Abstract
Objectives
(a) To estimate the risk of recurrent cervical intraepithelial neoplasia, grade 2/3 or worse (CIN2+/CIN3+), lesions within 5 years of follow-up in human papillomavirus-negative/human papillomavirus-positive cohorts; (b) to assess whether certain risk factors can predict the recurrence of CIN2+/CIN3+ lesions; and (c) to provide recommendations for follow-up after treatment of cervical intraepithelial neoplasia, grade 2/3 to prevent cervical cancer
Setting
Organized cervical cancer screening programme in Central Italy.
Methods
We included 1063 consecutive first excisional treatments performed between 2006 and 2014 for screening-detected cervical intraepithelial neoplasia, grade 2/3 lesions among women aged 25–65. The study population was divided into two groups according to the human papillomavirus test results performed 6 months after treatment: Human papillomavirus-negative and human papillomavirus-positive cohorts. The 5-year risk of developing cervical intraepithelial neoplasia, grade 2/3 or worse (CIN2+/CIN3+) was estimated using the Kaplan-Meier method and the Cox regression model.
Results
Among 829 human papillomavirus-negative and 234 human papillomavirus-positive women, six (0.72%; three cervical intraepithelial neoplasia, grade 2, three cervical intraepithelial neoplasia, grade 3) and 45 (19.2%; 15 cervical intraepithelial neoplasia, grade 2, 30 cervical intraepithelial neoplasia, grade 3), respectively, developed CIN2+ recurrence within 5 years of follow-up. The cumulative risks for CIN2+ and CIN3+ were 0.9% (95% confidence interval: 0.4%–2.0%) and 0.5% (95% confidence interval: 0.1%–1.4%), respectively, for the human papillomavirus-negative cohort, and 24.8% (95% confidence interval: 18.5%–32.7%) and 16.9% (95% confidence interval: 11.4%–24.5%), respectively, for the human papillomavirus-positive cohort. Risk factors associated with increased risk of recurrence were both margins positive for the human papillomavirus-negative cohort, and positive margins, cervical intraepithelial neoplasia, grade 3 lesions, high-grade cytology and high viral load for the human papillomavirus-positive cohort.
Conclusions
Human papillomavirus testing can identify women at increased risk of recurrence and this supports a recommendation for its use in the post-treatment follow-up of cervical intraepithelial neoplasia, grade 2/3 lesions.
Keywords
Introduction
Women treated for grade 2 or 3 cervical intraepithelial neoplasia (CIN2/CIN3) are at increased risk of developing pre-invasive and invasive cervical lesions.1,2 The frequency of recurrences may vary between studies depending on the follow-up tests, the follow-up interval, and the outcome considered (grade 2/3 CIN or worse (CIN2+/CIN3+)).3–5 The pooled risk of CIN2+ relapses in a systematic review of articles reporting human papillomavirus (HPV) testing in post-treatment surveillance was 4.8% (95% confidence interval (CI): 3.4%–6.8%), ranging from 0.4% to 19.5% in individual studies. 6 According to another systematic review, the estimated relative risk of cervical cancer among women treated for CIN was three times higher than in the general population and remained high for at least 20 years after treatment. 7 The evidence suggests that women treated for high-grade lesions require enhanced surveillance given their additional level of risk.4,5,7 The post-treatment surveillance most commonly applied in the past within organized screening programmes was cytology, with or without colposcopy. In recent years, HPV testing is being used increasingly in post-treatment surveillance.8,9 Several studies have demonstrated that HPV testing is more sensitive than cytology in identifying women with residual/recurrent disease and has a very high negative predictive value.2,5 Most post-treatment surveillance protocols use co-testing (HPV testing in combination with cytology), although the difference in sensitivity between HPV testing alone and co-testing is small and has not always been significant across studies.5,6,10,11 Many risk factors for recurrence after treatment of CIN2/3 have been evaluated, such as age, cone margin status, cytology, lesion grade, type of treatment, genotyping and viral load.3,4,10–12
The aims of this study were (a) to estimate the risk of recurrent CIN2+/CIN3+ lesions within 5 years of follow-up in HPV-negative/HPV-positive cohorts; (b) to assess whether certain risk factors can predict the recurrence of CIN2+/CIN3+ lesions; and (c) to provide recommendations for follow-up after treatment of CIN2/CIN3 to prevent cervical cancer
Materials and methods
Study population
This retrospective cohort study included 1063 consecutive first excisional treatments performed between 2006 and 2014 for screening-detected CIN2/CIN3 lesions, confirmed by histology, for women aged 25–65 from the Florence Screening Programme registry (Italy).
The inclusion criteria were (a) follow-up HPV testing at 6 months (± 2) after treatment (baseline); and (b) at least one subsequent “negative” follow-up test (Papanicolaou (Pap) test, colposcopy or HPV test) or a recurrence in the post-treatment follow-up period.
The follow-up period began on the date of the baseline HPV test and ended on the date of recurrence or the date of the last “negative” test performed during the first 5 years of follow-up. In any case, the follow-up period was terminated on December 31, 2016.
When the follow-up period was longer than 5 years, we looked for the first negative follow-up episode after 5 years but not later than 7 years, and we then attributed 5 years of follow-up.
To identify recurrent CIN2+/CIN3+ lesions diagnosed between 2006 and 2016, the records of the study cohort were merged with the Florence Screening Programme registers, the Tumor Registry of the Tuscany Region and the local neoplastic pathology reports. Although CIN3+ alone may be regarded as the relevant outcome, since it is unlikely to regress, we included CIN2+ because of the low number of recurrences in the negative HPV cohort. However, CIN2+ is an important clinical result because it represents the reference threshold to recommend treatment, and we can make comparisons with other studies in the literature that also included CIN2+.
