Abstract
Objective
Evidence of reduction in colorectal cancer (CRC) mortality following CRC screening based on the faecal immunochemical test (FIT) is insufficient. This study aimed to analyse if CRC mortality was reduced after implementing FIT-based screening.
Setting
The Danish national CRC screening programme.
Methods
This nationwide cohort study included residents aged 50–71 years invited to the prevalence round of the screening programme. Invitation order was decided by randomising on birth month; the first two birth months to be invited were classified as invited and the five last were classified as not-yet-invited and given a pseudo invitation data. Follow-up was from (pseudo)invitation date until 31 December 2017, emigration or death. Relative risk (RR) of CRC death was calculated with 95% confidence intervals (CIs).
Results
A total of 897,812 residents were included (29% invited and 71% not-yet-invited). The median follow-up was 3.3 years. The RR of CRC death at end of follow-up was 0.83 (95% CI 0.66; 1.03) among those invited to screening compared with those not yet invited. For men aged 60–71 years, this RR was 0.68 (95% CI 0.49; 0.94). For those participating in screening compared with a similar group of not-yet-invited residents, the RR was 0.71 (95% CI 0.46–1.08). For male participants aged 60–71 years, this RR was 0.49 (95% CI 0.27−0.89). For women and men aged 50–59 years, RRs were small and statistically non-significant.
Conclusion
This nationwide study showed that even within a median follow-up of only 3.3 years, implementing FIT-based CRC screening reduced CRC mortality among older men.
Introduction
Accounting for approximately 10% of all incident cancers and 10% of all cancer-related deaths, colorectal cancer (CRC) accounts for a considerable part of the global cancer burden among men and women alike. 1 To reduce morbidity and mortality from CRC, screening programs have been implemented in many countries. 2 Screening modalities differ regionally, with colonoscopy being the preferred test in the USA and faecal occult blood tests being preferred in Europe and Australia. 3
Biennial screening with guaiac faecal occult blood test (gFOBT) reduces the relative risk (RR) of mortality from CRC by 15% among invited individuals and by 25% among those attending at least one round of screening. 4 However, the more recent faecal immunochemical test (FIT) is superior to gFOBT with respect to detection rate, positive predictive value, and participation rate.5–9 Even so, three recent systematic reviews conclude that evidence of the impact of FIT-based screening on cancer-specific mortality is insufficient.10–12
Previous research shows that after implementing FIT-based screening, a larger proportion of screening invitees were diagnosed in early stages compared to a not-yet-invited population, indicating a better prognosis. 13 This nationwide cohort study including almost 900,000 Danish residents aged 50 to 71 years aimed to analyse if CRC mortality was reduced after randomized implementation of FIT-based CRC screening overall, and separately for sexes and particular age groups. Furthermore, we analysed if actual participation in CRC screening reduced CRC mortality.
Methods
Setting
The Danish CRC screening program was implemented from March 2014 through 2017 (prevalence round). For those aged 50 to 74 years on 1 January 2014, the order of invitation to participate in CRC screening was decided using a computer-generated randomization list of birth months created on 3 May 2013 using Stata 11.2 (STATA Corp., College Station, TX, USA). Residents turning 50 during the prevalence round were invited just before their birthday as were those turning 75 if they had not been invited earlier. The invitation was mailed directly to the resident's home address along with the screening kit (OC Sensor system (Eiken Chemical Company, Tokyo, Japan)), instructions on how to collect the faecal sample, and a return envelope. Non-participants received one reminder after 45 days. In the case of a positive result (≥100 ng haemoglobin/ml buffer), an appointment was booked to have a colonoscopy within 14 days. The invitation letter specified that participants in surveillance for CRC or adenomas should not participate and that patients with ulcerative colitis or Crohn's disease should discuss with their physician whether participation would be relevant. Participation and subsequent colonoscopy and treatment are free of charge in Denmark.14,15 There are no opportunistic routes for CRC screening of asymptomatic individuals outside the national screening programme.
Study design and population
This nationwide, register-based cohort study included all residents aged 50 to 71 years on 1 January 2014. The study period equalled the prevalence round of the screening program, which was from March 2014 to 31 December 2017.
Those born in April and June (the first two birth months to be invited) constituted the intervention group and were defined as the invited residents. Those born in February, March, July, October, and December (the five last birth months to be invited) constituted the control group and were defined as the not-yet-invited residents. In order to achieve the same follow-up time for intervention and control groups, we randomly allocated a pseudo-invitation date to the control population. The distributions of invitation and pseudo-invitation dates were exactly identical.
Finally, those born in January, May, August, September, and November were excluded to ensure a maximum of time between inviting the intervention group and the control group. Those aged 72 to 74 years on 1 January 2014 were excluded since they would otherwise be invited before turning 75 years old, thereby breaking the randomized invitation order. Residents who were diagnosed with CRC or who emigrated/died before the (pseudo)invitation were excluded.
