Abstract
Background
Vulnerable women face a higher risk of cervical cancer and reduced access to screening. Self-sampling may increase access to human papillomavirus screening. In this article, we report the co-creation of a document to guide the implementation of human papillomavirus self-sampling in Italy.
Methods
Working CollaBoratively with vulnerable women to Identify the best implementation Gains by SCREEning cervical cancer more effectively in European countries (CBIG-SCREEN) project, a Collaborative User Board has been set up in Italy, including national decision-makers, healthcare providers, and vulnerable women. To ensure that women's direct perspectives were incorporated into the process, informal individual interviews were carried out and brought to the Collaborative User Board meetings. The Collaborative User Board discussion was based on systematic reviews, screening program reports, and Italy's self-sampling pilots. Results were summarized with a matrix of vulnerable groups and barriers to evaluate interventions’ potential ability to remove barriers. Considerations were collected in a document for screening program coordinators and decision-makers.
Results
The Collaborative User Board identified major barriers to cervical cancer screening among different vulnerable groups and assessed five evidence-based strategies to address them: multilingual structured invitation letters, group training led by cultural mediators, training for healthcare professionals, training for third-sector operators, and self-sampling. Evaluating the potential of these strategies to remove barriers led to a focus on self-sampling. Mailing the device to all non-responders was the only strategy with sufficient evidence to increase participation in Italy. An opportunistic offer in a setting attended by vulnerable women is promising. The Collaborative User Board underscored the importance of improving healthcare providers’ communication.
Conclusions
The final document, created through a transparent and collaborative bottom-up process, collects considerations for implementing self-sampling for human papillomavirus testing.
Background
Cervical cancer rates in Europe are declining both in terms of incidence and mortality. The World Health Organization (WHO) aims to eliminate this cancer as a public health issue by 2030, reducing its status to that of a rare disease. 1 With this aim, WHO launched the strategy aiming for 90% vaccine coverage, 70% of women screened with at least one highly effective test by the age of 35 and again by the age of 45, and 90% of women with precancer or cancer receiving adequate treatment. 1 In many European countries, screening and treatment coverage already exceed the thresholds, but the incidence remains above the WHO elimination threshold of 4 per 100,000. Annual world age-standardized incidence rates vary from 6.8 per 100,000 women in Western Europe to 16 per 100,000 women in Central and Eastern Europe.2,3 Implementing vaccination will take several years before it substantially impacts incidence rates across all age groups. Moreover, coverage of screening services varies among women. Those in more vulnerable circumstances—such as women living with human immunodeficiency virus, women with substance or alcohol use disorders, female sex workers, and women in low socio-economic conditions—are often at an increased risk of developing cancer and typically have less access to screening facilities 4 (Box 1). Cervical cancer incidence and mortality rates vary significantly between different subgroups within countries and among European Union countries. Current programs frequently fail to engage the vulnerable subpopulations of women who account for a large portion of both cases and fatalities (Table 1).
The picture of screening coverage inequalities in Italy.
Screening test coverage is lower among women with limited education and economic hardship. Cervical cancer mortality is also markedly higher in women with low educational attainment compared with those who hold a university degree. 15
Women with a migrant background show lower screening coverage than Italian-born women; however, this gap narrows as the duration of residence in Italy increases. 16 Among women who have lived in Italy for more than 10 years, no differences in coverage are observed.
There is substantial heterogeneity in how screening programs are organized across Italian regions, and this variability affects participation rates. In areas where programs are less accessible to the general population, participation becomes even more challenging for migrant women. In these regions, the private sector plays a more prominent role, but it remains less accessible to immigrants, resulting in a larger coverage gap than in regions where public programs are more accessible.17,18
Relative risks of cervical cancer and estimates of the number of cancers for some vulnerable women subgroups.
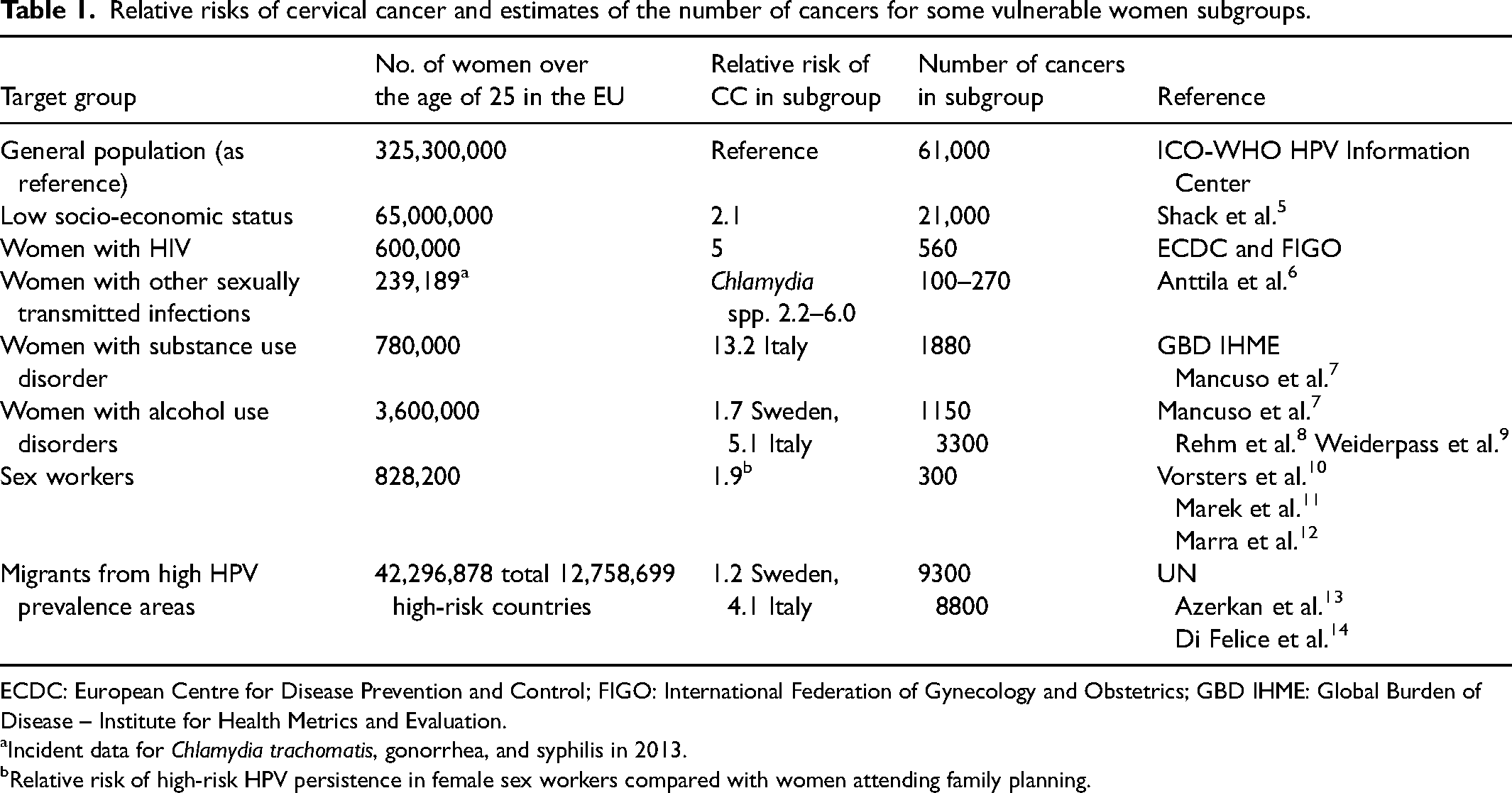
ECDC: European Centre for Disease Prevention and Control; FIGO: International Federation of Gynecology and Obstetrics; GBD IHME: Global Burden of Disease – Institute for Health Metrics and Evaluation.
Incident data for
Relative risk of high-risk HPV persistence in female sex workers compared with women attending family planning.