Failure was defined as a residual/recurrent (recurrent) CIN2+ or a CIN3+ lesion histologically confirmed in the follow-up period (within 5 years from baseline). Women with a negative test (co-testing: HPV and Pap test performed on the same date, colposcopy, HPV or Pap test) at the last follow-up episode were considered negative for recurrent lesions (negative exit test).
The study population was divided into two groups according to the baseline HPV test result: HPV-negative (HPV-) and HPV-positive (HPV+) cohorts. This strategy was necessary in order to minimize the risk of “ascertainment” bias (more tests done, more chance of finding lesions) due to a more intensive surveillance protocol being used for HPV+ compared to HPV- women.
Some risk factors were examined: Age, type of treatment, margin status, lesion grade (CIN2/CIN3), HPV/Pap test results, and Hybrid Capture 2 (HC2) semi-quantitative viral load for HPV+ women.
Type of treatment included laser conization, large loop (electrosurgical) excision of transformation zone (LLETZ) with laser vaporization, loop electrosurgical excision procedure, and cold-knife conization. The latter two procedures were evaluated together because of the small number of cases. Histological confirmation of high-grade CIN was taken from the worst histology result of either punch biopsy and excisional specimen examination. Histological margin status was classified as negative, one or both margins positive (endo and/or exocervical), or not specified. An HPV-HC2 test with viral load < 1 relative light unit/mean control (RLU/CO) was considered negative. The semi-quantitative viral load for HPV+ testing was classified into three categories: 1–9, 10–99 or ≥ 100 RLU/CO. Pap tests were classified into three categories: Not performed, negative (-) or positive (+; atypical squamous cells of undetermined significance or more severe (ASC-US+) by the Bethesda Reporting System
13
). The positive Pap test was classified in two categories: low-grade (ASC-US, low-grade squamous intraepithelial lesion: LSIL) and high-grade (atypical glandular cells: AGC
Follow-up protocol in the Florence
Screening Programme
In the period 2006–2010, a woman treated for CIN2/CIN3 was required to undergo six-month and 24-month follow-up HPV testing. If the HPV test was positive, the woman concerned was referred for a colposcopy, with the routine screening interval being resumed if all tests were negative. Since 2011, a new protocol has been implemented recommending 6-, 24- and 48-month co-testing, with the routine screening interval being resumed if all tests are negative (see Supplemental File 1: Figure S1).
Statistical analysis
The cumulative probability of developing a CIN2+ or CIN3+ lesion within 5 years of follow-up was estimated using the Kaplan-Meier method. The log-rank test was used to compare Kaplan-Meier failure curves. Cox's model was used to estimate the hazard ratios (HRs) for recurrence of CIN2+/CIN3+ lesions within 5 years of follow-up in univariate and multivariable analysis. In multivariable analysis, we used the likelihood ratio test to compare the goodness of fit of statistical models. Step by step, we dropped the variables that were not significant at 0.05 level, with the exception of age. To account for the intensity of follow-up, we calculated the median follow-up time by cohort and the mean number of follow-up tests (co-testing, HPV or Pap test) performed. A p-value < 0.05 was considered significant. Confidence intervals were calculated using the binomial exact method when the failures were zero in Kaplan–Meier estimates. 14 Statistical analyses were performed using STATA version 16.0 (StataCorp LP, College Station, TX, USA).
Ethical approval
The study was approved by the Area Vasta Centro Section of the Tuscany Ethics Committee (Number 19998_oss).
Results
A cohort of 1063 women aged 25–65 underwent an excisional treatment for screening-detected CIN2/CIN3 lesions. Among these patients, 829 (78%) and 234 (22%) women were HPV- and HPV+, respectively, at baseline.
HPV negative cohort
The HPV- cohort was monitored for up to 5 years, with a median follow-up time of 42 months after baseline (interquartile range (IQR): 1.6–4.8 years). About 40% of cases (333/829) did not undergo co-testing at baseline. Only 2.6% (13/496) of women undergoing co-testing at baseline were HPV-/Pap+, while 97.4% (483/496) were HPV-/Pap- (see Supplemental File 2: Table S2). Six women developed recurrence within 5 years of follow-up (three CIN2 and three CIN3) and none developed cervical cancer. Overall, the cumulative probability of developing a recurrent lesion within 5 years of follow-up was 0.9% (95% CI: 0.4%–2.0%) and 0.5% (95% CI: 0.1%–1.4%) for CIN2+ and CIN3+ lesions, respectively (Table 1; Figure 1A, B). In Cox's multivariable analysis, adjusted by age, only patients with both margins positive were significantly associated with an increased risk of developing CIN2+/CIN3+ lesions: HRs were 21.3 (95% CI: 2.2–210.6) for CIN2+ and 33.4 (95% CI: 2.8–398.8) for CIN3 + (Table 2).
