Abstract
Background
Numerous studies have shown that yoga can aid patients with Parkinson’s disease (PD) in managing their motor function, functional mobility, balance, anxiety, and overall quality of life. However, most of the reported research either needed more power or evaluated the results during or after the intervention. Due to the long-standing nature of PD, there are still few longitudinal studies investigating the potential of yoga to decrease the progression of the disease.
Purpose
We study whether sustained adjunct yoga intervention slows PD progression.
Methods
Over 18 months, this study will investigate the long-term benefits of yoga-based intervention in Parkinson’s patients. The concept of “intention-to-treat (ITT)” will be employed. From the outpatient clinic of the Swami Vivekananda Yoga Anusandhana Samsthana in India, 160 patients with PD will be chosen. They will then be randomly assigned to either the interventional or control groups (1:1). The adjunct intervention group will participate in 21 daily contact yoga sessions (60 min) at the University Medical Center, followed by continued tele-supervised home practice and at-home self-practice. The effectiveness of the adjunct intervention will be evaluated at four different time points (baseline, 3-, 9-, 12-, and 18-month post-randomization). The primary endpoint will be the difference in the Movement Disorder Society-Unified Parkinson Disease Rating Scale (MDS-UPDRS) Part III score at baseline and after 18 months of intervention. Secondary outcomes will include subscores of MDS-UPDRS I, II, and IV, Parkinson’s Disease Questionnaire (PDQ)-39 scores, treatment adherence, the PDQ-39, the Depression, Anxiety, and Stress Scale-21, and the Scales for Outcomes in PD-Sleep Scale (SCOPA-S). The primary outcome will be analyzed using mixed-effect models for repeated measures, adjusted for covariates as fixed effects.
Results
The protocol was conceptualized in October 2021 and approved by the Institutional Ethics Committee of SVYASA. Recruitment began in February 2022 and is underway with patient enrollment.
Conclusion
To our knowledge, this is the first controlled study to examine the long-term effects of yoga as an adjuvant therapy on the progression of PD.
Key message
This would be the first long-term trial to assess the potential of an adjunct yoga intervention to modulate disease progression in early Parkinson’s patients.
Introduction
Exemplified as a movement disorder, Parkinson’s disease (PD) is one of the most rapidly growing neurodegenerative conditions. The rising prevalence of PD has been projected to have an epidemiological match with pandemics. 1 The manifestations of PD are multifaceted and involve both motor and nonmotor symptoms. 2 Slow movement (bradykinesia), tremor, poor postural stability, muscular stiffness (rigidity), ataxia, abrupt freezing of movement, and lack of spontaneous muscular movements (akinesia) are the hallmark motor signs of PD. 2 Significant progress has been made in understanding the pathobiology of PD. 3 The major core motor features manifest due to the loss of dopaminergic neurons in the substantia nitre pars compacta, which project axons to the striatum. 4 Therefore, the underlying degeneration of dopaminergic nigrostriatal neurons and subsequent striatal dopamine deficiency form the basis for pharmacotherapy for PD.4−6 Several symptomatic therapies, such as dopamine replacement and deep brain stimulation, greatly benefit PD patients.4−6 However, the effectiveness of these drug treatments is limited to the early stages of disease progression and is further complicated by the development of medication-resistant symptoms associated with disease progression. 