Abstract
Objectives
DOX can promote liver cell inflammation and lead to liver cell death. Ka protects and stabilizes liver cells for the treatment of hepatitis, cirrhosis, and other diseases. However, there is no evidence to suggest that Ka is associated with chemotherapy-related liver inflammation.
Materials and Methods
Treat mice with DOX or Ka to induce or treat liver inflammation. Then, the body weight, liver weight, morphological changes, and liver inflammation of the mice were measured. Western blotting and RT-PCR were used to evaluate the AMPKα/SIRT1/NF-κB inflammatory signaling pathway and inflammatory gene expression. Finally, the above signaling pathways were verified in liver cells.
Results
DOX causes liver function damage and liver inflammation in mice. The specific manifestations are abnormal liver tissue structure in DOX mice; abnormal elevation of serum liver function markers ALP, ALT, AST, and GGT levels; abnormal elevation of serum inflammatory factors IL-1β, IL-6, IL-10, and TNF-α levels; and increased expression of liver inflammatory genes NF-κB, IL-1β, IL-6, TNF, and VCAM-1. Ka can effectively prevent and treat these changes. However, there was no significant change in the glucose and lipid metabolism levels of each group of mice. Further research suggests that the inhibitory effect of Ka on DOX-induced liver inflammation is mediated by the AMPKα/SIRT1/NF-κB signaling pathway. Primary liver cell studies have also confirmed the involvement of these signaling pathways and proteins.
Significance
This study demonstrates that Ka can improve DOX-induced liver inflammation, including changes in inflammatory factors or genes in serum and liver tissue. Further research has found that its potential mechanism may be related to the AMPKα/SIRT1/NF-κB signaling pathway.
Introduction
Chemotherapy drugs have always been the fundamental drugs for the treatment of malignant tumors (Knezevic & Clarke, 2020). However, the toxic side effects of chemotherapy drugs are increasingly prominent, such as hepatotoxicity, cardiotoxicity, nephrotoxicity, and bone marrow toxicity (Krzyzanowska et al., 2021). Studies have shown that chemotherapy drugs can damage cellular structure and function, cause systemic inflammation, and lead to irreversible damage and dysfunction of tissue structure (Behranvand et al., 2022). Therefore, in-depth research on the mechanism of inflammation caused by chemotherapy drugs and early prevention and treatment has extremely important theoretical and clinical significance. DOX is a commonly used anthracycline-based anticancer chemotherapy drug with high inhibitory effects on various tumors, but its application is often limited by liver damage (Aljobaily et al., 2020). Research has shown that DOX can promote liver cell inflammation, damage liver cells, and ultimately lead to various types of liver cell death (Aljobaily et al., 2020).
AMPKα is a key molecule widely expressed in metabolic organs that regulates energy metabolism (Rafiei et al., 2019). It is mainly influenced by the ratio of AMP/ATP and can be activated by various stimuli, including cell pressure, exercise, and hormones (Lin & Hardie, 2018; Stephenne et al., 2011). After activation, AMPKα phosphorylates downstream molecular signals, regulating energy metabolism and inflammatory status in the body, and has become a therapeutic target for various liver diseases, such as non-alcoholic fatty liver disease, cirrhosis, liver cancer, and drug-induced liver disease (Bian et al., 2022; Li et al., 2019). On the other hand, AMPKα is the activation target of SIRT1, which can lead to the increase of the NAD+/NADH ratio and the decrease of nicotinamide by activating the NAD + biosynthesis enzyme nicotinamide phosphoribosyltransferase, thus leading to the activation of SIRT1 (Li et al., 2019; Morales-Alamo & Calbet, 2016). SIRT1 can inhibit the activity of NF-κB and regulate the progression of inflammation in liver diseases (Li et al., 2019). Research has shown that SIRT1 can inhibit TNF-α-induced NF-κB activation and downstream reactions by deacetylation of lysine at the 310th position of the Rel/p65 dimer and can also inhibit NF-κB by interfering with IKK activity (Li et al., 2019, 2021). NF-κB activation can promote the expression of adhesion factors; mediate the adhesion of neutrophils, monocytes, T cells, etc., to endothelial cells; and produce inflammatory infiltration (Wang et al., 2022). NF-κB b and p38 MAPK signaling pathways are considered to be related to the intracellular inflammatory process (Wang, Zhao, et al., 2021). The activation of SIRT1 can not only promote the phosphorylation of p38 MAPK in the cytoplasm but also inhibit the expression of NF-κB in the nucleus, thereby reducing the liver inflammatory response (Zhao et al., 2022).
