Abstract
Background:
Plants belonging to the genus Viola and family Violaceae are well known in Ayurvedic and Unani medicine for their pharmaceutical importance. Viola, commonly known as Banafsha, has been traditionally used as an anti-malarial. The goal of this study was to determine the anti-Plasmodial activity of the cyclotide-rich crude methanol/water extract of leaves from Viola canescens Wall and to analyze its constituents by mass spectrometry (MS).
Materials and Methods:
Liquid chromatography-electrospray ionization-mass spectrometry (LC-ESI-MS) involving quadrupole time-of-flight (Q-ToF) and matrix-assisted laser desorption/ionization (MALDI)-time-of-flight-mass spectrometry (ToF-MS) were applied to investigate the cyclotides rich in methanol/water extract. The intact molecular masses were observed in the range of 2,800–3,400 Daltons, suggesting the presence of peptides. Since various head-to-tail cyclized and intramolecularly disulfide-bonded peptides known as cyclotides have been identified in the Violaceae family, we wanted to verify whether cyclotides are present in our extract. So, the intact masses inferred from the observed spectra were interrogated with Cybase (
Results:
We observed promising anti-Plasmodial activity (IC50 44 µg/ml) in the cyclotide-rich methanol/water extract prepared from the leaves of the Himalayan V. canescens Wall. The output obtained from Cybase confirmed the presence of about 20 different cyclotides in our extracts. However, no sequence information could be obtained from Cybase for 14 intact molecular masses, which were inferred from the observed LC-ESI-MS data. Hence, these could correspond to novel peptide/cyclotide sequences, which might be characteristic of Himalayan V. canescens.
Conclusion:
This is the first report on the anti-Plasmodial activity of the cyclotide-enriched methanol/water crude extract of the leaves of V. canescens.
Introduction
Viola, a herb commonly known as wild violet or sweet violet, with nearly 600 species is distributed in temperate regions. Many of its species are well-known for their importance in Ayurvedic and Unani medicine systems. These species are used for treating a range of disorders related to the respiratory system, nervous system, skin, and blood.1, 2 Furthermore, a few species of Viola are astringent, anti-inflammatory, diaphoretic, diuretic, emollient, expectorant, antipyretic, laxative, anticancer, and purgative.1, 3, 4 Banafsha, an Ayurvedic syrup, is traditionally prepared from Viola odorata.4, 5 However, it is not correct to say that Banafsha specifically refers to V. odorata. Indeed, other species of Viola, for example, Viola betonicifolia and Viola canescens, are also recognized as Banafsha or Banfosha.2, 4–7 For instance, the roots and fruits of V. betonicifolia Sm. have been utilized for kidney diseases, pneumonia, and bronchitis, while the flowers have been used for lung troubles, coughs, and colds. 8 A few studies have reported the anti-malarial activity of the Banafshas prepared from Viola websteri and V. canescens.7, 9 About 15–20 species of the Viola genus have been reported from India,10–12 which occur widely in the Himalaya and Western Ghats, with some being endemics. A preliminary survey has been done on the distribution of species of Viola in Uttarakhand and Himachal Pradesh. 13
A special class of head-to-tail backbone cyclized and intramolecularly disulfide-bonded peptides, known as “cyclotides,” occur in plants belonging to the Violaceae family, among other families. 14 These circular peptides are gene-encoded products synthesized ribosomally as longer precursor proteins, which upon proteolytic processing give rise to approximately 30 amino acid residues long cyclotides. 