7 The motor symptoms can be effectively treated with drugs such as levodopa, especially in the early stages of PD8–12; however, as the disease progresses, medication-resistant symptoms, such as postural instability, become more challenging to treat. No pharmacological treatments have been shown to delay the disorder’s progression or alter the disease course, especially in the early stages.8−12 On the contrary, there is strong evidence that regular and long-term exercise can slow the progression of PD.8, 13 The potential benefits of complementary therapies are supported by several lines of evidence, particularly those related to exercise-based modalities, including yoga, tai-chi, and tango, against both motor and nonmotor symptoms of PD.13−16 In this regard, yoga has been mainly considered a helpful intervention in PD patients, given its mild approach and the restricted ability of PD patients to engage in complex or intense exercises.17, 18 Several studies support the idea that yoga practice could significantly improve the scores on Unified Parkinson Disease Rating Scale (UPDRS)-III.19−22 A recent meta-analysis also supported the beneficial influences of yoga on motor coordination, balancing function, functional mobility, anxiety, depression, and the overall quality of life of PD patients. 22 Moreover, stretching exercises and extended physical positions in yoga align with the recommendation for stretching in the rehabilitation of PD to alleviate rigidity.19, 23, 24 The practice of yoga stretching postures has also been proposed to aid in the muscle strength of the erector spinae, gluteus maximus, and rectus abdominis, increasing the stretching distance of the body, expanding the range of joint activities, and enhancing the flexibility of the lumbar, knee, and ankle joints. 25 Based on the abovementioned results, yoga can make the limb and joint activities of individuals with PD more flexible, reduce bradykinesia and rigidity, and increase muscle strength and power. At the molecular level, yoga has been proposed to improve oxidative stress in different disease settings effectively. 20 Overall, yoga has clear short-term benefits for PD symptom management. However, the literature on yoga research is limited by a lack of research protocols and trial registration information reporting. 22
Given the long-standing nature of PD and the progressive deterioration of symptoms in the motor and nonmotor domains, it becomes imperative to conduct longitudinal and long-term studies to establish the influence of the intervention on disease progression. However, none of the previous trials on yoga reported the effects on disease progression based on a longitudinal assessment of relevant clinical outcomes, such as the Movement Disorder Society-UPDRS (MDS-UPDRS) Revision. Thus, we aim to conduct a randomized, controlled clinical trial to investigate the long-term effect of yoga on disease progression in early PD patients.
Methods
Study Design
The study will be a prospective, parallel-group, single-center, and two-arm randomized controlled trial. The subjects will be randomly allocated to two groups: adjunct yoga and standard of care. The protocol has been drafted following Consolidated Standards of Reporting Trials (CONSORT) (Figure 1). 26 The study will be conducted between March 2022 and March 2024. A total of 160 adults (aged ≥18 years) with a clinical diagnosis of idiopathic PD will be recruited from the outpatient department of Swami Vivekananda Yoga Anusandhanan Samsthana, Bangalore. Eligible participants will be randomized 1:1 to adjunct yoga or standard of care. An external statistician not involved in the study will do the randomization procedure through randomization software. The allocation sequences will be sealed, and immediately after baseline assessments, the participants will be informed about the further process. Investigators involved in evaluating outcome measures would be blinded to the randomization groups.
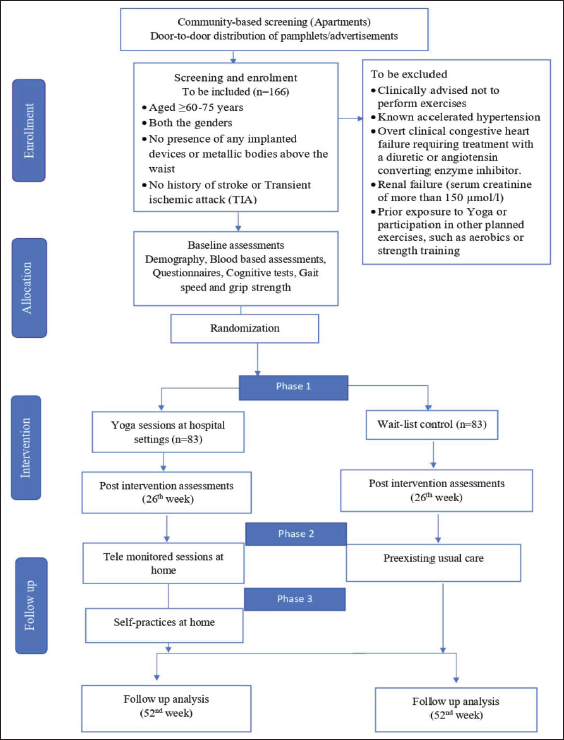
Primary and secondary outcomes will be assessed at baseline (before intervention) and at 3-, 6-, 12-, 18-month visits over a 545-day timeframe (with a 60-day window of flexibility) (see Figure 1). All PD participants will follow their regular medication scheme during the study period. This protocol has been approved by the Ethics Committee of Swami Vivekananda Yoga Anusandhana Samsthana (RES/IEC-SVYASA/179/2020). All participants who meet the inclusion criteria will sign informed consent prior to the study.
