated Mice
A skin histology study using H&E and Masson’s staining was performed to evaluate the protection of EUSM on mice skin. As shown in Figure 5A, these photographs indicated that, compared with the UVB group, supplementation with EUSM significantly reduced the UVB-induced formation of wrinkles and erythema. H&E staining illustrated the increased epidermal thickness of mouse skin, and treatment with EUSM significantly reduced epidermal thickness in UVB-induced mice skin (Figures 5B and D). Besides, Masson’s trichrome staining showed that collagen fibers in the control group were ordered and wavy. Comparatively to the control group, the UVB group showed collagen fiber breakage, which changed the morphology of the epidermis and dermis in the mouse skin tissue. Treatment with EUSM (100 mg/mL) significantly reduced collagen degradation (Figures 5C and E).
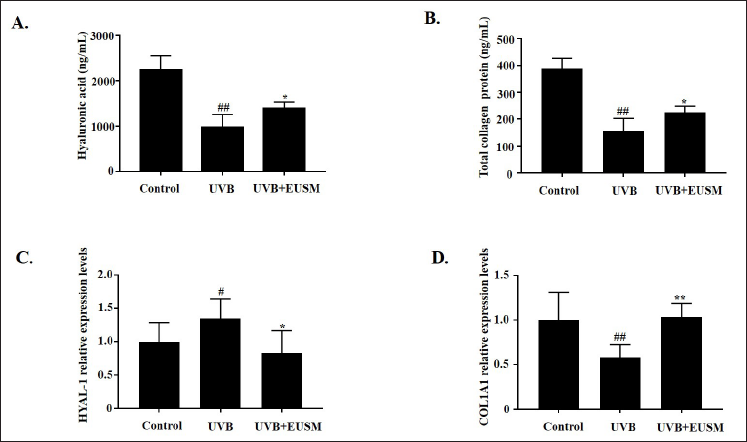
Effects of EUSM on Skin HA and Total Collagen Protein Production in UVB-treated Mice
As shown in Figures 6A and B, in the UVB + EUSM group, the levels of HA and total collagen protein were significantly increased compared to the UVB group. Moreover, the effects of EUSM on the levels of HYAL-1 and COL1A1 in skin tissue were examined using qRT-PCR. EUSM decreased the expression of HYAL-1 (Figure 6C) and increased the expression of COL1A1 in skin tissue (Figure 6D).
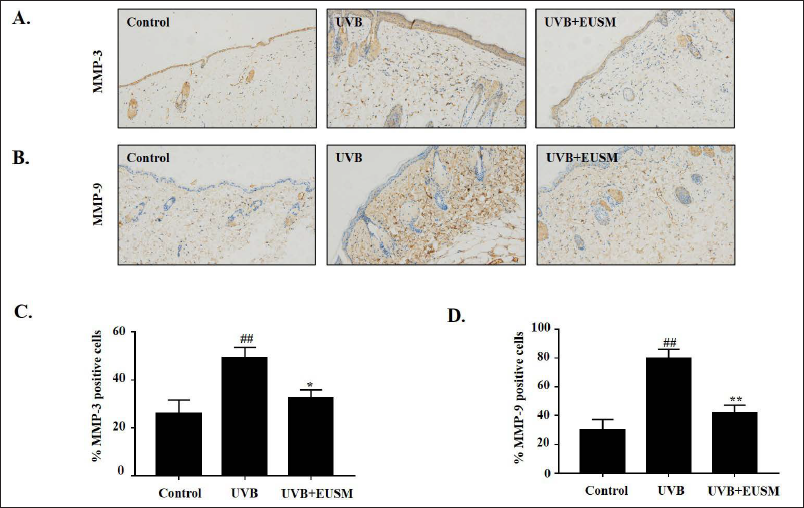
Effects of EUSM on MMP-3 and MMP-9 in Mice Skin
EUSM on UVB-exposed expression of MMP-3 and MMP-9 was assessed using immunohistochemical analysis. The UVB group clearly showed increased expression of proteins for MMP-3 and MMP-9, showing higher brown staining. However, EUSM (100 mg/mL) topical treatment prevented the UVB-mediated expression of proteins for MMP-3 and MMP-9 in the mouse skin (Figure 7).
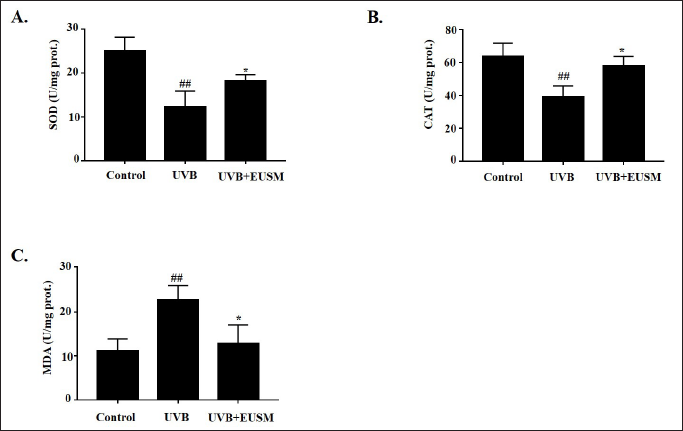
Effects of EUSM on SOD, CAT, and MDA Levels in UVB-induced Mice
Figures 8A and B shows that compared with the control group, the UVB group’s SOD and CAT levels were significantly lower (p < 0.01). After treatment with EUSM, the contents of SOD and CAT in skin tissue were significantly increased (p < 0.05). Figure 8C shows that the UVB group had considerably higher levels of MDA expression, but EUSM can significantly reverse this change (p < 0.05).
