Abstract
Background
Hyperoside is a potential liver cell and mitochondrial protector, but there is no evidence to suggest that hyperoside can effectively treat doxorubicin (DOX) related liver toxicity.
Objectives
This study aims to determine the role of hyperoside in DOX related liver toxicity and liver cell damage in vivo and in vitro.
Materials and Methods
A mouse model of liver toxicity was induced by intraperitoneal injection of DOX, and hyperoside was administered orally for treatment. During this period, the weight and food intake of the mice were recorded. The morphology of mouse liver tissue was observed by hematoxylin and eosin staining. Oxidative stress and inflammatory status in mice were observed through serum inflammatory factors and liver oxidative stress markers. Mitochondrial damage is determined by the degree of mitochondrial DNA damage. Western blotting and reverse transcription polymerase chain reaction (RT-PCR) were used to detect the expression of pyroptosis and genes in the liver and liver cells.
Results
Hyperoside can effectively treat DOX-induced oxidative stress and inflammatory status, liver function damage, and hepatocyte necrosis in mice. Further research suggests that the beneficial effect of hyperoside is achieved by inhibiting mitochondrial DNA damage in liver cells.
Conclusion
This study indicates that hyperoside improves DOX-induced liver toxicity by inhibiting mitochondrial DNA damage.
Introduction
Although doxorubicin (DOX) is an antineoplastic drug used to treat various types of cancer, it also exerts a certain degree of toxicity on the liver while delivering its therapeutic effects (Almajidi et al., 2023; Ashrafizadeh et al., 2021). The hepatotoxicity of DOX, manifested primarily through hepatocellular injury and abnormal liver function, arises from either its direct toxic effects on hepatocytes or its disruption of the liver’s metabolic and detoxifying processes (Alherz et al., 2023; Prasanna et al., 2020). In addition to directly damaging hepatocytes and interfering with liver metabolism, DOX may also cause hepatotoxicity through other pathways. For example, DOX may cause immune-mediated liver damage or interact with other drugs, leading to hepatotoxicity (Sasmito et al., 2017). The hepatotoxicity mechanism of DOX may be related to its chemical structure and metabolic processes (Sasmito et al., 2017; Sritharan et al., 2022). In the liver, DOX undergoes a series of enzymatic reactions and is converted into various metabolites, some of which may have direct toxic effects on hepatocytes, leading to hepatocyte damage and abnormal liver function (Sritharan et al., 2022; Zhou et al., 2021). In addition, DOX may also interfere with the metabolism and detoxification function of the liver, leading to abnormal metabolism of other drugs or chemicals in the body, thereby causing hepatotoxicity (Zhou et al., 2021).
Hyperoside is an effective component of Traditional Chinese Medicine extracted from the perennial herbaceous plant of Hypericum in the family Liliaceae, a flavonoid compound with multiple biological activities (Wang et al., 2021). In recent years, hyperoside has been found to have a protective effect on hepatocytes. It can inhibit the apoptosis of hepatocytes, promote hepatocyte regeneration, alleviate liver inflammation, and additionally inhibit the occurrence and alleviate the process of liver fibrosis (Wang et al., 2021; Zeng, Ma et al., 2023). In addition to its protective effect on hepatocytes, hyperoside also has the effect of improving liver function (Wang et al., 2021). It can reduce serum transaminase levels and restore normal liver function (Shi et al., 2019). In addition, hyperoside can also eliminate free radicals in the liver, resist oxidation, and prevent oxidative damage to the liver (Kwon et al., 2019).
