Abstract
Objectives
This study is aimed at identifying critical therapeutic targets of Astragalus membranaceus (Huangqi (HQ)) and investigating the effects and mechanisms of HQ treating uveitis. The potential drug targets of HQ and main active ingredients were obtained from the Traditional Chinese medicine (TCM) systems pharmacology database and analysis platform (TCMSP,
Materials and Methods
Cytoscape software was used to identify the disease targets of uveitis. Drug targets and disease targets were compared, and intersected hubs were applied for the active ingredient-target network and protein-protein-interaction (PPI) network construction. Signaling pathway enrichment annotation was performed to identify possible signaling involved in uveitis treatment. An endotoxin-induced uveitis (EIU) model was established, and the therapeutic effects of total flavonoids of Astragalus (TFA) on uveitis were investigated by examining the improvement of eye symptoms, histopathological alterations, and the levels of cytokines.
Results
Based on network pharmacological analysis, HQ could modulate the initiation and progression of uveitis by reducing the production of cytokines and regulating cell apoptosis via the NOD-like receptor (NLR), apoptosis, and toll-like receptor (TLR) signaling pathways. Based on animal experiments, high-dose TFA could reduce rat’s iris congestion, reduce anterior chamber exudation and pus, restore pupil size, and decrease the release of inflammatory factors IFN-γ and IL-10. Network pharmacological and experimental analyses revealed that TFA regulates the release of inflammatory factors through the NLR and TLR signaling pathways, thus regulating the immune system of EIU rats and ultimately relieving inflammation responses in uveitis rats.
Keywords
Introduction
Uveitis is an inflammatory lesion involving the uvea, retina, retinal vessels and vitreous humor, which can be seen in different age groups (Chang & Wakefield, 2002; Tsirouki et al., 2018; Wakefield & Chang, 2005). Uveitis, often involving both eyes, can have a serious impact on the patient’s visual ability, with a long course, a high recurrence rate, and a series of complications and sequelae, eventually leading to blindness in severe cases (de Smet et al., 2011; Munoz-Fernandez & Martin-Mola, 2006).
The etiology and pathogenesis of uveitis are not fully understood. Endogenous and exogenous antigens and triggers such as infections, trauma, tumors, and autoimmune reactions cause antigenic antibody reactions in the eye and manifest immunogenic uveitis (Barisani-Asenbauer et al., 2012; Krishna et al., 2017). The treatment principle of uveitis is to control intraocular inflammation, prevent relapses of inflammation, and reduce drug-related side effects. Currently, corticosteroids and immunosuppressants remain the mainstay of treatment drugs, which sometimes fail to control inflammation and frequently cause well-known ocular and systemic adverse effects (Miloslavsky et al., 2017; Niederer et al., 2017; Sen et al., 2014; Suhler et al., 2017). Therefore, it is urgent to identify more effective and safer therapies that target specific immune response mediators to achieve and maintain inflammation remission. Unlike single-target chemical pharmaceuticals, Traditional Chinese medicines (TCM) have the benefits of multi-component, multi-pathway, and multitarget synergy with fewer adverse effects and more therapeutic outcomes (Zhang et al., 2016). TCM believes that the pathogenesis of recurrent uveitis is mainly the result of Yin deficiency of the liver and kidney, accompanied by Qi stagnation and blood stasis, which is a syndrome of endogenous heat due to Yin deficiency.
TCM has achieved good results in the treatment of uveitis by mediating Qi and Yin deficiency and fully considering the principle of benefiting Qi and nourishing Yin (Davatchi et al., 2017; Zhou et al., 2012). Jing et al. (2019) built a Qinghuo Rougan Mingmu formula (QHRGF)-compound target-uveitis network and found that QHRGF reduced local inflammation in experimental autoimmune uveitis (EAU) mice by regulating natural killer T cells and blocking the mitogen-activated protein kinase (MAPK) signal pathways. Qingkailing injection alleviated autoimmune uveitis in rats by inhibiting the development of Th1 and Th17 effector cells as well as the release of key cytokines (Tian et al., 2012). Another herbal extract mixture often used in TCM, Longdan Xiegan Tang, can effectively relieve EAU symptoms, prevent the differentiation of uveitogenic CD4+T cells, and diminish the release of proinflammatory cytokines including IFN-γ, IL-17, and TNF-α (Tang et al., 2016). Astragalus membranaceus (Huangqi (HQ)) is a sweet-flavored and warm-natured TCM herb that goes to the lung, spleen, liver, and kidney meridians. HQ has Qi-benefiting and spleen-invigorating effects, detoxicating and granulation-promoting effects, diuresis-inducing and edema-reducing effects, Qi-invigorating and superficies-consolidating effects, and Yang-raising and prolapsed zang-fu organs-lifting effects. Clinically, HQ is often used to prevent and treat a variety of diseases, such as immune dysfunctional diseases, cardiovascular and cerebrovascular diseases, promote intellectual development, delay aging and anti-tumor, with precise efficacy and fewer side effects (Chen et al., 2020; Fu et al., 2014; Liu et al., 2017; Yue et al., 2017). Given all these previous findings, HQ may also be used in the treatment of uveitis, but its potential targets and mechanisms of action are unknown.