Ka is mainly derived from the rhizome of the ginger plant Kaempferol galanga L., which has been widely used in the food and pharmaceutical product industry (Alam et al., 2020). Research has confirmed that kaempferol can treat diseases in various organs, including liver fibrosis, heart failure, and acute kidney injury (Ashrafizadeh et al., 2020; Xu et al., 2019, 2023; Zhang et al., 2019). The main advantages of Ka as a common active ingredient in traditional Chinese medicine are its easy availability, low price, and the ability to prevent diseases through dietary therapy, but it has not yet been included in the clinical treatment of liver diseases. If its pharmacological effects and main mechanisms of action can be further elucidated, clinical indications can be expanded, and more valuable clinical drugs can be developed, which may bring more benefits to patients.
This evidence strongly confirms that Ka can become a potential liver-protective drug, but its ability to protect the liver, exert anti-inflammatory effects as a key target, and ultimately prevent chemotherapy-related liver damage has not been reported, and research is urgently needed to confirm this.
Materials and Methods
Reagents
Ka (HY-15449) and DOX (HY-15142A) were provided by MCE. HE Staining Kit (C0105M) was purchased from Shanghai Biyuntian Biotechnology Co., Ltd. (Shanghai, China). The ALP (A059-2-2), ALT (C009-2-1), AST (C010-2-1), GGT (C017-2-1), TC (A111-1-1), TG (A110-1-1), HDL (A112-1-1), LDL (A113-1-1), IL-1β (H002-1-2), IL-6 (H007-1-1), IL-10 (H009-1-2), and TNF-α (H052-1-2) assay kits were purchased from Nanjing Jiancheng Bioengineering Institute (Nanjing, China). The primary antibodies p-AMPKα (50081) and AMPKα (5832) were purchased from CST, and SIRT1 (13161-1-AP) and GAPDH (10494-1-AP) were purchased from Wuhan Proteintech Biotechnology Co., Ltd. (Wuhan, China).
Animal
20 male Sprague Dawley rats (180–220 g, eight weeks) were obtained from Beijing Vital River Laboratory Animal Technology Co., Ltd. The rats were randomly divided into four groups: (a) Control group: solvent control, (b) Ka group: 10 mg/kg Ka once a day, (c) DOX group: 3 mg/kg DOX once a week, and (d) Ka + DOX group: 3 mg/kg DOX once a week + 10 mg/kg Ka once a day. DOX was administered intraperitoneally, while Ka was administered by gavage. All the animals were kept in a standard laboratory (12-hour light/dark cycle, 22℃, 24-hour ventilation) and had free access to water and feed. Weight and food intake were measured weekly.
Liver and Blood Sample Collection
Rats were anesthetized by intraperitoneal injection of pentobarbital (50 mg/kg) and then euthanized using the cervical dislocation method. Rat blood was extracted from the rat aorta (3–5 ml), and the conditions for obtaining serum were room temperature, 3,000 rpm, and 20 minutes. The liver was isolated from the abdominal cavity of rats under sterile conditions.
Histopathological Examinations
The liver was immersed in a 4% paraformaldehyde solution for 48 hours and dehydrated in an ethanol solution. Then, it was embedded in paraffin and cut into 3–5 µM slices. According to the operating instructions of the kit, the sections were stained with HE for two hours, and the specimens were examined under an optical microscope (Olympus BX-50; Olympus, Tokyo, Japan).
Serum, Liver, and Hepatocyte Biochemical Markers
According to the operating instructions of several kits, the contents of TC, TG, HDL, LDL, ALP, ALT, AST, GGT, IL-1β, IL-6, IL-10, and TNF-α in serum were measured, and the absorbance values of each hole were read at 450 nm in the microplate reader.
Hepatocyte Grouping
The hepatocyte line Buffalo Rat Liver (BRL, SNL-270) was purchased from Shane Biotechnology Co., Ltd. Hepatocytes were divided into four groups: (a) Control group, (b) Ka group, (c) DOX group: 3 Μm DOX, and (d) Ka + DOX: 10 Μm Ka + 3 Μm DOX. The cultivation conditions were DMEM (Gibco) + 10% FBS (Gibco) + 1% P/S (Shanghai Biyuntian Biotechnology Co., Ltd.).
