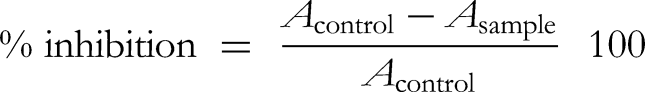Abstract
Chronic ultraviolet (UV) exposure produces oxidative stress, molecular damage, and aging-related signal transduction, all of which contribute to skin photoaging. In this study, the antioxidant and anti-inflammatory activities of Entada phaseoloides are reported. High-performance liquid chromatography (HPLC) detected 7 phenolic compounds: gallic acid, protocatechuic acid, 4-hydroxybenzoic acid, quercetin, luteolin, kaempferol, and apigenin. We investigated the antioxidant and protective effect of the acetone extract of E. phaseoloides leaves (AEP) on UVB-irradiated human epidermal keratinocytes (HaCaT cells). AEP showed antioxidant activity in the 2,2-diphenyl-1-picrylhydrazyl (DPPH) and 2,2-azinobis-3-ethylbenzothiazoline-6-sulfonic acid (ABTS) assays. AEP at a concentration of 40 μg/mL increased cell survival rate of the UVB-damaged cells. Moreover, AEP blocked gene expression of nitric oxide synthase (iNOS) and cyclooxygenase-2 (COX-2) in UVB-induced HaCaT cells and decreased UVB-induced apoptosis in HaCaT cells by regulating the gene expression of caspase-3. These results suggest that AEP has the potential to protect against UVB irradiation and antioxidant.
Introduction
Ultraviolet (UV) radiation is one of the major extrinsic factors that induce skin aging, which has been known to result from chronic exposure to solar radiation. 1 UV radiation can be divided into 3 types based on the wavelengths: UVA (315-400 nm), UVB (280-315 nm), and UVC (200-280 nm). Approximately 95% of UVA and 5% of UVB of total UV energy can reach the earth and are able to react with proteins and lipids leading to the formation of reactive oxygen species (ROS). UVB irradiation has been linked to skin inflammation and apoptosis due to oxidative stress caused by ROS. 2 Endogenous factors like the hydroxyl radical, superoxide anion radical, hydrogen peroxide, singlet oxygen, nitric oxide radical, and hypochlorite radical, as well as exogenous factors like smoking, ionizing radiation, pollution, and UV radiation, can cause oxidative stress, which can damage nucleic acid, proteins, enzymes, and other small molecules, causing structure and function loss. The number of free radicals created and the amount of antioxidants produced in the human body are in balance. 3
Human skin serves as the primary barrier against environmental pollutants while also providing external beauty. Extrinsic skin aging, also known as skin photoaging, is caused by prolonged exposure to external stimuli, with UVB and UVB irradiation being one of the most damaging culprits. UVB increases ROS generation while lowering endogenous antioxidants, causing molecular damage, and triggering aging-related signal transduction. UVB-induced changes can cause additional skin problems, including skin cancer, in addition to photoaging. 4
Entada phaseoloides (L.) Merr. is a very large, woody climber, stems often spirally twisted, belonging to the Leguminosae family. E. phaseoloides, found in tropical forests and lowland coastal forests of Africa, Asia, and Australia, contains triterpene saponins, flavonoids, and phenolic compounds. In 1989, Ikegami et al reported the basic component of E. phaseoloides leaves as entadamide C. 5 Phenolic compounds and flavonoids isolated from E. phaseoloides stems have displayed anti-oxidation properties in 2,2-dipheynyl-1-picrylhydrazyl (DPPH) assay. The seeds of Entada species are utilized as a folk medicine to treat skin disease and as a soap in Indonesia, Thailand, and other tropical countries.5,6 However, the biological activities of the leaves of E. phaseoloides are barely mentioned. To reduce intracellular ROS levels could be a good way to avoid UVB-induced human epidermal keratinocytes (HaCaT cells) apoptosis and skin damage. Thus, this study investigated the antioxidant activity and protective effect of an acetone extract of E. phaseoloides leaves (AEP) on UVB-induced HaCaT cells.