14 The cyclic cystine knot (CCK) structural motif (Figure 1a) is a major attribute that renders additional robustness to the three-dimensional molecular structure of these cyclic mini-proteins.15, 16 Consequently, they possess extraordinary stability against thermal denaturation, chemical, or enzymatic degradations.14, 16, 17 In addition to the popularly known uterotonic activity of kalata B1, several of these peptides exhibit anti-bacterial, anti-HIV, insecticidal, hemolytic, and anti-tumor activities, among others.18–24 Endowed with versatile physico-chemical and biological properties, including the ability to cross the blood-brain barrier, 25 cytotoxic activity against cancer cells,26–30 and ability to inhibit enzyme activity, 31 these peptides are attractive agents for therapeutic purposes and also possess potential applications in agriculture.18, 21, 32–35
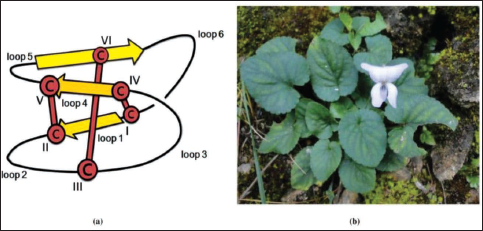
Certain regions of cyclotide sequences have been found to act as promiscuous templates for drug design and development, whereby the pharmaceutically relevant bioactive peptide sequences can be grafted into cyclotide frameworks that can offer stability and also retain the desirable biological activity.36, 37 However, reports on the anti-Plasmodial activity of cyclotides or cyclotide-enriched fractions are scarce. 38 Furthermore, there are only a few studies on the identification of cyclotides from Indian medicinal plants.39–41 Therefore, we wanted to investigate Himalayan species of Viola with the aim to explore cyclotides that might possess anti-Plasmodial activity. We found that the cyclotide-rich ‘methanol/water’ extract prepared from the leaves of Himalayan V. canescens Wall (Figure 1b) exhibited promising anti-Plasmodial activity. Mass spectrometry-based analysis of this extract revealed the presence of a wide diversity of peptides, some of which were identified as cyclotides.
Materials and Methods
Collection of Samples and Extraction
Leaves of V. canescens were collected from Pauri (30.1471°N, 78.7745°E) and Almora (29.5892°N, 79.6467°E), Uttarakhand, India. The species was identified with the help of relevant literature and confirmed by the Botanical Survey of India, Dehradun.7, 42 The specimen was deposited in the Herbarium of the Department of Botany, University of Delhi (Accession Number: DUH 14782). For the extraction of compounds, the procedure reported by Chen et al. (2005) was used. 43 Fresh plant material (369 gm) was grinded using pestle and mortar in the presence of liquid nitrogen (~3 L). The powder obtained was grinded with methanol:dichloromethane (DCM) (1:1, v/v) (1,126 mL) and kept for stirring overnight. The next morning, on transferring the solution to a separating funnel, two distinct layers were seen, of which the lower layer was dark green and the upper layer was light green. The top layer was filtered through cotton wool to remove traces of plant debris and then passed through fine filters using a sintered funnel with Whatman filter paper no. 1. The filtrate so obtained was then mixed with 400 mL of chloroform and 50 mL of water was added to let the phases separate in a separating funnel: the upper phase was brownish and the lower phase (containing chlorophyll) was greenish. The upper phase was subjected to rotary evaporation at 38°C to remove traces of chloroform/DCM and the aqueous layer was subjected to freeze drying to obtain 9 g of dry powder, which was designated as crude methanol/water extract. The solvents used for extraction were of analytical grade.
Anti-Plasmodial Activity of the Crude Methanol/Water Extract of V. canescens
This crude extract was tested for anti-Plasmodial activity using the SYBR Green microtiter plate assay as described by Smilkstein et al. 44
Ex vivo Cultivation and Maintenance of Plasmodium falciparum Culture
Chloroquine (CQ)-sensitive (Pf3D7), strain of P. falciparum, was cultivated ex vivo by the method of Trager and Jensen. 45 Cultures were maintained in fresh O+ve human erythrocytes at 4% hematocrit in complete medium at 37°C under reduced O2 (gas mixture 5% O2, 5% CO2, and 90% N2). A 0.05% of Albumax-supplemented cRPMI was used for cultivation of P. falciparum strains. Medium was changed daily (every 24 h), and the parasitemia was maintained at 5%–10%. Cultures at high % parasitemia (>10%) were diluted to low % parasitemia using fresh RBCs. Culture was synchronized at the ring stage using 5% sorbitol. 46