Western Blot
An appropriate amount of tissue (ground by a homogenizer) or cells (broken by an ultrasonic crusher) was taken and immersed in RIPA protein lysis buffer containing 1% protease inhibitor (Wuhan ServiceBio Biotechnology Co., Ltd.) for 1 hour (4℃). Then, it was centrifuged in a 4℃ centrifuge (12,000 rpm) for 15 minutes to obtain the protein stock solution. Protein concentration testing was performed according to the operating instructions of the BCA protein concentration detection kit (Wuhan ServiceBio Biotechnology Co., Ltd.). It was mixed with protein-loaded buffer (Wuhan ServiceBio Biotechnology Co., Ltd.) and soaked in water at 100℃ for 30 minutes. Then, 12% SDS-PAGE, membrane transfer, and protein blockade were performed using protein lysates. Subsequently, it was incubated overnight (4℃) in primary antibodies (AMPK, 1:1,000), (p-AMPK, 1:1,000), (SIRT1, 1:1,000), and then incubated in secondary antibodies for 1.5 hours. After cleaning, the protein imprinting results were analyzed using BeyoECL-Plus and ImageLab software. GAPDH (1:5,000) served as an internal reference.
PCR
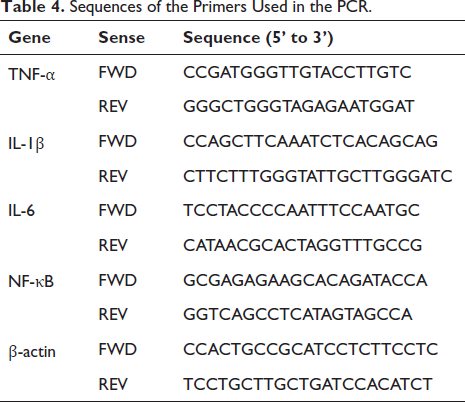
Invitrogen (Shanghai, China) provided TRIzol reagents for this study and isolated RNA from tissues or cells according to the user manual. Takara Bio, Inc. provided the PrimeScript reverse transcriptase kit for this study and synthesized RNA Cdna according to the instruction manual. Similarly, Takara Bio, Inc. provided QRT-PCR using the SYBR PremixEX Taq kit for this study. The thermal cycling conditions of QRT-PCR are as follows: initial denaturation for 10 minutes (95℃), followed by cyclic denaturation for 15 seconds (45℃) and the last 30 seconds (64℃). Adopt 2-ΔΔCT calculation was used to calculate the relative number of tested genes. The internal control was β-actin. The primer sequences used in this study were all provided by Sangon Biotech, as shown in Table 4.
Statistical Analysis
The normal distribution and homogeneity of variance of the data were detected by one-way or two-way ANOVA, and the significant differences between the groups were detected by Tukey’s multiple comparison post hoc test. p ≤ 0.05 was considered statistically significant.
Results
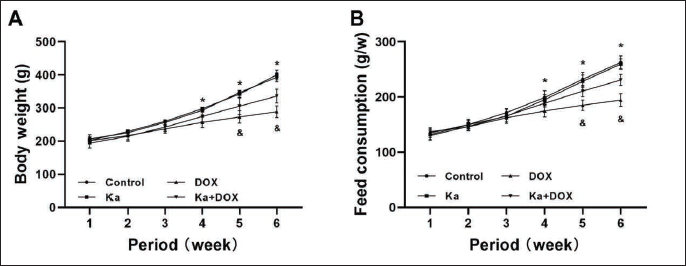
Ka Inhibits the Abnormality of Body Weight and Feed Consumption Caused by DOX
During the six-week model establishment period, the body weight of rats in the DOX group decreased significantly (Figure 1A) compared to the control group, and the weight of rats in the Ka + DOX group increased significantly (Figure 1A) compared to the DOX group. The feed consumption of rats in the DOX group decreased significantly (Figure 1B) compared to the control group, and the feed consumption of rats in the Ka + DOX group increased significantly (Figure 1B) compared to the DOX group. When Ka was used alone, there was no significant difference in body weight and food consumption.