Participants with idiopathic PD will be enrolled using convenience sampling. A clinical diagnosis of PD will be made according to the UK Brain Bank criteria and confirmed by two movement disorders specialists. Patients aged ≥18 years will be eligible if they are on stable dopaminergic medication for 4 weeks before enrolment, with a disease duration of fewer than 3 years from diagnosis, mild to moderate disease severity (Hoehn and Yahr stage 2–3 mild to moderate disease), 27 not having practiced any form of yoga regularly in the past 6 months, not currently participating in a supervised exercise program for more than 2 days a week, and no history of falls in the past 3 months.
Patients will be followed for 18 months or until they withdraw from the study. Patients will be excluded if they have a neurological history other than PD; significant arthritis; those with uncontrolled cardiovascular, musculoskeletal, or nerve disease; have not been permitted for exercise by their physician; and those who had any recent surgical interventions.
Study Outcomes and Assessments
The study’s primary goal is to compare the effectiveness of an adjunct yoga intervention to the standard of care in recruited participants with PD at week 72. The primary outcome is the difference from baseline in the MDS-UPDRS III at week 72 for adjunct yoga compared to standard of care. As shown in the CONSORT, other than baseline, assessments will occur at 6, 12, and 18 months over a 545-day timeframe (with a 60-day window of flexibility) (Table 1). The endpoint of the 18th month has been chosen as the longer-term outcome. The MDS-UPDRS Part III (motor examination) is a 33-item rater-assessed evaluation of motor signs, with each item rated 0–4. 28 The motor examination scores will be created by summing the ratings, with higher scores indicating worse motor signs. Scores for one side of the body range from 0 to 44, with higher scores indicating more significant motor impairment, and include subitems for rigidity, bradykinesia, and tremor. (Stages I–V, with stage I indicating unilateral involvement only and stage V indicating confinement to a bed or wheelchair.) At baseline and other study time points, the subscores of the MDS-UPDRS Parts I (non-motor experiences of daily living), II (motor experiences of daily living), and IV (motor complications) will also be collected. These subscores will be followed up depending on symptoms encountered the week before the assessment. The same rater evaluating the baseline MDS-UPDRS scores will also assess all subsequent follow-up evaluations. At each appointment, adverse events and current drugs will be noted. Additional outcome measures will be the percentage change in levodopa equivalent dose (SURE-PD D), the 39-item Parkinson’s disease Questionnaire (PDQ-39) and the Scales for Outcomes in PD-Sleep (SCOPA-SLEEP). At baseline, participant’s demographics and disease-related data will be collected. The dosages of the medications will be expressed as their daily SURE-PD Ds since the patients will be taking different types of anti-Parkinson drugs. The prior data and the reported conversion factors will be used to calculate the SURE-PD D for specific medicines. 29
Schedule of Enrolment, Interventions, and Assessments, According to SPIRIT 2013 (40) Guidelines.