The CBIG-SCREEN project (working CollaBoratively with vulnerable women to Identify the best implementation Gains by SCREEning cervical cancer more effectively in European countries), funded by the European Union, aims to enhance the involvement of the most disadvantaged populations in the cervical cancer screening process, which includes stages from human papillomavirus (HPV) testing to eventual triage, assessment, and treatment. By doing so, the project seeks to significantly reduce inequalities in both the incidence and mortality rates of cervical cancer across European countries and expedite the elimination of this disease. 19
The project, therefore, aims at identifying and developing interventions to facilitate the participation of the most vulnerable women from the initial test to possible treatment through continuous collaboration with operators, decision-makers, and women, answering their specific needs.
In December 2022, the European Council issued new recommendations regarding cancer screening for member states. For cervical cancer, the European Council recommends implementing screening with an HPV test from 30 to 65 years of age, with at least a five-year interval. They also suggest considering the use of self-sampling to increase coverage, especially for non-responders to screening invitations. 20
The present article presents the final considerations and outlines how the CBIG-SCREEN project in Italy, through the work of the Italian Collaborative User Board (CUB), led to the co-creation of a document, 21 providing guidance for the implementation of the European Council's recommendations on HPV self-sampling within Italian screening programs. Coordinated by the National Centre for Screening Monitoring (Osservatorio Nazionale Screening, ONS), a technical body of the Italian Ministry of Health (https://www.salute.gov.it/imgs/C_17_notizie_6748_0_file.pdf), the process made use of stakeholder and user involvement initiatives following the methodology defined by the CBIG-SCREEN project.
Since the process was not organized as a guideline development project, it could not produce formal clinical recommendations. Instead, it focused on developing considerations for implementing the European Council's recommendations, identifying organizational and training needs to improve vulnerable women's participation in cervical cancer screening, identifying research priorities, and framing clinical questions to guide future recommendations on self-sampling within the Italian cervical cancer prevention guidelines. 22
Methods
Setting and actors
The process and the contents of the ONS document 21 have been developed within the Italian CUB 23 constituted for the CBIG-SCREEN project, funded by the European Commission. 24 This was one of the seven CUBs conducted in seven European countries (Denmark, France, Italy, Estonia, Romania, Bulgaria, and Portugal) during CBIG-SCREEN. The process was performed with the scientific support of the CBIG-SCREEN partners under the coordination of the University Research Clinic for Cancer Screening (UNICCA, Denmark).25,26 The coordinator of the CBIG-SCREEN project is INSERM (French National Institute for Health and Medical Research), supported by the technology-transfer subsidiary INSERM TRANSFERT SA.
ONS and the Local Health Authority of Reggio Emilia, as the Italian partner of the CBIG-SCREEN project, promoted and established a CUB involving decision-makers, professionals, and potential screening users.
The composition of the Italian CUB (Supplemental Material 1) included three levels of stakeholders:
Macro: Actors involved in decision-making at the national and regional levels. Meso: Actors responsible for implementing healthcare decisions, such as screening program coordinators, and other professionals working in the healthcare and social sectors. Micro: Women belonging to the vulnerable subgroups for whom the services are directed, providing indirect input that helped inform the process.
The work of the CUB was coordinated and facilitated by researchers from the Local Health Authority-IRCCS in Reggio Emilia (two epidemiologists, one cultural anthropologist, and one sociologist, experts in qualitative research).
The micro-level involvement
The CUB participants were identified and invited to participate by the ONS to guarantee diversity in views by role, profession, geographical area of origin, and expertise, following the methodology previously described. 25 Women in vulnerable situations, along with two cultural mediators who had experience at both micro and meso-levels, found it challenging to engage in the online meeting. They consented to a brief interview, which was presented during the first CUB meeting. Furthermore, for the fourth meeting, micro-level feedback about the possible use of self-sampling was brought into the CUB through the meso-level stakeholders who work closely with them. Briefly, the CUB members in direct contact with vulnerable women asked semi-structured open questions (Supplemental Material 2). The questions aimed to understand needs, behaviors, habits, and ideas regarding the possibility of using self-collection. The meso operators reported the answers using structured templates, which were then analyzed with the help of qualitative researchers and discussed in one of the CUB meetings.
