Abstract
Background:
Cloves mean the dried flower bud harvested from a tree of Syzygium aromaticum L. (SA).
Objectives:
The current study was to identify the anti-inflammatory effects of SA on lipopolysaccharide (LPS) (0.2 µg/mL)-treated RAW264.7 cells.
Materials and Methods:
WST-8 cell proliferation assay kit (WST-8) and lactate dehydrogenase (LDH) assays were completed to observe cell proliferation and cytotoxicity effects of SA. Other biochemical analyses analyzed releases of nitric oxide (NO) and the production of dichlorofluorescin diacetate (DCF-DA). Besides, inflammation-related factors and Sirt1 were carried out through immunohistochemistry (IHC) and immunofluorescence (IF).
Results:
LPS exposure not only caused abnormal elevations of both NO and reactive oxygen species (ROS) secretions, but also markedly increased inflammation-related proteins including inducible NO synthase (iNOS), cluster of differentiation (CD) 14, and toll-like receptor (TLR) 4, and nuclear factor-κB (NF-κB). The above-listed alterations were notably attenuated by SA pretreatment in a dose-dependent manner. We further figured out that, SA particularly activated AMPKα/ Sirt1 signaling pathway. Furthermore, SA dramatically inhibited interleukin (IL)-1β production, which was mediated by NLR family Pyrin domain containing (NLRP) 3 mediated inflammasome signaling pathway.
Conclusion:
SA inhibits LPS-induced inflammation in RAW264.7 cells via the TRL4/CD14/NF-κB pathway and it can lead to the reduction of IL-1β release by the inhibition of NLRP3 inflammasome. Such an anti-inflammatory effect was closely related to AMPKα/Sirt1 activation. Taken together, the findings of this study support both the inhibition of NLRP3 inflammasome and activation of Sirt1 can alleviate inflammation.
Introduction
Inflammation refers to extremely complex physiological responses to external noxious materials, which are deeply involved in the pathogenesis of the chronic disease. 1 Various factors are broadly and intricately associated with diverse diseases due to the activations of macrophages. 2 These cells are known to be innate immune cells and phagocytic functions against xenobiotic invasions to the host as a primary role of defence. 3 As a result of the above actions, activated macrophages accelerate acute and chronic inflammatory responses by excessive production of oxidative stress mediators such as nitric oxide (NO), reactive oxygen species (ROS), and the release of inflammatory cytokines. 4
On the other hand, lipopolysaccharide (LPS), endotoxin, which is the major component of the gram-negative bacterial cell walls, has been commonly used as a stimulator of the inflammatory response and is closely related to the Toll-like receptor (TLR) 4. 5 Several studies have demonstrated that the LPS-induced TLR4 activation can sequentially lead to activation of nuclear factor-κB (NF-κB) transcription signaling, thereby LPS-mediated local inflammation, tissue damage even death in diverse organs. 1 During the NF-κB transcription response, its activation induces proinflammatory cytokine secretion mainly in macrophages. The cell surface protein CD14 regulates the LPS-induced activation of the TLR4/NF-κB pathway and exists on the surface of many TLR4-expressing cells, including macrophages, monocytes, dendritic cells, also epithelial cells. 6
According to recent studies, it is well documented that the blockade of NLR family Pyrin domain containing (NLRP) 3 inflammasome activation in LPS-induced inflammation ameliorated inflammation severity. The reason is that NLRP3 leads to the maturation and activity of interleukin (IL)-1β.7, 8 Accordingly, IL-1β can be strictly regulated by inflammasomes because the knockdown of NLRP3 considerably attenuated IL-1β release. 9 Recently, various studies have suggested a close relationship between Sirt1 and inflammation. In particular, the regulation of Sirt1 through the deacetylation of histones and transcription factors can modulate inflammation. 10 In other words, Sirtuin1 (Sirt1), a class III histone deacetylase, is an epigenetic regulator which influences various pathophysiological processes such as cellular inflammation and metabolism, respectively. Additionally, Sirt1 activation can play a critical role in metabolic homeostasis, modulation of oxidative stress, and protection of mitochondrial functions. 11 Sirt1 not only attenuates LPS-induced NLRP3 inflammasome activation but also inhibits inflammatory response and IL-1β secretion by reducing oxidative stress. 12
On the other hand, herbal medicinal plants have been widely accepted as natural products with lesser side effects and improved efficacy than their synthetic products. 13 Among these plants, Syzygium aromaticum L. (SA), commonly known as cloves, is the dried flower bud and it is commercially used as a medicinal agent, perfume, and preservative for meat processing. 14 Biological activities such as antioxidant, anti-inflammatory, anti-cholinesterase, antiangiogenic, anticancer, and analgesic activities,15–17 have been reported in various scientific studies, however, the anti-inflammatory effect of SA and its fundamental mechanism have not been fully explained.