Materials and Methods
Chemicals and Reagents
Acetone, methanol (MeOH), and dimethyl sulfoxide (DMSO) were purchased from Wako Pure Chemical Industries (Osaka, Japan), gallic acid and 4-hydroxybenzoic acid from Wako Pure Chemical Industries (Osaka, Japan), quercetin and luteolin from Sigma-Aldrich (St. Louis, MO, USA), and protocatechuic acid, kaempferol, and apigenin from Tokyo Chemical Industry (Tokyo, Japan).
Plant Material and Preparation of Acetone Extract of E phaseoloides Leaves
Leaves of E. phaseoloides were collected from Kanchanaburi Province, Thailand, in June 2019 and authenticated by Mr Ratanachai Suthima, a staff member of the Forestry Center in Kanchanaburi. The sample was dried in the shade for 4 days and then ground into a fine powder. The powdered leaves (200 mg) were extracted in a sonicator bath at 50 °C with acetone, four times (300 mL, 60 min each). The acetone extract was evaporated under reduced pressure, using a rotary vacuum evaporator at 45 °C to give the acetone extract.
HPLC Analysis
The secondary metabolites of AEP were analyzed by high-performance liquid chromatography (HPLC) using standard compounds such as gallic acid, protocatechuic acid, 4-hydroxybenzoic acid, quercetin, luteolin, and apigenin. All experiments were performed on a 1260 Infinity II LC (Agilent Technologies) with a 1290 Infinity II ELSD (Agilent Technologies). The acetone extract was dissolved in MeOH at a concentration of 3 mg/mL, then 5 μL of each was injected; the flow rate was 0.4 mL/min. The separation was performed on a YMC-Triart C18 column (4.6 × 150 mm, 5 μm) (YMC Company) at 40 °C with a gradient system of 0.1% formic acid in water (solvent A) and 0.1% formic acid in MeOH (solvent B) as follows: 0 to 2 min, 20% B; 2 to 22 min, 20% to 100% B; 22 to 30 min, 100% B, and finally the B content was decreased to the initial conditions in 5 min and the column re-equilibrated for 5 min. The diode array detector (DAD) was measured over the range of 200 to 600 nm. The evaporation temperature of the evaporative light scattering detector (ELSD) detector was set at 80 °C, and the nebulizer temperature at 30 °C. The gas flow ratio was 1.6 mL/min.
Total Phenolic Content Assay
The amount of total soluble phenolics was determined according to the Folin–Ciocalteu method 7 with some modifications. After AEP had been diluted with MeOH stepwise, 100 μL of diluted sample was pipetted into a 96-well plate. Fifty μL of Folin–Ciocalteu reagent (Merck, Darmstadt, Germany), diluted fivefold with 70% MeOH, and 50 μL of 7% (w/v) sodium bicarbonate (NaHCO3, purchased from Wako Pure Chemical Industries) were added to each well, and the plate was incubated at room temperature for 5 min. Color development at 650 nm was then determined using a BioTek Synergy HTX Multi-Mode Reader. The color development intensities of the AEP is shown as gallic acid equivalents (GAE) per mg of AEP (μg GAE/mg AEP).
Scavenging Activity of DPPH Radical
The DPPH• (2,2-dipheynyl-1-picrylhydrazyl purchased from Wako Pure Chemical Industries, Osaka, Japan) scavenging ability of AEP was evaluated by mixing 50 μL of DPPH• MeOH solution (0.06 mM) with 50 μL of either AEP or ascorbic acid (AA as positive control) dissolved in MeOH at different concentrations. The absorbance was measured at 517 nm (BioTek Synergy HTX Multi-Mode Reader) after 45 min of incubation at room temperature in the dark. A negative control was produced with 50 μL of MeOH mixed in 50 μL of DPPH• and maintained under the same conditions as the sample.
8
The antioxidant activity of AEP and AA were calculated by using the following equation:
Scavenging Activity of ABTS Radical
The ABTS•+ (2,2-azinobis-3-ethylbenzo-thiazoline-6-sulfonic acid purchased from Sigma-Aldrich, St. Louis, MO, USA) scavenging ability of AEP was evaluated by ABTS assay.