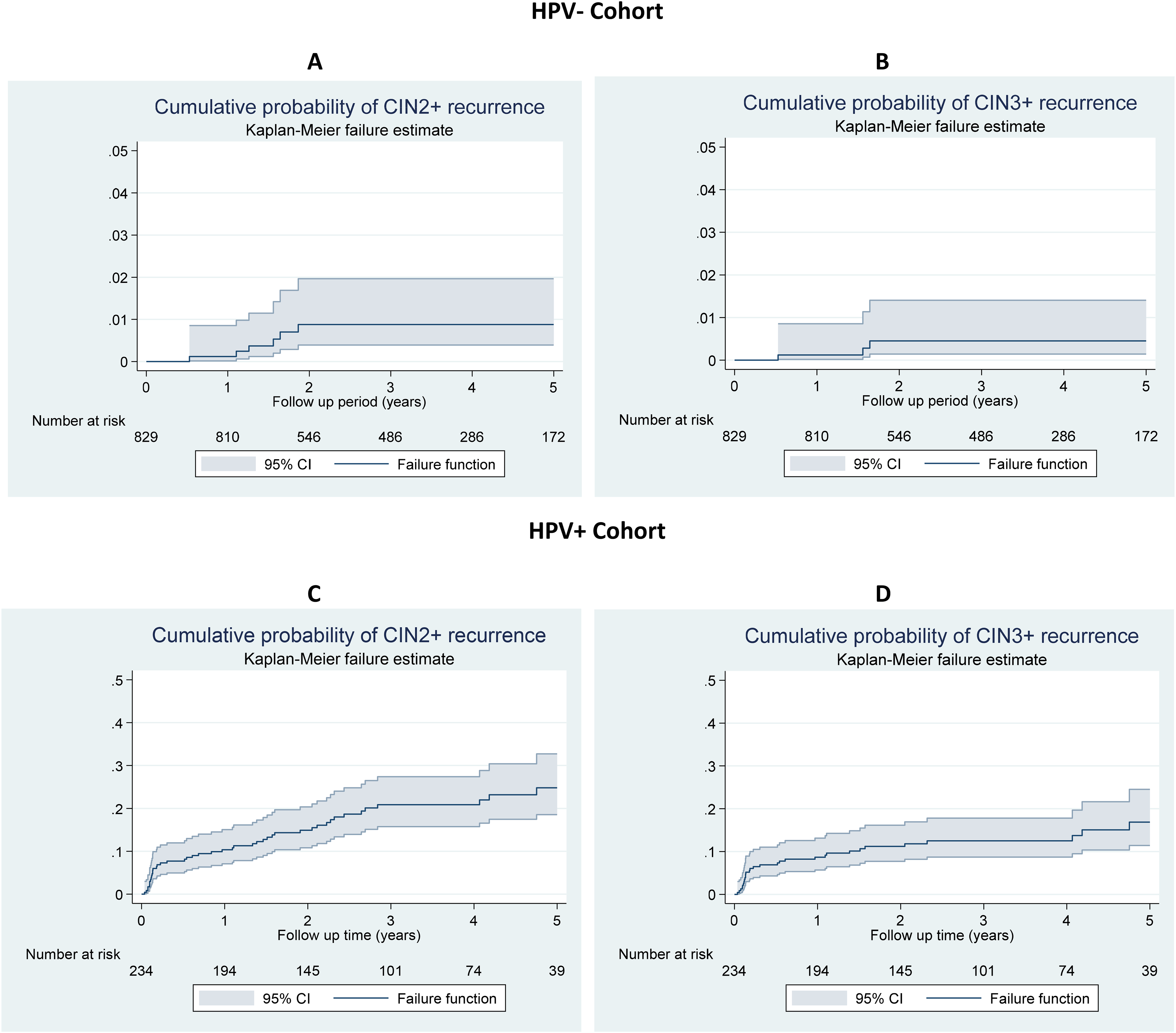
Cumulative probability of developing a CIN2+/CIN3+ lesion within 5 years of post-treatment follow-up of CIN2/CIN3 lesions for HPV- cohort (for CIN2+: A, for CIN3+: B) and HPV+ cohort (for CIN2+: C, for CIN3+: D). Note: different Y-scales were used for HPV+ and HPV- cohorts. HPV: human papillomavirus; CIN: cervical intraepithelial neoplasia.
Cumulative probability (%) of developing a recurrent CIN2+ or CIN3+ lesion within 5 years of follow-up using the Kaplan–Meier method, with 95% confidence intervals in the HPV- and HPV+ cohorts, by Pap test result, margin status and viral load (for HPV+ cohort).
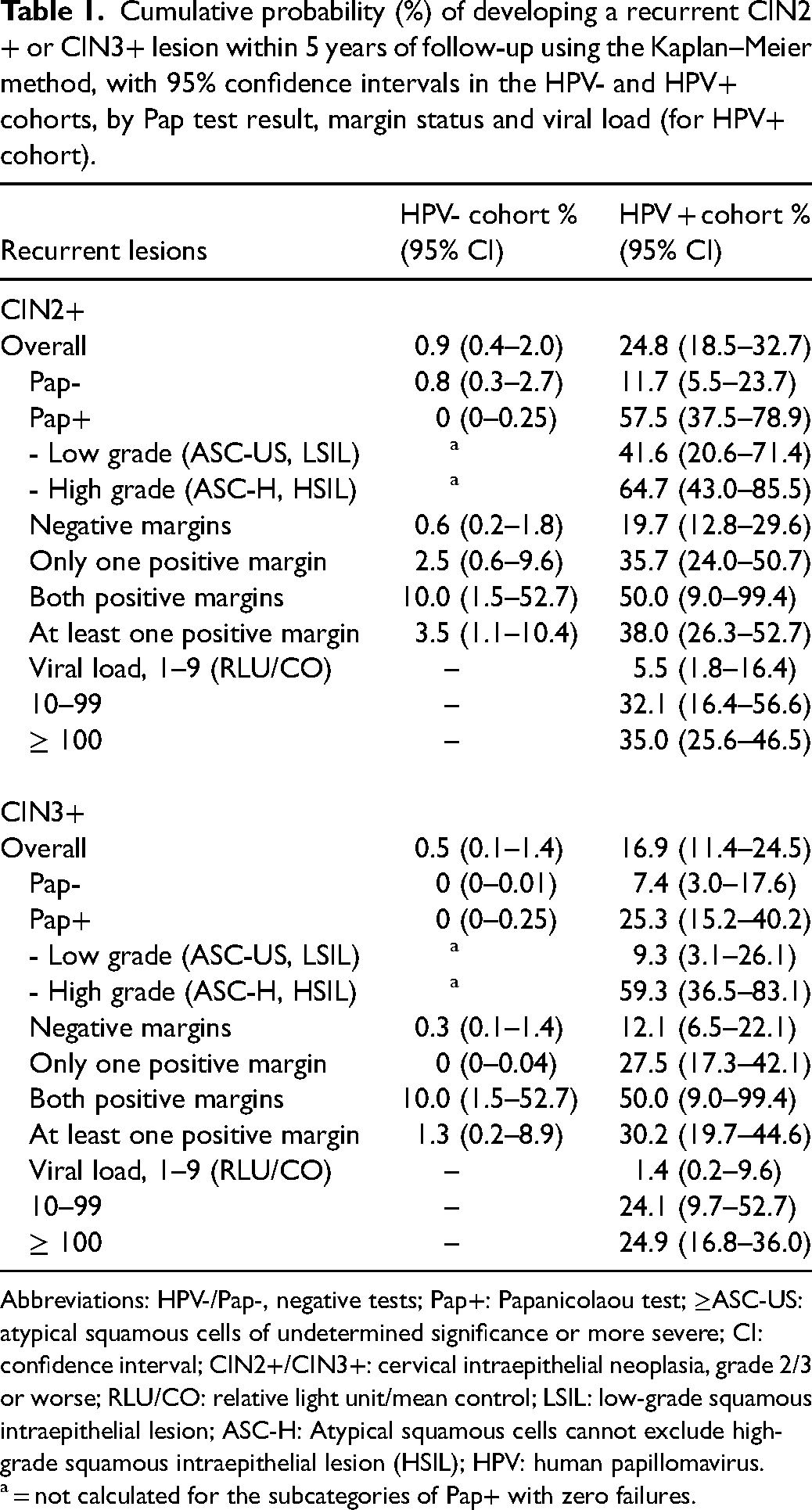
Abbreviations: HPV-/Pap-, negative tests; Pap+: Papanicolaou test; ≥ASC-US: atypical squamous cells of undetermined significance or more severe; CI: confidence interval; CIN2+/CIN3+: cervical intraepithelial neoplasia, grade 2/3 or worse; RLU/CO: relative light unit/mean control; LSIL: low-grade squamous intraepithelial lesion; ASC-H: Atypical squamous cells cannot exclude high-grade squamous intraepithelial lesion (HSIL); HPV: human papillomavirus.