The main chemical components of HQ are flavonoids, Astragalus polysaccharides, Astragalus saponins, amino acids, and trace elements, among which the total flavonoid component of Astragalus is one of the main bioactive components. In this study, network pharmacology was taken as an entry point to investigate the effective active ingredients and action targets of HQ for the treatment of uveitis and to reveal its molecular mechanism of action, which is of great significance for its clinical promotion.
Materials and Methods
Retrieval of Compound Information in Radix Astragali (Huangqi (HQ))
The chemical ingredients of HQ were collected using the TCM System Pharmacology Database (TCMSP,
Compounds Targets and Uveitis Disease Target Retrieval
The protein targets of the active compounds in HQ were obtained from the PubChem database (
Construction and Analysis of Networks
To comprehensively understand the molecular mechanisms of HQ for uveitis, Cytoscape_v3.7.1 was used to construct the compound-target-disease networks (Smoot et al., 2011). In these graphical networks, the compounds, targets, or diseases were represented as nodes, while the compound-target or target-disease interactions were represented as edges.
Protein-Protein-Interaction Network
The genes obtained from the intersection were applied to the STRING database to construct the Protein-Protein-Interaction (PPI) network which is involved in the HQ treatment of uveitis in vivo.
Signaling Pathway and Functional Enrichment Annotation
A Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment analysis and gene ontology (GO) functional enrichment analysis were performed on 18 potential targets in the DAVID database. GO enrichment analysis includes cellular component (CC), molecular function (MF), and biological process (BP). A threshold value of p < 0.05 was set, and the top 20 results were screened by ranking according to the number of targets involved.
Induction of Rat EIU
A total of 30 healthy male SD rats aged 6−8 weeks with body masses of 150−180 g were purchased from Spelford (Beijing) Biotechnology Co., Ltd. (SCXK (Beijing) 2016-0002). All procedures were approved by the Institutional Animal Care and Use Committees of the Affiliated Cancer Hospital of Xiangya School of Medicine, Central South University. A total of 30 SD rats were randomly divided into four groups (n = 6): the normal control group (NC group), the model control group (endotoxin-induced uveitis (EIU) group), the low-dose total flavonoids of Astragalus (TFA) treatment group, and the high-dose TFA treatment group. The EIU model was established by injecting 1 g/L of Salmonella typhi (0.1 mL) LPS physiological saline solution in the left plantar aspect of all rats. Sterile PBS (1X; 135 mM NaCl, 4.7 mM KCl, 10 mM Na2HPO4, 2 mM NaH2PO4, and pH 7.4) was injected alone as a control. EIU model rats in the low-dose TFA treatment group were intraperitoneally injected with TFA 50 mg/kg 1 h after modeling. EIU model rats in the high-dose TFA treatment group were intraperitoneally injected with TFA 500 mg/kg 1 h after modeling.
Morphological Observation and Histological Analysis
Rats’ eyes were observed at 2, 4, 6, 8, and 10 h after modeling with a slit lamp biomicroscope; changes in clinical signs were recorded and photographed; and the intensity of inflammation was classified from grade 0 to grade 5 according to the scoring criteria as the previous description (Okada et al., 1998). Briefly, grade 0, no inflammation; grade 1, discrete dilation of the iris and conjunctival vessels; grade 2, moderate dilation of the iris and conjunctival vessels; grade 3, intense iris vessel dilation and with flare in the anterior chamber; grade 4, more severe clinical signs than grade 3, with or without the formation of hypopyon; and grade 5: formation of fibrin in the pupillary area and miosis or total seclusion of the pupil.