Structure of the process
Four online CUB meetings were conducted. The first two focused on stakeholders and women's experiences of the barriers that hinder access to screening and identified the vulnerable groups of women to be considered. The second two meetings focused on possible solutions to remove the identified barriers (Figure 1).
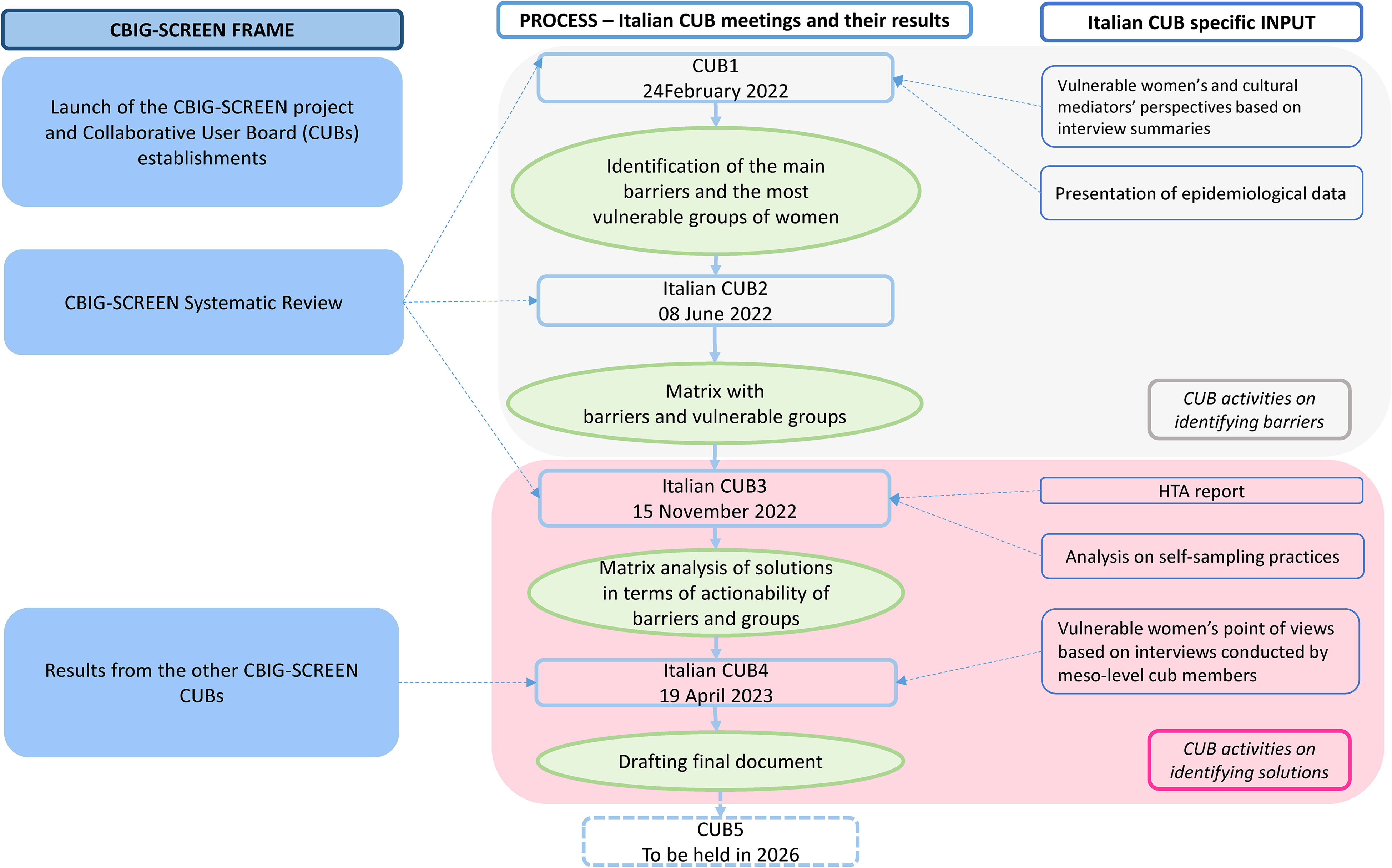
Main steps of the process, inputs, and instruments used during the Collaborative User Board (CUB) meetings and analytical theme.
Experts in qualitative research techniques supported the CUB activities. The facilitators adopted tools to encourage active participation in the discussion, such as electronic dashboards and a matrix to organize the inputs for the board participants. Furthermore, a back-office activity made it possible to document discussions through minutes, helped introduce and structure the discussion topics, and summarized the board's outputs.
During the first meeting, CUB participants were informed about screening coverage and inequalities in screening access in Italy through a presentation with infographics reporting data from the ONS 27 and from the PASSI survey, a National Health Interview 17 (Box 1).
To initiate the barrier analysis, the participants were encouraged to discuss the screening coverage and cervical cancer prevention inequalities in Italy (Box 1), the systematic review of the barriers conducted by CBIG-SCREEN investigators (Figure 2), and the interviews of three of the micro and meso-level participants.