= not calculated for the subcategories of Pap+ with zero failures.
Univariate and multivariable analysis of the probability of developing a recurrent CIN2+/CIN3+ lesion within 5 years of follow-up after treatment, HPV-negative cohort.
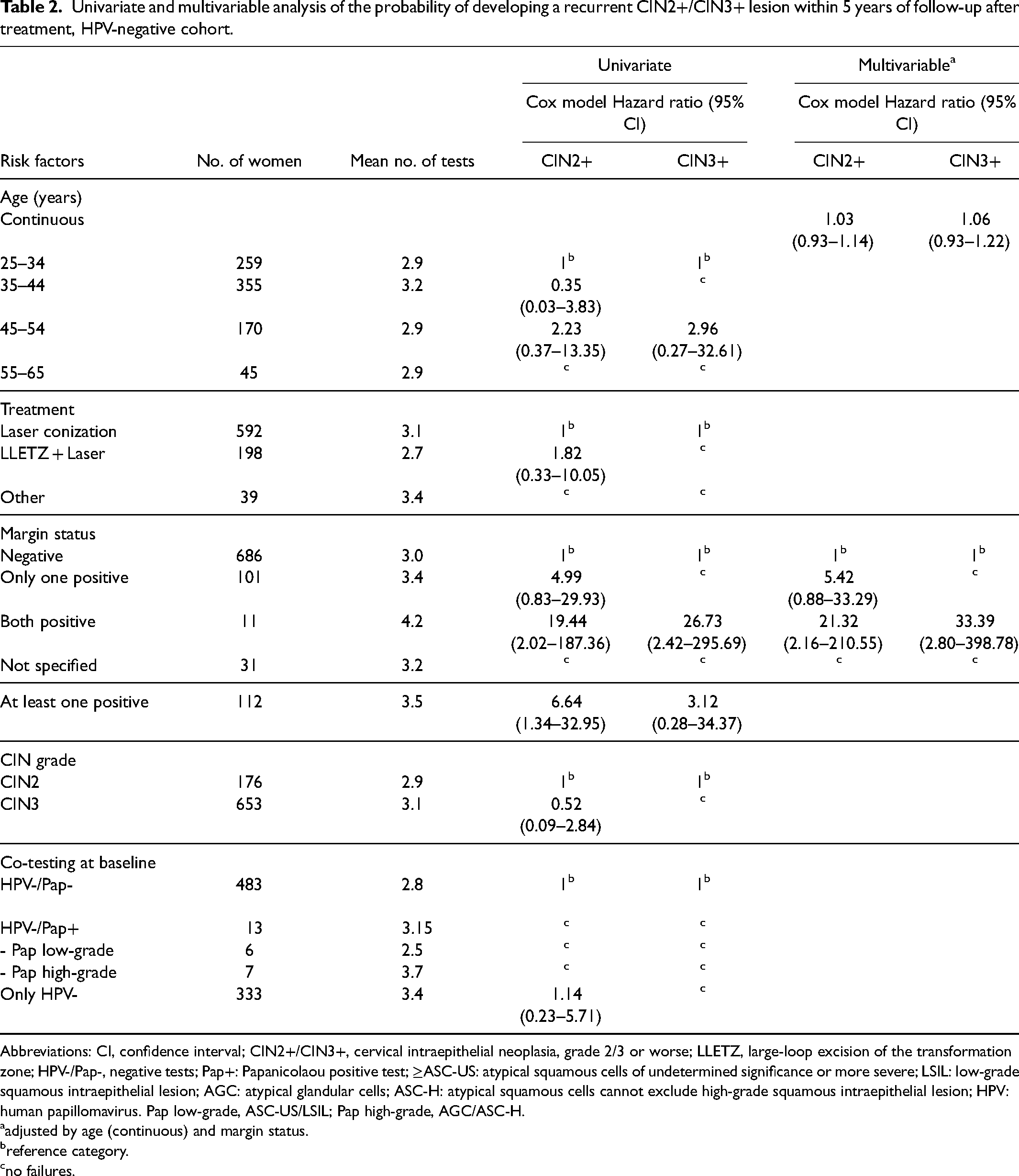
Abbreviations: CI, confidence interval; CIN2+/CIN3+, cervical intraepithelial neoplasia, grade 2/3 or worse; LLETZ, large-loop excision of the transformation zone; HPV-/Pap-, negative tests; Pap+: Papanicolaou positive test; ≥ASC-US: atypical squamous cells of undetermined significance or more severe; LSIL: low-grade squamous intraepithelial lesion; AGC: atypical glandular cells; ASC-H: atypical squamous cells cannot exclude high-grade squamous intraepithelial lesion; HPV: human papillomavirus. Pap low-grade, ASC-US/LSIL; Pap high-grade, AGC/ASC-H.
adjusted by age (continuous) and margin status.
reference category.
no failures.