8
To produce ABTS•+ radical cations, 7 mM ABTS stock solution was mixed with 2.45 mM potassium persulfate (K2S2O8) at room temperature in the dark for 16 h. The ABTS•+ solution was diluted with MeOH to obtain an absorbance of 0.70 ± 0.02 at 734 nm (BioTek Synergy HTX Multi-Mode Reader). Then, 20 μL of either AEP or AA (as positive control) dissolved in MeOH at different concentrations was added to 180 μL of the ABTS•+ solution. The absorbance was measured after 6 min at room temperature in the dark. The antioxidant activity of AEP and AA was calculated by using the following equation:
Cell Culture
Human spontaneously transformed keratinocyte (HaCaT) cells were cultured in Dulbecco's Modified Eagle's medium (DMEM, purchased from Wako, Osaka, Japan) high glucose supplemented with 10% heat-inactivated fetal bovine serum (FBS, purchased from Wako, Osaka, Japan) and 100 U/mL penicillin-streptomycin (Wako, Osaka, Japan) at 37 °C in a 5% CO2 humidified incubator.
Cell Viability Assay
Bioassay of cytotoxic activity was performed in vitro by a colorimetric method that measures the reaction of 3-(4,5-dimethylthiazol-2-yl)-2,5-dipheynyltrazoliumbromide (MTT, purchased from Tokyo Chemical Industry, Tokyo, Japan) with mitochondria. MTT is reduced to dark purple formazan, followed by solubilization and measurement by spectrophotometry. Generally, reduction of MTT occurs in metabolically active cells. The level of activity is thus measured from the viability of the cell, which is proportional. Cells were cultured in a 96-well plate at a density of 1 × 105 cells/mL for 24 h. Then, HaCaT cells were treated with different acetone extract concentrations, dissolved in DMSO. After 24 h, cell viability was determined using MTT reagent as follows: 10 μL of MTT solution (5 mg/mL PBS) was added to each well, followed by a 4-hour incubation at 37 °C in 5% CO2. The supernatant was discarded, and 40 mM HCl isopropanol (100 μL) was added to dissolve the formazan crystals. The absorbance was measured at 570 nm using a Microplate Reader 310-Lab (Corona Electric Company).
UV Irradiation
Cells were cultured in 96-well plates for 24 h and then pre-treated with different concentrations of acetone extract for 24 h at 37 °C in 5% CO2. A monolayer of HaCaT cells was washed once with phosphate buffer saline (PBS purchased from Wako, Osaka, Japan). After that, a thin layer of PBS was added to expose the cells to UVB irradiation (30 mJ/cm2). UV irradiation was performed with UVP CL-1000 Ultraviolet Crosslinker (Vilber Lourmat, Torcy, France). After removal of PBS, serum-free DMEM was added to the cells, which were incubated for 24 h. Then, MTT assay was performed.
Real-Time Quantitative PCR
HaCaT cells were seeded at 1 × 105 cells/mL in a 24-well plate and incubated at 37 °C for 24 h in 5% CO2. Then, the cells were pre-treated with acetone extract (10, 20, and 40 μg/mL). The cells were exposed to UVB (30 mJ/cm2, 20 s) and further incubated with serum-free medium for 24 h. After cell collection, total RNA was prepared from cells using an RNeasy Mini kit (Qiagen), following the manufacturer's instructions. cDNA was synthesized from the extracted total RNA by ReverTra Ace qPCR RT Master Mix with gDNA Remover (TOYOBO). Real-time quantitative PCR was performed using the synthesized cDNA as a template with an AriaMX (Agilent Technologies). THUNDERBIRD SYBR qPCRMix (TOYOBO) was used for the real-time PCR reaction. The real-time PCR reaction conditions were initial denaturation at 95 °C for 60 s. cDNA samples were amplified for 40 cycles (95 °C for 15 s and 60 °C for 60 s). Real-time quantitative PCR was performed using specific forward and reverse primers (Table 1).
Sequences of Primers (Human) Used in Real-Time Quantitative PCR.

Abbreviations: COX-2, cyclooxygenase-2; iNOS, nitric oxide synthase.
Statistical Analysis
All data are expressed as mean ± SD. Significance was analyzed by two-way analysis of variance (ANOVA) test. A P value of * < .01 or ** < .05 was considered statistically significant.
Results and Discussion
Determination of Total Phenolic Content
The content of total phenolics in AEP was estimated using a modified Folin–Ciocalteu method; a value of 83.53 ± 3.02 μg GAE/mg AEP was obtained. In the previous reports, many active compounds have been isolated from E. phaseoloides seeds and stems such as quercetin, luteolin, apigenin, and kaempferol. 6 To propose the active compounds in AEP, its profile was analyzed by HPLC.