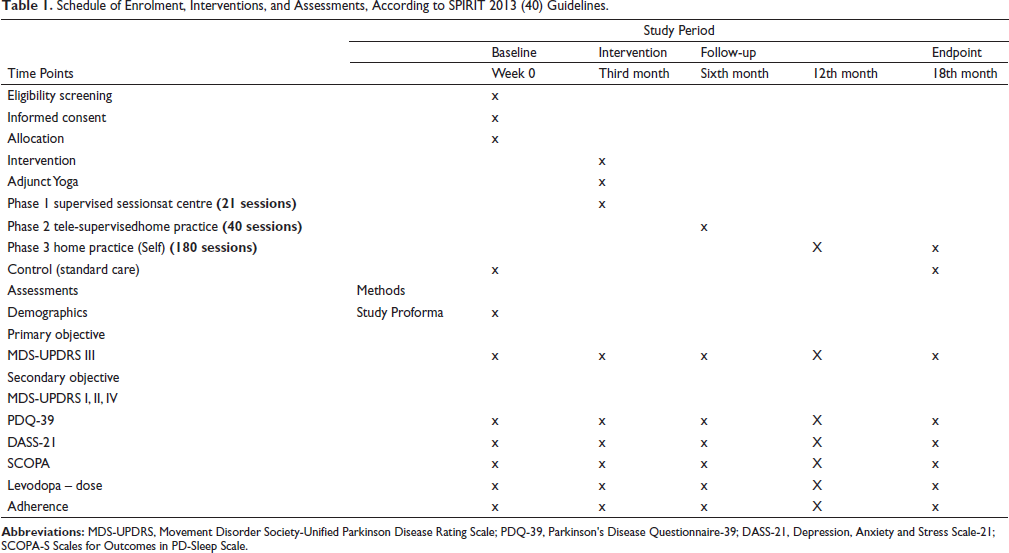
Quality of life will be assessed using the PDQ-39; scores range from 0 to 100, with higher scores indicating poorer well-being. Patients will evaluate their health-related quality of life (HRQoL) using the PDQ-39. 30 The PDQ-39 consists of 39 items organized into eight distinct sections, covering mobility, daily living activities, stigma, social support, cognition, communication, and physical discomfort. Higher scores indicate significantly worsened HRQoL; the final score is calculated by adding the scores of all items, dividing by the highest score allowed, and multiplying the result by 100.
The SCOPA-Sleep is a PD-specific rating scale for assessing nighttime sleep (NS) problems and daytime sleep (DS) problems in the past month. 31 It includes 12 items to measure sleep quality, NS disturbances, and DS. We will use the 21-item Depression, Anxiety, and Stress Scale (DASS-21), where the 21 items are distributed into three subscales to assess symptoms related to depression, anxiety, and stress in the last week. 32 The intensity of symptoms is scored for each item, ranging from 0 (absent) to 3 (highly frequent) to assess the intensity. The total score for each subscale was obtained by summing the item scores and multiplying the result by 2. For depression, a score <9 was normal, 10–13 mild, 14–20 moderate, 21–27 severe, and >27 extremely severe. A score <7 was normal for anxiety, 8–9 mild, 10–14 moderate, 15–19 severe, and >19 extremely severe. Regarding stress, a score of <14 was normal, 15–18 mild, 19–25 moderate, 26–33 severe, and >33 extremely severe.
Adherence is calculated based on participation in sessions. The trainer will maintain a logbook during phase 1 and phase 2; notepads will be provided to the individual participants to preserve records by themselves during phase 3. All these will be collected during follow-up assessments. Attendance rates for individual participants will be calculated by dividing the number of sessions attended by the total number of sessions conducted. Overall attendance will then be calculated by averaging all participants’ attendance rates. Attrition rates will be reported, defined as the number of individuals who dropped out of the study divided by the total number of enrolled participants. The reasons cited for study dropout will also be summarized.
Intervention
The adjunct yoga group participants will be given intervention 3–5 days a week. Qualified yoga professionals will deliver the intervention (Table 2). In the initial orientation at the center, participants will be given two 45-min group-based sessions, which will consist of a 1-min introduction followed by 5 min of gentle loosening movements, 30 min of postures followed by 2 min of relaxation, followed by 12 min of breathing practices, and ending with guided meditation (5 min). Overall, the adjunct yoga intervention will be divided into three phases: Phase 1 includes supervised sessions at the center, including the orientation. In Phase 2, tele-supervised at-home practice (40 sessions), participants will be monitored from their home settings in tele-mode through online platforms for 40 sessions, and Phase 3 comprises only at-home yoga practice for the next 9 months (180 sessions) (Table 2).
Schedule of Yoga Sessions.
