Abstract
Introduction
Inflammation is a normal protective reaction that occurs following trauma, infection, or tissue injury. 1 In the inflammation process, nitric oxide (NO) is produced and activated inflammatory cells are increased dramatically. 2 NO is produced by inducible nitric oxide synthases (iNOS) in macrophages, hepatocytes, and renal cells, under the stimulation of LPS, tumor necrosis factor-alpha (TNF-α), interleukin-1 (IL-1), or interferon-gamma (IFN-γ). 3 The overproduction of NO by iNOS has been implicated in the pathology of several inflammatory disorders, including septic shock, tissue damage after inflammation, and rheumatoid arthritis.4–6 Therefore, NO production induced by LPS through iNOS can reflect the degree of inflammation, and a change in the NO level by inhibiting iNOS enzyme activity could be a proxy with which to assess the anti-inflammatory effects of plant extracts.
During the screening program, we found that the ethyl acetate (EtOAc) fraction from the heartwood of Vietnamese Dalbergia oliveri Gamble ex Prain (Fabaceae) exhibited appreciable inhibitory activity in LPS-induced NO production in RAW264.7 cells in vitro. The Dalbergia genus consists of 274 accepted species and is widely distributed in tropical and sub-tropical regions. 7 Numerous Dalbergia species are used for the treatment of several diseases in the traditional medicine system.7–9 D. oliveri is a woody climber, and is widely distributed in southeast Asian countries, including in Myanmar, Thailand, Laos, Cambodia, and Vietnam.10,11 D. oliveri has been used to treat chronic ulcers, 10 and has been reported to possess various pharmacological activities, such as anti-tumor promoting, 12 cytotoxic and anti-fungal, 13 larvicidal,14,15 and cell proliferation activity. 16 Previous studies on chemical constituents of D. oliveri showed the presence of isoflavones, isoflavanones, isoflavans, neoflavones, coumestones, coumarins, flavanones, chalcones, and pterocarpans.12–17 Despite the number of studies, there has been no investigation of chemical constituents and inhibitory activity of NO production of Vietnamese D. oliveri. For that reason, this paper describes the isolation and structural elucidation of the isolated compounds as well as evaluating their inhibitory action on NO production.
Experimental
General experimental procedure
1H NMR (500 MHz) and 13C NMR (125 MHz) spectra were measured on a Bruker Avance 500 MHz spectrometer (Bruker Daltonics, Ettlingen, Germany). Chromatography column (CC) was carried out on silica gel Si 60 F254, 40–63 mesh (Merck, St Louis, MO, USA), YMCGEL ODS-A, 12 nm S-150 μm (YMC Co Ltd, Kyoto, Japan), and Sephadex LH-20 (Sigma–Aldrich, MO, USA). Compounds were visualized under UV radiation (254, 365 nm) and by spraying plates with 10% H2SO4 followed by heating with a heat gun.
Plant material
The heartwood of D. oliveri was collected at Dak Lak Province, a central province of Vietnam. Botanical identification was performed by Nguyen Quoc Binh. PhD, Vietnam National Museum of Nature, Vietnam Academy of Science and Technology (VAST). A voucher specimen (Cam Lai-CL-Lõi) is deposited with the Natural Product Chemistry Lab, Tay Nguyen University, Vietnam.