Herein, we established RAW264.7 cell models of inflammation induced by 0.2 µg/mL LPS and exposed cells to SA. Thereafter, we hypothesized that cloves treatment can indicate the anti-inflammatory effect through the inhibition of NLRP3 inflammasome via AMPKα/Sirt1 activation in LPS-induced inflammation.
Materials And Methods
Cell Culture and Reagents
The murine macrophage cell line RAW 264.7 was obtained from the American Type Culture Collection (ATCC, Rockville, MD, USA) cell bank. Cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM) high glucose (4.5 g/L) (Hyclone, Logan, UT, USA) supplemented with 10% fetal bovine serum (FBS) (Gibco, CA, USA) and antibiotics (1%) respectively. Cells were incubated at 37°C under a humidified atmosphere containing 5% CO2 in a cell incubator. LPS from Escherichia coli (E. Coli) O55:B5 was obtained from Sigma–Aldrich (catalogue no. L-2880, St. Louis, MO, USA) and was dissolved in PBS at a concentration of 0.2 µg/mL.
Extraction of Cloves
Extraction of 300 g of dried cloves (Quanzhou Bainian Xiuben Tea Industry Co., Ltd, Quanzhou, Fujian, China) was performed twice with distilled water, for 3 h each time. The two extracts were combined and then dried in a rotary evaporator (R1020, Zhengzhou Greatwall Scientific Industrial and Trade Co., Ltd., Zhengzhou, Henan, China) to obtain the clove extract. The powder of SA was a yield of 20.87% by weight and kept at −80°C before use.
Cell Proliferation Assay
RAW264.7 cells were transferred to the 96-well microplate (4 × 103 cell/well). After cells adhered various concentrations of SA were pretreated 6 h before with or without LPS treatment (0, 25, 50, 100, and 200 µg/mL). Cells were further incubated for 24 h and Water-Soluble Tetrazolium 8 (WST-8) reagents (#10010199, Cayman Chemicals, Ann Arbor, MI, USA) were added to the well and further incubated for 90 min in a cell incubator. The supernatants were measured using a microplate reader (Versa Max, San Jose, CA, USA) under the wavelength of 450 nm and 600 nm.
Measurement of Lactate Dehydrogenase (LDH) Release
The LDH activity was determined using a commercial LDH assay kit (#K726, BioVision, Waltham, MA, USA) according to the manufacturer’s instructions. First, 40 µL cell-free supernatant or cell lysate was mixed with nicotinamide-adenine dinucleotide (NADH) and 2,4-dinitrophenylhydrazine and incubated at 37°C for 15 min. Next, 0.4 mol/L NaOH was added to the samples and incubated at 37°C for 3 min. Finally, the absorbance of each sample was recorded at 440 nm using a UV spectrophotometer (Tecan’s Infinite M200, Salzburg, Austria).