Preparation of Stocks and Dilutions
The methanol/water crude extract of the leaves of V. canescens was dissolved in DMSO to make a 25 mg/ml stock solution. CQ diphosphate salt (1 mM) stock solution was prepared in water (Milli-Q grade). The initial stock of 25 mg/mL DMSO was then diluted tenfold in an aqueous medium such that the DMSO concentration was now reduced to 10% and the test sample was now at 2.5 mg/mL. All subsequent twofold dilutions of this stock were made in 10% DMSO, such that the test samples got diluted to (mg/mL) 1.25, 0.625, and so on, all in 10% DMSO. When 4 µL of the test samples are diluted to 100 µL with the parasite culture, both the test sample and the 10% DMSO are diluted by 25 times. Thus, the 2.5 mg/mL stock in 10% DMSO is diluted to 100 µg/mL (test sample) in 0.4% DMSO. This concentration of DMSO allows full growth of the malaria parasite. The test samples and the standard antimalarial drug CQ (positive control) were placed in 96-well flat-bottom tissue culture grade plates to yield triplicate wells in a final well volume of 100 µL for 72 h for anti-Plasmodial activity evaluation. Parasite growth in the absence of CQ (100% growth) in the presence of CQ (zero growth) and test sample-treated cultures was monitored by the SYBR green I lysis method. 44
Chromatographic Fractionation of Methanol/Water Extract of Leaves of V. canescens
The crude hydroalcoholic extract was applied on a C18 resin packed in a glass column, and step-gradient elution was performed using increasing concentrations of methanol in water. The chromatographic fractions eluted using 60% methanol/water and 70% methanol/water were designated as “60%V” and “70%V,” respectively. Subsequently, the methanol in each fraction was rotary evaporated, and the residual aqueous fraction was subjected to lyophilization. The resulting fractions obtained in the form of powders were re-dissolved in methanol or in a combination of methanol and water for mass spectrometry experiments.
Mass Spectrometric Profiling of Crude Extracts
The profiling of 60%V and 70%V was done by reverse phase-high-performance liquid chromatography (RP-HPLC)-coupled to electrospray ionization-mass spectrometry (LC-ESI-MS) as well as by matrix-assisted laser desorption/ionization-mass spectrometry (MALDI-MS). LC-ESI-MS was carried out on a hybrid quadrupole time-of-flight mass spectrometer (6545 Q-TOF; Agilent Technologies, Singapore), which was attached to an LC equipment (1290 Infinity II; Agilent Technologies, Singapore). Liquid chromatography was carried out on a reverse-phase C18 column (2.1 mm × 100 mm; particle size 2.6 µm; Phenomenex) using water (solvent A) and methanol (solvent B), each containing 0.1% acetic acid. Starting at 65% solvent B, a linear gradient elution program was performed till 95% solvent B for 30 min, and the flow rate of the mobile phase was maintained at 0.1 mL/min. 10 µL of the sample was injected on the column. This liquid chromatograph was equipped with a diode array detector (DAD), using which the absorptions of the eluents were measured at 214 and 280 nm. The LC-ESI-MS data were processed and analyzed using Agilent Mass Hunter Workstation Software, Qualitative Analysis Version B.07.00. Liquid chromatography-mass spectrometry (LC-MS) grade solvents were used for LC-ESI-MS data acquisitions.
Using α-cyano 4-hydroxy cinnamic acid (α-C) as the matrix compound, these samples were also analyzed on a MALDI-ToF/ToF spectrometer (Axima Performance, Shimadzu Biotech, Manchester, United Kingdom). The solution of this matrix was prepared using a combination of acetonitrile and water in the ratio 1:1 (v/v). Trifluoroacetic acid (TFA) at 0.1% (v/v) was added to this matrix solution. The matrix, α-C was purchased from Sigma-Aldrich (Merck).