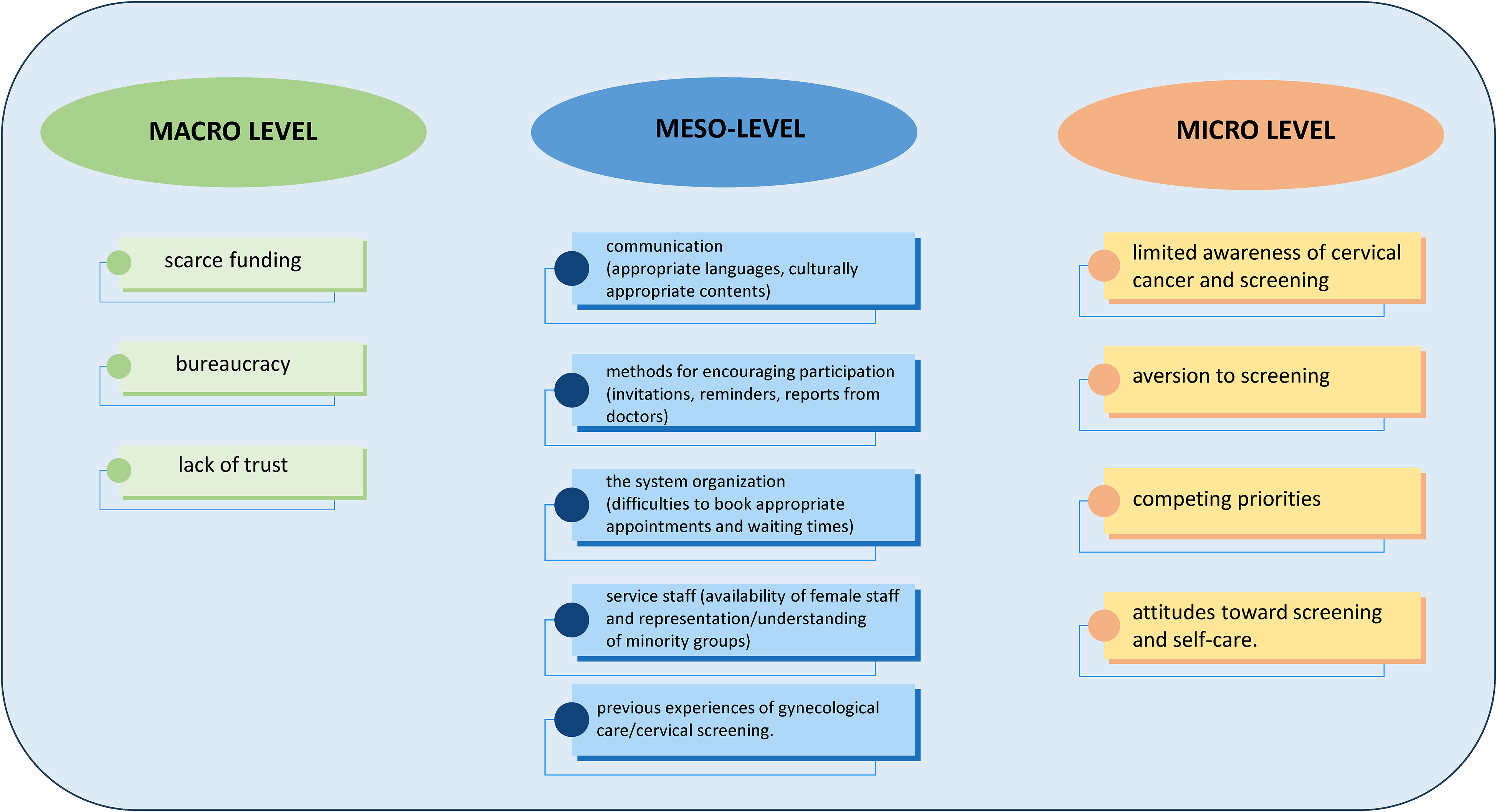
Barriers to cervical cancer screening in vulnerable women identified by the CBIG-SCREEN systematic review. 28
Furthermore, the Italian CBIG-SCREEN investigators collected and summarized the results of all the published and unpublished studies and pilot programs on self-sampling. 29 The results were presented during the third CUB meeting with infographics and facilitation by the investigators of the Italian studies. Moreover, to complete the overview of the effective interventions to increase participation among vulnerable and disadvantaged women, CUB participants received a short presentation of the executive summary of the Health Technology Assessment (HTA) report funded by the Italian Ministry of Health on methods to increase participation in screening 30 (Box 2).
Interventions aimed at increasing screening coverage.
What works:
Postal reminders Telephone reminders, but have only been evaluated in small studies that do not consider feasibility and sustainability in population programs The signature of the general practitioner on the invitation letter The scheduled appointment Interventions to bring the provider closer and reduce logistical barriers Self-sampling for HPV testing Specify that the cervical sample will be carried out by a woman
What does not work:
Long and overly detailed letters The addition of brochures and information material Offer the option between Fecal Occult Blood Test and sigmoidoscopy
What is not known whether it works or not:
Reporting (reminder) to the family doctor that women are late in carrying out checks Mass information campaigns.
Reminder and incentives for women (invitation letter) Group training and individual training (education provided in a targeted language, accessible teaching material, adapted, and individual consultation) Feedback and incentives for healthcare professionals (reminder and doctor commitment) Reduction of structural barriers and out-of-pocket expenses for the assisted (self-collection or self-sampling).
Data synthesis
The barriers and vulnerable groups identified by the literature and integrated by the CUB participants were used as columns (vulnerable groups), rows, and cells (barriers) of a matrix. This matrix was subsequently used to identify particular mechanisms and phenomena through which barriers affect a specific group of vulnerable women.
The matrix was then used to define the theoretical usefulness of a putative solution to remove the barriers. For each cell of the matrix, containing a specific barrier, the participants discussed whether the proposed solution would make the specific barrier actionable for the different vulnerable groups and at which level of resolvability/actionability (low, medium, or high). The discussion was then summarized in a qualitative color code, following a traffic light system (red: low actionability; yellow: medium actionability; and green: high actionability).
Results
During the first CUB meeting, three interviews were analyzed: one with a woman connected to pathological addiction services and two with cultural mediators. We recognized two major barriers:
In some contexts, the idea of secondary prevention is uncommon, and undergoing a health visit/test without a health problem seems unreasonable. The health service organization is unclear, and the invitation with a letter is ineffective because it is difficult to understand, and arranging an appointment is troublesome.