Mitochondria are the main energy production sites within cells and are one of the key factors for cell survival and death (Kramer & Bressan, 2018). When DOX enters cells, it may bind to the mitochondrial membrane, leading to mitochondrial dysfunction or death (Ding et al., 2023). This kind of damage may be due to the interference of DOX with electron transfer in the respiratory chain of mitochondria or its interaction with mitochondrial DNA, resulting in DNA damage or mutation (Zhang et al., 2020). In addition, mitochondrial damage may also trigger inflammatory reactions and oxidative stress, further exacerbating cellular damage and disease progression (Li et al., 2020). Hyperoside can eliminate free radicals in cells, reduce lipid peroxidation on mitochondrial membranes, protect mitochondria from oxidative stress and apoptosis, and thus protect their structure and function (Li et al., 2008). In addition, hyperoside can also inhibit the release of proapoptotic factors in mitochondria, block the transduction of proapoptotic signals, and further protect mitochondria from damage (Boukes & van de Venter, 2016). Mitochondrial damage can also lead to cell pyroptosis. Cellular pyroptosis is a programmed necrotic cell death mediated by gasdermin, characterized by the formation of inflammasomes and the activation of caspase and gasdermin, accompanied by the release of a large number of proinflammatory factors such as interleukin (IL)-1 and IL-18, which amplifies the inflammatory response (Tan et al., 2021; Yuan & Ofengeim, 2024). When mitochondrial damage occurs, the inner and outer membranes are permeable, electron transport is disrupted, reactive oxygen species and mitochondrial autophagy are induced, and soluble mitochondrial proteins and DNA are released, accelerating and enhancing cell apoptosis (Liu, Xia et al., 2021; Tan et al., 2021). The excessive activation of cell pyroptosis can trigger an inflammatory cytokine storm in the body, leading to fatal inflammatory diseases such as sepsis (Wei et al., 2023).
However, it has not been reported whether hyperoside can prevent and treat DOX-related liver toxicity by protecting mitochondria and regulating hepatocyte pyroptosis.
Materials and Methods
Reagents
Hyperoside (HY-N0452) was purchased from MCE. DOX (A183027) was purchased from Shanghai Aladdin Biochemical Technology Co., Ltd. The alanine aminotransferase (ALT) (C009-2-1), aspartate aminotransferase (AST) (C010-2-1), total cholesterol (TC) (A111-1-1), triglycerides (TG) (A110-1-1), high-density lipoprotein (HDL) (A112-1-1), low-density lipoprotein (LDL) (A113-1-1), IL-1β (H002-1-2), IL-6 (H007-1-1), and tumor necrosis factor-alpha (TNF-α) (H052-1-2) ELISA kits were purchased from Nanjing Jiancheng Bioengineering Institute (Nanjing, China). The primary antibodies NLR family pyrin domain containing 3 (NLRP3) (68102-1-Ig, 1:500), IL-1β (16806-1-AP, 1:1000), IL-18 (10663-1-AP, 1:1000), apoptosis-associated speck-like protein containing a CARD (ASC) (10500-1-AP, 1:5000), Caspase 1(p20, 1:1000) (22915-1-AP), and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) (10494-1-AP, 1:1000) were purchased from Wuhan Proteintech Biotechnology Co., Ltd. (Wuhan, China).
Animal
Twenty male C57BL/6 mice, weighing between 18 and 20 g and aged 6–8 weeks, were acquired from Chengdu Enswell Biotechnology Co., Ltd. The mice were randomly assigned to four distinct groups: a control group receiving only the solvent, a hyperoside group receiving 50 mg/kg of hyperoside in addition to the solvent, a DOX group receiving 3 mg/kg of DOX with the solvent, and a combined hyperoside + DOX group receiving both 3 mg/kg of DOX and 50 mg/kg of hyperoside. DOX was administered intraperitoneally, while hyperoside was given through gavage. All animals were housed in a standard laboratory environment, maintained at 22℃ with a 12-h light/dark cycle and continuous ventilation. They had unrestricted access to water and food. Weekly measurements were taken to monitor their weight and food intake.
Liver and Serum Sample Collection
A blood sample of 0.5 mL was extracted from the orbital vein and centrifuged at 4℃ and 300 g for a duration of 20 min, resulting in the isolation of the supernatant, known as serum. Following this, the chest and abdominal cavity of the mice were surgically accessed under stringent sterile conditions. Subsequently, sterile perfusion of phosphate-buffered saline (PBS) was initiated from the left ventricle, continuing until the liver turned white, indicating that the blood had been successfully washed out. Finally, liver samples were collected for further analysis.
Histopathological Examinations
Immerse the liver sample in a 4% paraformaldehyde solution for a duration of 24 h to fix it, and subsequently dehydrate it using an ethanol solution. Proceed by embedding the sample in paraffin and slicing it into thin sections ranging from 3 to 5 µm. Adhering to the kit’s instructions, stain the sections with hematoxylin and eosin (HE) for 2 h, and finally inspect the specimens under an optical microscope for analysis.
Serum Biochemical Markers
Adhering to the operational guidelines of multiple kits, the serum concentrations of TC, TG, HDL, LDL, ALT, AST, IL-1β, IL-6, and TNF-α were accurately measured. The absorbance values for each well were subsequently recorded at 450 nm using a microplate reader.