Results
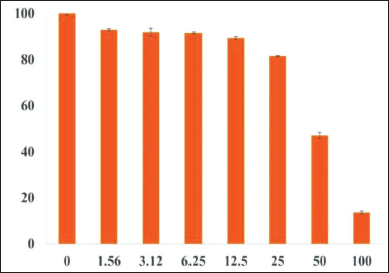
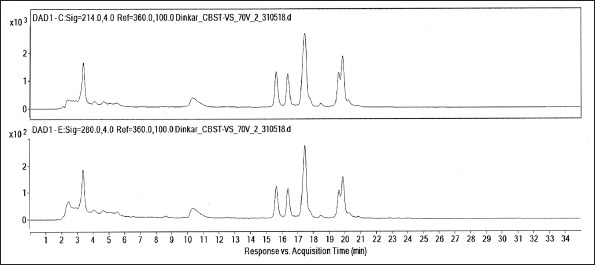
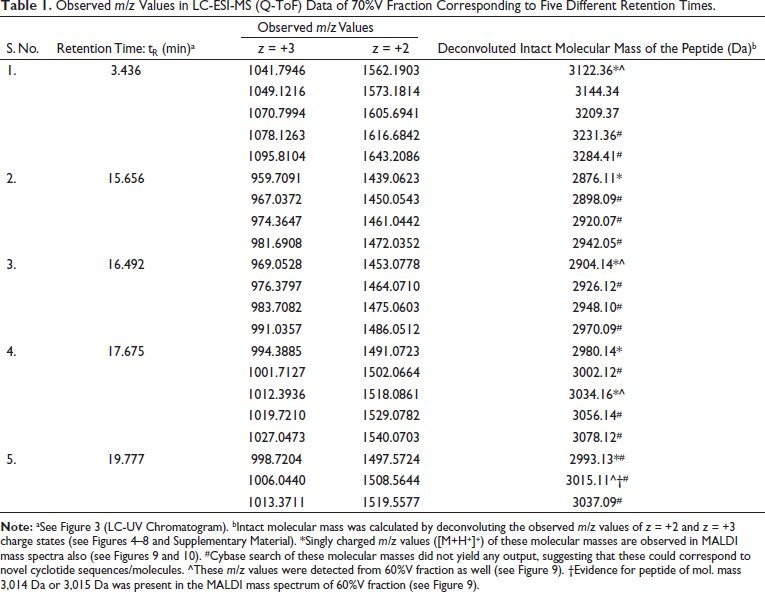
The crude methanol/water extract prepared from the leaves of V. canescens showed a dose-dependent inhibition of the growth of the malaria parasite grown ex vivo in human red blood cells with an IC50 44 µg/mL (Figure 2). This motivated us to identify the molecular constituents in the extract. Accordingly, the crude extract was subjected to column chromatography (see “Materials and Methods” section), and two different fractions at 60% and 70% methanol were collected. Subsequently, these two fractions were investigated by LC-ESI-MS. In the LC-ESI mass spectrometer (6545 Q-TOF, Agilent Technologies), prior to mass spectrometric detection, it was also possible to acquire UV chromatograms, because of the availability of DAD (see “Materials and Methods” section). The DAD-derived analytical UV chromatograms of the 70%V fraction contained a rich diversity of peaks having higher intensities than the corresponding UV chromatograms of the 60%V fraction (see Supplementary Material, Figure S1). Hence, only the 70%V fraction was taken for further analysis. Based on the analytical UV chromatogram of the 70%V sample (Figure 3), certain key retention times were chosen to categorize the ESI mass spectral data as shown in the LC-ESI-MS data of 70%V (Table 1). The m/z values of multiply protonated ions, specifically of charge states z = +2 and z = +3, were scrutinized in the ESI-MS data and deconvoluted to obtain the respective neutral intact molecular masses (Figures 4–8 and Supplementary Material, Figures S2–S17). Some intact molecular masses inferred from deconvolution of LC-ESI mass spectra were equivalent to certain masses interpreted from MALDI mass spectra (Figures 9 and 10). The deconvoluted mass values were in the range typical of cyclotides’ molecular masses. Tandem mass spectrometry (MS/MS) is a method of choice to elucidate peptide sequences.47–49 However, prior to MS/MS, we first wanted to verify, whether the deconvoluted intact molecular masses obtained (Table 1) correspond to the molecular masses of some known cyclotides or not. So, it was decided to analyze our data using Cybase (
Observed m/z Values in LC-ESI-MS (Q-ToF) Data of 70%V Fraction Corresponding to Five Different Retention Times.