Discussion
Photoaging, also known as extrinsic skin aging, is primarily brought on by ultraviolet irradiation on the skin (Grether-Beck et al., 2014). UV irradiation of the skin can produce highly reactive free radicals that, through oxidation or crosslinking, destroy the antioxidant system of the skin, which can trigger the NF-κB signaling pathway and promote the release of inflammatory factors. Then it induces MMPs production and DNA damage, finally causing damage, mutation, and death of cells in the skin (Petruk et al., 2018). EUSM treatments showed anti-photoaging effects on UVB-irradiated HaCaT cells and mice skin in this study.
UV irradiation can induce skin changes in mice, such as hyperkeratosis, acanthosis, inflammatory cell infiltration, and collagen fiber damage (Ghorbanzadeh et al., 2019). In this study, Figure 5 shows that UVB irradiation caused significant histopathological changes in the mouse skin tissues, including a large number of hyperkeratoses and decreased collagen fiber levels. EUSM attenuated these histopathological changes by reducing epidermal changes and increasing the distribution of collagen fibers. However, the skin absorption of EUSM should also be detected in further studies.
As is well known, keratinocytes account for nearly 90% of the epidermis, which is crucial for maintaining the skin barrier and skin elasticity (Than et al., 2019). For this reason, we used HaCaT cells, a human keratinocyte cell line, to evaluate EUSM’s antiaging potential. In this study, UVB-induced HaCaT cells are protected by EUSM (Figure 3).
COL1A1 is the main component of type I collagen, whose loss contributes to increased skin sagging (Zhang et al., 2018). In addition, hyaluronidase (HYAL) can degrade HA in skin tissues, thereby reducing skin hydration (Maytin, 2016). In this study, total collagen and HA contents were significantly lowered by UV irradiation (p < 0.05). EUSM can increase the levels of HA and total collagen in HaCaT cells and mice skin tissues (Figures 4A and B, 6A and B). Otherwise, the relative mRNA expression levels of HYAL-1 and COL1A1 in skin tissue were further investigated, and our results showed that EUSM improved these two indicators (Figures 6C and D).
MMPs are major participants in the aging of the skin and can degrade ECM proteins in the skin (Sárdy, 2009). When exposed to UV radiation results in a significant amount of ROS, matrix metalloproteinase-3 (MMP-3) participates in the activation of matrix metalloproteinases (TIMPs), and gelatinase B (MMP-9) can degrade denatured collagen, gelatin, and many ECM molecules, including natural collagen IV, V, and XI (Pittayapruek et al., 2016). Here, we found that EUSM has antioxidant effects, which reduced the level of MDA and increased the contents of SOD and CAT in UVB-irradiated mice skin (Figure 8). In addition, EUSM significantly reduced the expression levels of the mRNA for MMP-3 and MMP-9 in HaCaT cells (Figure 4C and D) and decreased the expression of the proteins for MMP-3 and MMP-9 in mouse skin tissues (Figure 7).
E. ulmoides has been used in traditional medicine (Wang et al., 2019). However, the potential use of E. ulmoides seed meal in the cosmetic industry has yet to be explored. In this study, we identified six active components, namely, medicagenic acid, azelaic acid, gingerol, carnosic acid, ursolic acid, and sclareol, in EUSM using LC-MS analysis (Figure 1). Among these, ursolic acid has been reported to reduce the UVB-induced photooxidative damage of human skin fibroblasts by decreasing inflammation and oxidative stress (Samivel et al., 2020). Also, sclareol reduced the MMPs expression levels in UVB-induced HS68 cells and reduced facial wrinkles in clinical trials to achieve anti-photoaging effects (Park et al., 2016). And according to LC/MS, monomer components were screened, and the results showed that the sclareol (10 µM) and medicagenic acid (10 µM) increased cell viability significantly (p < 0.05 or p < 0.01, Supplementary Figure S1). We believe that these two EUSM components are extremely likely to be the key active components for anti-photoaging actions. As a result, we have provisionally assessed the content of medicagenic acid and ursolic acid present in EUSM and will continue to detect more active components (Figure 2). Also, we assessed the safety of EUSM for animals. The results of mouse liver H&E staining revealed that EUSM (100 mg/mL) had no evident histological alterations on the liver (Supplementary Figure S2).
Conclusion
Our findings suggest that EUSM increased the antioxidant level and reduced the production of MMPs in UVB-induced HaCaT cells and mouse skin to increase the contents of HA and collagen. This study demonstrates that EUSM can protect against UVB-induced photoaging, which both saves plant resources and excavates the potential value for the application of EUSM in the cosmetics field.
Footnotes
Authors Contribution
X.W. and S.H. designed the experiments and wrote the manuscript. T.W. and M.X. performed biochemical assays and conducted data analysis. L.G. and B.L. assisted in the design of the cell experiments and data analysis. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
All animals were cared for in strict accordance with the Guide for the Care and Use of Laboratory Animals (NIH Publication No. 85-23, revised 1996), and the experimental design was approved by the Use Committee of Qingdao University of Science and Technology (Approval no. ACQUST-20221-127).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by National Natural Science Foundation of China (82074578, 81960775, and 81960781) and Shandong Provincial Natural Science Foundation (ZR2021LZY032).
Informed Consent
There are no human subjects in this article, so informed consent is not applicable.