Hepatocytes Grouping
After anesthetizing the C56BL/6 mice, they were humanely euthanized by decapitation. Subsequently, the liver was perfused with physiological saline to efficiently eliminate blood and other impurities. Then, collagenase was utilized to digest the liver tissue blocks, facilitating the breakdown of intercellular adhesions. The liver cells were subsequently separated by centrifugation and cultured in designated groups. Hepatocytes were categorized into four distinct groups: (a) control group, (b) hyperoside group, (c) DOX group treated with 1 µM DOX, and (d) hyperoside + DOX group treated with 50 µM hyperoside and 1 µM DOX.
Western Blot
Take an appropriate quantity of tissue (thoroughly homogenized using a homogenizer) or cells (disrupted by an ultrasonic crusher) and immerse it in RIPA protein lysis buffer containing 1% protease inhibitor (sourced from Shanghai Biyuntian Biotechnology Co., Ltd.) for 1 h at 4℃. Subsequently, centrifuge the mixture in a 4℃ centrifuge at 12,000 rpm for 15 min to isolate the protein stock solution. Proceed with protein concentration testing by adhering to the operating instructions provided with the BCA protein concentration detection kit (supplied by Shanghai Biyuntian Biotechnology Co., Ltd.). Mix the protein stock solution with the protein loading buffer (from Shanghai Biyuntian Biotechnology Co., Ltd.) and incubate in water at 100℃ for 30 min. Following this, perform electrophoresis using 12% SDS-PAGE, membrane transfer, and protein blochyperosidede utilizing the prepared protein lysates. Then, incubate the membrane overnight at 4℃ with the primary antibodies, followed by a 2-h incubation with the secondary antibodies. After thorough washing, analyze the protein expression patterns using BeyoECL-Plus and ImageLab software. GAPDH serves as an internal reference for normalization.
PCR
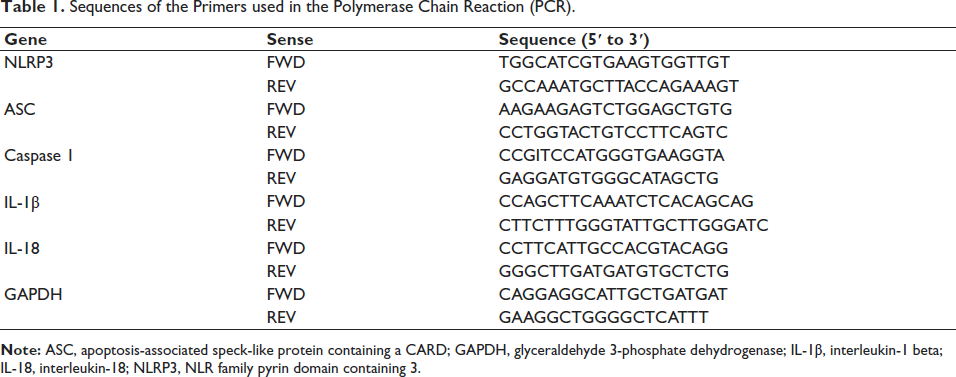
Wuhan Saiweier Biotechnology Co., Ltd., supplied TRIzol reagents for the study, facilitating the isolation of ribonucleic acid (RNA) from tissues or cells in accordance with the manufacturer’s instructions. Additionally, Tahyperosidera Bio, Inc., contributed to the PrimeScript reverse transcriptase kit, enabling the synthesis of RNA complementary DNA (cDNA) as per the provided guidelines. Furthermore, the SYBR PremixEX Taq kit, also supplied by Tahyperosidera Bio, Inc., was utilized for qRT-PCR in this study. The qRT-PCR thermal cycling protocol comprised an initial denaturation step of 10 min at 95℃, followed by repeated cycles of denaturation for 15 s at 45℃ and extension for 30 s at 64℃. Relative gene expression levels were calculated using the 2−∇∇CT method, with β-actin serving as the internal reference gene. The primer sequences utilized in this study were sourced from Sangon Biotech and are presented in Table 1.
Sequences of the Primers used in the Polymerase Chain Reaction (PCR).
Statistical Analysis
The normality and homogeneity of variance of the data were verified through one-way or two-way analysis of variance (ANOVA). Significant differences among groups were further analyzed using Tukey’s multiple comparison post hoc test. Statistical significance was set at p < 0.05.