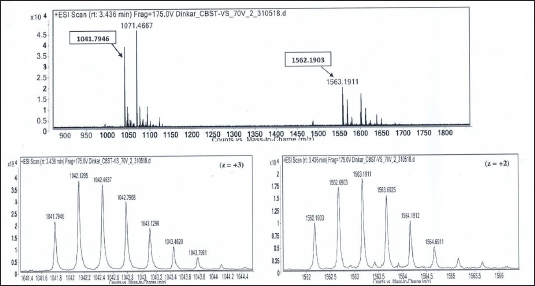
Isotope Peaks of m/z 1041.7946 (charge state, z = +3) and m/z 1562.1903 (charge state, z = +2), Respectively, are Zoomed-in and shown below. Deconvoluted Mol. Mass = 3122.36 Da.
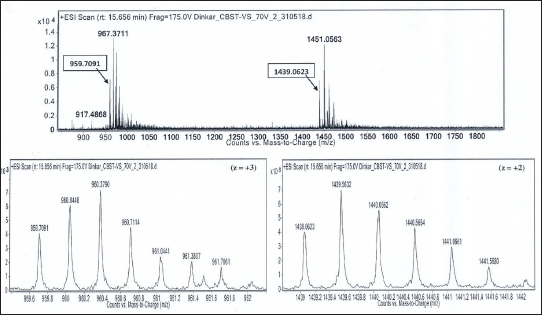
Isotope Peaks of m/z 959.7091 (charge state, z = +3) and m/z 1439.0623 (charge state, z = +2), Respectively, are Zoomed-in and Shown Below. Deconvoluted Mol. Mass = 2876.11 Da.
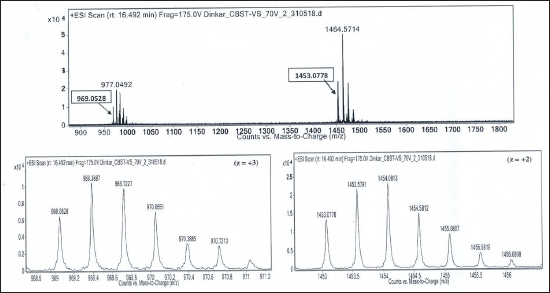
Isotope Peaks of m/z 969.0528 (charge state, z = +3) and m/z 1453.0778 (charge state, z = +2), Respectively, are Zoomed-in and Shown Below. Deconvoluted Mol. Mass = 2904.14 Da.
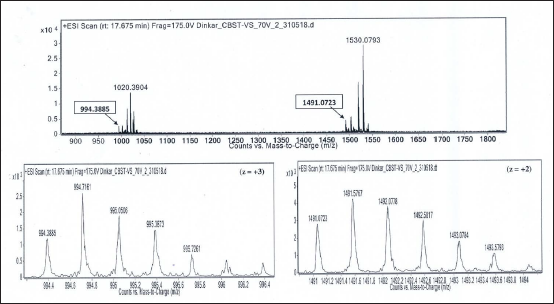
Isotope Peaks of m/z 994.3885 (charge state, z = +3) and m/z 1491.0723 (charge state, z = +2), Respectively, are Zoomed-in and Shown Below. Deconvoluted Mol. Mass = 2980.14 Da.
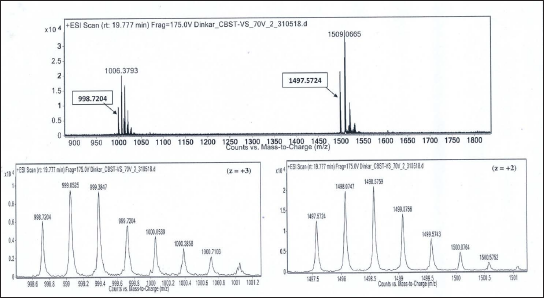
Isotope Peaks of m/z 998.7204 (charge state, z = +3) and m/z 1497.5724 (charge state, z = +2), Respectively, are Zoomed-in and Shown Below. Deconvoluted Mol. Mass = 2993.13 Da.
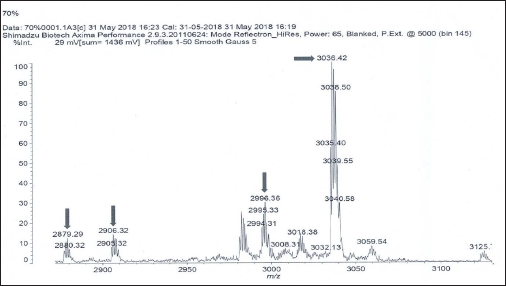
MALDI Mass Spectrum of 60%V Fraction.