After a structured brainstorming activity and back-office work conducted between the first and second meetings on barriers, the CUB participants defined the matrix of barriers and vulnerable groups, which served as the basis for conducting an analysis on solutions.
An initial list of the vulnerable groups was defined by the project; the National CUBs had the possibility to adapt the list to their local context by adding or removing groups. The structured brainstorming defined a list of barriers and grouped them according to the setting where the barriers originate or the mechanism of action of the barrier. CUB participants suggested that each barrier could affect different groups in distinct ways, both qualitatively and quantitatively. As a result, each single barrier could be placed in a matrix in which the kind of barriers were listed in columns and the vulnerable groups were reported in rows.
Starting from the solutions identified by the systematic reviews 28 and considering the Italian HTA report on methods to increase screening participation, 30 summarized in Box 2, the CUB participants defined a short list of interventions that were consistent with the Italian health system and had evidence of efficacy to increase vulnerable women's participation in screening.
Five types of interventions were identified:
Structured Invitation letters in multiple languages with explanatory material in various languages. Group training carried out by cultural mediators and community reference people, such as immigrants, peers, and healthcare workers. Training of general practitioners (GPs)/other primary care workers, with an active role of the GP/primary care workers in inviting the woman. Third-sector training, non-healthcare association services. Self-sampling was sent to all women, with explanations in multiple languages and easy-to-read information material.
The CUB participants then analyzed the first four interventions using the barrier/vulnerability groups matrices. For each intervention, the CUB participants discussed its potential ability to act on the barriers identified in every single cell of the matrix. They summarized this judgment in the red, yellow, green traffic light code. This was a qualitative assessment conducted by consensus among the board participants: red signifies a limited capacity of the intervention to surmount the barrier, yellow indicates a moderate ability, while green denotes a capability to effectively dismantle the barrier. The four matrices are reported in Supplemental Material 3.
The focus on self-sampling as a possible intervention
To better define how self-sampling could be implemented, the CUB analyzed the results of international literature,28,30,31,32 Italian studies,33,34 and the pilots.29,35 For the latter, at the time of the CUB, the results were not published in peer review journals, so they were in part reported directly by the investigators that conducted them, and in part summarized by the facilitators (Figure 3). The strategies targeting the general population in Italy were all shown to be ineffective in increasing participation or even had a negative effect.29,35 Among the strategies targeting non-responders and eventually applicable specifically to vulnerable women, the strategy of mailing the self-sampler to all women (often referred to as an opt-out strategy, because the woman has to actively ask to not receive the sampler) was shown to be efficacious in increasing participation in Italy and, consistently, in the international literature. For the strategies requiring an active request by the woman to receive the sampler (often called opt-in), there are conflicting results. In Italy, the option to collect the sampler at a pharmacy had different results in different geographic areas, 34 while the option to call the screening center to receive it at home did not increase the participation in any Italian center that tested the strategy. 33 Finally, at the time of the CUB, for the active offering of the sampler in settings where vulnerable and underserved women can be opportunistically contacted, the systematic review (green) retrieved only one study 36 with promising results.
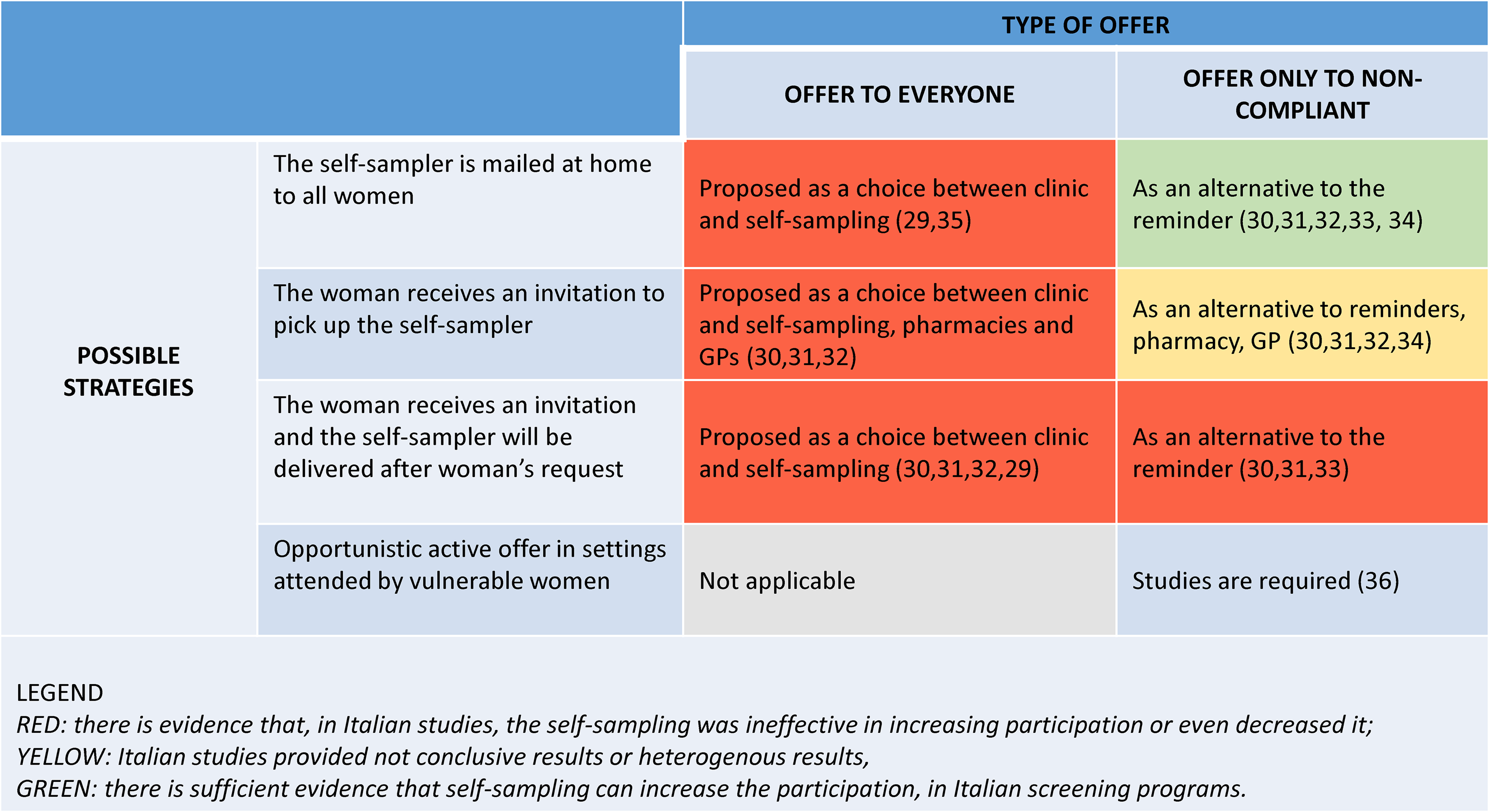
Self-sampling: possible strategies and populations, and evidence of effectiveness in increasing participation based on Italian and international studies.