Effects of Ka and DOX on (A) Body Weight and (B) Food Consumption in Rats During Six Weeks of Model Establishment. (Values are expressed as mean ± SD. *p < 0.05 vs. control, & p < 0.05 vs. DOX.)
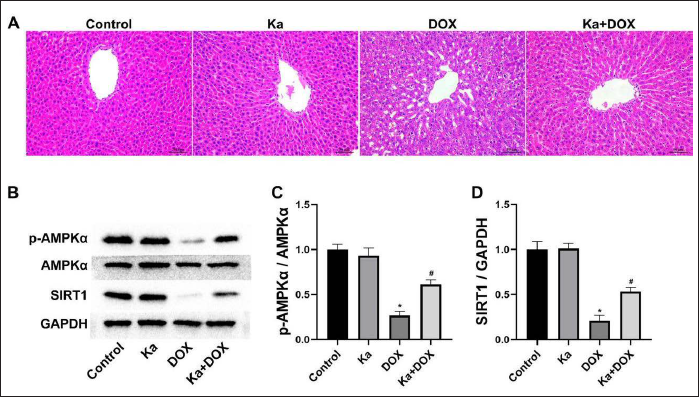
Ka Inhibits the Abnormality of Liver Morphology, Metabolism, and Function Caused by DOX
When observing liver tissue with HE staining, it was found that, compared to the control group, the liver cells in the DOX group had unclear boundaries, fusion, disordered arrangements, increased gaps, and nuclear abnormalities. The above changes in the liver of rats in the Ka + DOX group were relatively mild (Figure 2A).
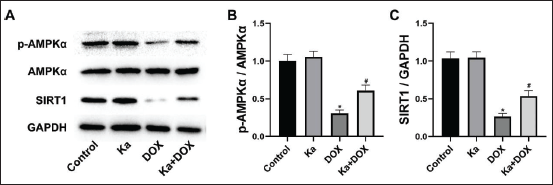
Effects of Ka and DOX on (A) Liver Morphology and (B) AMPKα/SIRT1 Signaling Pathway in Rats. Semiquantitative analysis of AMPKα (C) and SIRT1 (D) protein expression. (Values are expressed as mean ± SD. *p < 0.05 vs. control, & p < 0.05 vs. DOX.)
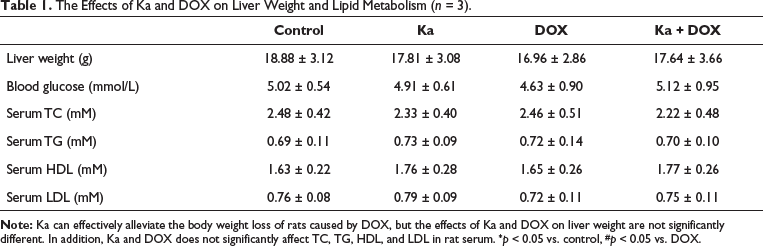
At the end of the experiment, the detection of liver metabolism in rats showed no significant difference in liver weight, and there were no significant changes in blood glucose, serum TC, serum TG, serum HDL, and serum LDL (Table 1). However, the detection of liver function indicators in rat serum revealed a significant increase in serum ALP, serum ALT, serum AST, and serum GGT in the DOX group compared to the control group (Table 2). However, compared to the DOX group, the above changes were relatively mild in the Ka + DOX group rats (Table 2).
The Effects of Ka and DOX on Liver Weight and Lipid Metabolism (n = 3).
Ka Inhibited Liver Function Deterioration Caused by DOX (n = 3).