Results
Hyperoside inhibited the Abnormality of Body Weight and Feed Consumption caused by DOX
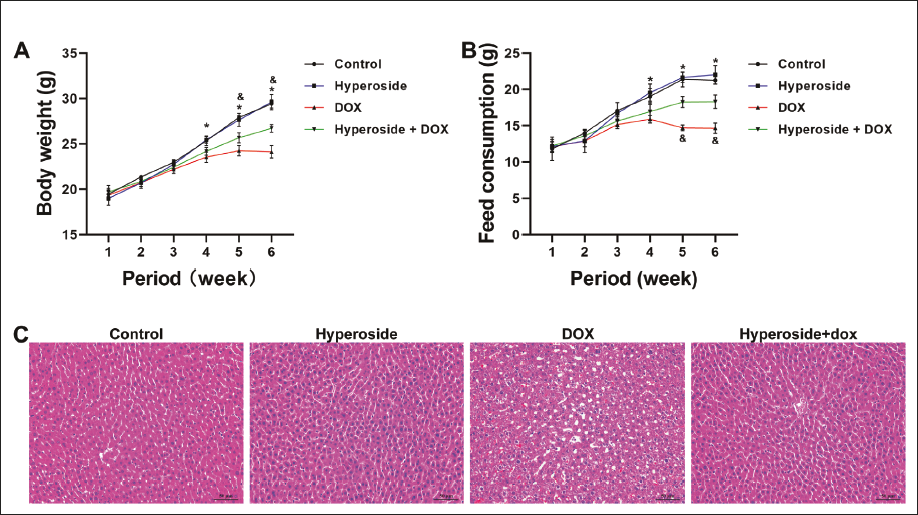
During the 6-week experimental period, DOX administration resulted in a gradual decrease in mouse weight, commencing from the 4th week as depicted in Figure 1A (p < 0.05, DOX vs. Control). Conversely, the therapeutic effects of hyperoside became evident in the 5th week, resulting in a notable increase in the weight of the mice (p < 0.05, hyperoside + DOX vs. DOX). Analogously, DOX administration caused a reduction in food intake among the mice, as shown in Figure 1B (p < 0.05, DOX vs. Control). However, hyperoside administration also had a positive impact, increasing the food intake of the mice, as illustrated in Figure 1B (p < 0.05, hyperoside + DOX vs. DOX).
Hyperoside improves Liver Abnormalities caused by DOX
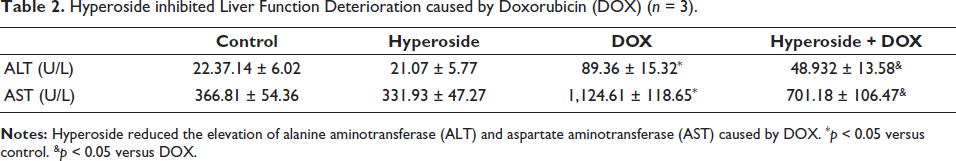
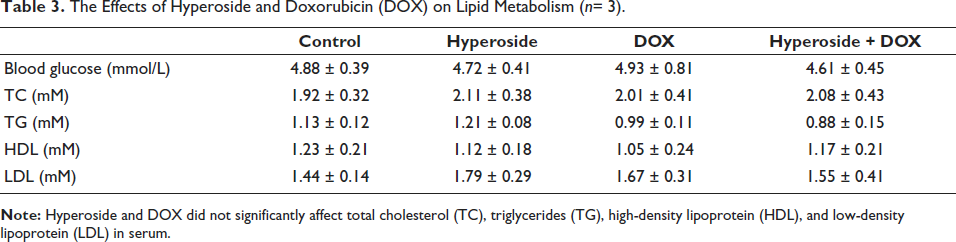
Research has revealed that DOX administration in mice results in unclear cell boundaries, cell fusion, disordered cell arrangement, enlarged intercellular gaps, and abnormal cell nuclei within the liver, as observed in Figure 2A (p < 0.05, DOX vs. Control). Notably, hyperoside administration effectively reversed these pathological changes (Figure 2A, p < 0.05, hyperoside + DOX vs. DOX). Furthermore, liver function testing of mouse serum indicated that DOX significantly elevated serum ALT and AST levels (Table 2, p < 0.05, DOX vs. Control), whereas hyperoside effectively reduced these liver function indicators (Table 2, p < 0.05, hyperoside + DOX vs. DOX). It is worth mentioning that DOX did not significantly alter glucose and lipid metabolism indicators in the mouse liver, including blood glucose, TC, TG, HDL, and LDL (Table 3, p < 0.05).