No recurrence was diagnosed among women who had two consecutive negative co-testings (baseline and subsequent), i.e., 410 of 483 (84.9%) women with HPV-/Pap- at baseline.
An average of 3.0 tests (co-testing, HPV or Pap test) were performed in the follow-up period (Table 2).
HPV positive cohort
The HPV+ cohort was monitored for up to 5 years, with a median time of 30 months after baseline (IQR: 1.4–4.4 years). About 40% (92/234) of the women in this cohort did not undergo co-testing at baseline. Among women who did undergo co-testing at baseline, 35.2% (50/142) were HPV+/Pap+ and 64.8% (92/142) were HPV+/Pap- (see Table S2: characteristics of the cohort). Forty-five women developed recurrences within 5 years of follow-up (15 CIN2 and 30 CIN3) and none developed cervical cancer. Overall, the cumulative probability of developing a recurrent lesion within 5 years of follow-up was 24.8% (95% CI: 18.5%–32.7%) and 16.9% (95% CI: 11.4%–24.5%) for CIN2+ and CIN3+ lesions, respectively (Table 1; Figure 1C, D). The cumulative probability by margin status and viral load is shown in Table 1 and Figure 2.
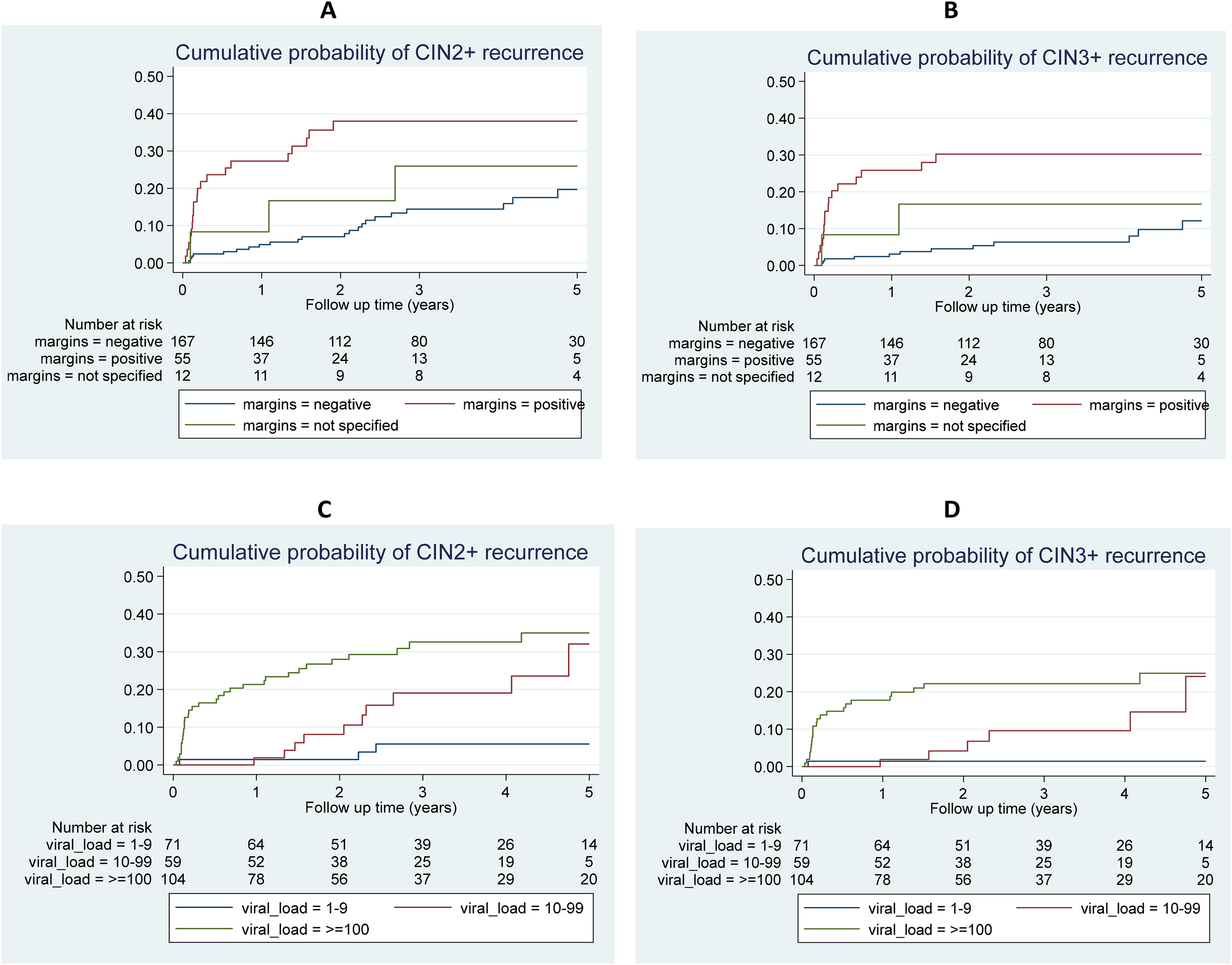
Cumulative probability of developing a CIN2+/CIN3+ lesion within 5 years of post-treatment follow-up of CIN2/CIN3 lesions for HPV+ cohort: By margin status (for CIN2+: A, for CIN3+: B), and baseline viral load (for CIN2+: C, for CIN3+: D). HPV: human papillomavirus; CIN: cervical intraepithelial neoplasia.