The intervention and control groups were followed for CRC death from the (pseudo)invitation date to 31 December 2017, emigration or death, whichever came first To achieve the longest possible follow-up, both groups were followed until the end of the prevalence round, meaning that the control group was invited to screening at the end of the follow-up period. This invitation date was ignored and the controls kept their pseudo-invitation date, which was early in the prevalence round.
Data
The study cohort was identified using the Danish Central Registration System, which holds information on date of birth, sex, and the unique identification number of everyone residing in Denmark. 16
The date of the screening invitation was retrieved from the Danish CRC Screening Database, which monitors the quality of the national CRC screening program and includes everyone invited to participate in the program.17,18
Information on death during the study period was retrieved from the Danish Register of Causes of Death. 19 When a person in Denmark dies, a doctor performs a post-mortem examination. The doctor fills in a death certificate and reports the death by submitting the certificate to the Danish Health Authority. Causes of death are coded according to the International Classification of Diseases, 10th version (ICD10) (CRC codes: DC18* and DC20*).
To exclude those with CRC prior to invitation, information on CRC diagnoses was retrieved from the Danish National Cancer Register. 20 From 1943 to 1978, CRC diagnoses were classified according to the ICD8 (codes: 1530, 1534, 1535, 1540, 1549, 2530-2534, 4530-4535, 4538, 4540, 4549, 8530-8535, 8540, 8549), thereafter according to the ICD10 (codes: DC18*, DC19* (until 2008), DC20*). For sensitivity analyses, data on ulcerative colitis (ICD10: DK51) and Crohn's disease (ICD10: DK50) were collected from the Danish National Patient Register.21,22
To test comparability between the groups, data on demographic and socioeconomic position were obtained from Statistics Denmark. 23 Country of origin was classified as either Danish, Western (EU, Andorra, Australia, Canada, Iceland, Liechtenstein, Monaco, New Zealand, Norway, San Marino, Switzerland, and the USA), or non-Western (others). Marital status was classified as cohabitating or living alone. Educational attainment was classified according to the United Nations Educational, Scientific and Cultural Organization (UNESCO)'s classification as low (≤10 years), middle (11–15 years), or higher education (>15 years). 24 Family disposable household income, based on the Organisation for Economic Co-operation and Development (OECD)-modified equivalence scale, was used as an income measure. 25 Using tertiles, we categorized income as low (lowest 33%), middle (33-66%), or high (highest 33%).
Missing data are negligible in the Danish clinical databases 26 and rare among the demographic and sociodemographic variables (all high-quality, thoroughly documented variables from Statistics Denmark with few missing values).
Analyses
Descriptive statistics (numbers and proportions) were used to compare baseline demographic and socioeconomic characteristics of the invited residents, participants, and not-yet-invited residents.
In the primary analyses, the number of CRC deaths from the date of invitation to end of follow-up was calculated per 100,000 invited and not-yet-invited residents. Kaplan-Meier survival estimates were calculated from date of (pseudo)invitation and illustrated in Kaplan-Meier survival curves. The non-proportional survival curves were compared at end of follow-up using the pseudo value approach. 27 The resulting RRs of CRC deaths for the invited compared with the not-yet-invited residents were presented along with 95% confidence intervals (CIs).
A sensitivity analysis excluding those diagnosed with ulcerative colitis or Crohn's disease was performed using the same methods.
In the secondary analyses, the RR of CRC death for participants compared with a similar group of not-yet-invited residents was calculated using the methods presented by Duffy et al. 28 This method takes into account the healthy-user bias by adjusting the RR for participants compared with the not-yet-invited residents for the RR for non-participants compared with the not-yet-invited residents.
All analyses were stratified according to sex and age groups (50 to 59 years and 60 to 71 years).
Analyses were performed using SAS version 9.4 or STATA version 15 (STATA Corp., College Station, TX, USA).