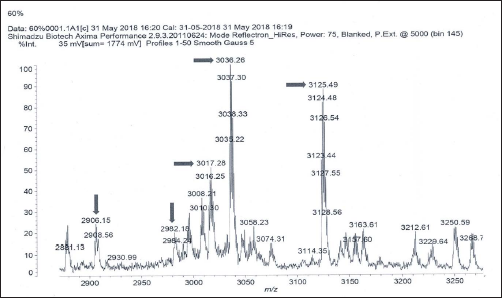
MALDI Mass Spectrum of 70%V Fraction.
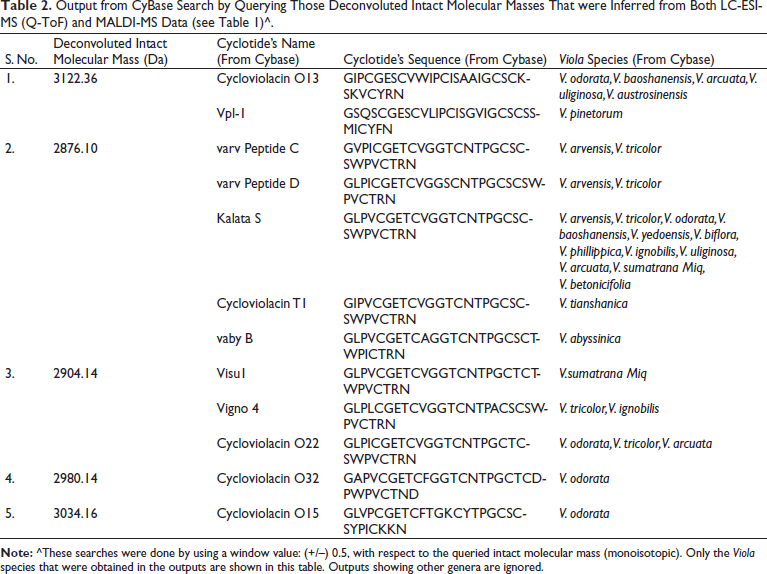
Output from CyBase Search by Querying Those Deconvoluted Intact Molecular Masses That were Inferred from Both LC-ESI-MS (Q-ToF) and MALDI-MS Data (see Table 1)^.
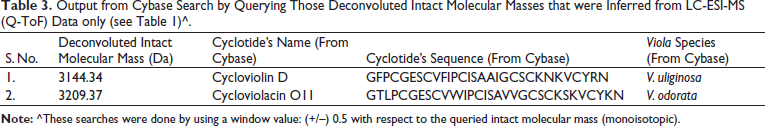
Output from Cybase Search by Querying Those Deconvoluted Intact Molecular Masses that were Inferred from LC-ESI-MS (Q-ToF) Data only (see Table 1)^.
Discussion
The finding of promising anti-Plasmodial activity (Figure 2) in the methanol/water extract of leaves of V. canescens motivated us to investigate this extract to know its molecular constituents. Crude natural extracts or partially purified chromatographic fractions can be directly analyzed by LC-ESI-MS and MALDI-MS for the analytical identification of molecules, prior to preparative or semi-preparative chromatographic purification of the molecules.41, 51–54 So, herein, we first wanted to obtain an analytical profile, viz., mass spectrometric molecular fingerprinting of the crude extract prepared from the leaves of the V. canescens by LC-ESI-MS and MALDI-MS, before semi-preparative chromatographic purification. Search in Cybase using the observed mass spectral data clearly indicated that our hydroalcoholic extract was indeed rich in cyclotides (vide supra). Further, survey into the Cybase, Knottin,55, 56 and Uniprot KB databases yielded no cyclotide sequences from V. canescens. Therefore, this is the first study reporting cyclotides from V. canescens. The sequences retrieved from Cybase using our mass spectral data were from the species of Viola other than V. canescens (Tables 2 and 3). However, the same sequences may be found in V. canescens as well, for which further investigations are warranted based on polymerase chain reaction (PCR) and MS/MS, so as to elucidate and validate the sequences.