After 24 h of modeling, the rats were executed under chloral hydrate anesthesia and the eyeballs were removed and collected. Tissues were routinely fixed, placed in gradient doses of ethanol solution for 30 min for dehydration, placed in xylene for 4 min for transparent treatment, and immersed in paraffin for 2 h. Then, tissues were embedded, cut into 4-µm sections, and subjected to hematoxylin and eosin (H&E) staining. Slices were observed under a light microscope, and inflammation grading was performed as described in the previous description (Shi et al., 2020). Briefly, the histopathological grade was classified from grades 0 to 4. Grade 0, normal tissue; grade 1, dilation of iris vessels, thickened iris stroma, and the anterior chamber with exudate, protein, and/or a few scattered inflammatory cells; grade 2, infiltration of inflammatory cells in the stroma of the iris and/or ciliary body, and the anterior chamber with a moderate number of inflammatory cells; grade 3, the iris stroma and ciliary body with heavy infiltration of inflammatory cells and the anterior chamber with heavy infiltration of inflammatory cells; and grade 4, the anterior chamber with dense protein aggregation and infiltration of cells and the corneal endothelium with infiltration of inflammatory.
Serum IL-10, TGF-β, IFN-γ and IL-17 Detected by ELISA
Corresponding ELISA kits for IL-10, TGF-β, IFNγ, and IL-17 levels were used (Cat. JL13427-48T, JL13643-48T, JL13987-48T, and JL20879-48T, Jianglai, China). Samples were incubated with horseradish peroxidase-labeled detection antibodies. After washing to remove the unbound material, horseradish peroxidase-labeled streptavidin was added. After washing, the chromogenic substrate TMB was added, and the color was developed away from light. The reaction was terminated by adding the termination solution, and the absorbance value was measured at 450 nm.
Statistical Analysis
SPSS 21.0 statistical software was used for data processing and statistical analysis. Experimental data from individual experiments were presented as the mean ± standard deviation (SD). Comparisons between conditions were performed by a Student’s t-test or one-way ANOVA with Tukey’s post hoc analysis. A p-value greater than 0.05 or 0.01 means that no effect was observed.
Results
Screen of Active Components of HQ
The active components of HQ were screened through the TCMSP database, and a total of 87 active components were obtained. Next, 20 active components of HQ were screened with OB ≥ 30% and DL ≥ 0.18 (Table 1). The action targets of these active components were investigated, and 97 action targets were obtained after deduplication.
Main Active Ingredients in HQ According to TCMSP Database.

Targets of Uveitis
The targets of uveitis were retrieved from international databases (such as Drugbank, TCMSP, and OMIM), with “uveitis” as the keyword. As a result, a total of 605 therapeutic targets for uveitis were obtained.
Construction of the HQ Active Components-Potential Targets-Uveitis Network
After taking the intersection of 97 targets of HQ active components and 605 targets of uveitis, 18 potential targets of HQ in uveitis treatment were identified (Figure 1A). Next, the network of HQ active components, potential targets, uveitis was constructed using the Cytoscape 3.7.1 software (Figure 1B). The network consisted of 24 nodes, of which 1 node represented uveitis, 1 node represented HQ, 4 nodes represented different HQ active components, and 18 nodes represented potential targets at the intersection of HQ active component targets and uveitis targets (Figure 1B).