Estimation of Active Compounds from AEP
AEP was filtered through Millipore 0.20 μm filters (Millex-LG, Japan) before injection. The HPLC method coupled with DAD and ELSD were conducted for determination of the compounds in AEP. Its components were proposed by comparing their retention times with those of the following reference standards: gallic acid (9.181 min), protocatechuic acid (13.173 min), 4-hydroxybenzoic acid (16.079 min), quercetin (22.955 min), luteolin (23.607 min), kaempferol (24.610 min), and apigenin (24.987 min) (Figure 1).

Chemical structures of the compounds identified are as follows: gallic acid (
Antioxidant Potential of AEP
The radical scavenging activity of DPPH• and ABTS•+ were found to be positively correlated with the concentration of AEP. IC50 values of the AEP evaluated in this study are shown in Table 2. These tests evaluated the ability of AEP to donate an electron or hydrogen to DPPH• and transfer an electron to ABTS•+. The results showed that AEP presents antioxidant properties in the methodologies evaluated.
Antioxidant Activity of AEP and AA According to Different Cell-Free Antioxidant Assays.

Abbreviations: AA, ascorbic acid; AEP, acetone extract of E. phaseoloides leaves; DPPH, 2,2-diphenyl-1-picrylhydrazyl; ABTS, .2,2-azinobis-3-ethylbenzothiazoline-6-sulfonic acid.
Values are the average of three replicates ± SD and expressed in IC50 in μg/mL. IC50: inhibitory concentration to 50%
Effect of AEP on Cell Viability of HaCaT Cells
The cytotoxic effect of AEP on the HaCaT cell line was investigated with different concentrations (eg 2.5, 5.0, 10.0, 20.0, and 40.0 μg/mL) for 24 h, and cell viability was examined by the MTT assay. As shown in Figure 2, AEP did not show any toxicity up to a concentration of 10 μg/mL. At concentrations of 20 and 40 μg/mL, AEP displayed around 80% cell viability. Therefore, a concentration of 40 μg/mL was considered the highest concentration for further experiments.

Cell viability of AEP was evaluated by 3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT) assay. HaCaT cells (1 × 105 cells/mL) were seeded into a 96-well plate and pretreated with AEP at concentrations of 2.5, 5, 10, 20, and 40 μg/mL. Cell viability was performed by MTT assay. The data are expressed as mean ± SD of at least 3 independent experiments in each group.
Effect of AEP on UVB-Induced Cell Oxidative Damage
To determine the UVB intensity used in the analysis, the cell viability of the cells was examined by UVB irradiation. Cells were irradiated with UVB at 30 mJ/cm2 for 10, 20, 30, 40, 50, and 60 s. Cell viability was evaluated by MTT assay after irradiation for 24 h. The survival rate of HaCaT cells in the UVB irradiation was in a time-dependent manner, as shown in Figure 3. The cell survival rate of the HaCaT cells was about 86% for UVB irradiation for 10 s, and 78% for 20 s. UVB irradiation showed a significant reduction in cell viability in an intensity-dependent manner. Thus, we used 30 mJ/cm2 for 20 s for our UVB dose in all further experiments. In our study, AEP was evaluated for its protective effect on keratinocyte cells. Pretreatment with AEP increased cell viability of UVB-irradiated HaCaT cells at the dose of 30 mJ/cm2 for 20 s. The cell viability was 82% and 65% at concentrations of 40 μg/mL and 2.5 μg/mL, respectively. These results suggest that AEP abolished the effects of UVB-induced HaCaT cell damage (Figure 4).

Cell viability of HaCaT cells was irradiated with UVB for different irradiation times. HaCaT cells were seeded followed by incubation for 24 h. HaCaT cells were irradiated with 30 mJ/cm2 of UVB and then cultured with DMEM serum-free medium for 24 h. After 24 h irradiation, the cell viability was measured by MTT assay. The data are expressed as mean ± SD of at least 3 independent experiments in each group.