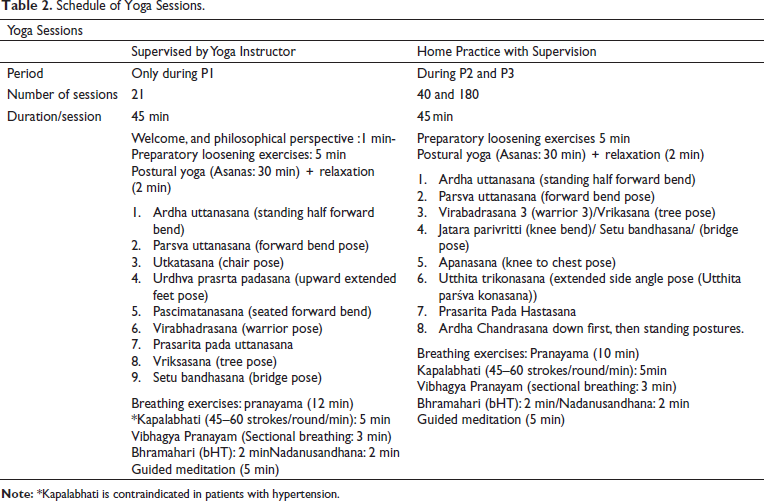
The protocol has been designed to incorporate standing postures to strengthen the quadriceps, tibialis anterior (TA), and gastrocnemius (GA) to target gait and balance deficiencies. TA activation, in particular, has been targeted in most of the pharmacological interventions for PD, based on the idea that TA may be targeted more because it has more inputs from the cortex to its motor neurons than other lower limb muscles.33, 34 Electromyography-based experiments have indicated the impact of several poses on the activation of the TA and GA. Those postures are the tree pose (Vriksashana), half-moon posture (Ardha Chandrasana), and warrior III (Virabhadrasana)]. 25 The tree pose also requires the participants to attain and maintain unilateral standing. 35 Other poses included in the protocol, such as adho mukha svanasana (downward-facing dog pose), have been reported to increase upper-extremity weight bearing and promote stretching of the GA/soleus groups and hamstrings, and bandha konasana (bound angle pose) to improve hip range of motion. 35 Setu Bandhasana (bridge pose) was included based on the reported strengthening effect on the overall lower extremity.35, 36 An earlier study verified that Warrior 3 has unique training effects on the hamstring. 36
Based on our prior experiences with yoga-based interventions, we also aim to incorporate the following strategies to reduce attrition and minimize loss to follow-up: being responsive to participants or spouse/care partner questions and the yoga coordinators meeting with participants throughout the study, occasionally traveling to the participant’s fitness facility or home, which should enhance adherence to the programmer and allow the development of a strong rapport. To reduce the travel burden on participants in light of holidays, business commitments, or other factors impacting adherence to the study, we have also added periodic tele-supervisions.
Statistical Analysis
Sample Size
The study sample size was calculated to achieve 80% power at α = 0.05 using standard methods for a two-sided pairedt-testwith standard deviation (SD) estimates, and target mean approximated from prior PD intervention study findings of SURE-PD report. 37 Adopting the same, we assumed that the study would have 80% power with n = 170 subjects randomized 1:1 to yoga or standard of care if the effect of the yoga-based adjunct intervention were to reduce the 1.5-year increase in MDS-UPDRS by a 15% reduction in the expected rate of progression.
After screening for inclusion and exclusion criteria, participants will be invited to baseline data collection yoga classes for 21 days after that second-time data collection, then continue 3 months again third-time data collection, again yoga class up to 9 months after that data collection. Data will be extracted based on the standard scoring key and the appropriate procedure described in the commercial kits. Data will be analyzed by using Microsoft Office Excel and R Studio software.
Baseline characteristics will be presented using appropriate descriptive statistics. Using the Kruskal–Wallis analysis, variable distributions will be checked before analysis to ensure that the normality assumptions are met. The independent test for continuous variables and the chi-square or Fisher’s exact test for categorical variables will be used to compare differences between the two groups. The baseline characteristics of the participants who completed the study will be compared with dropouts. The following factors will be included in the model for effectiveness analyses of each continuous endpoint, primary, secondary, or exploratory outcome; the background therapy at baseline age, sex, and SURE-PD D. Pearson’s regression will also link intervention outcomes with participant scores for high, medium, and poor adherence.