Measurement of ROS Secretion
Cellular ROS levels of the SA-treated RAW264.7 cells were by application of DCFDA/H2DCFDA—Cellular ROS Assay Kit (ab 113851, Cambridge, UK). Briefly, treated RAW 264.7 cells (4 × 104 cells/well/mL) were transferred to a 96-well microplate and various doses of SA were pretreated prior to 6 h of LPS stimulation. After incubation for 24 h, cells were washed with 10 mM PBS (pH 7.3) twice then DCFH-DA (10 µM) was added and incubated for 40 min at 37°C in the dark. The final product of ROS was measured by using a microplate reader by applications of fluorescence-filtered conditions (excitation/emission at 485 nm/535 nm).
Measurement of NO Production
The NO production was determined using a commercial NO assay kit, the CytoTox-96® Non-Radioactive Cytotoxicity Assay (Promega Corporation, Madison, WI, USA) according to the manufacturer’s instructions and following a previously published method. 18
Analysis of Immunohistochemistry and Immunofluorescence
Immunohistochemistry (IHC) analysis was performed for detecting RAW264.7 cells. We further completed IF analyses for observing CD14 (#11-0141-82, Thermo Fisher Scientific, Waltham, MA, USA), phosphor-AMPKα (#50081, CST, Danvers, MA, USA), Sirt1(#8469, CST), Ki67(#9129, CST), NLRP3 (#MA-32255, Thermo Fisher Scientific), TLR4 (#MA5-16216, Thermo Fisher Scientific), and IL-1β (#12242, CST). Respectively. Briefly, cells were seeded at 1 × 105/wells and pretreated with SA prior to 6 h of LPS treatment. After 24 h incubation, cells were washed with 10 mM PBS (pH 7.3) twice, then fixed in 4% paraformaldehyde (PFA) overnight at RT. Discarded PFA, cells were washed three times with 10 mM PBS then incubate at 0.05% PBST for 5 min at RT to increase permeabilities of the cell, and cells were blocked by applications of 2.5% of normal horse serum then directly applied primary antibodies using blocking buffer for overnight at 4°C.
For IHC analysis, cells were washed with 10 mM PBS and applied secondary antibody and amplifying buffer using a universal IHC assay kit (VECTASTAIN® Elite ABC-HRP Kit, Peroxidase (Standard), Cat. #PK-6100, Vector laboratories, Burlingame, CA, USA). The products of the final reactions were using ImmPACT® DAB EqV Peroxidase (HRP) Substrate (#SK-4103, Burlingame, CA, USA). Hematoxylin was applied as counterstaining. The signals were detected under the light microscopy condition (Leica, Wetzlar, Germany).
For immunofluorescence (IF) analysis, cells were applied donkey-rabbit-488 or 594/goat-mouse-488 or 594 antibodies and DAPI was applied for counterstaining. The fluorescence signals were detected under the fluorescence filtered equipped microscopy condition.
Protein intensity of each IHC and IF for positive signals was applied by Image J analysis (ver. 1.64, NIH, Bethesda, MD, USA).
Statistical Analysis
Experimental data are expressed as mean ± SEM. We performed one-way ANOVA followed by Tukey’s post hoc tests using SPSS 26 software (Chicago, IL, USA). The statistical significance (p values) was defined as less than 0.05. All analyses were performed three times.