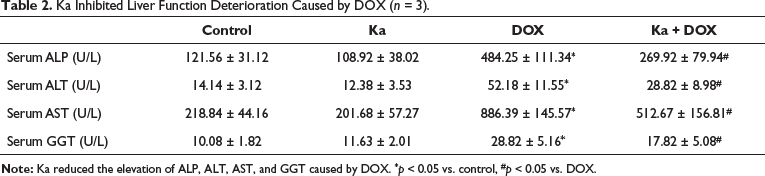
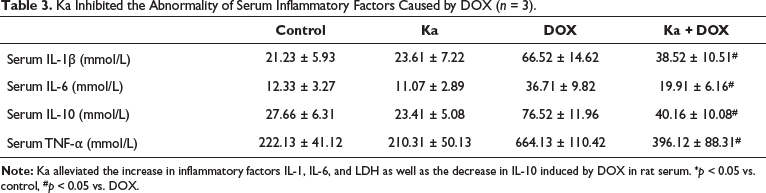
Ka Inhibited the Abnormality of Serum Inflammatory Factors Caused by DOX
As shown in Table 3, DOX significantly increased serum IL-1β, serum IL-6, and serum TNF-α, while serum IL-10 significantly decreased compared to the control group. The serum indicators in the Ka + DOX group showed significant relief compared to the DOX group. When Ka was used alone, there was no significant impact on the aforementioned inflammatory indicators.
Ka Inhibited the Abnormality of Serum Inflammatory Factors Caused by DOX (n = 3).
Sequences of the Primers Used in the PCR.
Ka Regulated DOX-induced Liver AMPKα/SIRT1 Pathway Abnormalities
As shown in the WB results, the expression of AMPKα protein and SIRT1 protein (Figure 2B) in the liver of DOX group rats decreased compared to the control group. However, Ka alleviated the decreased expression of AMPKα protein and SIRT1 protein in the rat liver (Figure 2B) compared to the DOX group. The semi-quantitative analysis further confirmed the above results (Figure 2C, D).
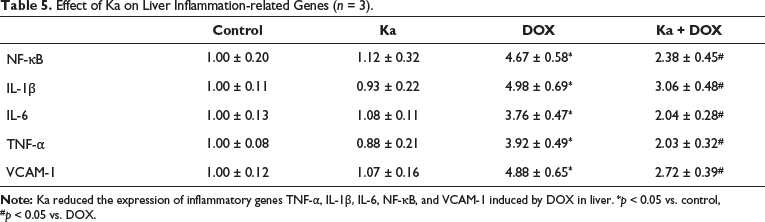
Ka Inhibited DOX-induced Liver Inflammatory Genes Abnormalities
As shown in Table 5, DOX significantly increased NF-κB, VCAM-1, IL-1β, IL-6, and TNF-α genes in the liver compared to the control group. These genes in the Ka + DOX group showed significant relief compared to the DOX group. When Ka was used alone, there was no significant impact on the aforementioned inflammatory genes.
Effect of Ka on Liver Inflammation-related Genes (n = 3).
Ka Regulated DOX-induced Hepatocyte AMPKα/SIRT1 Pathway Abnormalities
As shown in the hepatocyte WB results, the expression of AMPKα protein and SIRT1 protein (Figure 3A) in the hepatocytes of DOX group rats decreased compared to the control group. However, Ka alleviated the decreased expression of AMPKα protein and SIRT1 protein in the rat liver (Figure 3A) compared to the DOX group. The semi-quantitative analysis further confirmed the above results (Figure 3B,C).
Effects of Ka and DOX on (A) AMPKα/SIRT1 Signaling Pathway in Hepatocytes. Semiquantitative analysis of AMPKα (B) and SIRT1 (C) protein expression. (Values are expressed as mean ± SD. *p < 0.05 vs. control, & p < 0.05 vs. DOX.)
Ka Inhibited DOX-induced Hepatocyte Inflammatory Genes Abnormalities
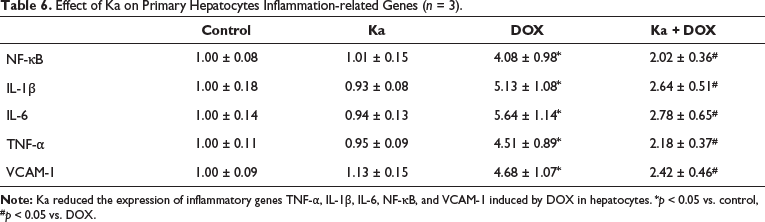
As shown in Table 6, DOX significantly increased NF-κB, VCAM-1, IL-1β, IL-6, and TNF-α genes in hepatocytes compared to the control group. These genes in the Ka + DOX group showed significant relief compared to the DOX group. When Ka was used alone, there was no significant impact on the aforementioned inflammatory genes.
Effect of Ka on Primary Hepatocytes Inflammation-related Genes (n = 3).