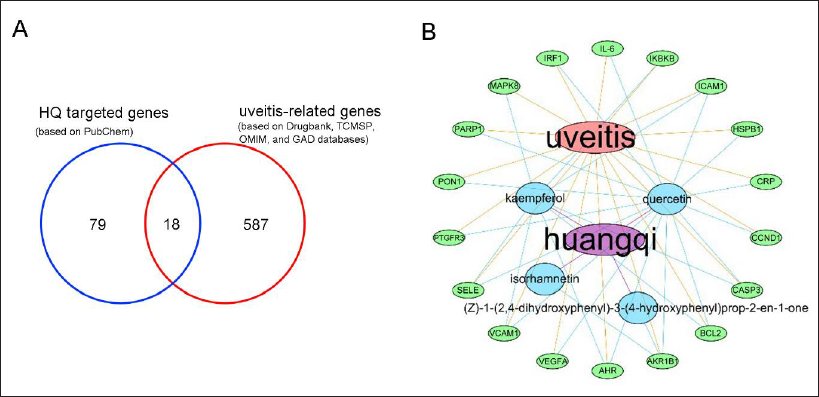
PPI Network of HQ in Uveitis Treatment was Constructed
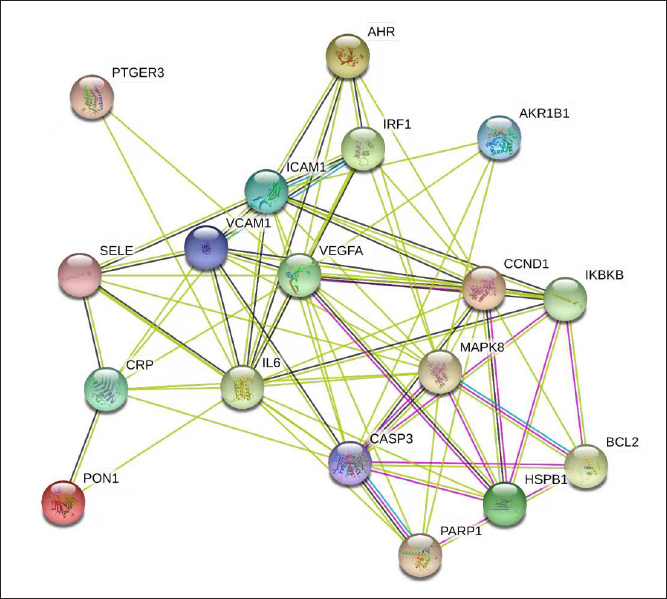
Subsequently, 18 target genes were introduced into the STRING database to construct the in vivo PPI network of HQ in uveitis treatment. This PPI network consisted of 18 nodes and 75 sidelines, with an average node degree of 8.33 (Figure 2).
KEGG Pathway Enrichment Analysis
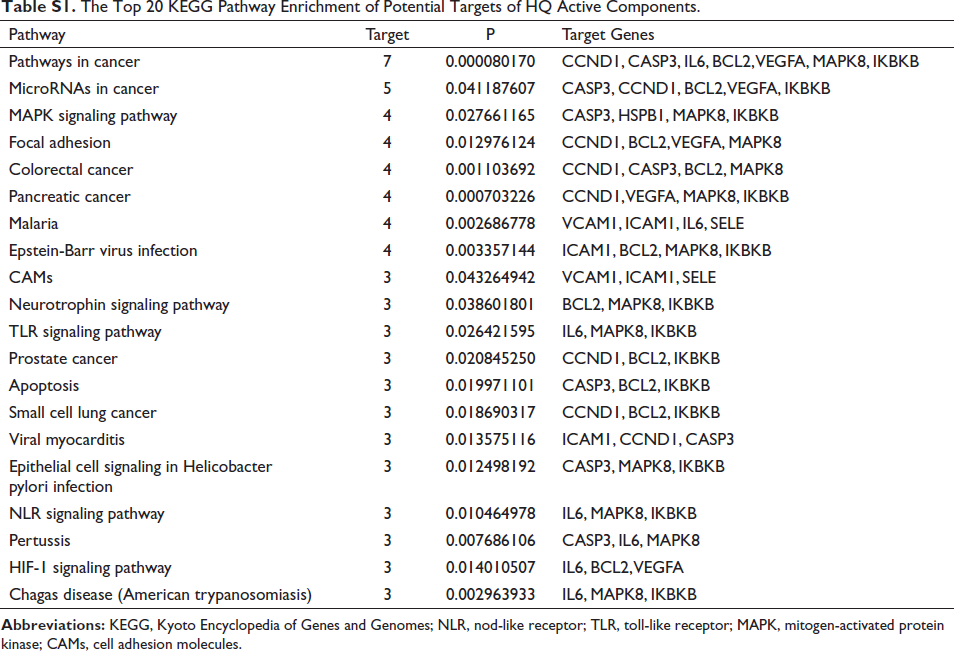
The KEGG pathway enrichment analysis was performed on the potential targets of HQ active components. The first 20 pathways were screened out in descending order according to the target numbers, with p < 0.05 as the screening criteria (Table S1). Finally, five uveitis-related pathways were identified: the NOD-like receptor (NLR) signaling pathway, apoptosis, the toll-like receptor (TLR) signaling pathway, the MAPK signaling pathway, and the cell adhesion molecules (CAMs). The detailed KEGG pathway enrichment analysis results are displayed in Table 2.