Results
SA Pretreatment Shows Cell Proliferation Inhibitory Effect in RAW264.7 Cells
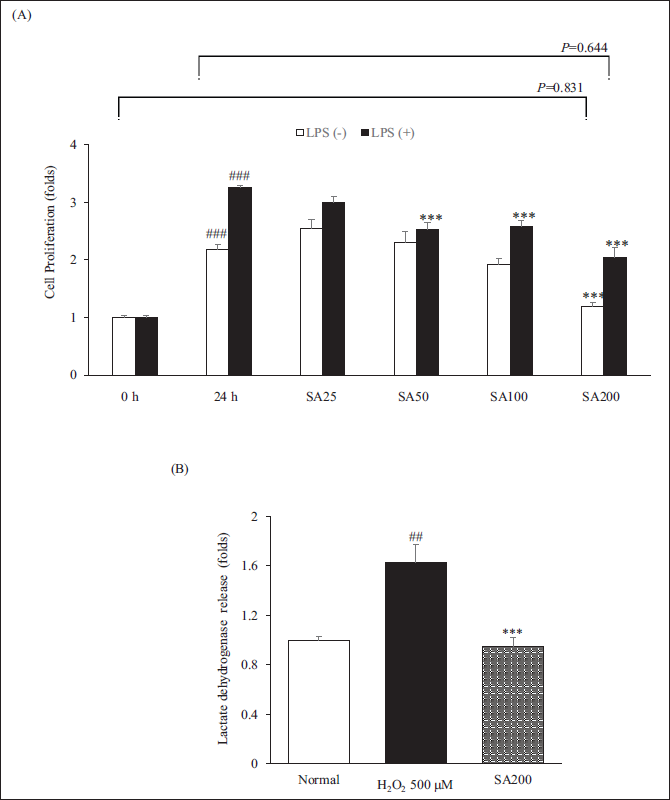
The inhibitory effect of SA on RAW264.7 cell proliferation was assessed using WST-8 assays. As shown in Figure 1A, SA significantly inhibited RAW264.7 cell activation from 50 to 200 µg/mL when LPS treatment and also prevented RAW264.7 cell proliferation with or without LPS. According to the results of the LDH release assay, SA did not display any cytotoxic effects. Especially, the LDH release level by SA200 treatment was significantly reduced to the level of the normal group (p < 0.001). This implies that SA plays a crucial role in the inhibition of RAW264.7 cells proliferation by LPS stimulation without cytotoxicity (Figure 1B).
SA Suppresses LPS-induced Both NO and ROS in RAW264.7 Cells
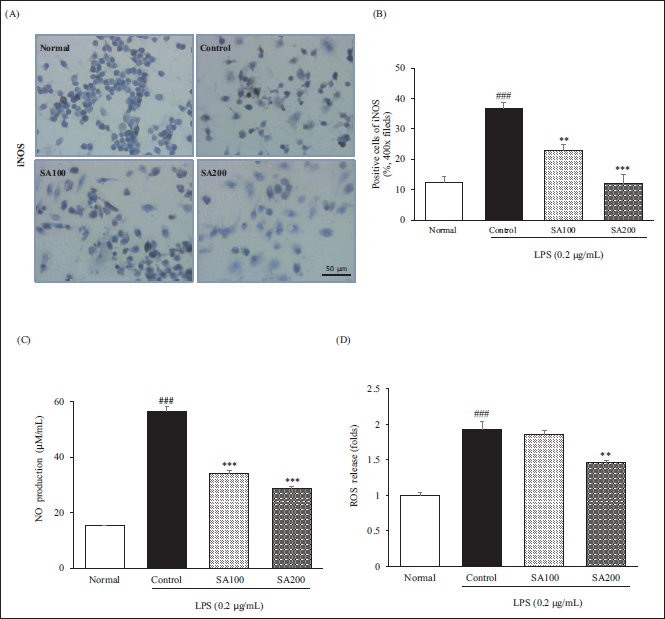
To investigate the inhibitory effect of oxidative stress of SA, factors including NO and ROS in LPS-stimulated RAW264.7 cells were first determined. iNOS that produces NO induced by LPS stimulation dramatically increased in the control group (p < 0.001), whereas the elevated level by SA treatment was reduced in dose-dependent (SA100, 37.4%, p < 0.01; SA200, 67.3%, p < 0.001). Moreover, the obvious increase of NO level from LPS stimulation was suppressed by SA in a dose-dependent manner (SA100, 39.6%, p < 0.001; SA200, 49.0%, p < 0.001). However, in terms of ROS generation, only SA200, not SA100, significantly decreased ROS generation due to LPS. Collectively, these results suggest that SA exhibits an anti-inflammatory effect through the inhibition of oxidative stress (Figure 2).