Discussion
In this study, DOX-induced hepatotoxicity in mice showed abnormal elevation of ALT, AST, ALP, GGP, and other indicators, which are often used in clinical practice to evaluate the liver function status of patients (Liu et al., 2018; Protchenko et al., 2021; Wang et al., 2003; Wang, Cai, et al., 2021). In clinical practice, when a liver injury occurs, inflammatory markers can often serve as early prediction and efficacy evaluation indicators for liver toxicity, such as elevated CRP, IL-1, IL-6, MCP-1, TNF-α, etc., while decreasing IL-10 (Handa et al., 2019; Ligthart et al., 2018). Therefore, we speculate that reducing liver inflammation levels may be the key to treating DOX-related hepatotoxicity and have added detection of inflammatory factors in mouse serum and liver in the study. To investigate how inflammatory factors in liver cells erupt, we tested AMPKα/SIRT1. AMPKα is widely expressed in various cells, and after activation, it can further activate downstream molecular signals and regulate cell energy metabolism and inflammatory status (Ma et al., 2022). In research on liver diseases, it has been found that AMPKα is a key target for treatment (Chen et al., 2022). On the other hand, AMPKα can activate SIRT1 by activating the NAD + biosynthesis enzyme Nampt, which can inhibit the activity of NF-κB and TNF-α, ultimately regulating the progression of inflammation (Morales-Alamo & Calbet, 2016). Numerous studies have also demonstrated that regulating AMPKα/SIRT1/NF-κB can effectively treat liver inflammation caused by various reasons (Li et al., 2019; Xu et al., 2021; Tian et al., 2016).
The more important finding of this study is that Ka, as a potential therapeutic drug for various liver diseases, can effectively alleviate DOX-induced liver injury by activating the expression of AMPKα/SIRT1 and inhibiting the production of inflammatory factors. In this study, the levels of CRP, IL-1, IL-6, MCP-1, TNF-α, etc., in the liver tissue of rats treated with Ka decreased, while the levels of IL-10 increased. As a star product among the effective ingredients of Traditional Chinese Medicine, Ka has been proven to have various beneficial effects, such as lipid-lowering, anti-inflammatory, antioxidant, and anti-apoptotic effects in various organs (Jin et al., 2021). Ka plays a cardioprotective role in ventricular remodeling after acute myocardial infarction through the NF-κB/NLRP3/Caspase-1 pathway (Hua et al., 2022). Some studies have also pointed out that Ka is a potential natural agonist of AMPKα, which can improve cerebral ischemia-reperfusion injury, hyperglycemia, and amyotrophic lateral sclerosis by activating AMPKα and inhibiting inflammation (Ueda et al., 2013, et al., 2017; Velagapudi et al., 2019). In addition, in the study of liver cancer cells, it was also found that Ka can induce cell cycle arrest and autophagic cell death in SK-HEP-1 human liver cancer cell G2/M through AMPKα/PTEN/PI3K-Akt/TOR signaling and CDK1/cyclin B expression (Huang et al., 2013). These pieces of evidence strongly confirm the strong anti-inflammatory potential of Ka, and AMPKα may be a potential target for its anti-inflammatory effects.
Conclusion
Our study suggests that Ka inhibits the liver inflammatory response by activating AMPKα/SIRT1 and ultimately prevents DOX-induced hepatotoxicity. Currently, many plants and their components with anti-inflammatory activity have been studied for the treatment of liver inflammation-related diseases. AMPKα/SIRT1 may only be a way for Ka to inhibit inflammation, and other targets or pathways may also have an impact on inflammation. These findings will help us further understand the toxic side effects of chemotherapy drugs and the potential for prevention and treatment of Ka and identify new targets and drugs for the treatment and prevention of DOX-induced liver disease.
Authors’ Contributions
QL and DC are responsible for the conception and design of the research.
QL, PZ, and NT have made substantial contributions to data acquisition, analysis, and interpretation.
QL, JG, and ZL participated in the drafting of the manuscript.
QL, XL, and ZZ are responsible for the key revision of the knowledge content.
DC approves the final manuscript version to be published and confirms the authenticity of all original data.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
All experimental procedures in this study follow the guidelines of the Research Ethics Committee of the First People’s Hospital of Zunyi.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Zunyi Science and Technology Bureau (HZ-2022-74).