Hyperoside inhibited Liver Function Deterioration caused by Doxorubicin (DOX) (n = 3).
The Effects of Hyperoside and Doxorubicin (DOX) on Lipid Metabolism (n= 3).
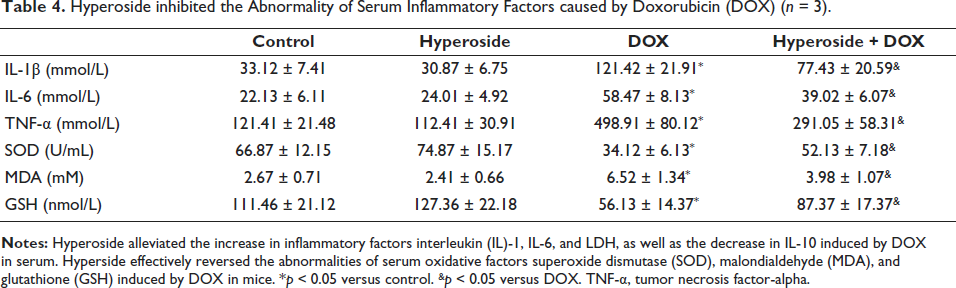
Hyperoside inhibited the Abnormality of Serum Inflammatory Factors and Oxidation Factors caused by DOX
As depicted in Table 4, exposure to DOX notably elevated the levels of serum IL-1β, serum IL-6, serum TNF-α, and serum MDA, while concurrently decreasing serum SOD and GSH levels in comparison to the control group. Remarkably, the hyperoside + DOX group exhibited significant alleviation in these serum indicators when compared to the DOX group alone. When hyperoside was administered solely, it did not exert a significant influence on the aforementioned biomarkers.
Hyperoside inhibited the Abnormality of Serum Inflammatory Factors caused by Doxorubicin (DOX) (n = 3).
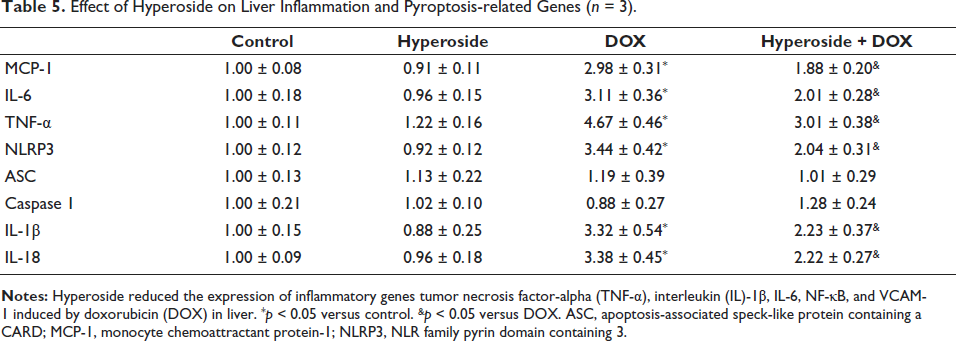
Hyperoside-inhibited DOX-induced Liver Inflammation and Pyroptosis Gene Abnormalities
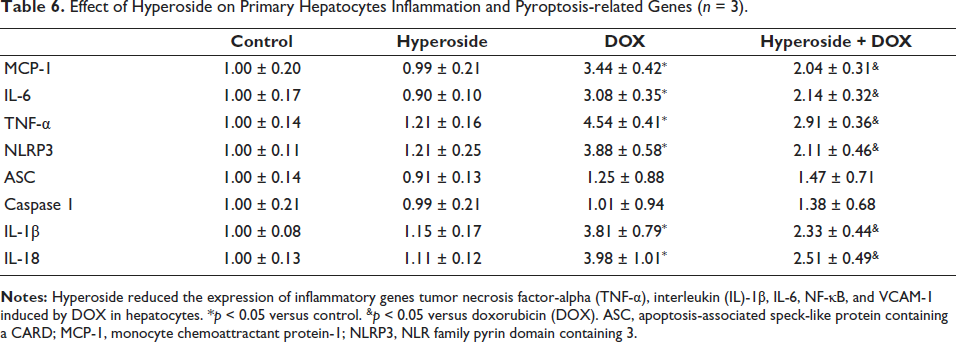
As shown in Table 5, DOX significantly increased liver inflammatory genes MCP-1, IL-1β, IL-6, and TNF-α genes and pyroptosis genes NLRP3, IL-1β, and IL-18 compared to the control group. These genes in the hyperoside + DOX group showed significant relief compared to the DOX group. However, there was no significant change in the mRNA levels of ASC and Caspase1, regardless of whether they were treated with DOX and/or hyperoside. When hyperoside is used alone, there is no significant impact on the aforementioned inflammatory genes.