According to the multivariable model (Table 3), the recurrence risk for CIN2+ and CIN3+ for positive endocervical margins was 3.88 and 5.07 times higher, respectively, than for negative margins. The risk of developing CIN2+ and CIN3+ recurrences according to CIN grade at the time of treatment was 3.03 and 11.47 times higher for CIN3 grade than for CIN2 grade, respectively. The risk of developing CIN2+ and CIN3+ recurrences was 5.88 and 13.62 times higher for a viral load ≥ 100 RLU/CO compared to the category with viral load < 10 RLU/CO, respectively. In addition, the risk of developing CIN2+ and CIN3+ recurrences was 13.04 and 10.06 times higher for high-grade cytology, respectively, compared to HPV+/Pap- category. In a further multivariable analysis not shown in Table 3, the risk of developing CIN2+ and CIN3+ recurrences for the category of at least one positive margin was 2.8 (95% CI: 1.5–5.4) and 3.9 (95% CI: 1.8–8.6) times higher than for negative margins, respectively.
Univariate and multivariable analysis of the probability of developing a recurrent CIN2+/CIN3+ lesion within 5 years of follow-up after treatment, HPV-positive cohort.
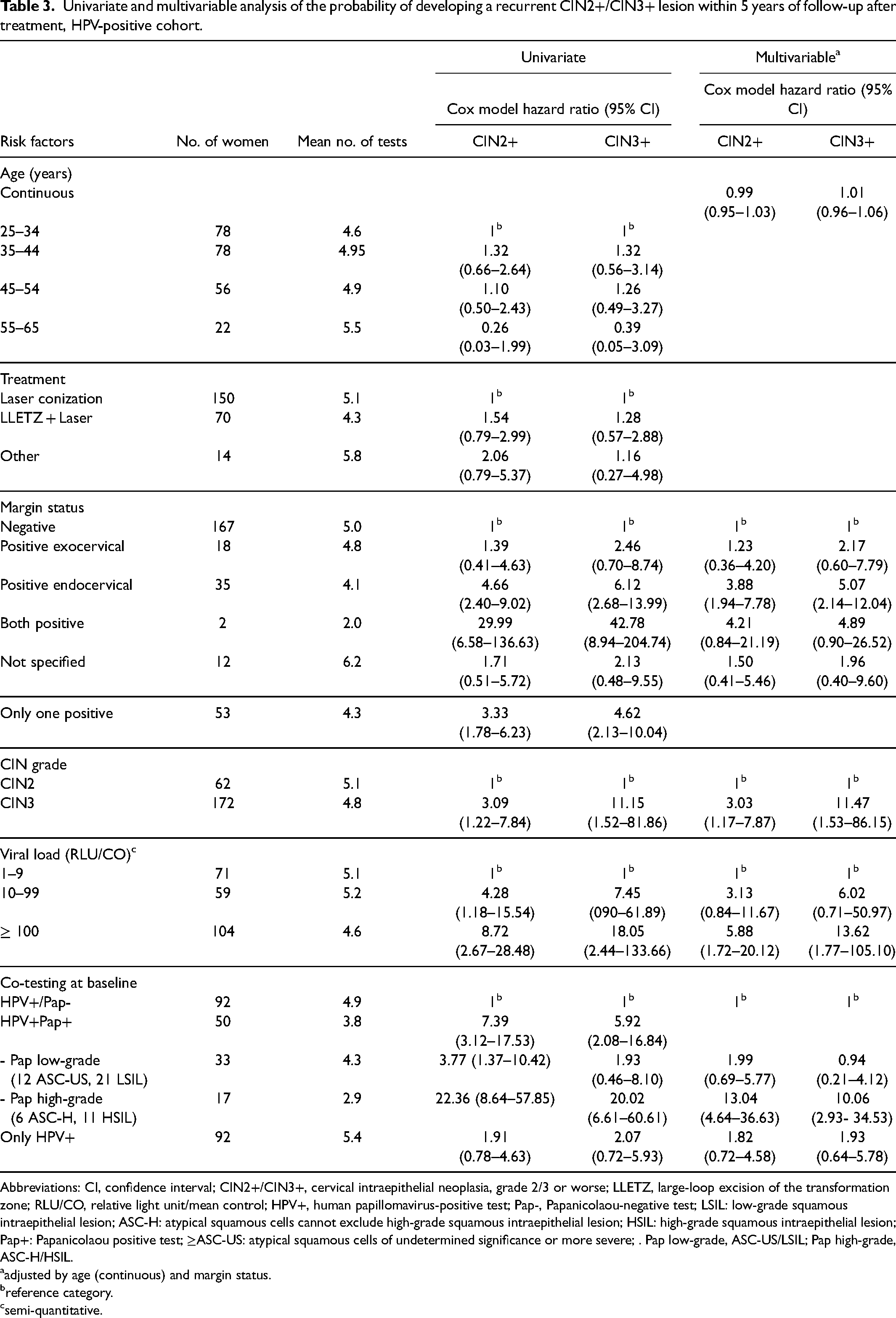
Abbreviations: CI, confidence interval; CIN2+/CIN3+, cervical intraepithelial neoplasia, grade 2/3 or worse; LLETZ, large-loop excision of the transformation zone; RLU/CO, relative light unit/mean control; HPV+, human papillomavirus-positive test; Pap-, Papanicolaou-negative test; LSIL: low-grade squamous intraepithelial lesion; ASC-H: atypical squamous cells cannot exclude high-grade squamous intraepithelial lesion; HSIL: high-grade squamous intraepithelial lesion; Pap+: Papanicolaou positive test; ≥ASC-US: atypical squamous cells of undetermined significance or more severe; . Pap low-grade, ASC-US/LSIL; Pap high-grade, ASC-H/HSIL.
adjusted by age (continuous) and margin status.
reference category.
semi-quantitative.