Results
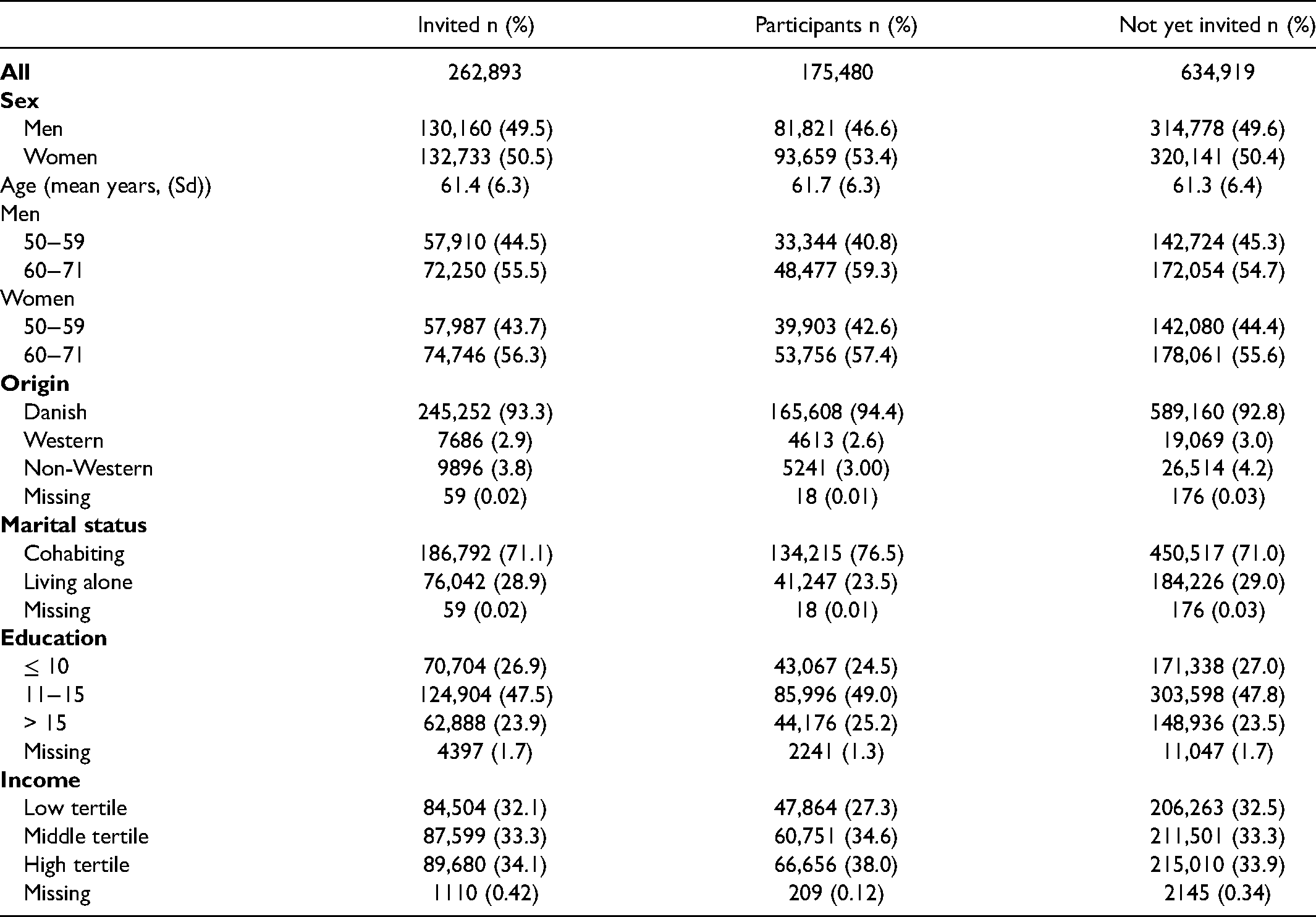
Among 1,548,097 residents eligible for inclusion, 897,812 were included in the study population. Of these, 262,893 (29%) were invited for CRC screening and 634,919 (71%) comprised the not-yet-invited group (Figure 1). The two groups were largely comparable on all socioeconomic and demographic characteristics apart from the fact that the invited residents included slightly fewer non-Western immigrants. Among the invited residents, 175,480 (67%) participated in CRC screening, constituting a group of individuals tending to be older, of Danish origin, and with a higher socioeconomic status (Table 1). Individuals in the study population were followed for up to 3.8 years; the median follow-up was 3.3 years for all groups ranging from 2.0 to 3.8 years for those not censured. A total of 534 CRC deaths were registered during the study period, 141 among invited (63 of those among participants) and 393 among not yet invited.