KEGG Pathway of HQ Active Components/Disease Target Genes.
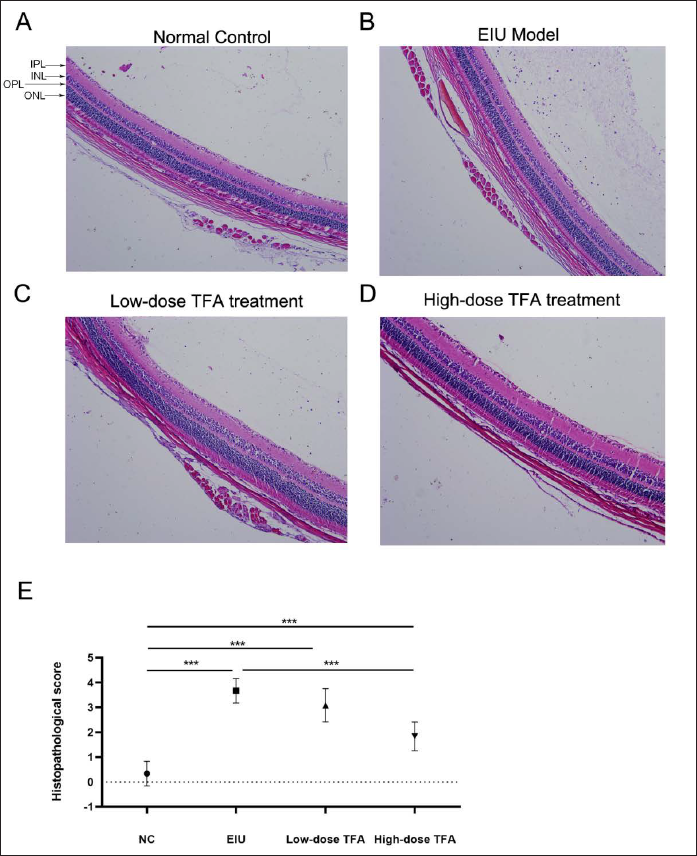
Histopathology of Rats’ Retinas in Each Group Was Observed Using H&E Staining
As indicated by the H&E staining results (Figure 3A−D), there was no inflammatory cell infiltration and vascular congestion in the retina and vitreous cavity in the NC group. For rats in the EIU model group, there was a small amount of inflammatory cell infiltration and vascular dilatation in the vitreous cavity and retina. In the low-dose total TFA treatment group, slight inflammatory cell infiltration and a few vascular congestions were present in the retina and vitreous cavity. In the high-dose TFA treatment group, there were still a few inflammatory cells infiltrated; lymphocytes were observed in individual choroidal vessels, together with a few vascular dilatations; however, the numbers of inflammatory cells and dilated vessels were lower than those in the EIU model group. Correspondingly, the histopathological score significantly increased in the EIU model group compared with the NC group. TFA treatment significantly reduced the score, especially in the high-dose TFA treatment group (Figure 3E).
Effect of the Low and High Doses of TFA on Blood Cytokines Levels in EIU Rats
The contents of IFN-γ, IL-10, IL-17, and TGF-β were analyzed using ELISA. It was found that the EIU model rats exhibited notably higher IFN-γ content than NC rats (p < 0.05); rats intraperitoneally injected with low-/high-dose TFA in treatment groups showed effectively reduced IFN-γ content compared with rats in the model group (p < 0.05). Additionally, IL-10 and IL-17 contents in the model group were remarkably higher than those in the NC group (p < 0.05). Moreover, the low-dose TFA treatment group showed significantly higher IL-17 content than the NC group (p < 0.05). The results are shown in Table 3.
The Effect of TFA on IFN-γ, IL-10, IL-17, and TGF-β Levels in Rats Blood Sample (
± s, pg/ml).