Extraction and isolation
The heartwood of D. oliveri (4.0 kg) was reflux extracted with 96% ethanol (EtOH) for 1 h. The extract was then filtered before being evaporated under reduced pressure to give a crude EtOH extract. The EtOH extract (150 g) was then suspended in hot water and partitioned with ethyl acetate (EtOAc) successively. The resulting fraction was concentrated under decreased pressure to give EtOAc (120 g) and water (H2O) fractions. Following activity-guided fractionation, the EtOAc soluble fraction (120 g) was chromatographic on a silica gel (Kieselgel 60, 230–400 mesh) CC (15 × 50 cm) using a stepwise gradient of CH2Cl2-MeOH (100:1 to 0:1, each 3.0 L), to yield 14 fractions (E1–E14) based on their TLC profiles. Fraction E4 (8.2 g) was applied to a silica gel (Kieselgel 60, 230–400 mesh) CC (6 × 50 cm) and eluted with a gradient of n-hexane-EtOAc (5:1 to 1:1) to obtain four fractions (E4.1–4.4). Further purification of sub-fraction E4.3 (800 mg) by Sephadex LH-20 (25 μm particle size) CC (3 × 90 cm), eluting with a gradient of MeOH:H2O (1:1), yielded compound
Daidzein (
Formononetin (
3,7-dihydroxy-4′-methoxyflavone (
Liquiritigenin (
3′-methoxydaidzein (
Dalbergin (
Butin (
Sativanone (
Isoliquiritigenin (
Biological assay
Cell culture, cell viability assay and determination of NO production were performed according to the methods previously described.18,19
Statistical analysis
The NO production inhibitory activity assay was performed in triplicate. The results are presented as the means ± standard error of the mean.
Results
Chemical structure identification
The ethanol extract was separated with EtOAc to obtain EtOAc soluble fraction, which was then subjected to column chromatography using silica gel and Sephadex LH-20 to obtain nine secondary metabolites ( Structure of compounds 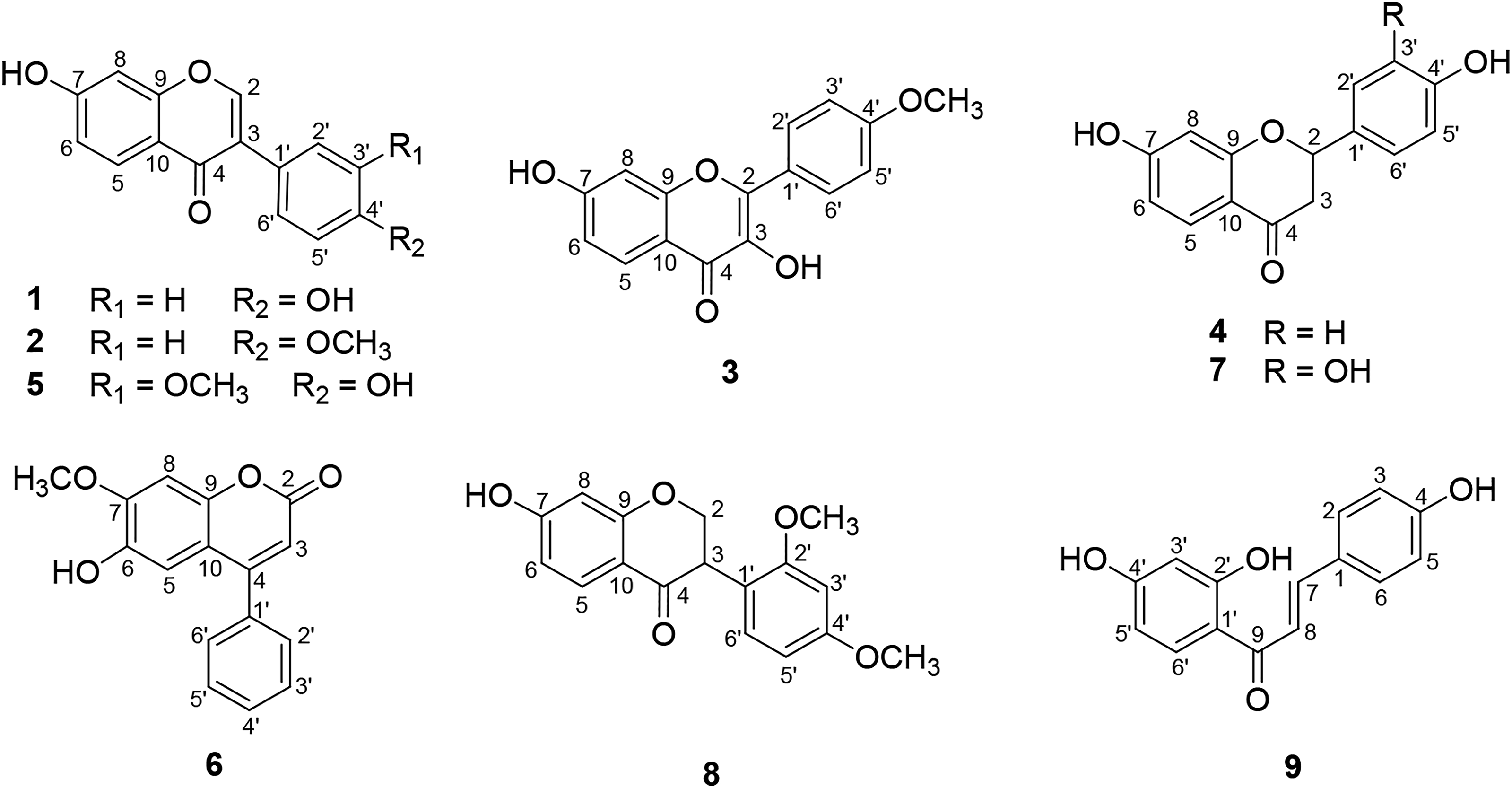
The 1H-nuclear magnetic resonance (NMR) spectrum of compounds
Compound
Both compounds
Compound
Compound
Compound
Anti-inflammation activity
In the preliminary test, a cytotoxic assay was performed to define the safe and non-toxic concentration of isolated compounds ( Effect of compounds 