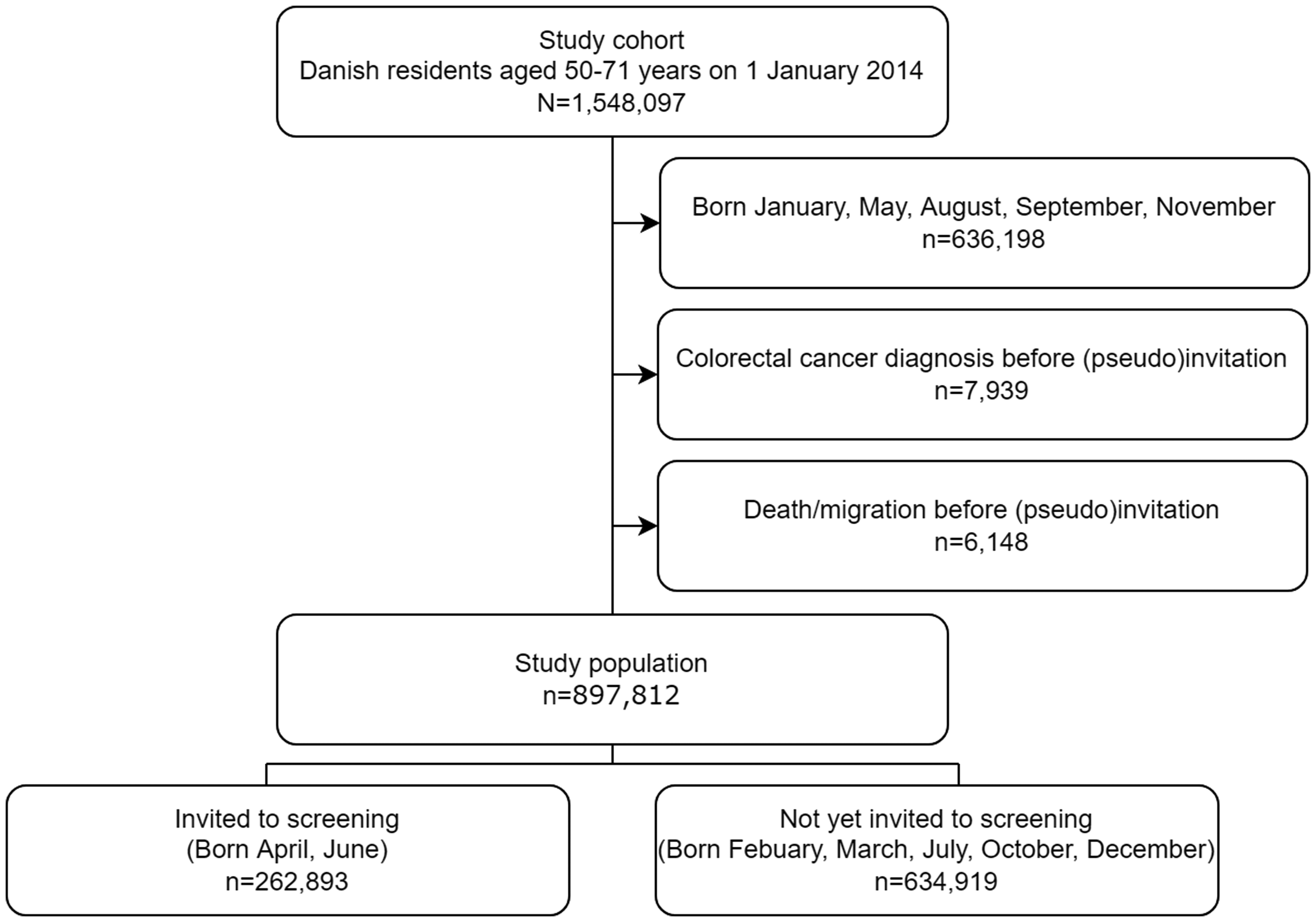
Study inclusion flowchart.
Demographic and socioeconomic characteristics of invited residents, participants (67% of the invited), and not-yet-invited residents (N = 897,812).
The overall RR of CRC death was 0.83 (95% CI 0.66;1.03), corresponding to a statistically non-significant 17% RR reduction in CRC mortality at end of follow-up among those invited to screening compared with those not yet invited (Table 2). Kaplan-Meier survival curves illustrate that survival was similar within the first two years after (pseudo)invitation, whereupon the curves separate (Figure 2).
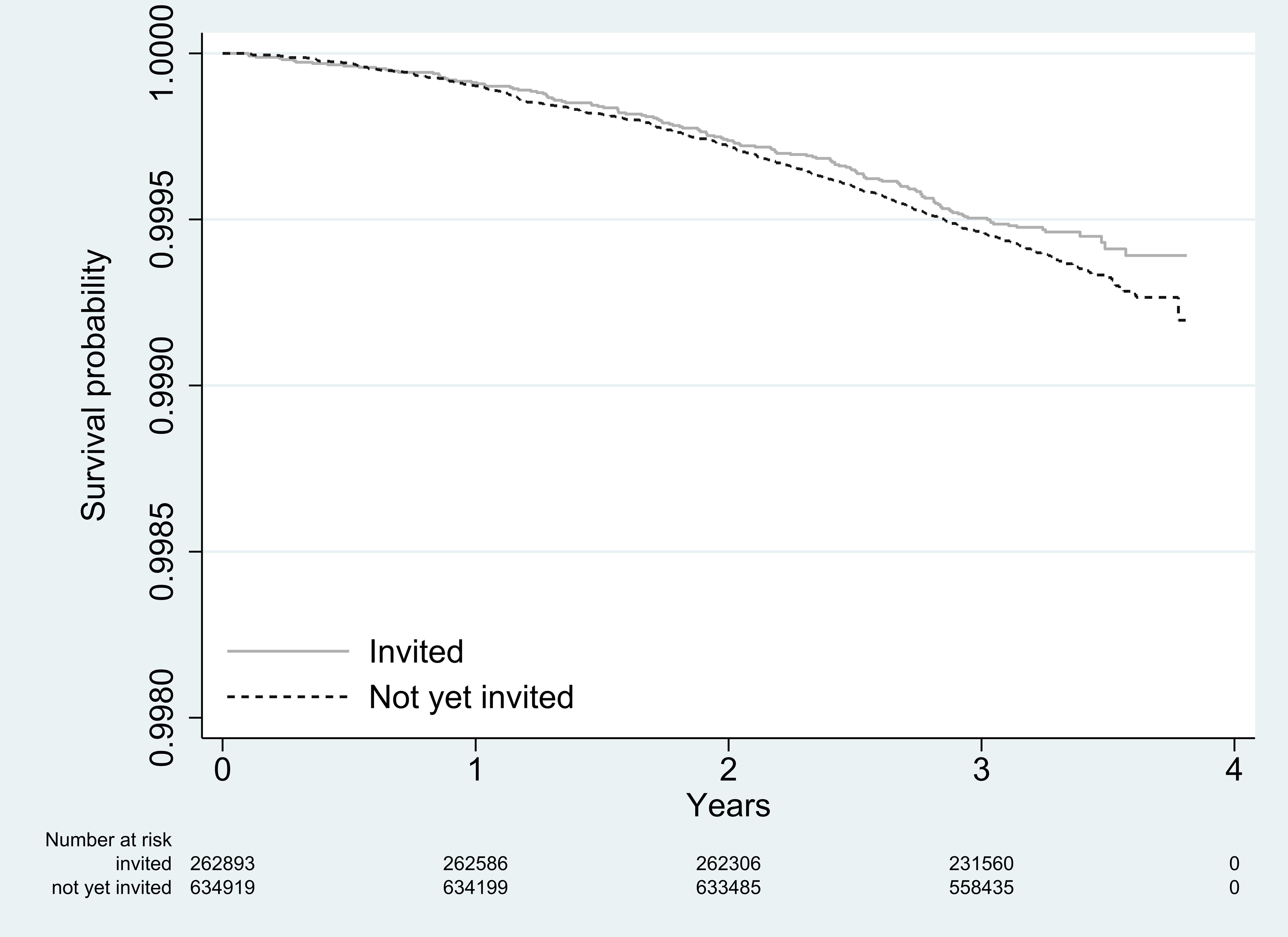
Kaplan-Meier survival curves for invited (solid line) and not-yet-invited (dotted line) residents.
Total number of CRC deaths and number of colorectal cancer (CRC) deaths per 100,000 invited residents, participants, and not-yet-invited residents along with Kaplan-Meier survival estimates and relative risk (RR) of death at end of follow-up with 95% confidence intervals (CI) (N = 897,812).
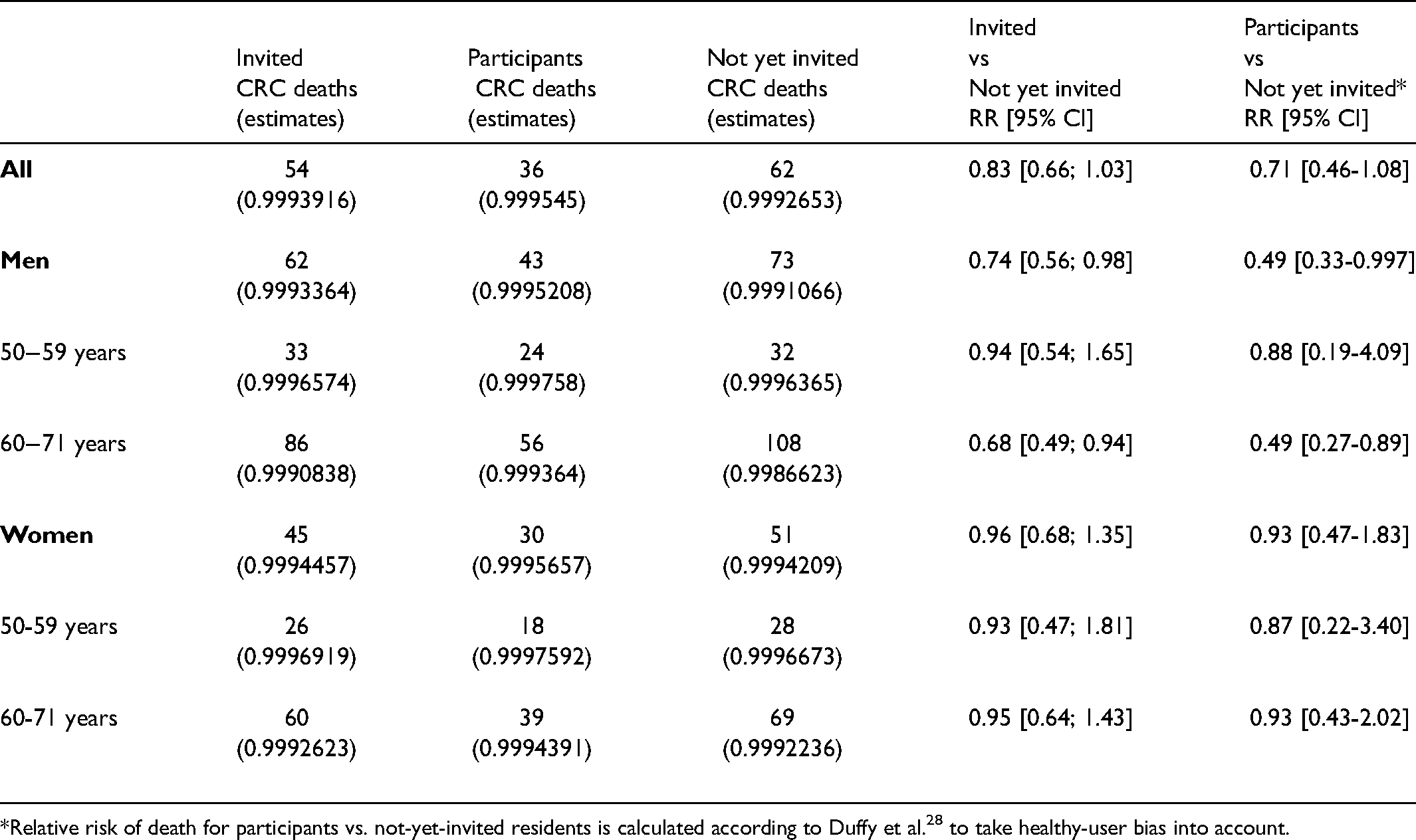
*Relative risk of death for participants vs. not-yet-invited residents is calculated according to Duffy et al. 28 to take healthy-user bias into account.