Effect of the Administration Route and Dose of TFA on Improving Clinical Symptoms in EIU Rats
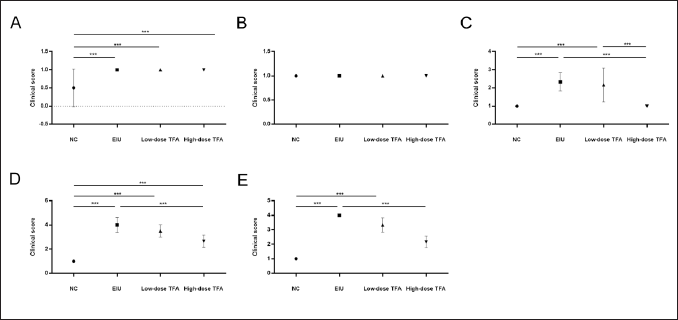
The eyes of rats in three different groups (NC group, EIU model group, and low-/high-dose TFA treatment group) were scored every 2 h. According to the results, at 2 h, the clinical eyeball score of rats in the NC group was notably lower than that in other groups (p < 0.01). At 6 h, rats in the NC group exhibited a dramatically lower clinical eyeball score than rats in the EIU model group and low-dose TFA treatment group (p < 0.05); rats in the EIU model group showed a significantly higher clinical eyeball score compared with those in the high-dose TFA treatment group (p < 0.01); rats intraperitoneally injected with low-dose TFA had a notably higher clinical eyeball score than those intraperitoneally injected with high-dose TFA (p < 0.01). At 8 h, the clinical eyeball score of the NC group was remarkably lower than that of other groups except for the high-dose TFA treatment group (p < 0.01); the EIU model group had a noticeably higher clinical eyeball score than the high-dose TFA treatment group (p < 0.01). At 10 h, it was found that the rat eyeball score of the NC group was dramatically lower than that of the other 3 groups (p < 0.05); the low-dose TFA treatment group exhibited a significantly higher clinical eyeball score of rats than the high-dose TFA treatment group (p < 0.05). The results are shown in Figure 4.
Eyeball Scores of Rats in Each Group at 5 Time Points. (A) 2 h; (B) 4 h; (C) 6 h; (D) 8 h; (E) 10 h.
Discussion
Uveitis is accepted as a common ophthalmic disease in clinics. According to TCM theory, recurrent uveitis is caused by Qi- and Yin-deficiency. Therefore, the principles of Qi-reinforcing and Yin-nourishing should be fully considered in uveitis treatment. HQ, widely accepted as a kind of Qi-reinforcing TCM, can strengthen the middle warmer and invigorate the spleen, as well as reinforce Wei and consolidate the superficial. The main bioactive components of HQ are flavonoids. Recently, it has been shown that TCM has obvious curative effects on uveitis; TCM also has significant advantages in reducing inflammatory recurrence, hormone dependence, and toxic side effects (Hopkins, 2007). However, due to its unclear mechanism and complex components, TCM has not been widely used in clinics.
Network pharmacology is a research method based on the theory of system biology in the field of TCM that analyzes the network of biological systems and selects disease-related signal nodes for the molecular design of target drugs. Network pharmacology analyzes the drug-body interaction according to the molecular targets, thereby guiding the discovery of new drugs, which has become a new research model in the field of TCM (Li et al., 2010). In the present study, based on the analysis using network pharmacology, 84 targets of the active components of TFA were predicted. Among these 84 targets, 22.6% were found to be related to uveitis treatment after searching using DrugBank and other databases, which confirmed the rationality of HQ in treating uveitis. The relevant signaling pathways of HQ in uveitis treatment were obtained following the enrichment analysis of HQ active components and therapeutic targets. The results showed that the TLR signaling pathway and the NLR signaling pathway may be tightly implicated in uveitis treatment by HQ. Moreover, it was found that, in the therapeutic targets of HQ in uveitis treatment, flavonoids accounted for a large proportion. Therefore, we speculated that HQ treatment of uveitis may be mediated by flavonoids.
TLRs and NLRs are pattern-recognition receptors that play important roles in the body’s resistance to foreign microbial infection, which are important components of human innate immunity. TLR4, as the first discovered member of the TLR family, is a crucial molecule in initiating the cellular innate immune response (Qiu et al., 2016). Under the action of TLR4, LPS activates macrophages and enhances their phagocytic activity, which induces the reaction of the body with the originally tolerated autoimmune antigen, thereby resulting in the initiation of uveitis (Li et al., 2013).