Collecting women's voices: Summary of the interview analysis results
CUB participants at the meso-level collected (Supplemental Material 2) and reported the perspectives of 41 women, reflecting micro-level insights on self-sampling. These included women with mental disabilities cared for by the Services of the Mental Health Department, women guests with their children in residential communities (often single mothers, without family support, and often in conditions of poverty), women with addiction problems, and young Nigerian women who had recently arrived in Italy.
GPs were perceived as key points of reference when women experienced a health problem, sought routine health check-ups, or wished to obtain health-related information. In contrast, participants reported that gynecologists primarily serve to direct individuals toward completing Pap tests or HPV tests, particularly following an invitation letter for screening.
About half of the women (
Although GPs emerged as a point of reference, participants rarely mentioned GPs as a channel for either collecting the self-sampling kit or returning the completed test. Women expressed a preference for receiving the kit at home, collecting it at a pharmacy or clinic, and returning it to a healthcare facility or clinic.
Considerations of the CUB participants
Key considerations and reflections from the Italian CUB participants highlight barriers, facilitators, and potential strategies in cervical cancer screening, including the effectiveness of communication, access to free screening, opportunities to raise awareness and identify vulnerabilities, and the use and acceptability of self-sampling in specific settings, taking into account the particular characteristics of different vulnerable groups and associated barriers.
- Communication and relationships should be ongoing, occurring at all stages of the screening process. Clear communication helps reveal specific vulnerabilities. Identifying and engaging community leaders to promote awareness and spread information is crucial. Additionally, the relational aspect involves paying close attention to how services receive and respond to communication. - Free screening: The free-of-charge offer is important not only for overcoming economic barriers but also as a tangible aspect of the importance given by institutions to screening, helping to overcome barriers due to low priority or lack of awareness of prevention. - Leverage opportunities to increase awareness and detect vulnerabilities: Interviews reveal that pregnancy is often the entry point into prevention programs, enabling the collection of information and screening. This contact also provides a chance to engage with health services and professionals, especially when challenging situations arise, to raise awareness and promote screening. - Resistance to the choice of self-testing and the proposal in an opportunistic setting: Literature and recent Italian experiences have shown some critical issues when self-testing is offered as a first choice. The interviews confirm resistance in this regard (even those who favor self-testing request that explanation/training should be given with direct contact). Self-sampling as a first choice could, however, be provided in opportunistic settings, where information given directly by the operator is possible and thus reaches those who otherwise might not have found other channels of access to screening.
The matrix in Figure 4 summarizes the considerations deriving from CUBs on the potential utility of self-sampling in removing barriers. This intervention, when coupled with the specific training of the healthcare operators, is shown to have the highest potential to remove time-related, limited action, and healthcare service-related barriers. In considering these results, it is essential to remember that what is presented here is only a synthesis of reasoning and reflections. Furthermore, each identified category of vulnerability has very different internal specificities, and barriers have emerged both from representatives of the categories and from members of the CUB who directly or indirectly deal with people from those categories. CUB participants highlighted the risk of erroneous, incomplete, or stereotyped interpretations of the barriers that could prevent us from tracing the most relevant and decisive aspects, as well as those most in line with the realities of experience.