NO production inhibition in RAW264.7 cells of isolated compounds.

aThe inhibitory effects are represented as the molar concentration (μM) giving 50% inhibition (IC50) relative to the vehicle control. Values are mean ± SD (n = 3).
bPositive control.
Neither LPS nor the samples were added to the control group. Thus, the inhibitory effects of these compounds on NO production were not attributable to any cytotoxic effect. After LPS (1 μg/mL) stimulation, NO production increased approximately 12-fold after 24 h in the control. Compounds Inhibitory effect of 
Discussion
Lipopolysaccharide inflammation stimuli, known as lipoglycans or endotoxins, stimulate macrophages to induce the expression of iNOS protein to produce NO. NO has been found to be an inflammatory mediator by acting as an activator of macrophages to kill microorganisms through signal transduction.
29
On the other hand, the overproduction of NO in the immune system might cause immune hypersensitivity reactions that are subsequent to tissue or cell injury.
19
Thus, the inhibition of iNOS activity or downregulation of iNOS expression could be beneficial to inhibit inflammatory responses. From our results, the flavanone butin (
Formononetin (
In this study, we obtained some positive results, such as compounds
Conclusion
In conclusion, based on a bioassay-guided extraction and isolation, nine natural compounds including daidzein (
Supplemental Material
Supplemental Material - Anti-inflammatory activity of ingredients from the heartwood of Vietnamese Dalbergia oliveri Gamble ex Prain
Supplemental Material for Anti-inflammatory activity of ingredients from the heartwood of Vietnamese Dalbergia oliveri Gamble ex Prain by Ngu Truong Nhan, Phi Hung Nguyen, Dam Thi Bich Hanh, Dang Thi Thuy My, Nguyen Phuong Dai Nguyen, Truong Ba Phong, Nguyen Thi Mai Huong, Manh Hung Tran, Nguyen Thị Thu Tram and Dao Cuong To in European Journal of Inflammation
Footnotes
Author’s contributions
PHN, NPDN, NTTT designed and planned the experiments. NTN, DTBH, DTTM, TBP, NTMH implemented the experiments. MHT, DCT investigated the data and wrote the manuscript. All the authors agreed the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