MyD88 is validated as a main adaptor protein in the TLR signal transduction pathway, and two TLR signaling pathways have been found so far: a MyD88-dependent signaling pathway and a MyD88-independent signaling pathway. A previous study (Hsu et al., 2015) has pointed out that TLR4 activates its downstream signal molecules via the MyD88-dependent pathway during uveitis pathogenesis; following the activation of corresponding ligands, tumor necrosis factor-associated factor 6 (TRAF-6), crucial for the MyD88-dependent pathway, is activated through the signal cascade, which promotes the activation of protein kinase by mitogens and then the activation of the NF-κB pathway.
As has been evidenced previously, NF-κB is tightly implicated in uveitis pathogenesis, and its activation affects the transcription of inflammatory mediators (such as IL-1β, IL-12, and IL-6) and contributes to the amplification and persistence of the inflammatory process through a feedback loop (Lu et al., 2008). It has been shown that the inflammatory mediator IL-12 can induce helper T cells to differentiate into Th1 cells, thereby promoting the expression of IFN-γ, which can activate inflammatory cells and induce autoimmune diseases. IL-6 and IL-1β can stimulate helper T cells to activate and secrete the key inflammatory factor IL-17 (Kuwabara et al., 2017).
Tang et al. (2018) have reported that blocking the NF-κB pathway using inhibitors helps to suppress the expression of key inflammatory factors, thereby alleviating ocular inflammation. Qiu et al. (2016) have pointed out that isorhamnetin, one of the TFAs, exerts protective effects on the kidney of diabetic nephropathy rats, which may be related to reducing the production of inflammatory mediators and oxidative stress via inhibiting NF-κB signal transduction activity in glomerular mesangial cells. Moreover, a study by Li et al. (2013) has also suggested that TFA may inhibit the NF-κB signaling pathway to suppress the expression of proinflammatory cytokines, thereby playing an anti-inflammatory role in endotoxin-induced mastitis. From all of the above, it was indicated that TFA may participate in body immune regulation via negatively regulating the NF-κB pathway. Therefore, it was speculated that the therapeutic effects of high-dose TFA on uveitis development may be mediated by the inhibition of the NF-κB signal transduction process.
In this study, SD rats were induced for EIU model establishment by injecting S. typhi LPS normal saline solution into rat plantars. Model rats were intraperitoneally injected with different low-/high-dose TFA for treatment, with NC rats and EIU model rats as controls, and the curative effect of TFA on uveitis was evaluated. IFN-γ, TGF-β, IL-10, and IL-17 contents were measured using ELISA. The eyeball cell structure of rats was observed using H&E staining. A previous study has pointed out that IFN-γ and IL-17, as the marker cytokines of Th1 and Th17, respectively, can enhance the inflammatory response and then aggravate EAU development, with their high concentrations indicative of inflammation aggravation and their development trends consistent with disease development trends (Bing et al., 2019). IL-10, secreted by regulatory T (Treg) cells and Th2 cells, inhibits inflammatory cell aggregation and the functional expression of inflammatory cells and slows down inflammation development, and elevated IL-10 is positively correlated with disease remission (Rizzo et al., 1998). TGF-β is an immunosuppressive cytokine that plays a key role in Th17 and Treg cell differentiation (Lee, 2018). TGF-β and IL-10, as the main effector factors of regulatory effector T cells, play roles in negative immune regulation and maintaining autoimmune tolerance in uveitis. During uveitis development, serum TGF-β and IL-10 concentrations increase first and then remain at a high level in the recovery period, which is one of the main mechanisms mediating the natural recovery of EAU (Ke et al., 2011). In the present study, the results showed that EIU model rats exhibited aggravated ocular inflammation as well as elevated IFN-γ, TGF-β, IL-10, and IL-17 contents compared with NC rats, which indicated the high activation of helper T cells and Treg cells in patients. Following high-dose intraperitoneal injection of TFA, IFN-γ and IL-17 levels were notably decreased and basically returned to the normal level in model rats, along with the alleviation of uveitis; this suggested that high-dose TFA contributed to downregulating the high activity state of Th1 and Th17, thereby alleviating uveitis. Additionally, the IL-10 level was markedly higher in the model group than that in the NC group, which was then decreased to a certain extent after high-dose TFA treatment. However, inconsistent with the existing research results, little change in TGF-β level was observed after different treatments, which may be due to the fact that TFA can effectively inhibit the inflammatory response of uveitis too early, thereby shortening the disease course and reducing body autoimmune activity, which therefore caused a reduced IL-10 level and no significant change in TGF-β level. From all of the above, TFA can alleviate uveitis progression, and its mechanism may be closely related to the NLR signaling pathway, the TLR signaling pathway, and NF-κB signal transduction. High-dose TFA promised to be a new choice for uveitis control.