SA Inhibits LPS-induced Inflammation via CD14/TLR4/NF-κB Pathway in RAW264.7 Cells
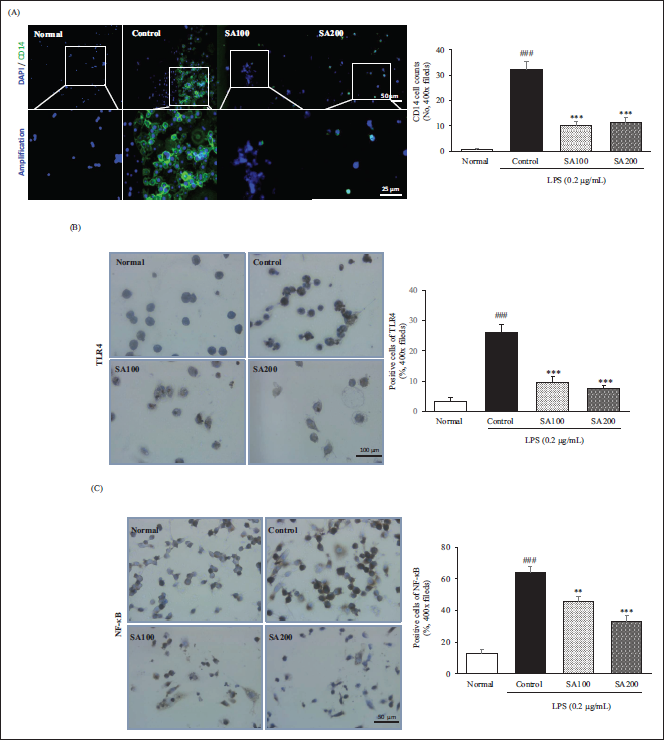
Both CD14 and TLR4 play an important role in sensitizing the cells as pattern recognition receptors (PRRs) on the cell membrane that recognize LPS during the activation of RAW264.7 cells. This is the onset of the classical inflammatory process. Here we exposed LPS-induced RAW264.7 cells to SA (Figure 3). Our results indicated that SA pretreatment decreased activation of the NF-κB signaling pathway by inhibiting CD14 and TLR4 on the surface of RAW264.7 cells. The elevated levels of CD14, TLR4, and NF-κB after LPS stimulation showed 43, 7.4, and 5.1 folds compared with that of the normal group, respectively. Interestingly, NF-κB inactivation via the inhibition of both CD14 and TLR4 suppressed 28.8% (SA100 group) and 48.2% (SA200 group) compared with that of the control group, respectively.