When stratifying for sex and age, we found a 26% CRC mortality reduction among invited men (RR 0.74 (95% CI 0.56; 0.98)), covering a 32% reduction among invited men aged 60-71 years (RR 0.68 (95% CI 0.49; 0.94)) and a statistically non-significant 6% mortality reduction among invited men aged 50-59 years (RR 0.94 (95% CI 0.54; 1.65)) (Table 2). For invited women compared with not-yet-invited women, only small and statistically non-significant differences were found both overall and in the two age groups (Table 2). For men aged 60-71 years, survival estimates were similar within the first two years after (pseudo)invitation, whereupon the curves separate and become statistically significantly different after 3.5 years (Figure 3).
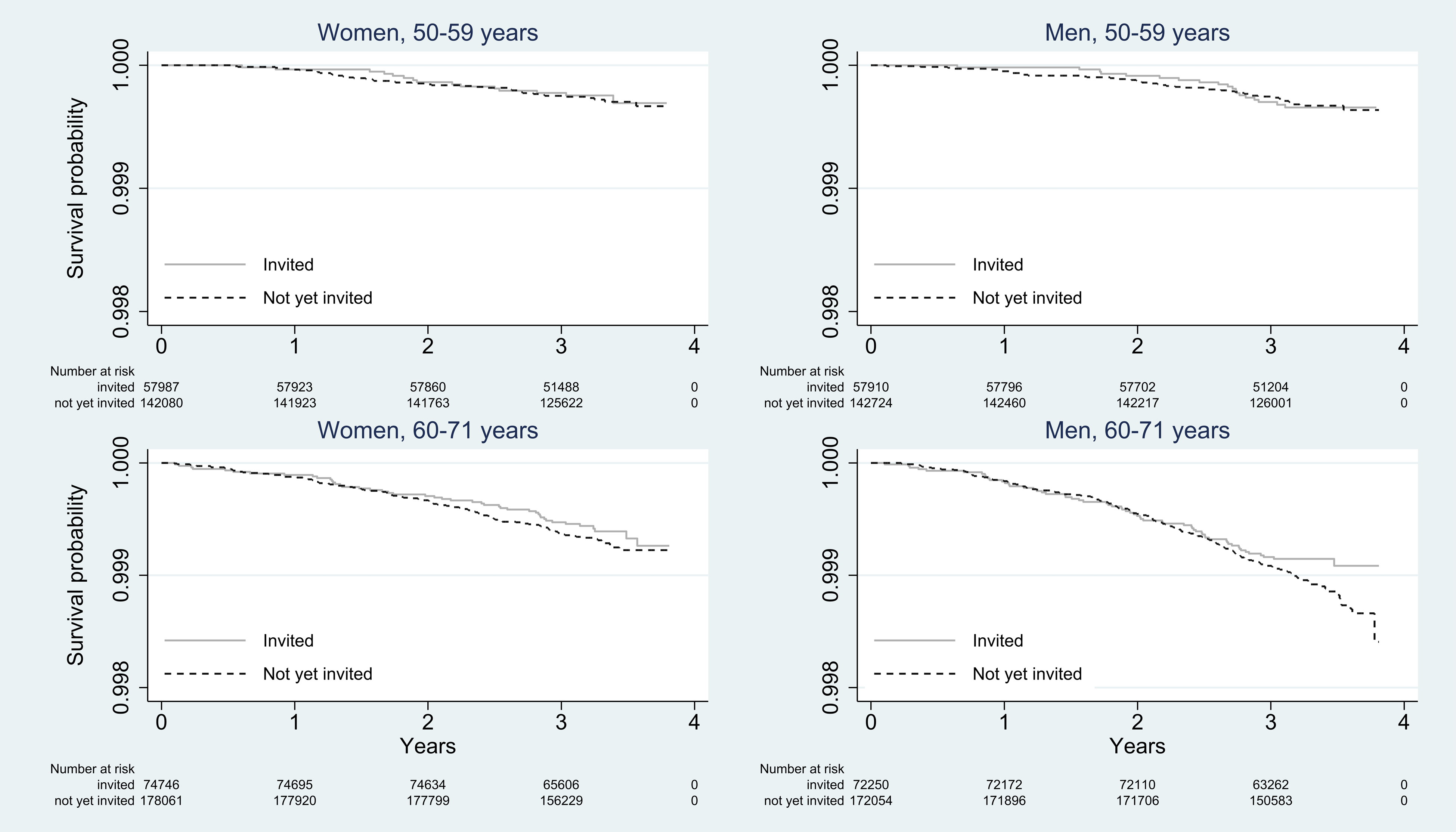
Kaplan-Meier survival curves for invited (solid line) and not-yet-invited (dotted line) male and female residents, aged 50-59 and 60-71 years.
The sensitivity analyses showed that excluding those with ulcerative colitis and Crohn's disease did not alter our conclusions (data not shown).
For those participating in screening compared with a similar group of not-yet-invited residents, we found a statistically non-significant 29% overall reduction in CRC mortality (RR 0.71 (95% CI 0.46−1.08)) when the healthy-user bias was taken into account. For male participants aged 60 to 71 years, we found a 51% reduction in CRC mortality (RR 0.49 (95% CI 0.27−0.89)). For participating women, we found small and statistically non-significant reductions in CRC mortality both overall and in each age group (Table 2).
Discussion