Conclusion
This study explored the molecular targets and pathways of TFA in uveitis treatment using network pharmacology technology. Based on animal experiments, it was found that TFA could effectively reduce uveitis-caused eye tissue damage and reduce IFN-γ, TGF-β, IL-10, and IL-17 levels. Furthermore, the underlying mechanism was investigated by combining the results of network pharmacology and experimentation. The results suggested that TFA may inhibit the related inflammatory factors in uveitis treatment via regulating the NLR signaling pathway and the TLR signaling pathway. This study verifies the effectiveness of HQ in uveitis treatment and provides a new perspective for the clinical therapy of uveitis. However, there are some limitations to this study. We only confirmed the involvement of the NLR signaling pathway and the TLR signaling pathway in the therapeutic effects of TFA in uveitis, and we failed to further clarify the specific mechanism. In addition, more comprehensive and in-depth in vitro and in vivo experiments will be needed in the future to further explore the related mechanisms, thereby providing a more reliable theoretical basis for uveitis therapy with TFA.
The Top 20 KEGG Pathway Enrichment of Potential Targets of HQ Active Components.
Footnotes
Summary
HQ may modulate uveitis progress via the NLR, apoptosis, TLR, MAPK, and CAM signaling pathways based on network pharmacological analysis.
High-dose TFA could improve clinical symptoms in EIU rats.
High-dose TFA could decrease the serum level of the inflammatory factors IFN-γ and IL-10 in EIU rats.
Abbreviations
EIU: Endotoxin-induced uveitis; HQ: Huangqi; PPI: Protein-protein Interaction; TFA: Total flavonoids of Astragalus; TCM: Traditional Chinese medicine; QHRGF: Qinghuo Rougan Mingmu Formula; EAU: Experimental autoimmune uveitis; OB: Oral bioavailability; DL: Drug-likeness; CC: Cellular component; MF: Molecular function; BP: Biological process; H&E: Hematoxylin and eosin; KEGG: Kyoto Encyclopedia of Genes and Genomes; NLR: Nod-like receptor; TLR: Toll-like receptor; MAPK: Mitogen-activated protein kinase; CAMs: Cell adhesion molecules; IPL: Inner plexiform layer; INL: Inner nuclear layer; OPL: Outer plexiform layer; ONL: Outer nuclear layer; NC: normal control.
Acknowledgments
The authors would like to thank the OriEditing team for polishing the English language.
Authors’ Contributions
Jing Lu designed the research and wrote the manuscript. Yujie Wu, Yijing Yang, and Jing Zhou performed the majority of experiments. Bo Huang and Chengfeng Xie performed the figures and the statistical analysis. Qinghua Peng and Xiaohuan Zhang developed the conceptual design of the study. All authors read and approved the final manuscript.
Declaration of Conflicting of Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All the procedures were approved by the Institutional Animal Care and Use Committees of the Affiliated Cancer Hospital of Xiangya School of Medicine, Central South University (approval number: 2019-14).
Funding
This study was supported by the National Natural Science Foundation of China (82104936), the Natural Science Foundation of Hunan Province (2021JJ30498), the Hunan Provincial Postgraduate Innovation Fund Key Project (CX2018B461), the 2019 Hunan University Student Innovation and Entrepreneurship Training Program Project (1686), the 2020 Hunan Provincial University Students Innovation and Entrepreneurship Training Program (2599), the Hunan University of TCM Fundamental Medicine First-Class Discipline Open Fund (2018JCYX13), the Academician Liu Liang Workstation Guidance Project (22YS003), the Central Finance Supports Local University Construction Project, the State Administration of Traditional Chinese Medicine Key discipline construction project of ophthalmology, and the Hunan province key discipline construction project of TCM and five sense organs.