Effect of Hyperoside on Liver Inflammation and Pyroptosis-related Genes (n = 3).
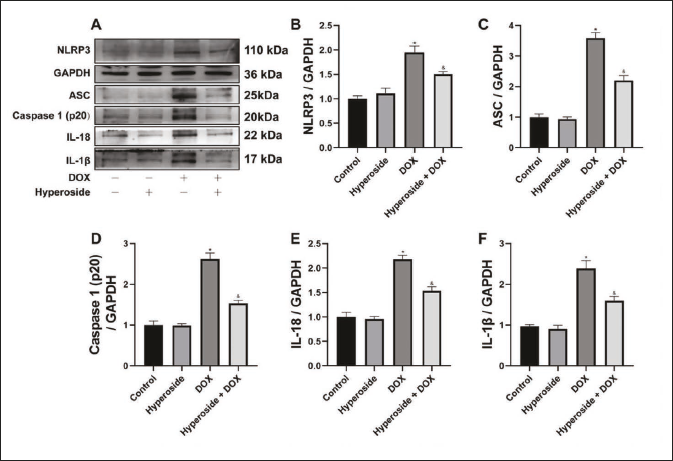
Hyperoside regulates DOX-induced Hepatocyte Pyroptosis Pathway Abnormalities
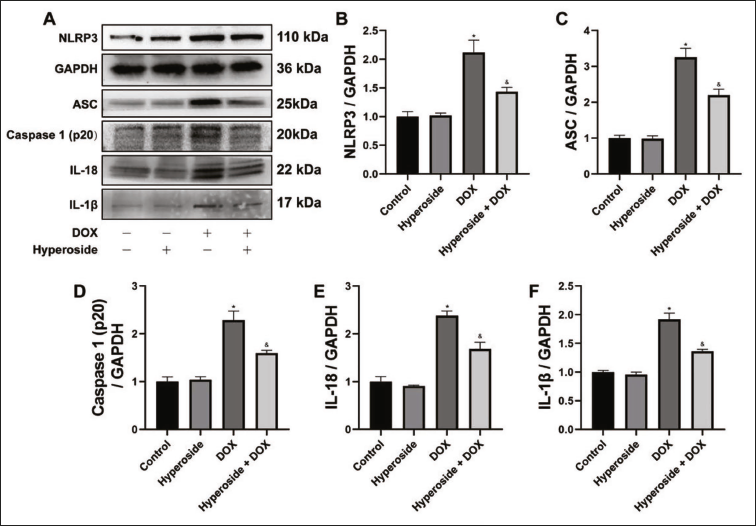
As shown in the hepatocyte WB results, the expression of pyroptosis protein (Figure 2A) in the hepatocyte of DOX group increased compared to the control group, such as NLRP3, ASC, Caspase 1(p20), IL-1β, and IL-18 (Figure 2B–F). However, hyperoside alleviated the increased expression of pyroptosis protein in the mice liver compared to the DOX group.
Hyperoside inhibited DOX-induced Hepatocyte Inflammatory and Pyroptosis Genes, and Pyroptosis Protein Abnormalities
DOX significantly increased inflammatory genes MCP-1, IL-1β, IL-6, and TNF-α genes and pyroptosis genes NLRP3, IL-1β, and IL-18 compared to the control group in hepatocytes. These genes in the hyperoside + DOX group showed significant relief compared to the DOX group in hepatocytes. However, there was also no significant change in the mRNA levels of ASC and Caspase1, regardless of whether they were treated with DOX and/or hyperoside. When hyperoside is used alone, there is no significant impact on the aforementioned inflammatory genes. As shown in the hepatocyte WB results, the expression of pyroptosis protein (Figure 3A) in the hepatocyte of DOX group increased compared to the control group, such as NLRP3, ASC, Caspase 1(p20), IL-1β, and IL-18 (Figure 3B–F). However, hyperoside alleviated the increased expression of pyroptosis protein in the mice liver compared to the DOX group.