This nationwide register-based cohort study showed that implementing FIT-based CRC screening in Denmark reduced CRC mortality, especially among older men. With a median follow-up of 3.3 years after invitation, invited men aged 60 to 71 years had already achieved a 32% RR reduction in CRC mortality; and when participating in screening, they had achieved a 51% RR reduction. Among women and younger men, CRC mortality was not significantly reduced by implementing FIT-based CRC screening.
This study provides high-level evidence of the effectiveness of implementing a FIT-based CRC screening program. The main strengths were the randomized invitation order ensuring comparability among those invited and those not yet invited and the register-based approach using variables from Danish databases proven to be of high validity and completeness. 26 Thus, both known and residual confounding was minimized.
Our estimates of CRC mortality reduction were undoubtedly underestimated due to several factors. Since the not-yet-invited residents were invited in the latest part of the prevalence round, few of the not-yet-invited residents may actually have benefitted from screening. The pace of invitations was continuously increased as colonoscopy capacity increased. Thus, the vast majority of not-yet-invited were invited within the last year of the prevalence round (mean 260 days before 31 December 2017). However, this bias may be negligible due to the limited effect on mortality within the first year after invitation. Further, the incidence of CRC is higher in the invited than not-yet-invited population, 13 and since prior diagnoses may influence doctors’ coding in the death certificate a slight possibility therefore exists that those invited to screening are more likely to be registered with CRC as cause of death. Most importantly, the relatively short follow-up of a maximum of 3.8 years limited the number of CRC deaths and allowed us to estimate the short-term effectiveness of CRC screening only.
The short-term effectiveness is probably primarily a result of screen-detected cancers being detected at an earlier stage. We have previously shown that the RR of being diagnosed in stage I for invited versus not-yet-invited women and men was 3.39 and 3.71, respectively. 13 Long-term effectiveness is expected to increase because mortality reductions cumulate over screening rounds 29 and the full effect of removal of adenomas will not present itself within this follow-up due to the natural history of CRC.30,31 In an earlier study, we showed a high potential for prevention of CRC owing to removed adenomas, 32 but this impact can be seen only when the follow-up time is longer than the present one. However, it is not possible to prolong the follow-up since there will be no un-screened control group after the programme is fully implemented.