SA Activates AMPKα/Sirt1 Signaling Pathway in RAW264.7 Cells
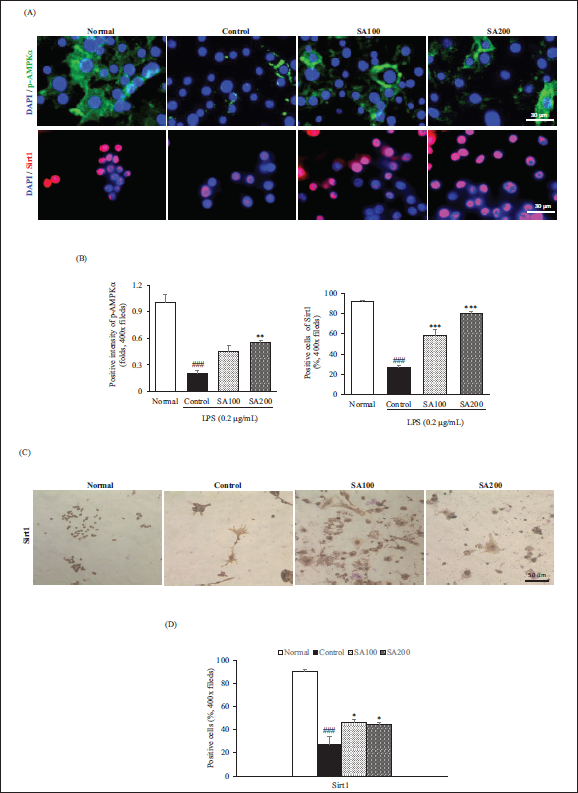
We performed further analyses on the AMPK/Sirt1 signaling pathway. As shown in Figure 4 (A–D), compared with the Control group, SA treatment showed significantly beneficial expression of p-AMPKα (SA100, 2.3 folds; SA200, 2.8 folds) and Sirt1 (SA100, 2.1 folds; SA200, 2.9 folds). Additionally, when Sirt1 was analyzed through IHC, the Sirt1 level in the control group was significantly reduced by 69.4% compared with that of the normal group, whereas, SA100 and SA200 treatment restored 1.7 folds, 1.6 folds compared with the control group, respectively. These findings imply that SA activates AMPKα/Sirt1 signaling pathway in RAW264.7 cells (Figure 4).
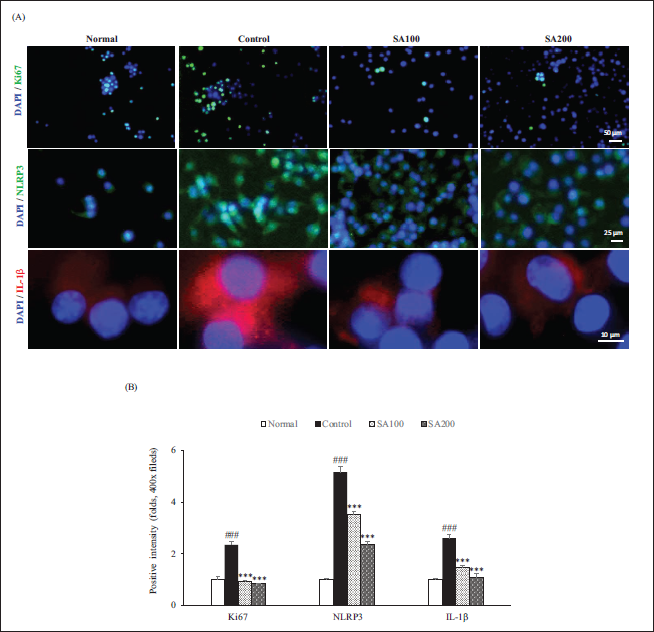
SA Downregulates the Expression of Ki67 and NLRP3/IL-1β Pathway in RAW264.7 Cells
We also confirmed the expression of Ki67, NLRP3, and IL-1β through IF staining (Figure 5). The value of proliferation marker Ki67 by LPS challenge significantly increased 2.3 folds compared with that of the normal group. However, the elevated values dramatically decreased below the normal group by SA treatment. Also, SA significantly attenuated NLRP3-induced IL-1β release in a dose-dependent manner. When SA200 was administrated, two factors significantly downregulated 54.3% and 58.5%, respectively. These data confirmed that SA effectively disrupted the NLRP3 inflammasome/IL-1β positive feedback loop in RAW264.7 cells.
Discussion
This study selected RAW 264.7 cells for the establishment of an inflammation model and discovered the possible mechanism of SA as a medical material to relieve inflammation. Here, we revealed that SA ameliorated LPS-induced inflammation via CD14/TLR4/NF-κB pathway and also led to the decrease of the NLRP3 inflammasome by Sirt1 activation in Raw 264.7 cells as evidenced by IHC and IF staining. LPS, an external noxious factor, can cause tissue damage, along with abnormally released genes or proteins. 19 Inflammation, if not controlled at the proper time, may provoke life-threatening outcomes. 20 SA has potential anti-inflammatory effects to various reports.21, 22 Accordingly, we explored the fundamental mechanism of SA in suppressing inflammation, aiming to provide accurate information.