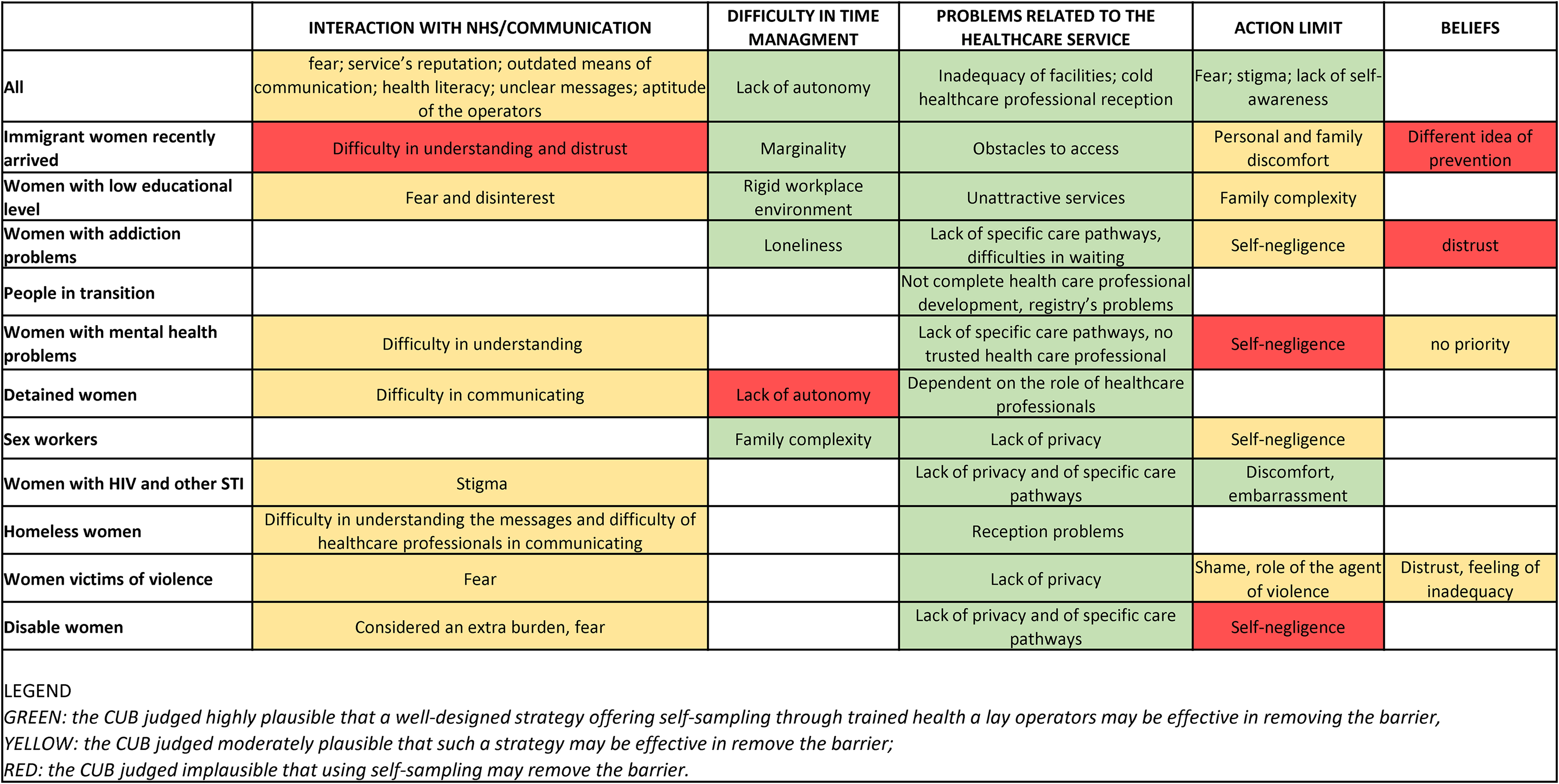
Systematization of barriers by vulnerable groups and categories of barriers identified by a Collaborative User Board (CUB), and judgment on potential plausibility that interventions using self-sampling will remove each specific barrier in a specific group of vulnerable women.
Discussion and conclusions
The process described in this article has led to final considerations on implementing self-sampling in Italian screening programs, which are included in a document edited by the ONS and endorsed by the Ministry of Health, 21 representing an important institutional outcome of the CBIG-SCREEN project in the Italian context. The final considerations reported in the results section on self-sampling implementation in Italian screening programs have been included in the document. The document is aligned with the European Council's recommendation on CCS and use of HPV self-sampling and provides guidance for its implementation within Italian screening programs, thus holding significant value for future program planning. The drafting of the document was shared among the CUB participants, and it includes their names and affiliations as a formal recognition of their expertise and contributions, a practice also applied in the present article (with further information provided in Supplemental Material 1).
The European Council recommendations 20 suggest self-sampling as a possible solution to increase the participation of women who do not comply with screening. In Italy, studies that sent the self-sampling kit to all non-responding women showed an increase in participation with consistent results, while the offer of self-sampling at the woman's request showed less or similar compliance with standard reminders. Intermediate solutions with the offer of collection of the self-sample in the pharmacy have shown heterogeneous results depending on the context and organizational methods.
Offering self-sampling as a first option to women who had previously signed up for screening achieved much lower participation than the standard invitation to the clinic, with homogeneous results across the different studies. In the Italian context, the offer of self-sampling to participating women must be made only in the context of controlled trials of innovative methods that have the potential to change the dynamics observed in previous trials.
Women who opted for self-sampling and tested positive participate in the follow-up, if appropriately contacted, in a similar way to those who participate in the clinic, and therefore, there is no rationale for adopting different algorithms for in-depth analysis, even if this involves an additional appointment for the cytological sampling.
Offering self-sampling to vulnerable women who have contact for other reasons (opportunistic offer) with specially trained operators is a promising way to increase coverage in this group and should be evaluated in appropriate trials.
It is a priority that recommendations suitable for the Italian context are developed and included in the National Guidelines System on how the potential of self-sampling can be exploited, starting from the conceptualization proposed in Figure 3.
Supplemental Material
sj-docx-1-msc-10.1177_09691413261429385 - Supplemental material for The co-creation of a document to guide the use of self-sampling for human papillomavirus screening in Italy
Supplemental material, sj-docx-1-msc-10.1177_09691413261429385 for The co-creation of a document to guide the use of self-sampling for human papillomavirus screening in Italy by Laura Bonvicini, Giusy Iorio, Luca Ghirotto, Noemi Auzzi, Marc Bardou, Rikke Buus Bøje, Pia Kirkegaard, Paola Mantellini, Paolo Giorgi Rossi, and in Journal of Medical Screening
Supplemental Material
sj-docx-2-msc-10.1177_09691413261429385 - Supplemental material for The co-creation of a document to guide the use of self-sampling for human papillomavirus screening in Italy
Supplemental material, sj-docx-2-msc-10.1177_09691413261429385 for The co-creation of a document to guide the use of self-sampling for human papillomavirus screening in Italy by Laura Bonvicini, Giusy Iorio, Luca Ghirotto, Noemi Auzzi, Marc Bardou, Rikke Buus Bøje, Pia Kirkegaard, Paola Mantellini, Paolo Giorgi Rossi, and in Journal of Medical Screening
Supplemental Material
sj-docx-3-msc-10.1177_09691413261429385 - Supplemental material for The co-creation of a document to guide the use of self-sampling for human papillomavirus screening in Italy
Supplemental material, sj-docx-3-msc-10.1177_09691413261429385 for The co-creation of a document to guide the use of self-sampling for human papillomavirus screening in Italy by Laura Bonvicini, Giusy Iorio, Luca Ghirotto, Noemi Auzzi, Marc Bardou, Rikke Buus Bøje, Pia Kirkegaard, Paola Mantellini, Paolo Giorgi Rossi, and in Journal of Medical Screening
Footnotes
Acknowledgements
The study was partially supported by the Italian Ministry of Health – Ricerca Corrente annual program 2026 and by the Regional Health Authority. It is part of the CBIG-SCREEN, an EU-funded 5-year project under the EU Horizon 2020 Research and Innovation Programme (Grant Agreement No. 964049).