Although statistically non-significant, the magnitude of the 17% overall RR reduction in CRC mortality for invited residents and 29% for those participating is in line with meta-analysis of screening based on gFOBT which demonstrates a 15% and 25% RR reduction in CRC mortality, respectively. 4 However, in the meta-analysis, median follow-up ranges from 11 to 18 years and covers 2-9 screening rounds whereas in our study the follow-up was median 3.3 years and encompassed a single screening round. Thus, the fact that our study demonstrated a similar RR reduction at a median follow-up of 3.3 years indicates that FIT-based screening may ensure larger improvements in CRC mortality in the future. However, cancers diagnosed in the first round of screening most likely differ from those diagnosed in later rounds, screening participation may change over later rounds as well as attention towards timely reaction to symptoms of CRC; all factors influencing how our estimates could have been with longer follow-up. Future research needs to determine how these and other factors will influence the relative reduction in CRC mortality.
The 17% overall RR reduction in the present study is lower than RR reductions reported in two other European studies on FIT-based screening with longer follow-up than in our study. Using an ecological design, an Italian study found a 24% RR reduction in CRC mortality within 10 years in areas initiating screening before other areas. 33 The study used age-period data, counting the number of CRC deaths in the 50-74-year age group and comparing them between the early and late screening areas. Thus, they did not follow residents on an individual level and reported only aggregated data. Using a before/after design, the other Italian study found a 36% reduction in CRC mortality within an eight-year follow-up period among 50-69-year-old men and women, comparing a cohort aged 50-69 years in 1997 (pre-screening) with a cohort aged 50-69 years in 2005 (screening). 34 A limitation of this study is that they did not adjust for temporal differences in mortality even though they demonstrated a 12% temporal difference in mortality in the population above the screening age. Even though the temporal risk reduction was not statistically significant, it may still be clinically relevant.
As expected and demonstrated in our study, those self-selected to participate in screening are generally more well off than the not-yet-invited group, which introduces a healthy-user bias when comparing the two groups directly. Therefore, we accounted for risk of healthy-user bias using the method described by Duffy et al. 28 when analysing the reduction in CRC mortality for those actually participating; we found an overall 29% RR reduction. This is slightly higher than the estimate from a Spanish study that did not correct for healthy-user bias in which FIT-based screening reduced CRC mortality at 5 years by 23%. 35 Yet, compared with our control group of randomly selected not-yet-invited residents, the control group in the Spanish study was very heterogeneous, constituting not-yet-invited individuals, individuals who were not invited by mistake, and self-selected non-participants.
Our results showed that screening was more effective among men than among women, whereas the Italian study, using an ecological design, found a larger mortality reduction among women than among men. 33 Several factors may explain sex differences in the effectiveness of screening. First, the difference may be explained by the underlying prevalence of undiagnosed CRC, where both prevalence and incidence are higher among men than among women and increase more with age for men than for women.36,37 This supports our finding of a higher effectiveness among men than among women, especially among older men. Second, effectiveness also depends on actual participation in screening, which is higher among women than among men in Denmark, 38 indicating that men have a potential for achieving an even higher reduction in CRC mortality. Third, women have a more active healthcare-seeking behaviour than men in general 39 and for alarm symptoms for cancer in particular. 40 We have previously shown that the RR of having a colonoscopy for invited versus not-yet-invited women was 2.58 (2.51−2.66) compared to 3.29 (3.20−3.39) for men, probably also indicating that women are more active in seeking colonoscopies outside the screening programme. 32 Even though screening is supposed to detect CRC before symptoms appear, its effectiveness would be greater among those not reacting promptly on symptoms. Women are almost twice as likely to react to symptoms within one month as men are. 40 Further studies are needed to determine how symptoms may affect participation in and effectiveness of screening differently among men and women.
In conclusion, even within a median follow-up of only 3.3 years, implementation of FIT-based CRC screening reduced CRC mortality among men aged 60 to 71 years. Yet it demonstrated limited effect among younger men and women in both age groups which may be explained by the relatively short follow-up. Future research needs to explore this.
Footnotes
Ethics
According to the EU's General Data Protection Regulation (Article 30), the project was listed on the record of processing activities for research projects in the Central Denmark Region (R. No.: 1-16-02-396-16). According to the Consolidation Act on Research Ethics Review of Health Research Projects, Consolidation Act number 1083 of 15 September 2017, Section 14 (2), notification of medical database research projects to the research ethics committee system is required only if the project involves human biological material.
Data availability statement
The data that support the findings of this study are available from The Danish Health Data Authority and Statistics Denmark. Restrictions apply to the availability of these data, which were used under license for this study. The data may be available upon reasonable request to The Danish Health Data Authority and Statistics Denmark.
Funding
Sisse Helle Njor was funded by the Health Research Fund of Central Denmark Region (no. A1518). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Disclosure
The authors declare no conflicts of interest
Author Contributions
BA, SHN, and MBL designed the study; SHN and BS analysed the data; and MBL drafted the manuscript. All authors revised the manuscript critically and approved the final version to be published. All authors are accountable for all aspects of the work.
Disclosure
The authors declare no conflicts of interest
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