First, we performed cell proliferation and cytotoxicity through WST-8 and LDH assay. RAW264.7 cells were incubated with different concentrations (25 µg, 50 µg, 100 µg, and 200 µg) of SA for 24 h (Figure 1). Cell proliferation due to the treatment of LPS showed a significant decrease in all concentrations of SA except for SA25 (p < 0.001) but showed an excellent reduction effect in SA200 treatment (37% reduction compared with the LPS 24 h). SA200 alone treatment did not show a significant change compared with that of 0 h (p = 0.831) and also SA200 with LPS treatment did not show a significant change when compared with that of 24 h (without LPS, p = 0.644). This result shows that SA200 effectively inhibited cell proliferation. The effect of SA treatment on the release of LDH was evaluated and exhibited a significant decrease in LDH release. Interestingly, the LDH level showed a dramatic reduction compared with that of the positive control (H2O2), which showed maximum levels of LDH (p < 0.001). Especially, such levels in the SA group exhibited a tendency to decrease slightly compared to the normal group. Through this, it was confirmed that SA treatment inhibits cell proliferation, but is safe because it is non-toxic.
Macrophages are deeply involved in the inflammation process through the production of inflammatory mediators during the immune response and inflammatory diseases. To be specific, macrophages regulate various inflammatory mediators such as NO, prostaglandin, and proinflammatory cytokines. 23 Macrophages are specialized in inducing a series of courses of inflammatory cytokines and mediators in response to LPS exposure. 24 As a result, inducible NOS (iNOS), a key enzyme in the macrophage inflammatory response, is expressed only in response to LPS stimulus in some cells. 25 LPS-induced iNOS is the source of NO that is powerfully induced in inflammatory responses. NO generated by iNOS is implicated in important functions such as immune regulation and microbial killing. 26 Besides, inflammatory cells including macrophages produce excessive ROS. Accordingly, interactions between NO and ROS in cellular redox signaling are intensive determinants of the inflammatory response. As shown in Figure 2, after LPS stimulation for 24 h, the activated RAW264.7 cells showed a significant increase in the release of both NO production and ROS release (p < 0.001), and pretreatment with SA prior to the LPS challenge effectively attenuated the enhancement of these factors. The NO production was 3.7 times higher in the control group than in the normal group. Whereas, SA100 and SA200 treatment significantly decreased by 39.6% and 49% compared with that of the control group, respectively. In addition, SA200 was found to inhibit LPS-induced ROS release by 47.8% more potently than SA100 (Figure 2). These results suggested that SA exerts anti-inflammatory activity via the suppression of NO generation and ROS release in LPS-induced RAW264.7 cells.
Many studies27, 28 revealed that CD14/TLR4/NF-κB pathway was closely related to inflammatory response when stimulation of LPS is given. CD14 and TLR4 are the first PRRs on the cell that bind to LPS; more precisely, CD14 and TLR4 are upstream molecules through which LPS induces the activation of NF-κB transcription. In the case of CD14 knockdown, even in the presence of LPS stimulation, it did not activate the TLR4/NF-κB pathway, resulting in no inflammatory response.20, 29 Accordingly, we further investigated how SA decreased LPS-induced activation of the CD14/ TLR4/NF-κB pathway. As expected, SA pretreatment inhibited the expression of CD14 and TLR4 in LPS-induced RAW264.7 cells and showed the subsequent suppression of NF-κB pathway activation. The elevated CD14 expression of the Control group was significantly inhibited by more than 65% by SA treatment. Moreover, the inhibition of NF-κB expression by SA treatment led to 28.8% and 48.2%, respectively, in a concentration-dependent manner, according to the inhibition of TLR4 (Figure 3). As a result, SA dramatically prevented the activation of the TLR4/NF-κB pathway by inhibiting CD14 expression in LPS-induced RAW264.7 cells. These results mean that it can effectively suppress the inflammation triggered by LPS.