A large body of research investigations on cyclotides from various plant families, such as Violaceae, Rubiaceae, etc. have been carried out in various countries. 14 The highlights of certain major studies are discussed herein. For example, in a multi-institutional large-scale study on plants collected from some countries in South America and Africa, cyclotides were identified from 22 different species of Rubiaceae family. 57 In the same large-scale study, cyclotides have also been identified in Apocynaceae, which is a family related to Rubiaceae and Violaceae. 57 Furthermore, cyclotides have been identified and characterized from Australian Hybanthus and Viola hederaceae,43, 58 Chinese Viola yedoensis, 23 Iranian Viola ignobilis,59, 60 Vietnamese Viola species (V. arcuata, V. tonkinensis, and V. austrosinensis), 61 and recently from Sri Lankan V. betonicifolia. 62 However, very little has been investigated about cyclotides from Indian plants. Thus far, Clitoria ternatea and V. odorata (Banafsha) are the only plants that have been studied in India with regard to cyclotides. Sudarslal et al. (2015) reported two novel cyclotides from C. ternatea in Kerala. 39 Known and novel cyclotides have been identified and compared from V. odorata that were collected from three different locations in India: Chennai, Nilgiris, and Harwan. 40 By transcriptomic profiling, Kalmankar et al. (2020) identified several novel cyclotides from C. ternatea. 41 Since cyclotides appear to be widespread in the Violaceae family, 14 we thought that our extracts might also contain cyclotides. Also, the North Indian medicinal plants have not been explored for cyclotides as yet. Therefore, hoping to find cyclotides, we focused on the hitherto unexplored Himalayan Viola species, and we chose the Himalayan V. canescens, for this investigation.
It is clear from the dose-response curve (Figure 2) that a blood concentration of ~200 µg/mL will be necessary to eliminate all parasite. Since a mouse has 2 mL of blood, the amount of extract required for intravenous or intraperitoneal routes of administration would be ~0.5 mg. As for the oral route of administration, the amount required could be slightly higher in view of the possibility of some degree of proteolysis and uncertainty about bioavailability. However, it may be noted that cyclotides are known to be remarkably stable to thermal and chemical denaturation and biological degradation by proteolytic enzymes. 17 These special features unique to cyclotides are likely to confer good pharmacokinetic properties to cyclotides, thereby enhancing their chances of becoming effective drugs. Nevertheless, it may be good to test different doses , e.g., 0.5 mg, 1 mg, 1.5 mg, and 2.0 mg to enable the determination of blood concentrations of cyclotides as a function of different oral doses. Depending on results, the dose corresponding to a blood concentration of 200 µg/ml could be chosen in the design of an in vivo antimalarial activity experiment. The chances of good in vivo activity are high, since studies have demonstrated the efficacy of cyclotides as specific CXCR4 imaging reagent for in vivo imaging, and the ability of cyclotides to cross cellular membranes has facilitated the study of modulation of intracellular protein-protein interactions both in vitro and in vivo.63, 64
Although there are no anti-malarial peptide-based drugs in clinical use, nature itself seems to use the rapidly available and fast-acting peptides to thwart malaria. The killing of the blood-stage malaria parasite by platelets is well-known to be mediated by the 70 amino acid residues long peptide PF4, which is a major CXC type chemokine stored in the granules of platelets.65, 66 This is the body’s innate immune response to fight malaria when infection is relatively low. Later, it was found that PF4 exerts potent antimalarial activity by disrupting the integrity of the parasite’s digestive vacuole. Indeed, taking cues from this natural peptide, a much smaller innovative cyclic PF4 peptide dimer (cPF4PD) was synthesized by covalently circularizing two peptides, each of which is a stretch of 14 amino acids, taken from the C-terminus of PF4. The more protease-resistant cPF4PD showed a similar antimalarial potency (EC50 = 4.3 µM) compared to that of PF4 (EC50 = 5 µM), as well as a similar mechanism of action as the much larger linear parent peptide. 67 Several antibiotic peptides with cationic, amphipathic helices have also been found to be selectively active against the malaria parasite. 