Phototoxicity of HaCaT cells (1 × 105 cells/mL) was pretreated with AEP at concentrations of 2.5, 5, 10, 20, and 40 μg/mL. After 24 h, the cells were irradiated with UVB (30 mJ/cm2) and cultured in serum-free medium for 24 h. Cell viability was performed by MTT assay. The data are expressed as mean ± SD of at least 3 independent experiments in each group.
Effect of AEP on UVB-Induced Gene Expression of COX-2, iNOS, and Caspase-3
UVB radiation plays an important role in ROS production compared with nonirradiated cells. ROS have a role in the cellular signaling pathway of UV-induced hypersensitivity, inflammation-related redness, and carcinogenic development by acting as a secondary messenger. Cyclooxygenase-2 (COX-2) and nitric oxide synthase (iNOS) gene expression are mediated by UVB-induced inflammation reactions and skin cell apoptosis. 9 Dong et al reported that phenolic compounds and flavonoids isolated from E. phaseoloides stems show radical scavenging activity in the DPPH assay. 10 Rheediinosides A and B isolated from kernels of Entada rheedei showed moderate cytotoxic potency and antioxidant activity. 11 Therefore, reducing intercellular ROS generation and inhibiting COX-2 and iNOS expression may represent an effective strategy for preventing diseases from photodamage. Real-time qPCR analysis was used to examine the ability of gene expression of COX-2 and iNOS. Pretreatment with AEP at a concentration of 40 μg/mL suppressed the expression of COX-2 when compared with non-treated cells (Figure 5A). The mRNA expression of iNOS, compared with the non-treated control after UVB irradiation, was increased in HaCaT cells. In the pretreatment group with AEP, after UVB irradiation, the expression of iNOS was downregulated, as shown in Figure 5B. Several studies have shown that UVB irradiation considerably augments both protein and mRNA levels of iNOS and COX-2. COX-2 and iNOS are inflammatory products triggered by external and endogenous stimuli such as growth factors, tumor promoters, and cytokines. 12 Thus, AEP can play a role in controlling inflammatory responses. UVB irradiation is thought to be a potent inducer of cell death. Internal cell death, also known as apoptosis, can be triggered by the production of ROS. 13 Real-time qPCR analysis showed increased expression levels of caspase-3 in UVB-induced HaCaT cells. Pretreatment of AEP suppressed the apoptotic death in UVB-irradiated HaCaT cells by downregulating the expression level of caspase-3 (Figure 5C). However, higher concentrations (20 and 40 μg/mL) interestingly showed an increased caspase-3 gene expression level compared to 10 μg/mL due to hormesis, which is a dose–response phenomenon explaining the inhibition caused by high dosage, as well as stimulation by low dosage. Hormesis is a redox-dependent aging process linked to the production of free radical species, which are inflammatory responses that have a role in neurodegenerative and neuroprotective pathways in Parkinson's disease neurobiology.14–17

Gene expression in UVB-irradiated HaCaT cells. AEP was added to HaCaT cells and incubated for 24 h. The cells were irradiated with 30 mJ/cm2 UVB and cultured with serum-free medium for 24 h. The gene expression was measured by RT-qPCR. (A) Gene expression level of COX-2, (B) iNOS, and (C) caspase-3. The data are expressed as mean ± SD of at least 3 independent experiments in each group.
Thus, these results suggest that AEP protected the cells by inhibiting caspase-3 activation since caspase-3 is the last effector caspase in the apoptosis pathway.
Conclusion
In this study, 7 phenolic compounds, namely gallic acid, protocatechuic acid, 4-hydroxybenzoic acid, quercetin, luteolin, kaempferol, and apigenin were identified by HPLC. We demonstrated for the first time that AEP had potential antioxidant properties and inhibited UVB-induced phototoxicity, which was verified by MTT assay. UVB irradiation (30 mJ/cm2) for 10 to 60 s significantly reduced HaCaT cell viability after 24 h incubation. AEP reduced ROS generation and downregulated the expression of COX-2 and iNOS in UVB-irradiated HaCaT cells. It also decreased the apoptotic cell death, which had been identified by a downregulated level of caspase-3 in UVB-irradiated HaCaT cells. However, further studies on the isolation of the active components of AEP and their skin protection on UVB-treated HaCaT cells should be carried out.
Footnotes
Acknowledgments
The first author would like to thank the Mitsubishi UFJ Trust Scholarship Foundation for the scholarship.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.