Hyperoside regulates DOX-induced Hepatocyte Mitochondrial DNA Damage
To further investigate how hyperoside regulates hepatocyte pyroptosis, we measured the level of mitochondrial DNA damage in hepatocytes. As shown in Tables 6 and 7, compared with the control group, the DOX group caused mitochondrial DNA damage of hepatocytes in vivo and in vitro. However, compared to the DOX group, hyperoside reduced mitochondrial DNA damage of hepatocytes in vivo and in vitro.
Effect of Hyperoside on Primary Hepatocytes Inflammation and Pyroptosis-related Genes (n = 3).
The Effect of Hyperoside on Mitochondrial DNA (mtDNA) Damage in Liver or Primary Hepatocytes (n = 3).
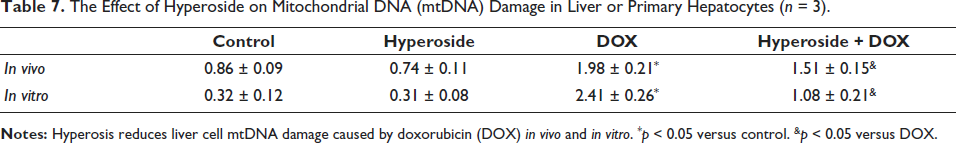
Discussion
As research delves deeper into the realm of hepatocyte pyroptosis, a mounting body of evidence underscores the pivotal regulatory role played by hyperoside in this process. Pyroptosis, a form of programmed cell death, is characterized by cellular swelling, membrane rupture, and DNA degradation (Boukes & van de Venter, 2016; Tan et al., 2021; Yuan & Ofengeim, 2024). In the context of hepatocytes, pyroptosis often correlates with the progression and exacerbation of liver diseases (Gan et al., 2022). Hyperoside, a compound with a broad spectrum of biological activities, plays a vital role in numerous biological processes, encompassing anti-inflammatory, antioxidant, and antitumor effects (Kong et al., 2020; Liu, Zhang et al., 2021; Zhang, Li et al., 2023). Current investigations reveal that hyperoside, at specific concentrations, can effectively inhibit cell pyroptosis (Zhang, Li et al., 2023). This inhibitory effect may stem from hyperoside’s antioxidant and anti-inflammatory properties, as it reduces the generation of reactive oxygen species and inhibits the release of inflammatory mediators, thereby mitigating hepatocellular damage (Liu, Zhang et al., 2021; Zhang, Shang et al., 2023). Furthermore, hyperoside exerts its inhibitory influence on hepatocyte pyroptosis by modulating certain signaling pathways, such as NF-κB and MAPK (Kim et al., 2022; Li et al., 2023). This intricate interplay underscores the multifaceted mechanisms by which hyperoside contributes to preserving hepatic health and mitigating the detrimental effects of pyroptosis.
DOX, a classic anthracycline anticancer drug, is notorious for its organ toxicity, particularly affecting the liver, heart, and bone marrow (Huang et al., 2024; Wei et al., 2019; Wu et al., 2022). While previous studies have predominantly focused on DOX’s most prevalent cardiotoxicity, its hepatic toxicity has often been overlooked, leading to severe consequences that further jeopardize cancer patients’ health. The hepatotoxicity induced by DOX primarily manifests as hepatocellular damage, potentially resulting in necrosis, steatosis, fibrosis, cholestasis, and vascular injury (Zhao et al., 2019; Zhou et al., 2021). These injuries, if left unchecked, can progress to liver failure (Sordi et al., 2024). The underlying mechanism of this toxicity may be attributed to the oxidative free radicals generated by DOX in vivo, which induce lipid peroxidation, disrupt cell membrane structures, and impair cellular metabolism and function (Afsar et al., 2019; Liu & Tan, 2003). Additionally, hepatocyte pyroptosis, a critical component in the development of DOX-related hepatotoxicity, may exacerbate liver damage by promoting inflammatory responses and tissue injury (Liu et al., 2022). During DOX metabolism, a surge of oxidative free radicals is generated, which elevates intracellular oxidative stress levels, thereby activating pyroptosis-related signaling pathways including the NLRP3 inflammasome, ultimately leading to cell pyroptosis (Tan et al., 2022; Zha et al., 2023). Furthermore, DOX-generated free radicals can damage mitochondrial structure and function, reducing mitochondrial membrane potential and damaging mitochondrial DNA, thereby facilitating cell pyroptosis (Ding et al., 2023; Li et al., 2008, 2020; Zhang et al., 2020). On the other hand, the pyroptotic process induced by DOX is accompanied by the release of profuse proinflammatory cytokines like TNF-α and IL-1β, which can exacerbate tissue damage and inflammatory reactions (Zeng et al., 2020). In this study, we established a mouse model of DOX-induced hepatic toxicity to investigate the effects of hyperoside on liver function, inflammatory responses, and hepatocyte pyroptosis. Our findings reveal promising therapeutic potential for hyperoside in mitigating DOX-induced liver injury. Specifically, hyperoside exerts its hepatoprotective effects primarily through anti-inflammatory, antioxidant, and antipyroptotic mechanisms.