AMP-activated protein kinase (AMPK) plays an important role in maintaining the balance of intracellular energy metabolism in the absence of ATP. 30 Once activated, AMPK primarily regulates the catabolic pathway, resulting in ATP production whereas inhibits the anabolic pathway, resulting in ATP consumption. Ultimately, this action leads to the restoration of energy homeostasis. 31 Moreover, the role of Sirt1 as an energy sensor on energy balance is newly lit. 32 The activation of both AMPK and Sirt1 in the control group was dramatically decreased compared with the normal group. Similar to previous studies, our results demonstrated that the downregulation of AMPK led to the downregulation of the expression of Sirt1 by LPS treatment.33, 34 Whereas, SA pretreatment promoted both p-AMPK and Sirt1 expressions. Unlike SA100, SA200 showed an excellent increase effect in both factors (Figure 4A, B). Interestingly, we confirmed through IHC that the decreased Sirt1 by LPS challenge was reversed by SA treatment (Figure 4C, D).
Ki67, which is one of the biological indices, plays an important role in mediating hyperproliferation, and also its level increases after LPS exposure. Accordingly, its low expression may be associated with a beneficial outcome. Consistent with these previous results, our data increased Ki67 expression in the control group; however, such increase was reversed by SA treatment.35, 36 (Figure 5). LPS is well known as a proven NLRP3 inflammasome activator. The NLRP3 inflammasome is activated by various external danger signals that can derive from a variety of inflammatory disorders, including arteriosclerosis, inflammatory bowel disease, gout, diabetes, and Alzheimer’s disease. When NLRP3 is activated, it leads to the secretion of the pro-inflammatory cytokines (IL-1β) via caspase 1 activation.37–39 In the current study, IL-1β secretion via NLRP3 activation induced by LPS treatment dramatically increased in the control group. The elevated NLRP3 expression of the control group was significantly inhibited by 54.3% by SA treatment. In contrast, SA pretreatment positively affected the secretion of IL-1β. Herein, the inhibition of IL-1β release by SA treatment led to 44.2 and 58.5%, respectively, in a concentration-dependent manner.
Conclusion
Our study demonstrates that SA inhibits LPS-induced inflammation in RAW264.7 cells via the CD14/TLR4/NF-κB pathway and also the inhibition of NLRP3 inflammasome led to the reduction of IL-1β release. In addition, SA affected an anti-inflammatory effect through the decrease of oxidative stress markers via the AMPK/Sirt1 pathway.
Footnotes
Authors’ Contributions
Writing-original draft, Writing-review, and editing, S.W. Seo; investigation and methodology, K. Kim; conceptualization and supervision, M. Shin. All authors have read and agreed to the published version of the manuscript.
Abbreviations
LPS: Lipopolysaccharide; LDH: Lactate dehydrogenase; DEX: Dexamethasone; NO: Nitric oxide; ROS: Reactive oxygen species; DCF-DA: Dichlorofluorescin diacetate; OS: Oxidative stress; PBS: Phosphate buffer saline; CD: Cluster of differentiation; TLR: Toll-like receptor; AMPK: AMP-activated protein kinase; Sirt: Sirtuin; IL: Interleukin; NLRP: NLR Family Pyrin Domain Containing.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (2021R1I1A1A01059605).
Statement of Informed Consent and Ethical Approval
Necessary ethical clearances and informed consent was received and obtained respectively before initiating the study from all participants.
Summary
The current study illustrated the inhibitory effect of SA via the CD14/TLR4/NF-κB pathway in LPS-induced inflammation. SA also suppresses the inflammatory response by disrupting NLRP3/IL-1β interaction via AMPK/Sirt1 activation. We conclude that SA may be a potential anti-inflammatory agent.