Study outcomes will be compared between groups based on the intention-to-treat (ITT) principle. A mixed-effect model for repeated measurements will examine the change in MDS-UPDRS Part III from baseline versus control, with covariates adjusted as fixed effects. The SURE-PD D, age (years), education (years), sex, smoking status, and baseline MDS-UPDRS sum of Part III scores will all be covariates. The model will also have terms for treatment-by-week interactions. All statistical analysis will be performed using IBM SPSS 24.0 software. Two-tailed tests will be used, and statistical significance will be set at a p-value <0.05.
Furthermore, assuming an attrition rate of 25% and missing data, we also aim to conduct sensitivity analyses using the multiple imputation method and subgroup analyses for age, gender baseline H and Y scores. A support staff member will carry out the data entry, and participants will be assigned specific numbers. Data access will be limited to the concerned research team.
Discussion
This study will add a dimension of sustenance of effects to the existing body of literature on the effectiveness of yoga against the management of PD symptoms. If found in the study, results will provide the first proof of evidence that yoga could aid in slowing down the disease progression of PD. This will be a valuable addition to the existing literature, given the need for such studies in yoga research in the purview of the long-lasting nature of PD. Recent research has also demonstrated that yoga promotes neuroplasticity in conditions like depression and can raise neurotransmitter levels in specific brain regions.
Secondarily, the study will shed light on the impact of adjunct yoga intervention—which has not been extensively studied in a longitudinal design on the quality of life and related sleep and anxiety issues in PD. This study is based on the proceeding from prior studies and has been designed with adequate sample size calculation, including long-term follow-up period along with a follow-up of 12 months from baseline, adding the required rigor to the study.
The corpus of studies on using yoga to treat both motor and nonmotor symptoms in patients with chronic illnesses, including neurodegenerative ailments, may be expanded due to this study. Yoga is a desirable adjunct therapy for PD patients who experience repetitive and progressive imbalances brought on by degenerating PD conditions while remaining active in daily and recreational activities, given that it has few side effects and is cost-effective compared to other current treatments. The validated outcomes will attest to the efficacy of the approach that will be examined and modified for additional chronic conditions that cause both motor and nonmotor deficits, such as stroke. This will also be the first report to provide exploratory insight into attrition probabilities and response rates in long-term yoga-based trials. Given that yoga-based interventions could be provided in home settings, the results will influence the current paradigm of long-term symptom management of PD. This is the first study that will use a mixed contact and home-based approach to deliver yoga intervention to manage the progression of PD.
Conclusion
Overall, this would be the first long-term trial to assess the potential of adjunct yoga intervention to modulate the disease progression in early Parkinson’s patients.
Footnotes
Abbreviations
PD: Parkinson’s disease;
UPDRS: Unified Parkinson Disease Rating Scale;
PDQ-39: Parkinson’s Disease Questionnaire-39;
DASS-21: Depression, Anxiety and Stress Scale-21
SCOPA-S: Scales for Outcomes in PD-Sleep Scale;
CONSORT: Consolidated Standards of Reporting Trials;
TA: Anterior Tibialis;
GA: Gastrocnemius;
NS: Nighttime sleep;
DS: Daytime sleep;
SURE-PD: D Levodopa Equivalent Dose;
ITT: Intention-to-treat;
Authors’ Contribution
PS: data collection, analysis, and project coordination; VM: study conceptualization, project supervision and manuscript writing; MNK: review of manuscript; KS: review of manuscript, study conceptualization; AS: data curation
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
ICMJE Guidelines
The manuscript complies with International Committee of Medical Journal Editors (ICMJE) guidelines.
Statement of Ethics
The study was initiated after approval from Institute’s Ethics Committee (RES/IEC-SVYASA/179/2020) and after obtaining registration of the clinical trial; Clinical Trial Registry of India (CTRI/2022/01/039116).