NLRP3 exerts its function in pyroptosis by assembling into the NLRP3 inflammasome, a multiprotein signaling complex comprising pattern recognition receptors (such as NLRP3), the adaptor protein ASC, and the pro-Caspase-1 (Zeng et al., 2020; Zhang, Shang et al., 2023). ASC, an apoptosis-associated speck-like protein, interacts with the CARD domains of NLRP3 and Caspase-1 through its own CARD domain, facilitating inflammasome assembly and Caspase-1 activation (Xu et al., 2019). Caspase-1, a member of the Caspases family, is paramount in the pyroptotic process, responsible for cleaving substrates like gasdermin D (GSDMD), thereby triggering pyroptosis (Fang et al., 2023). Upon cleavage, GSDMD forms pores in the cell membrane, leading to cell swelling, rupture, and the release of inflammatory mediators (e.g., IL-1β, IL-18), which ignite inflammatory responses and pyroptosis (Fang et al., 2023; Zeng, Ma et al., 2023). In this study, hyperoside treatment significantly downregulated the protein levels of NLRP3, ASC, Caspase-1, IL-1β, and IL-18 in the livers and hepatocytes of DOX-treated mice, revealing hyperoside’s potent therapeutic effect against DOX-induced hepatocyte pyroptosis. Further investigations demonstrated that hyperoside, through its remarkable antioxidant and anti-inflammatory capabilities, effectively reduced oxidative stress marker levels in the blood and decreased the expression of inflammatory gene mRNAs in liver tissues of DOX-induced mice, offering a novel perspective on hyperoside’s hepatoprotective role. Based on these discoveries, we conclude that hyperoside holds promise as a natural therapeutic agent for DOX-related hepatic toxicity.
Conclusion
Our study suggests that hyperoside inhibits liver inflammatory response by activating AMPKα/SIRT1 and ultimately prevents DOX-induced hepatotoxicity. Currently, many plants and their components with anti-inflammatory activity have been studied for the treatment of liver inflammation-related diseases. In fact, AMPKα/SIRT1 may only be a way for hyperoside to inhibit inflammation, and other targets or pathways may also have an impact on inflammation. These findings will help us further understand the toxic side effects of chemotherapy drugs, the potential for prevention and treatment of hyperoside, and the identification of new targets and drugs for the treatment and prevention of DOX-induced liver disease.
Abbreviations
ALT: Alanine aminotransferase; ASC: Apoptosis-associated speck-like protein containing a CARD; AST: Aspartate aminotransferase; DOX: Doxorubicin; GAPDH: Glyceraldehyde 3-phosphate dehydrogenase; GSDMD: Gasdermin D; GSH: Glutathione; HDL: High-density lipoprotein; HE: Hematoxylin and eosin; IL-1β: Interleukin-1 beta; IL-6: Interleukin-6; IL-18: Interleukin-18; LDL: Low-density lipoprotein; MCP-1: Monocyte chemoattractant protein-1; MDA: Malondialdehyde; mtDNA: Mitochondrial DNA; NLRP3: NLR family pyrin domain containing 3; PCR: Polymerase chain reaction; RT-PCR: Reverse transcription polymerase chain reaction; SOD: Superoxide dismutase; TC: Total cholesterol; TG: Triglycerides; TNF-α: Tumor necrosis factor-alpha; WB: Western blot.
Footnotes
Acknowledgments
The research was supported by the Chongqing Natural Science Foundation General Project of China.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
The entire study adhered to the ethical guidelines of the Research Ethics Committee of Chengdu University.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the Chongqing Natural Science Foundation General Project of China (CSTB2022NSCQ-MSX1490).