We observed 56 out of 234 women (23.9%) with two consecutive negative co-testing procedures at different points of follow-up: 31 (55.3%) at their second and third co-testings, 16 (28.6%) at their third and fourth co-testings, eight (14.3%) at their fourth and fifth co-testings, and one (1.8%) at fifth and sixth co-testings. We did not observe two consecutive negative HPV tests among women with recurrence within the follow-up period.
An average of 4.9 tests (co-testing, HPV or Pap test) were performed in the follow-up period (Table 3).
Discussion
The post-treatment surveillance protocol with HPV testing or co-testing adopted by the Florence Screening Programme has proven to be effective in preventing the occurrence of invasive cervical cancers within 5 years of follow-up. Identification of relapses or new pre-invasive lesions in the post-treatment follow-up period and subsequent treatment may have prevented the onset of invasive cervical cancer.
The overall cumulative probability of developing a recurrent CIN2+ /CIN3+ lesion within 5 years of follow-up for the HPV+ cohort was very high (24.8% and 16.9%, respectively), whereas, for the HPV- cohort, it was very low (0.9% and 0.5%, respectively), as confirmed in many other studies.3,6,10,11,15,16
The cumulative probability of recurrence for CIN2+ from a negative co-testing outcome (HPV-/Pap-) was very low (0.8%), in agreement with the literature.3,6,10,11,15,16 It was not possible to estimate the risk for HPV-/Pap+ co-testing, because the number of women with this outcome was too small (2.6% of co-testing procedures) and there were no failures. In our study, most women with baseline co-testing in the HPV-negative cohort had a negative Pap test (97%), so most women had no benefit from adding the Pap test. However, some studies have shown an increased risk of recurrence with HPV-/Pap+ compared with HPV-/Pap-, especially for high-grade cytology.11,15 In Egemen et al. 15 (Kaiser Permanente Northern California Cohort), the 5-year risk of CIN3+ in women with a negative HPV and high-grade cytology was 18%. Although the proportion of women in this category was very small (1.5%: 59/3864), the 5-year risk of CIN3+ was very high. This finding may suggest the use of co-testing in negative HPV women.
In the HPV- cohort, five out of six recurrences occurred in women who became HPV+ at their second HPV test (12/18 months from baseline), and no recurrences occurred after the two first consecutive negative co-testings. These findings could suggest the need for a subsequent check after the first HPV- test and a return to regular screening after two consecutive negative co-testings. In the UK, 1–2 negative co-testings within 24 months were considered as the ‘test of cure’ for returning to screening. 5 The Australian National Cervical Screening Programme Safety Monitoring Committee 17 reported that there were no incidents of subsequent cervical cancer, following two negative co-testings. These data supported the effectiveness of two negative consecutive co-tests as ‘test of cure’. Many researchers estimated the risk of recurrence in post-treatment surveillance of CIN2/CIN3 according to the number of negative tests. Kocken et al. 2 reported a risk of recurrence for CIN2+ within 5 years of follow-up of 1.0% with two negative episodes (co-testing) at 6 and 24 months after treatment, while the 5-year risk for CIN3+ was 0.0%. Katki et al. 3 showed that the risk of recurrence of CIN2+ within 5 years of follow-up was slightly lower after two episodes than after only one. The 2019 guidelines of the American Society for Colposcopy and Cervical Pathology present risk-based recommendations for the management of surveillance after treatment for CIN2/CIN3. 15 The estimated 5-year CIN3+ risk of recurrence was 0.68% or 0.91% for two negative co-testings or two negative HPV tests, respectively, resulting in 1-year follow-up, while the risk of recurrence for three negative co-testings or three negative HPV tests was 0.35% and 0.44%, respectively, resulting in 3-year follow-up management. 15 The recommendation to return to screening at 5 years was not triggered even with three negative tests.
Our cumulative 5-year risk of CIN2+/CIN3+ recurrence was 57.5%/25.3% for HPV+/Pap+ compared to 11.7%/7.4% for HPV+/Pap- women. Close surveillance should be carried out for HPV+/Pap+ women due to the very high risk of recurrence, as confirmed by other studies.11,15
Many studies have evaluated risk factors associated with an increased risk of recurrence after treatment of CIN2/CIN3 lesions, particularly for positive margins.11,18,19,20 In our study, the only risk factor associated with an increased risk of recurrence in the HPV- cohort was having both margins positive. This situation occurred in only 1.3% (11/829) of HPV- women (with one failure). In many articles, there were no significant risk variations for major risk factors among HPV- women, even when the margins were positive.11,18
In the HPV+ cohort, lesion grade, positive margins, co-testing results and viral load were associated with an increased recurrence risk within 5 years of follow-up. Only treatment type and age were not associated with this increased risk of recurrence. The Cochrane review 21 also found no significant differences in treatment failures between surgical techniques. Some studies found an association with age,4,7,22 while others found no differences.22,23 Our study was in agreement with several studies that reported a high risk of recurrence in HPV+ women with positive margins,11,18,19 particularly for endocervical or endo/exocervical margins. Women with a CIN3 lesion had a higher risk of recurrence than women with a CIN2 lesion, as found in other studies.3,12,19 In multivariable analysis, the risk of recurrence increased when viral load increased (≥ 100 RLU/CO), which is in agreement with some studies.12,23 Furthermore, women with HPV+/Pap+ outcomes showed a risk of recurrence of CIN2+ or CIN3+ lesions 4 and 2.8 higher, respectively, which tallies with other studies with HPV+ outcomes at baseline.11,15
Women treated for high-grade lesions have an increased risk of developing pre-invasive lesions and cancer compared to the general population.1,2,5,8 Therefore, women should be monitored during post-treatment follow-up using a very sensitive test. In the past, Pap tests were used in follow-up, but this tool has now been replaced by HPV testing. Two recent studies found that strategies with co-testing 24 or HPV testing alone 25 were cost-effective compared to the previous standard follow-up strategy (Pap test alone 24 or Pap test and colposcopy 25 ). HPV testing has a high negative predictive value,2,5 and there is thus a high probability that subjects with a negative HPV test result really do not have the disease. This means the screening programme can extend the follow-up interval and even recommend a return to routine screening. In most Italian screening programmes, co-testing (HPV and Pap tests) is preferred in the post-treatment follow-up of CIN2/CIN3, although the relative sensitivity of co-testing for CIN2+ is only marginally higher than for HPV testing alone.5,6,12,15
In our study, most women (78%) undergoing follow-up had a baseline HPV- test outcome, a very low-risk status, which could allow for less intensive post-treatment surveillance and a quick return to screening for at least three-quarters of the women undergoing such follow-up. Conversely, intensive surveillance is recommended in cases with a positive baseline HPV test, especially when the Pap test was also positive. Clinicians could use the information on other risk factors to customize the follow-up of the HPV+ cohort.