68 One of the reasons for this selectivity could be the fact that, in contrast to eukaryotic cells, bacterial cells have an anionic surface, which enables the selective ionic binding of cationic peptides to the bacterial membranes. It is interesting that unlike healthy red blood cells, the malaria parasite-infected red blood cells are annexin-positive suggesting membrane reorganization leading to the externalization of phosphatidyl serine in the malaria parasite infected RBC. Indeed, our studies have shown that the de novo designed lysine branched dendrimeric cationic peptide delFD, which contains helicogenic conformationally constrained amino acids e.g., didehydrophenylalanine, has potent anti-Plasmodial activity. This peptide (IC50 = 1.5 µM) was found to spare the healthy RBC and enter only the malaria-infected RBC. Furthermore, delFD was found to enter the nucleus of malaria parasite and bind to its DNA after crossing several membranes. 69 Additionally, the cyclic peptide micrococcin was reportedly shown to be a potent inhibitor of protein synthesis and thereby growth of P. falciparum, with IC50 = 35 nM, which was comparable to antimalarials such as mefloquine, pyrimethamine, and CQ, whose IC50 values are in the nanomolar range. 70 Likewise, cyclic hexadepsipeptides from the fungus Isaria were also found to inhibit the intra-erythrocytic growth of P. falciparum. 71 Fairlie et al. (2008) showed the importance of the cyclic structure formed by the disulfide bond for the inhibition of the malaria parasite. 72 These findings of the aforementioned investigations suggest that the conformation(s) arising due to backbone and/or sidechain cyclization can be useful for designing potent anti-malarial compounds of therapeutic value. 73 Therefore, it is apparent that the anti-Plasmodial activity observed by us in the cyclotide-rich methanol/water extract of Viola is of interest and deserves further studies on the potency, selectivity, oral bioavailability, and pharmacokinetics of every cyclotide from this valuable plant. It may be important to note that the anti-Plasmodial activity observed by us in the cyclotide rich hydroalcoholic extract could have contributions also from the non-peptidic small molecular weight specialized secondary metabolites that are the hallmarks of each plant. Besides, the possibilities of synergy across secondary metabolites and cyclotides in V. canescens need further exploration. In fact, nearly 200 small molecular secondary metabolites have been found in various plant species belonging to the genus Viola. 74
Conclusion
Although Banafsha generally refers to V. odorata, other species of Viola, for example, V. betonicifolia and V. canescens, are also recognized as Banafsha or Banfosha.2, 4–7 This is the first investigation showing that the cyclotide-rich methanol/water extract of the leaves of Himalayan V. canescens suppresses the growth of the malaria parasite in culture.
Nearly 20 different cyclotides seem to be present in the methanol/water extract of the leaves of V. canescens. These were detected by mass spectrometry and preliminarily identified by matching with the intact molecular mass values in Cybase. Interestingly, no cyclotide sequence information could be obtained from Cybase for 14 different intact molecular masses that were inferred from the observed LC-ESI-MS data. The absence of matching masses in the database seems to suggest that these novel peptide sequences might be characteristic of V. canescens. In summary, conventional mass spectrometry, in conjunction with the database search (Cybase), has indicated the presence of cyclotides in the extracts of the leaves of V. canescens. However, preparative scale separation and anti-Plasmodial activity-guided purification of each peptide will be required to identify the ones that may have promising activity against malaria. Further, in order to know the complete sequence and disulfide linkages, it is imperative to collate information from PCR-based sequence information, selective proteolysis-based peptide separation, and MS/MS studies.
Footnotes
Abbreviations
Acknowledgment
The authors thank Dr. R. Nagarjuna Chary, CSIR-IICT, Hyderabad for his help in mass spectral data acquisitions.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Neha Mann thanks the University Grants Commission for financial support. P. Vishnupriya thanks VIT, Vellore for the Teaching and Research Assistant (TRA) fellowship. Dinkar Sahal acknowledges ICGEB, New Delhi, for internal funding. S. Prabhakar thanks the Director, CSIR-IICT, Hyderabad for facilities and encouragement (IICT/Pubs./2021/351).
Statement of Informed Consent and Ethical Approval
Necessary ethical clearances and informed consent were obtained before initiating the study from all the participants.