Author contributions
PGR, LB, GI, NA, LG, and PM: designed the work, worked on the acquisition, and the analysis and the interpretation of data. PGR and LB: drafted the original version of the paper, and all authors reviewed the paper. MB, RBB, and PK: conceptualization and critical review. All authors approved the final version to be published and agree to be accountable for all aspects of the work, ensuring that questions related to the accuracy are addressed.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the European Commission with the project CBIG-SCREEN, an EU-funded 5-year project under the EU Horizon 2020 Research and Innovation Programme (Grant Agreement No. 964049), by Ministero della Salute (Ricerca Corrente - annual program 2026) and by the Regional Health Authority.
Supplemental material
Supplemental material for this article is available online.
Italian collaborative user board working group
Mental Health, ASL Firenze, Italy: Chiara Amendola. Cultural-linguistic mediation service, Dimora d’Abramo, Reggio Emilia, Italy: Philomena Ameyao. Screening and Secondary Prevention Unit, Istituto per lo Studio, la Prevenzione e la Rete Oncologica, Firenze, Italy: Noemi Auzzi, Paola Mantellini. Health promotion and prevention, Regione Lazio, Italy: Diego Baiocchi. Screening Program, Regione Lazio, Italy: Lilia Biscaglia. Caritas Reggiana, Reggio Emilia, Italy: Giovanna Bondavalli. Epidemiology Unit, Azienda USL-IRCCS di Reggio Emilia, Reggio Emilia, Italy: Laura Bonvicini, Paolo Giorgi Rossi. Qualitative Research Unit, Azienda USL-IRCCS di Reggio Emilia, Reggio Emilia, Italy: Luca Ghirotto, Giusy Iorio. Screening Center, Azienda USL-IRCCS di Reggio Emilia, Reggio Emilia, Italy: Cinzia Campari. Italian Society of Migration Medicine, Italy: Paola Capparucci. Prevention and Health Promotion, General Board of Health Prevention, Ministero Salute, Italy: Daniela Galeone, Maria Teresa Menzano Secondary prevention, Regione Sicilia, Italy: Lucia Li Sacchi. Health promotion and prevention, Regione Lazio, Italy: Alessandro Maltempi. Azienda ULSS 9 Scaligera, Verona, Italy: Gessica Martello. Screening Unit, Azienda Zero, Regione Veneto, Italy: Elena Narne. Mental Health Service – Addiction Unit, Azienda USL-IRCCS di Reggio Emilia, Reggio Emilia, Italy: Luana Oddi. National Federation of CREAI, Clichy, France: Sabrina Sinigaglia. Secondary Prevention, Regione Sicilia and Screening Center, ASP Palermo, Italy: Mario Valenza. Cooperativa Madre Teresa, Reggio Emilia, Italy: Lisa Vezzani.
CBIG-SCREEN consortium
CIC-P INSERM 1432, CHU Dijon-Bourgogne, Dijon, France: Marc Bardou. UNICCA- University Research Clinic for Cancer Screening, Randers Regional Hospital, Randers, Denmark: Berit Andersen, Pia Kirkegaard, Rikke Buus Bøje, Pernille Aakjær Mikkelsen. London School of Hygiene & Tropical Medicine, London, UK: Rosa Legood, Anna Foss, Martin McKee, Rachel Greenley, Liu Sun. Epidemiology Unit, Azienda USL-IRCCS di Reggio Emilia, Reggio Emilia, Italy: Paolo Giorgi Rossi, Letizia Bartolini, Laura Bonvicini, Olivera Djuric. Qualitative Research Unit, Azienda USL-IRCCS di Reggio Emilia, Reggio Emilia, Italy: Luca Ghirotto, Giusy Iorio. Screening and Secondary Prevention Unit, Istituto per lo Studio, la Prevenzione e la Rete Oncologica, Firenze, Italy: Noemi Auzzi, Paola Mantellini. Instituto de Saude Publica da Universidade do Porto, Porto, Portugal: Nuno Lunet, João FirminoMachado, Ana Fernandes, Margarida Teixeira, Firmino Machado, Mariana Amorim, Inês Baía, Romeu Mendes, Cláudia Gouvinhas. Tartu University, Tartu, Estonia: Anneli Uusküla, Anna Tisler. Universitatea Babes Bolyai, Cluj-Napoca, Romania: Adriana Baban, Diana Tăut, Nicoleta Jiboc. Institute of Oncology Cluj-Napoca, Cluj-Napoca, Romania: Florian Nicula, Alexandra Tolnai. European Institute of Women's Health, Dublin, Ireland: Rebecca Moore, Vanessa Moore. International Agency for Research on Cancer, Lyon, France: Partha Basu, Isabel Mosquera Metcalfe, Keitly Mensah, Eric Lucas. Ecole d’économie de Paris, Paris, France: Lise Rochaix, Camilla Fiorina, Violette Delisle. Health Psychology Research Center, Sofia, Bulgaria: Irina Todorova, Yulia Panayotova, Tatyana Kotzeva. European Cancer Leagues, Switzerland: David Ritchie, Helena Ros Comesana, Meritxel Mallafré-Larrosa, Ginevra Papi. Inserm Transfert SA, Paris, France: Christiane DascherNadel.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