In the HPV+ cohort, a quarter of women had two consecutive negative co-tests at different points of follow-up and without recurrences. This could be an important result with regard to return to screening, but the limited number of women in this condition and the insufficient length of follow-up after the two consecutive negative co-testings do not allow to draw practical conclusions.
A new approach to reducing the risk of post-treatment recurrence may come from prophylactic vaccination against HPV at the time when CIN2/CIN3 lesions are treated. 26 This strategy could provide new and more effective follow-up protocols after conservative treatment of CIN2/CIN3 lesions.
We cannot rule out the possibility that women who have not undergone an HPV test at 6 (± 2) months (inclusion criteria) may be at increased risk of developing invasive cervical cancer. Performing an intraoperative HPV test could provide an early estimate of the risk of recurrence, 27 and may overcome the problem of non-compliance with the 6-month follow-up HPV test requirement.
The strength of this study was that the data were taken from an organized population-based screening programme where women were monitored according to a follow-up protocol. The main limitation was that this study was not a randomized controlled trial, although most studies in this field have been observational studies.6,9,11,18 Another important limitation was the ascertainment bias: “the more tests a woman undergoes, the more lesions will be found”. This bias may be relevant in the context of a more intensive follow-up protocol for high-risk cases (HPV+ women) leading to the diagnosis of a higher number of pre-invasive lesions, but some of these lesions may spontaneously regress and might no longer be found with a less intensive protocol. To address this limitation we used several strategies: (a) we divided the cohort based on the result of the HPV test at baseline and we also calculated the average number of tests performed in each cohort; (b) we also estimated the risk of CIN3+ lesions, which should be less regressive than CIN2+; (b) every observation was truncated on the date of a negative exit test; iv) we attributed 5 years of follow-up only when the date of a negative exit test was more than 5 years but up to 7 years. Another limitation was that only women with a positive screening test had colposcopy, and therefore the possibility of a diagnosis of CIN2+, and this may have underestimated the risks. However, our findings could be compared with many other studies that have applied a similar follow-up protocol in the post-treatment of CIN2/CIN3 lesions.
Conclusions
This study confirms that HPV testing can identify women at increased risk of recurrent CIN2+/CIN3+ and support the recommendation for utilization of HPV tests in the post-treatment follow-up of CIN2/CIN3. The women who were HPV-negative at baseline could return to routine screening after two consecutive negative co-tests. Further studies are needed to determine the best follow-up protocol for HPV-positive women at baseline.
Supplemental Material
sj-doc-1-msc-10.1177_09691413231175630 - Supplemental material for The 5-year risk of recurrence of grade 2/3 cervical intraepithelial neoplasia after treatment in a population screening programme by human papillomavirus status: A cohort study in central Italy
Supplemental material, sj-doc-1-msc-10.1177_09691413231175630 for The 5-year risk of recurrence of grade 2/3 cervical intraepithelial neoplasia after treatment in a population screening programme by human papillomavirus status: A cohort study in central Italy by Carmen Beatriz Visioli, Anna Iossa, Giuseppe Gorini, Paola Mantellini, Lisa Lelli, Noemi Auzzi, Carmelina Di Pierro, Francesca Maria Carozzi and Marco Zappa in Journal of Medical Screening
Supplemental Material
sj-doc-2-msc-10.1177_09691413231175630 - Supplemental material for The 5-year risk of recurrence of grade 2/3 cervical intraepithelial neoplasia after treatment in a population screening programme by human papillomavirus status: A cohort study in central Italy
Supplemental material, sj-doc-2-msc-10.1177_09691413231175630 for The 5-year risk of recurrence of grade 2/3 cervical intraepithelial neoplasia after treatment in a population screening programme by human papillomavirus status: A cohort study in central Italy by Carmen Beatriz Visioli, Anna Iossa, Giuseppe Gorini, Paola Mantellini, Lisa Lelli, Noemi Auzzi, Carmelina Di Pierro, Francesca Maria Carozzi and Marco Zappa in Journal of Medical Screening
Footnotes
Acknowledgements
The authors thank Dr Giovanna Masala for her useful comments on early drafts of this manuscript.
Authors’ contributions
C.B.V., A.I. and M.Z. designed the study. C.B.V., A.I., C.D.P., L.L. and N.A. set up, collected and reviewed the data. C.B.V. prepared the data for analysis. C.B.V., A.I., G.G. and M.Z. carried out the analyses and drafted the manuscript. All authors contributed to data interpretation and preparation of the final manuscript. All authors read and approved the final manuscript.
Data availability
The datasets generated and/or analyzed during this study are not publicly available for confidentiality reasons.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
The study was approved by the Area Vasta Centro Section of the Tuscany Ethics Committee (Number 19998_oss). The study was performed in accordance with the Declaration of Helsinki.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
